Abstract
Acute inflammatory processes can trigger increased production of acute phase proteins (APPs) that can be useful biomarkers of inflammation. APPs are diverse and include proteins involved in coagulation, opsonization, iron regulation, and limitation of tissue injury. Haptoglobin, serum amyloid A, and alpha-1 acid glycoprotein have been proposed as useful APPs in goats. APPs can differ markedly by species, therefore species-specific reference intervals and studies are necessary. The objective of this study was to determine species-specific reference intervals for 4 APPs in goats. Haptoglobin, serum amyloid A, lipopolysaccharide binding protein, and alpha-1 acid glycoprotein were measured in in 54 clinically normal adult goats. APPs were measured using goat-specific commercial enzyme-linked immunosorbent assay kits. Results were analyzed by 1-way analysis of variance to compare sexes and breeding status. Reference Value Advisor was used to calculate reference limits according to the IFCC-CLSI guidelines. Only 1 APP was found to vary in healthy animals; serum haptoglobin was increased in lactating animals and decreased in pregnant does in their second trimester when compared with open, nonlactating does. No sex-based differences were seen for any of the APPs measured. We report normal reference intervals for 4 serum APPs that may be useful as disease markers. Haptoglobin should be interpreted with caution in animals with unknown pregnancy status. Further studies are needed to determine whether these APPs are useful biomarkers in goat disease states.
Keywords
Introduction
Any acute inflammatory process can trigger an acute phase response. Infectious causes are common, but many noninfectious causes (e.g., trauma, strenuous exercise) are also documented. The body’s response to an inflammatory insult is altered liver production of many proteins, termed acute phase proteins (APPs). Most of these proteins are termed positive APPs, meaning that production increases with inflammation. The positive APPs are a diverse group including proteins involved in coagulation, opsonization, iron regulation, and limitation of tissue injury. Protein electrophoresis can indicate increases in globulin fractions that are composed largely of APPs and nonspecifically indicate inflammation. Measurement of individual APPs can be useful diagnostically and is not limited to blood; assays for APPs in milk and saliva have also proven useful.4,10,11
Several studies document altered APP levels in goats in response to various disease states. A 2013 published study showed increases in haptoglobin (Hp), serum amyloid A (SAA), and lactoferrin in milk of goats receiving intramammary lipopolysaccharide (LPS) infusion. 18 Another study demonstrated significantly increased blood levels of Hp and SAA in experimentally induced subacute ruminal acidosis (SARA). 12 Additionally, Hp was increased in experimentally induced pregnancy toxemia. 8 These studies suggest that measurement of APPs may be clinically useful in the diagnosis of disease in goats. However, the clinical use of APP values requires the establishment of species-specific reference intervals. The objective of this project was to establish reference intervals for Hp, SAA, lipopolysaccharide binding protein (LBP), and alpha-1 acid glycoprotein (A1AGP) in clinically healthy goats in Missouri.
Haptoglobin is a positive APP and is the principal scavenger of free hemoglobin in blood. 16 Hp has a bacteriostatic effect by binding free Hb, making iron unavailable to bacteria. In an experimental model of inflammation using turpentine injections in goats, Hp was found to be significantly elevated above baseline at 72 hr postinjection. 9
Serum amyloid A proteins are involved in lipid transport and chemotactic recruitment of leukocytes and are implicated in the pathogenesis of chronic inflammation. In the turpentine-induced inflammation model in goats, SAA was significantly increased above baseline at 24 hr postinjection indicating that it may be useful as an early marker of inflammation. 9
Lipopolysaccharide binding protein is a soluble polypeptide that binds to bacterial LPS and enhances its pro-inflammatory activity 100- to 1,000-fold. 5 LBP can also bind lipoteichoic acid and augment TLR-2 signaling. 19
Alpha-1 acid glycoprotein has been reported to exhibit immunomodulatory functions in leukocytes, platelets, and endothelial cells. A1AGP is partially protective in several rodent models of septic shock by maintaining perfusion of vital organs. 15 However, A1AGP can potentially inhibit neutrophil migration to a site of infection, leading to inadequate bacterial clearance and resulting in increased risk of mortality.1,14
Materials and methods
Goat population and sampling
Fifty-five goats from 10 farms in Central Missouri were used for this study. Farm owners were invited to participate in the study via email correspondence; only herds within a 40-min drive were chosen to limit delays in sample processing. Adult goats, between 1 and 6 years of age based on dentition or producer records, were sampled (maximum of 7 animals per herd). Animals were apparently healthy on physical examination and had no current health concerns according to the owners or farm records. The color of the conjunctiva and third eyelid were evaluated using the FAMACHA system, and all goats enrolled had scores of 3 or less. 13 Female goats who had kidded within the previous week were excluded. Each goat was minimally restrained by a person holding its head. Blood was collected via jugular venipuncture using an 18- or 20-gauge needle and placed in commercial anticoagulant tubes containing lithium heparin. Samples were stored in an insulated container with cold packs for transport back to the diagnostic laboratory. The time elapsed between sample collection and arrival to the laboratory was less than 4 hr. On arrival at the laboratory, the samples were centrifuged at 1,500 × g for 10 min, and the plasma was recovered and frozen at −80°C until analysis. All goats were handled in accordance with the University of Missouri Animal Care and Use Committee guidelines.
The 55 goats sampled included 39 females, 11 intact males, and 5 castrated males. A total of 12 breeds were represented (Alpine, Pygmy, Kinder, Boer, Toggenburg, Nubian, Oberhasli, Kiko, Spanish, Fainting goat, LaMancha, and Nigerian Dwarf). The goats ranged from 1 to 6 years of age. The majority of female goats were in the first or second trimester of pregnancy and approximately 20% of the does were lactating. Table 1 shows sex and reproductive status of the goats sampled. One sample was not used for analysis due to hemolysis.
Sex and pregnancy status of goats sampled.

Acute phase protein assays
Haptoglobin, SAA, LBP, and A1AGP—were measured at a reference laboratory a using goat-specific commercial enzyme-linked immunosorbent assay (ELISA) kits. b All samples were run in duplicate with standard curve and recovery spike testing performed simultaneously.
Statistics
Reference Value Advisor, macroinstructions for Microsoft Excel guided by the IFCC-CLSI recommendations, was used for analysis of the data. 7 The data was tested for Gaussian distribution using the Anderson–Darling test. Outliers were identified using the Dixon–Reed test and removed from the calculations. 6 Reference limits (with 90% confidence intervals) were calculated using a nonparametric method. Results were analyzed by 1-way analysis of variance to compare sexes and breeding status.
Results
Figure 1 shows the results for the 4 APPs in clinically healthy animals. Only Hp was found to vary significantly between groups of healthy animals; values differed between pregnant and nonpregnant does. Serum Hp levels tended to differ within groups of does with increased values in lactating animals and decreased values in pregnant does in their second trimester when compared with open, nonlactating does (Fig. 2); a statistically significant difference was found only between lactating does and pregnant does in the second trimester. No sex differences were seen for any of the APPs measured. Table 2 shows the proposed reference intervals for the APPs. The reference interval for Hp is composed of values for the entire population, as too few does were available in each subgroup of pregnant, lactating and open, nonlactating does to construct subgroup-specific reference intervals. However, given the evidence that pregnancy and/or lactation status affects Hp values in does, this reference interval should be used with extreme caution.
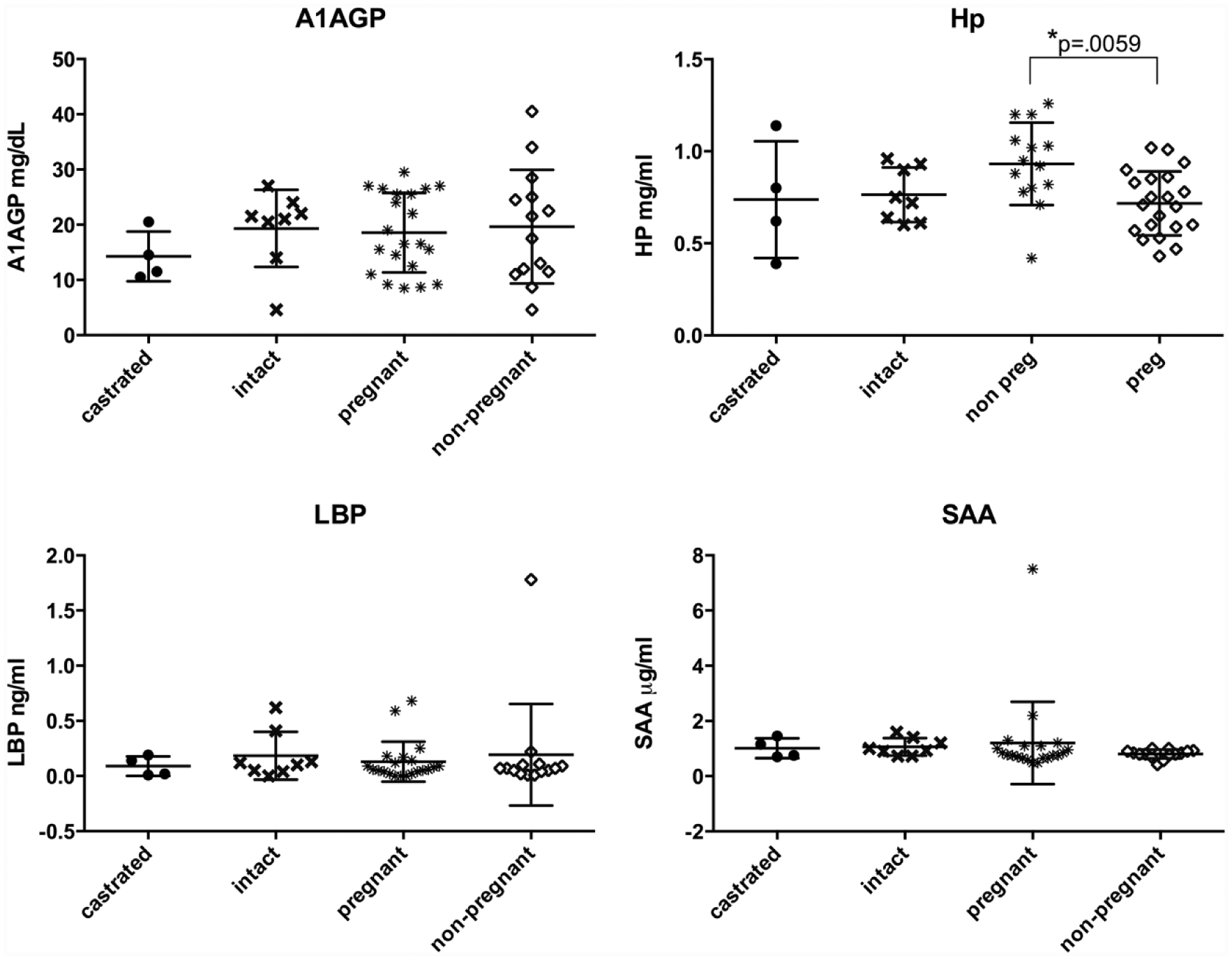
Serum levels of 4 acute phase proteins from healthy goats: lipopolysaccharide binding protein (LBP), haptoglobin (Hp), serum amyloid (SAA), and alpha-1 acid glycoprotein (A1AGP). No differences between sex or pregnancy status were found except for Hp where pregnant animals had significantly lower serum Hp levels than nonpregnant animals (P = 0.0059).
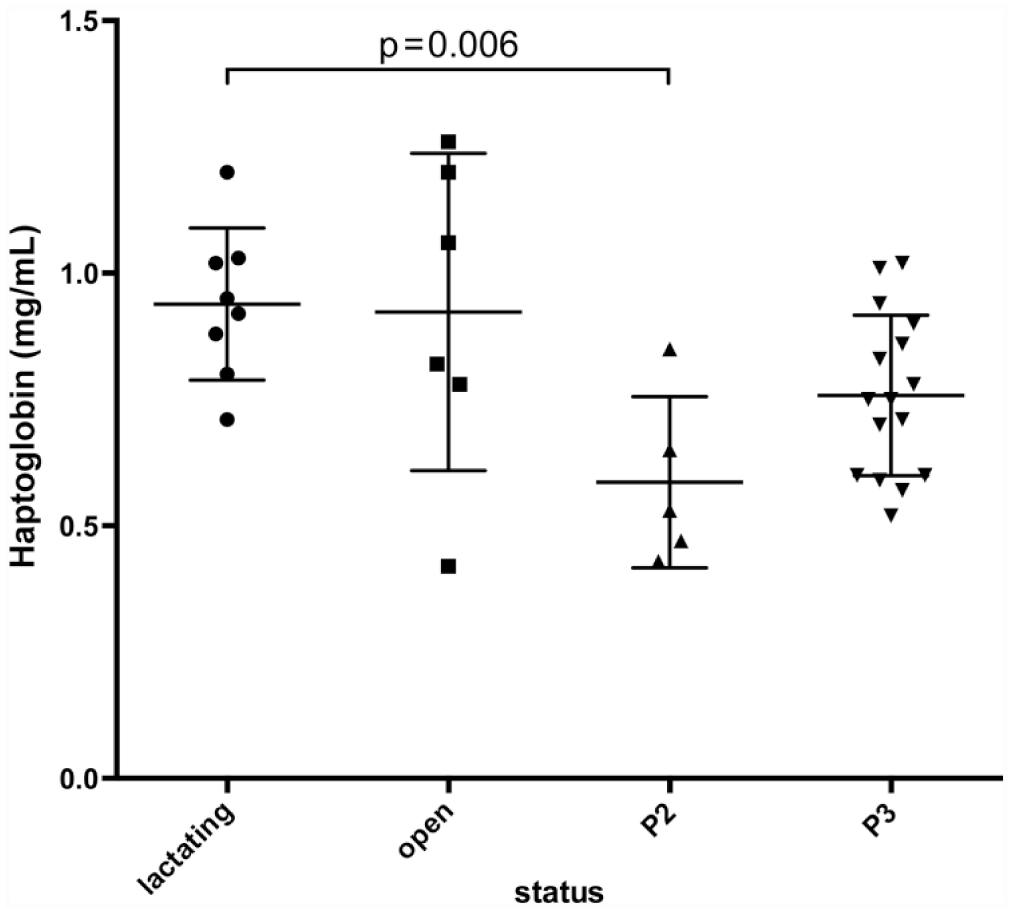
Serum haptoglobin (Hp) concentrations in female goats by pregnancy and lactation status. P2 = second trimester; P3 = third trimester. Goats in their second trimester of pregnancy had lower serum Hp than lactating animals (P = 0.006).
Proposed reference intervals for lipopolysaccharide binding protein (LBP), haptoglobin (Hp), serum amyloid (SAA), and alpha-1 acid glycoprotein (A1AGP) in healthy goats.*
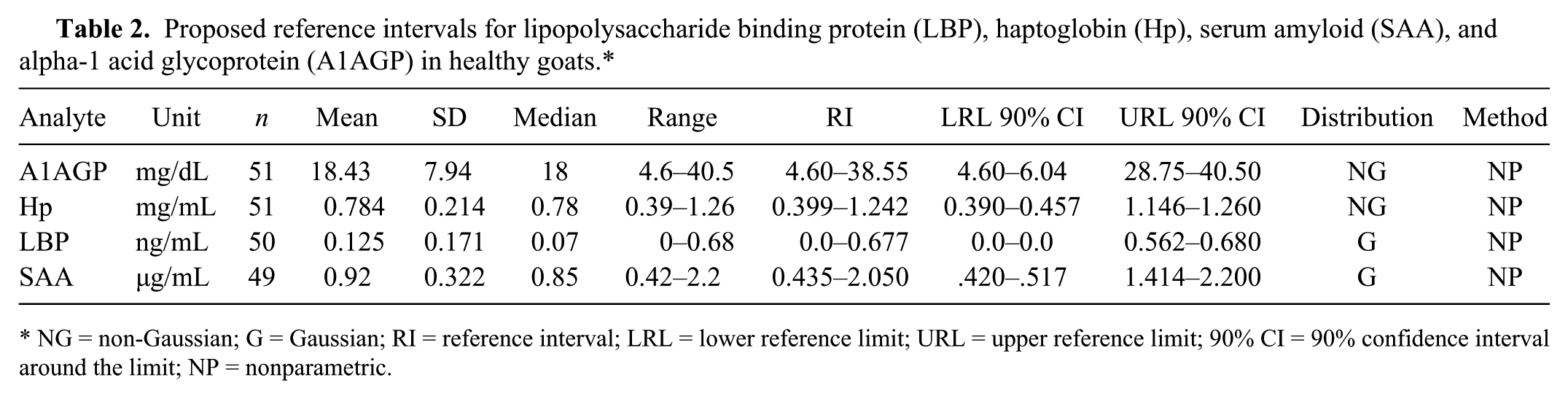
NG = non-Gaussian; G = Gaussian; RI = reference interval; LRL = lower reference limit; URL = upper reference limit; 90% CI = 90% confidence interval around the limit; NP = nonparametric.
Discussion
Few studies on goat APPs are available, and those that exist are based on relatively small sample sizes. In the study of turpentine-induced inflammation in goats, a total of 6 goats were evaluated. 9 The methods to measure A1AGP also differed from those used in the current study, precluding a direct comparison of data. In the turpentine study, SAA was measured using perchloric acid precipitation method and bicinchoninic acid protein assay reagent; in the current study, a goat-specific ELISA was utilized. In the same study, Hp and SAA were measured using ELISA kits from a different source than in the current study. SAA levels in untreated goats were generally higher than those reported herein. This possible discrepancy may be due to several factors including different kit manufacturers and differing animal populations. This highlights the point that direct comparisons of laboratory data should not be made when methods differ between studies and/or laboratories. Additionally, breed may play a significant role and should be taken into consideration, as the goat breeds evaluated in the prior studies (Murcia–Granada, and a “Chinese local breed”) differed from those in the current study.8,9,12
Haptoglobin production could potentially be decreased in pregnant animals as a component of systemic immunosuppression that occurs during pregnancy. 17 However, a study looking at Hp levels in healthy dairy cattle did not find any significant difference between healthy pregnant and nonpregnant animals that were followed for the first 2 trimesters. 3 In contrast, multiple studies have shown that APPs and specifically Hp increase in the peripartum period in cattle.2,20 One study looking at prepartum goats prior to experimental induction of pregnancy toxemia had Hp concentrations above those reported herein; however, researchers did not have nonpregnant goats to compare and the study used a different method assay method. 8 Given the results shown in Figure 2, Hp reference intervals specific to stage of pregnancy would be an extremely useful addition in the future.
Lipopolysaccharide binding protein has been investigated as a diagnostic tool in a number of human disorders including neonatal sepsis, and in rodent models of inflammatory diseases. It is a relatively new APP to be recognized as a potentially useful biomarker of inflammation in goats. The reference interval presented herein establishes a baseline for the future evaluation of LBP in natural and experimental disease of goats.
We report normal reference intervals for 4 serum APPs that may be useful as clinical biomarkers of disease in goats. Hp should be interpreted with caution in animals with unknown pregnancy status. Further studies are needed to determine whether these APPs are useful biomarkers for specific disease states.
Footnotes
Acknowledgements
The authors thank the Animal Diagnostic Lab in the Veterinary Service Center at Stanford University School of Medicine for their assistance with this project, and Dr. Holly Minard for assistance in sample collection.
a.
AniLytics Inc., Gaithersburg, MD.
b.
Cusabio Biotech Co. Ltd., Wuhan, China.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
Support for this research was derived from the Stanford University Department of Comparative Medicine, and the University of Missouri Department of Veterinary Medicine and Surgery.
