Abstract
Infestation with nonnative, “exotic” lice was first noted in Washington black-tailed deer (Odocoileus hemionus columbianus) in 1994 and has since then spread throughout the western United States. In California, infestation with the exotic louse Damalinia (Cervicola) sp. was first detected in black-tailed deer from northern California in 2004, and, in 2009, the exotic louse species Bovicola tibialis and Linognathus africanus were identified on mule deer (Odocoileus hemionus californicus) in central Sierra Nevada in association with a mortality event. Exotic lice have since been detected in various locations throughout the state. We describe the geographic distribution of these exotic lice within California, using data from 520 live-captured and 9 postmortem-sampled, free-ranging mule deer examined between 2009 and 2014. Data from live-captured deer were used to assess possible associations between louse infestation and host age, host sex, migratory behavior, season, and blood selenium and serum copper concentrations. Damalinia (Cervicola) sp. and B. tibialis lice were distinctively distributed geographically, with D. (Cervicola) sp. infesting herds in northern and central coastal California, B. tibialis occurring in the central coastal mountains and the Sierra Nevada, and L. africanus occurring only sporadically. Younger age classes and low selenium concentrations were significantly associated with exotic louse infestation, whereas no significant relationship was detected with serum copper levels. Our results show that exotic lice are widespread in California, and younger age classes with low blood selenium concentrations are more likely to be infested with lice than older deer.
Keywords
Introduction
Pediculosis caused by infestation with nonnative louse species (exotic pediculosis of deer, EPOD) and associated hair-loss syndrome (HLS) was first noted in Columbian black-tailed deer (BTD; Odocoileus hemionus columbianus) in western Washington State in 1995.1,11 Deer with exotic pediculosis exhibit various degrees of hair loss ranging from thinning to complete loss of the hair coat on much of the body, often accompanied by poor body condition and increased loads of pulmonary and gastrointestinal parasites. 2 Two chewing lice have been associated with the recent emergence of EPOD. Damalinia (Cervicola) sp. was first reported in BTD from western Washington and Oregon, 2 whereas Bovicola tibialis was found on Rocky Mountain mule deer × black-tailed deer hybrids in south-central Washington in 2005–2006. 18 In addition, the African blue louse (Linognathus africanus) caused alopecia and mortalities in deer in California in 1967 3 but is not considered a significant contributor to the recent emergence of EPOD.
The clinical manifestation of EPOD caused by D. (Cervicola) and B. tibialis seems to be similar, and infested animals may exhibit pruritus and groom and rub extensively because of the irritation caused by the chewing lice, causing hair damage, hair loss, secondary skin hyperpigmentation, and hyperkeratosis. 1 The syndrome is seen primarily in fawns and yearlings but also can affect adult animals. In Oregon and Washington, prevalences of hair loss as high as 74% in BTD fawns and 33% in does were observed in affected herds. 1
In California, EPOD was first recognized in 2004, when hair loss, poor body condition, and infestation with the exotic louse D. (Cervicola) sp. were noted in BTD deer in Humboldt County of northern California (D. Lancaster, California Department of Fish and Wildlife [CDFW], pers. comm.). Infestation with exotic lice received more attention in 2009, when unusual mortality was observed in a resident herd of California mule deer (CMD; Odocoileus hemionus californicus) in Tuolumne County in the central Sierra Nevada of California. Examined deer from the herd were infested with B. tibialis and L. africanus lice, 18 and had mild to severe hair loss, verminous pneumonia, and copper and selenium deficiencies (CDFW, unpublished observations).
The louse species B. tibialis and L. africanus are not new to California, but widespread infestations have not been observed previously. Bovicola tibialis was probably introduced with European fallow deer (Dama dama), and it was confirmed on BTD in Mendocino County in 1973 and on D. dama in Marin County in 2005.18,28 D. (Cervicola) sp. was also identified on Roosevelt elk (Cervus canadensis roosevelti) from Del Norte County in April 2005, 2 BTD in Siskiyou County in 2005, a BTD from Trinity County, and tule elk (Cervus canadensis nannodes) in March 2008 (J Mortensen, unpublished observations; Fig. 1a–d). D. (Cervicola) sp. lice were probably introduced into the Pacific Northwest with exotic Asian deer; however, the time, place, and origin of the introduction are unknown, and examination of archived louse specimens from British Columbia and Oregon failed to find any examples of D. (Cervicola) lice from before the 1990s. 2 The sucking louse (L. africanus), an ectoparasite of sheep and goats, was first reported on deer in the United States in association with mortalities in BTD and CMD in California, 3 and was also detected on deer from Lassen, Shasta, and Siskiyou Counties in 2005 (J Mortensen, unpublished observations; Fig. 1).
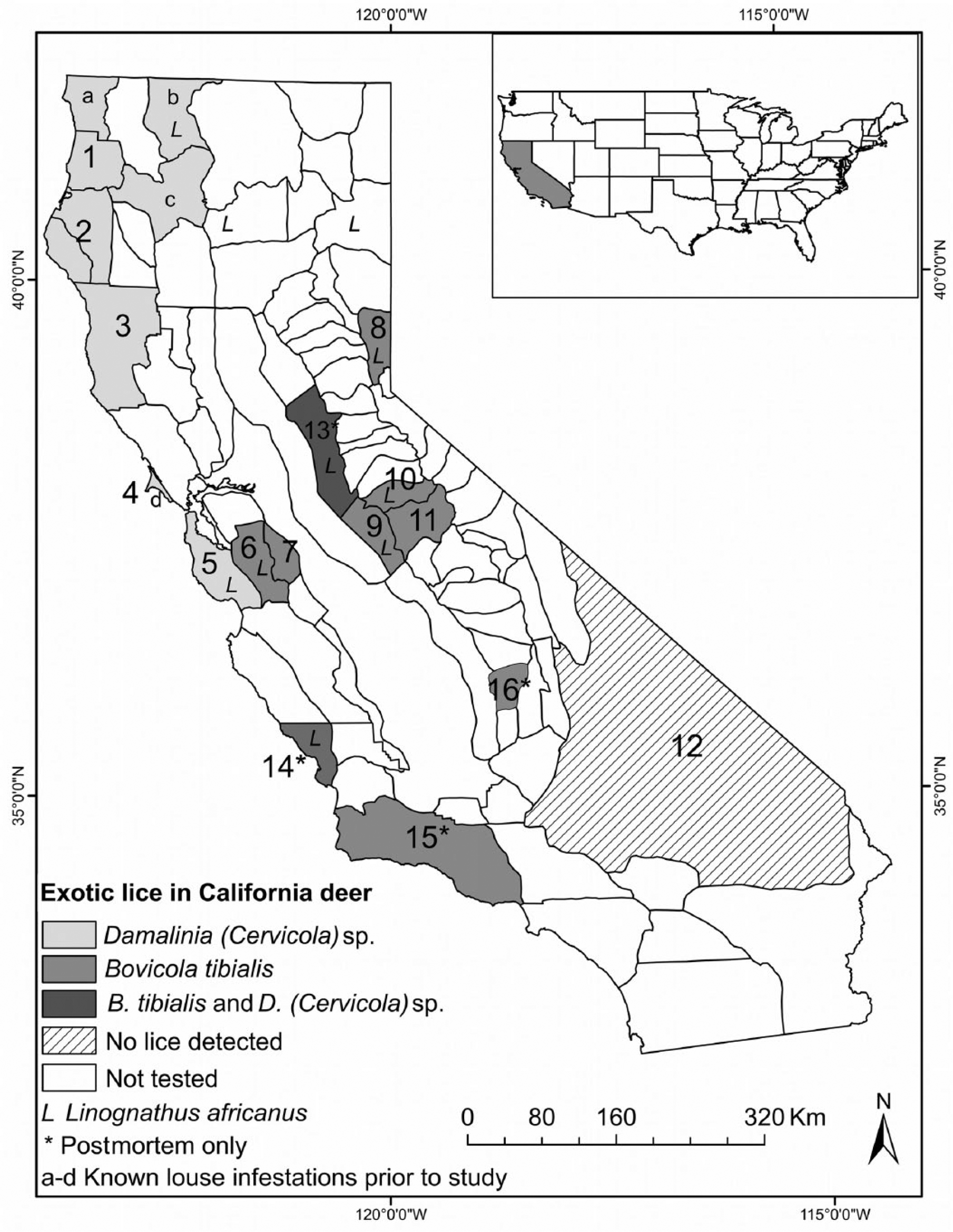
Geographic distribution of Damalinia (Cervicola) sp., Bovicola tibialis, and Linognathus africanus lice in 16 California deer herds (for details on herds 1–16, see Table 1) variously sampled between December and June of 2009–2013.
Risk factors for development of EPOD have been evaluated in Washington and Oregon deer, and, in addition to the presence of louse infestation and age of the animal, nutritional stress in the winter months was suggested as a contributing factor that could reduce host immunity and impair the deer’s ability to rid the lice.1,18 The cases of EPOD in 2009 in Tuolumne County, California, were associated with high mortality, and most afflicted deer were severely deficient in both selenium and copper. Such mineral deficiencies have been reported in California wildlife before the emergence of EPOD.7,9,12,14 However, because both trace minerals have important functions for the immune system, 13 we wondered whether their deficiencies could impair the immune response of infested deer to lice, and thereby increase the risk of explosive louse infestations and subsequent HLS. Our initial field observations additionally suggested that EPOD was most severe in resident deer populations in California. Resident deer often occur at higher densities and are at higher risk for seasonal malnutrition than are migratory deer, and both scenarios could be risk factors for flourishing pediculosis. Therefore, to investigate this hypothesis, we surveyed for the presence of B. tibialis and D. (Cervicola) sp. lice in a select subset of deer herds in California, and we assessed possible associations between louse occurrence and levels of whole blood selenium and serum copper, host age, host sex, season, and migratory behavior in these deer herds.
Materials and methods
We included data from a total of 520 live deer (148 BTD, 284 CMD, 23 Rocky Mountain mule deer [RMMD; Odocoileus hemionus hemionus], 65 desert mule deer [DMD; Odocoileus hemionus eremicus]), plus 9 postmortem-sampled BTD, which originated from 16 deer herds (Table 1, Fig. 1). Climate and vegetation types varied among geographical areas occupied by the deer, with conifer forest, mixed woodland, and high winter rainfall in the northeastern coastal mountains; mixed woodland, grassland, moderate winter rainfall, and more proximate urban areas in the central coastal mountains; arid scrubland in the southeastern desert areas; and conifer forest and mixed woodland with winter snowfall in the higher elevations of the Sierra Nevada. 20
Observed characteristics of 520 live and 9 postmortem-sampled deer from 16 California deer herds and their associated exotic lice sampled, between December and June of 2009–2013.*
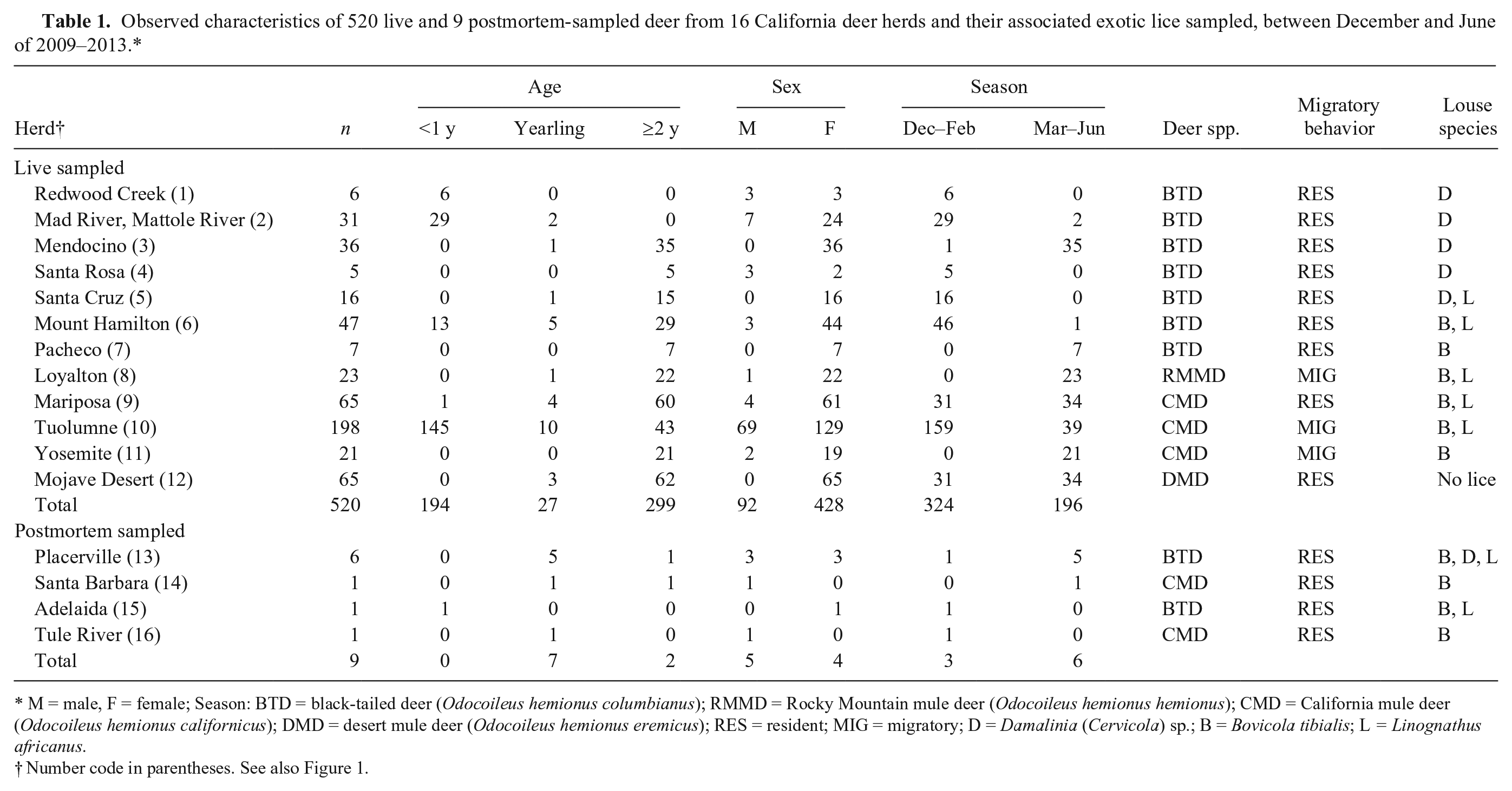
M = male, F = female; Season: BTD = black-tailed deer (Odocoileus hemionus columbianus); RMMD = Rocky Mountain mule deer (Odocoileus hemionus hemionus); CMD = California mule deer (Odocoileus hemionus californicus); DMD = desert mule deer (Odocoileus hemionus eremicus); RES = resident; MIG = migratory; D = Damalinia (Cervicola) sp.; B = Bovicola tibialis; L = Linognathus africanus.
Number code in parentheses. See also Figure 1.
The 520 live-captured, free-ranging deer from 12 herds were sampled in association with various research studies or management-related field activities conducted by CDFW between December and June in the years 2009–2013. The age and sex of the animals varied depending on the goal of the capture. Only data from captures conducted December–June were included because EPOD rarely occurs in the summer and fall months. The 9 postmortem-sampled deer originated from 4 additional deer herds, which were not part of the field studies (Fig. 1). They were submitted by biologists to the CDFW Wildlife Investigations Laboratory (Rancho Cordova, California) between December and June of 2009–2014 and were opportunistically included to describe the louse distribution by deer herd across the state, but not included in the risk factor data because fresh blood samples were not taken from them for analysis of trace minerals.
All animal captures were overseen and approved by CDFW. Capture methods included net-gunning from a helicopter, a clover trapping, 5 and free-range darting, b using a combination of 2.2 mg/kg of tiletamine plus zolazepam c and 2.2 mg/kg of xylazine. d Immobilized deer were hobbled and blindfolded. Blood samples were collected by jugular venipuncture. Age class (<1 year, yearling, or ≥2 years; estimated based on tooth wear), and sex, hair coat condition (normal, alopecia), deer subspecies, and migratory behavior (migratory, resident) were recorded for each animal. We classified sampled deer as migratory or resident types, based on specific knowledge of the local deer herd’s usual behavior. Migratory deer move seasonally between high-elevation summer ranges and low-elevation winter ranges, whereas resident deer largely remain in the same area throughout the year.
Ectoparasites were collected by hand or with forceps from the abdomen, flanks, and neck of the deer, and stored in 70% ethyl or isopropyl alcohol. The number of ectoparasites collected from each animal varied from 1 to several hundred, depending on the severity of infestation. Intensities of pediculosis were not assessed for most sampled deer; individual deer with any exotic lice (i.e., D. (Cervicola) sp., and/or B. tibialis), regardless of abundance, were scored positive for lice in our analysis. If no lice were detected on a deer, its record was so noted and scored as negative for lice. Other ectoparasites were collected opportunistically. All ectoparasite samples were stored or transported at ambient temperatures and identified by standard morphologic methods at the U.S. Department of Agriculture National Veterinary Services Laboratories in Ames, Iowa, where voucher specimens are retained.
Blood was collected in EDTA tubes and tubes without anticoagulant. EDTA tubes were stored at 4°C or frozen at −20°C until submission to the California Animal Health and Food Safety Laboratory (CAHFS) in Davis, California, for selenium analysis. Tubes without anticoagulant were allowed to clot at environmental temperature for 2–10 h and centrifuged at 1,808 × g for 10 min. Serum was then transferred into plastic cryovials, and refrigerated at 4°C or frozen at −20°C until submission to CAHFS for trace mineral analysis. Whole blood selenium concentrations were measured from EDTA (ethylenediamine tetra-acetic acid) blood by inductively coupled argon plasma–spectrometric analysis (ICP), e as described previously. 25 The limit of quantification of the assay was 0.01 mg/kg. Copper concentrations were measured in serum samples using an ICP emission spectrometer.e,17 Accuracy of ICP results for these elements was measured by analyzing quality assurance sera obtained from the Veterinary Laboratory Association Quality Assurance Program. f Data were accepted if analyzed quality assurance serum values were within 2 standard deviations of the reference values. 24 Mineral concentrations below the detectable limit were included in the analysis by assigning values of half the detection limit. 27
Statistical analysis
The percentages of sampled deer with hair loss and/or lice were calculated by herd, age class (<1 year, yearlings, and ≥2 years), sex, season (December–February, March–June) and migratory behavior (migratory, resident). The median and range of whole blood selenium (mg/kg) and serum copper (mg/L) were described by the same variables.
Mixed effects logistic regression models were developed to evaluate possible associations between louse presence (yes/no) and whole blood selenium, serum copper, host age class, host sex, season, or migratory behavior. The models were adjusted for dependence among deer sampled from the same herd by including a random effect for herd. The Akaike information criterion (corrected for small sample size, AICc) was calculated for each model, and models were ranked based on difference in AICc between the best approximating model and all other models (ΔAICc). Models with ΔAICc < 2 were deemed to have substantial levels of empirical support and considered to be competing models to the best model. 4 Model weights (ωi), which can be interpreted as the probability that a model is the best model given the candidate set of models, were calculated for each model. 4 Evidence ratios were calculated for the best-fitting model and competing models by dividing the Akaike weights of the top model with that of the competing model. 4 The probability of louse infestation was graphed for the top model by converting log odds into probability. 23 Statistical and mapping softwareg,h,i were used for the analyses and to generate a distribution map of exotic lice among deer sampled in California.
Results
Among the 520 deer sampled, 9% had hair loss, and 45% were exotic louse-infested (Table 2). Specifically, 63% of deer <1 year of age were louse-infested compared with only 32% of deer ≥2 year of age. Because the deer captures were conducted for various purposes with different age and sex combinations of the captured animals, it was difficult to compare the prevalences of louse infestation and hair loss across herds. Damalinia (Cervicola) sp. lice were predominantly detected in coastal deer herds, whereas B. tibialis lice were present in herds from the Sierra Nevada and coastal mountains (Table 1, Fig. 1). A small number of postmortem-sampled deer from the Placerville deer herd (herd 13) in the Sierra Nevada foothills had both D. (Cervicola) sp. and B. tibialis lice. To date, no lice have been detected on deer in the southern desert areas (Table 1, Fig. 1). Whole blood selenium and serum copper concentrations varied by herd, and half of the sampled herds had individual deer with undetectable selenium concentrations (Table 2). Linognathus africanus was detected in low numbers only in the coastal and Sierra Nevada deer herds together with D. (Cervicola) sp. or B. tibialis (Table 1). Other ectoparasite species detected opportunistically and in low numbers included Dermacentor albipictus, Dermacentor occidentalis, Euschoengastia numerosa, Ixodes pacificus, Lipoptena depressa, Neolipoptena ferrisi, Neoschoengastia fullbergae, Ornithodoros coriaceus, Pulex simulans, Solenopotes ferrisi, and Trichodectidae spp. Because these ectoparasites were not evaluated systematically, their geographical distributions could not be described. The model with the variables selenium, age, and season had the lowest AICc, and one model, with the variables selenium, age, season, and migration, was considered to have substantial support (ΔAICc < 2) compared with the top model (Table 3). The evidence ratio between these 2 competing models was 2.07, indicating that the most parsimonious model containing the independent variables selenium, age, and season was twice as likely to be the best model compared with the competing model (Table 3). Young age class and decreasing whole blood selenium concentrations were significantly associated with the presence of lice (Table 4). In the spring, deer <1 year of age with blood selenium <0.3 mg/kg, had ~20% higher probability of having lice than deer ≥2 years of age with the same selenium blood concentrations; deer <1 year of age with blood selenium concentrations ≤0.2 mg/kg had >30% higher probability of having lice than deer of the same age class with blood selenium concentrations ≥0.4 mg/kg (Fig. 2).
Percentage of deer with hair loss and exotic lice, as well as selenium and copper concentrations of 12 live-sampled California deer herds, with summaries according to host age, host sex, season, and behavior, between December and June of 2009–2013.*
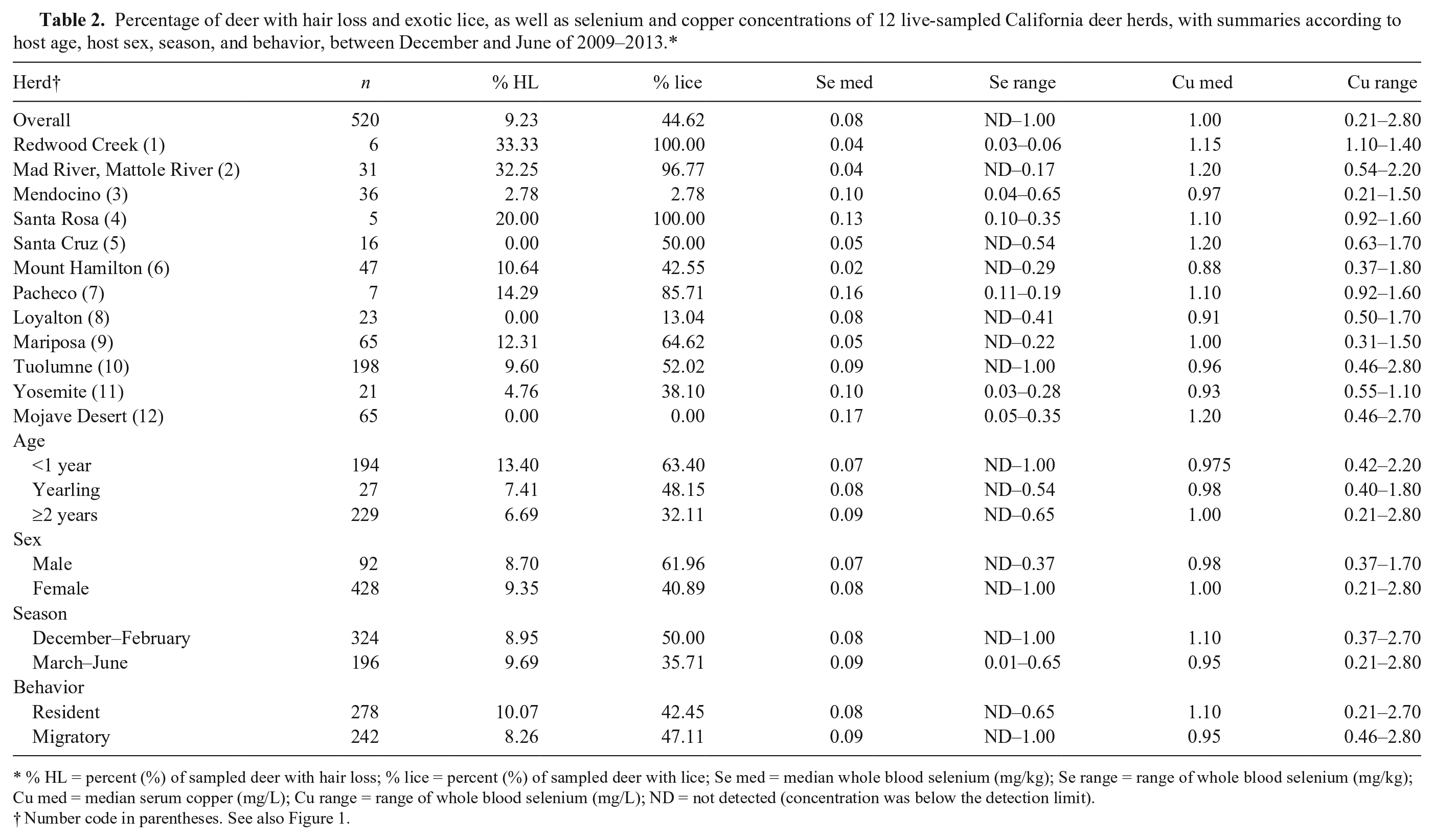
% HL = percent (%) of sampled deer with hair loss; % lice = percent (%) of sampled deer with lice; Se med = median whole blood selenium (mg/kg); Se range = range of whole blood selenium (mg/kg); Cu med = median serum copper (mg/L); Cu range = range of whole blood selenium (mg/L); ND = not detected (concentration was below the detection limit).
Number code in parentheses. See also Figure 1.
Rankings of mixed effects logistic regression models relating presence of exotic lice in California deer with various derived or observed herd characteristics, between December and June of 2009–2013.*
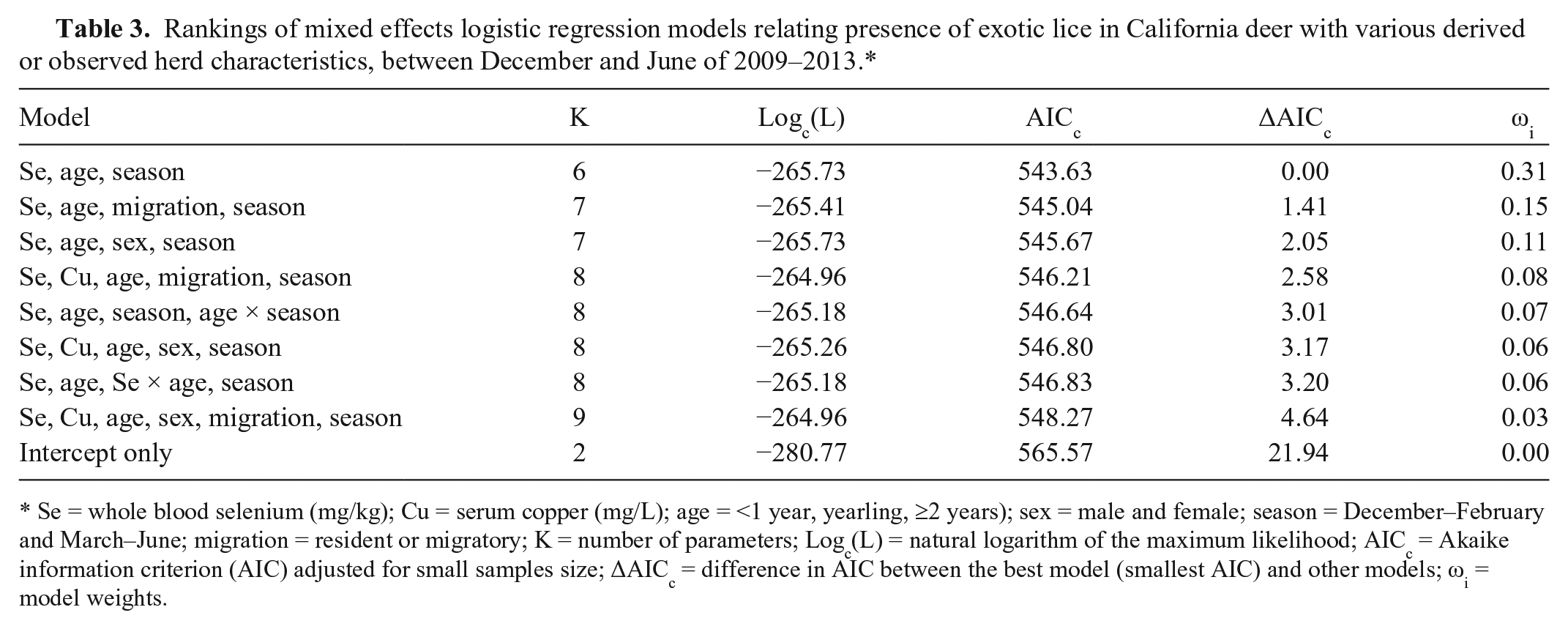
Se = whole blood selenium (mg/kg); Cu = serum copper (mg/L); age = <1 year, yearling, ≥2 years); sex = male and female; season = December–February and March–June; migration = resident or migratory; K = number of parameters; Logc(L) = natural logarithm of the maximum likelihood; AICc = Akaike information criterion (AIC) adjusted for small samples size; ΔAICc = difference in AIC between the best model (smallest AIC) and other models; ωi = model weights.
Mixed effects logistic regression model assessing the association between infestation with exotic lice and the independent variables whole blood selenium, age class, and season in California deer, between December and June of 2009–2013.*
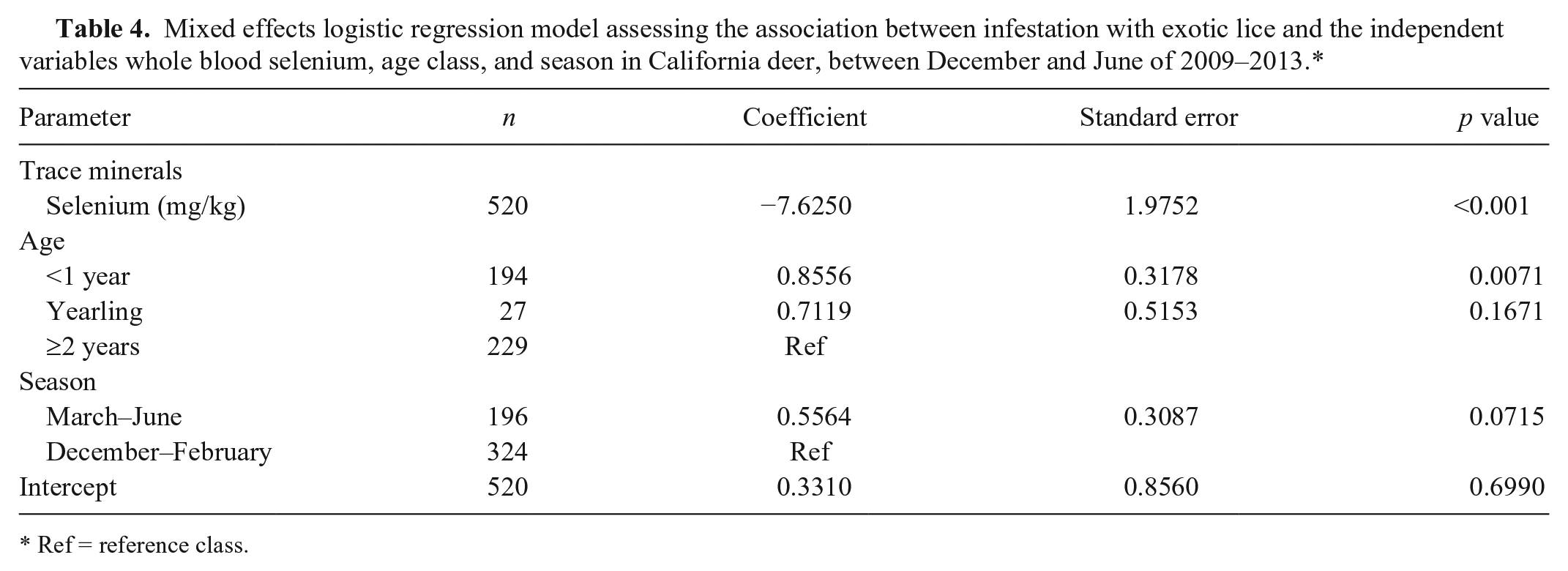
Ref = reference class.
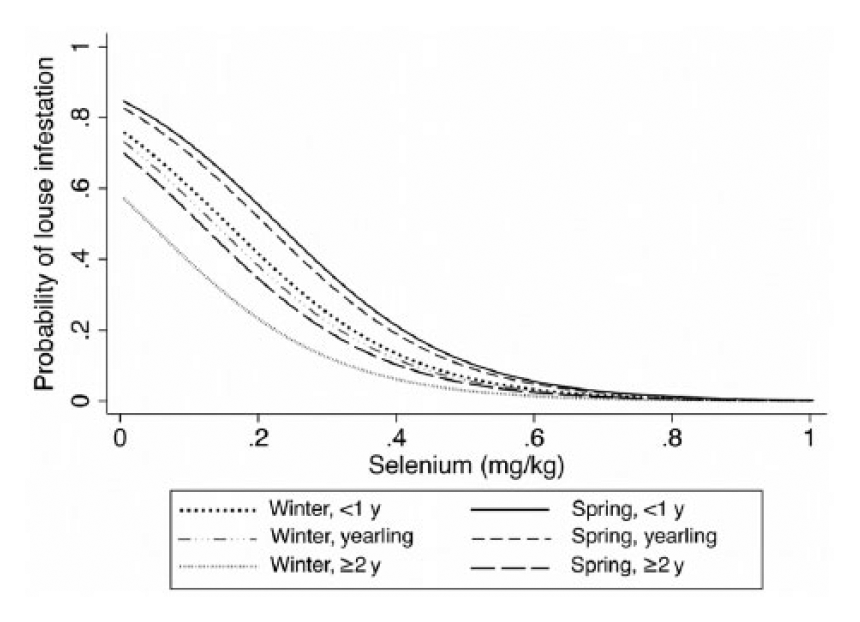
Probability of louse infestations for 3 age classes and 2 seasons (December–February, March–June) versus whole blood selenium concentrations (mg/kg) based on a mixed effects logistic regression model (shown in Table 4), assessing the association between infestation with exotic lice and the independent variables whole blood selenium, age class, and season in California deer in 2009–2013.
Discussion
In contrast to reports from Oregon and Washington, where D. (Cervicola) sp. is the predominant louse in BTD herds, we detected both B. tibialis and D. (Cervicola) sp. lice in BTD from the coastal mountain range. Experimental studies have shown that RMMD are susceptible to infestation with D. (Cervicola) sp. when in close contact with infested BTD (Robinson JA. Transmission of the chewing louse, Damalinia [Cervicola] sp., from Columbian black-tailed deer [Odocoileus hemionus columbianus] to Rocky Mountain mule deer [Odocoileus hemionus hemionus] and its role in deer hair-loss syndrome [master’s thesis]. Corvallis, OR: Oregon State University, 2007), and infrequent mixing between deer herds and subspecies may explain the geographical distribution of the lice. Additionally, hair loss has only been detected sporadically in elevations above 600 m for D. (Cervicola) sp.–infested populations. 2 To date, lice have not been detected in DMD herds in southeastern California, which may be attributed to the dry climate and isolated location. Deer in the Placerville herd should be sampled and monitored in order to better understand the unique occurrence of both louse species in that area.
The higher louse infestation in young deer with marginal or low selenium levels may reflect lowered immune function, as observed in cattle with selenium deficiencies, 13 or generally poorer body condition, which could impair the deer’s ability to rid the lice. Selenium plays an important role in the antioxidant defense system, and selenium deficiency may impair lymphocyte function and the microbicidal activity of phagocytes. 8 Furthermore, chronic inflammation may increase oxidative stress. 6 These and other factors may contribute to the association between low selenium and louse infestation, but based on our data, we cannot make any inference about the exact immunologic mechanisms behind the observed trends.
Selenium deficiencies and regional variations in selenium concentrations have previously been reported in free-ranging mule deer throughout California.7,9,19 A study conducted in 3 deer management groups in the central Sierra Nevada (total n = 276) in 1980–1988 detected mean blood selenium concentrations of 0.07–0.08 mg/kg, 19 and these values are largely comparable to the median selenium concentrations detected in the Mariposa, Tuolumne, and Yosemite herds, indicating that selenium concentrations have not significantly changed over time. Previous studies have reported ranges of selenium concentrations of deer in or near the Mt. Hamilton herd of 0.02–1.16 and 0.01–0.42 mg/kg, respectively.7,19 The upper ends of these ranges are higher than we observed in our study, but because the past studies did not report the sampling season(s) for individual herds, direct comparisons to past data are difficult. Although selenium deficiencies have frequently been reported in California deer, they cannot be considered the sole driver of EPOD emergence, but they may influence the host’s response to an exotic louse infestation. Definitive selenium reference ranges have not been established for deer. Suggested ranges include <0.05 mg/L as “extremely deficient” and 0.08–0.50 mg/L as “adequate” for plasma selenium concentrations, 10 and 0.06–0.15 mg/L as “adequate” for serum selenium concentrations. 21 Our findings indicate that the probability of exotic lice infestation is higher in young deer with selenium concentrations in the deficient and marginal ranges compared with deer with higher selenium concentrations and older deer.
Low serum copper was not associated with EPOD, although a few deer had serum copper concentrations far below the proposed adequate range of 0.60–1.30 mg/L for mule deer and BTD. 21 Serum is generally a less reliable sample to measure copper levels than liver tissue, 26 and the copper deficiencies detected in our original 2009 cases of EPOD in California were based on measurements from liver samples. As opportunities arise to collect postmortem samples, the role of copper and other nutritional factors as factors contributing to EPOD should be further examined. Little information is available on serum copper levels in free-ranging mule deer from California for comparison, but copper deficiencies have been implicated in unthriftiness and poor reproductive performance in tule elk in Point Reyes, 12 as well as in an abnormal rate of antler breakage in elk from Owens Valley. 14
Over the past 2 decades, EPOD in deer has spread from Washington to California, and the range of the lice continues to expand. In 2009 and 2011, respectively, B. tibialis was detected in Riggins in western Idaho, Box Butte County in Nebraska, Carbon County in Wyoming, 18 Fallon in western Nevada, Tonopah in central Nevada, and Baker in eastern Nevada (P Wolff, Nevada Department of Wildlife, pers. comm.). In 2015, B. tibialis was detected in areas south, west, and north of Salt Lake City, Utah (A Roug, unpublished observations), as well as near Denver, Colorado (K Fox, Colorado Parks and Wildlife, pers. comm.). It is unclear why the exotic lice apparently have spread so rapidly. The parthenogenetic reproduction of the Bovicola and D. (Cervicola) sp. lice, a mostly naïve deer population, and possibly environmental and immune-suppressive factors could play a role in the spread of exotic lice. Similar alopecia in Scandinavian moose, caused by infestations with native deer keds (Lipoptena cervi), as well as increasing incidences of winter tick (Dermacentor albipictus) infestation in moose in Alberta, Canada has been attributed to unusually warm weather.15,22
Limited information is available on possible population impacts of exotic louse infestations. A study in western Washington game management units reported that fawn production and preweaning survival was possibly reduced in deer with HLS, but fawn overwinter survival did not appear to be affected. 1 In contrast, a study in the northwestern Olympic Peninsula of Washington found decreased overwinter survival in fawns with HLS. 16 Without long-term population and demographic data from affected areas, it is difficult to determine what consequences exotic louse infestations will have for California deer populations. Except in unique circumstances, where the louse infestation is restricted to a single isolated deer herd, intervention through antiparasitic treatments to reduce louse loads is not practical in free-ranging populations because treatment of all affected animals is unlikely. During captures, deer or elk should be carefully examined for ectoparasites in order to detect new foci of louse infestations. Results of this study demonstrated that young deer with low blood selenium are most likely to be infested by exotic lice. Longitudinal studies are underway to provide further information concerning population impacts of lice combined with trace element deficiencies, and the possible effect of antiparasitic treatment and trace element supplementation on EPOD in California.
Footnotes
Acknowledgements
We thank the many CDFW biologists, environmental scientists, scientific aids, volunteers, and other field personnel, as well as the CAHFS Laboratory analytical chemists, for technical assistance and expertise needed for deer capture, blood and sample collection, sample preparation and analysis, data recording, and processing of the samples. Also thank you to Thomas Edwards, Utah State University, for advice on the statistical analysis.
Authors’ contributions
A Roug drafted the manuscript. A Roug, P Swift, G Gerstenberg, S Torres, B Puschner, and L Woods contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. JW Mertins contributed to conception of the study, and contributed to analysis and interpretation of data. CK Johnson contributed to analysis of data. J Mortensen contributed to acquisition of data. All authors critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Coda Netgun, Coda Enterprises, Mesa, AZ.
b.
Pneu-dart, Williamsport, PA.
c.
Telazol, Fort Dodge Animal Health, Fort Dodge, IA.
d.
AnaSed (100 mg/mL), Lloyd Inc., Shenandoah, IA.
e.
FISONS (Accuris model), Thermo Optek Corp., Franklin, MA.
f.
Genzyme Diagnostics, Blaine, MD.
g.
STATA version 13.3, StataCorp, College Station, TX.
h.
R-Studio version 0.98.939, R development core team, 2015, Boston, MA.
i.
ArcMap, version 10.2, ESRI Corp., Redlands, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the California Department of Fish and Wildlife.
