Abstract
Systemic inflammatory response syndrome (SIRS) and sepsis can be challenging to diagnose in cats. Retrospectively, we investigated the diagnostic and prognostic potential of serum amyloid A (SAA), a major feline acute-phase protein (APP), in a population of critically ill cats with SIRS related to trauma or sepsis. A total of 56 SIRS cats (trauma n = 27; sepsis n = 29) were included and compared with healthy controls (n = 18). SAA concentration was significantly increased in SIRS cats compared to controls, confirming its potential for the detection of systemic inflammation in this species. Significantly higher values of SAA were detected in cats belonging to the sepsis group; however, according to the results of the receiver operating characteristic curve analysis, the value of using SAA (>81 mg/L) to discriminate septic cats was only moderate (AUC = 0.76). Additionally, cats with sepsis had significantly higher serum bilirubin concentrations and toxic neutrophil changes compared to the trauma group. Overall, 38 of 56 cats were survivors; 18 of 56 were non-survivors, with 83% of the non-survivors (15 of 18) belonging to the sepsis group. Serum bilirubin concentration, but not SAA, was able to predict outcome. Prospective studies are needed to assess the potential of SAA in the diagnosis of feline sepsis and outcome prediction.
The systemic inflammatory response syndrome (SIRS) and sepsis are complex clinical syndromes in critically ill cats, and are associated with substantial disease effect and mortality. 3 Clinical response to sepsis is less predictable and specific in feline patients, potentially delaying early diagnosis and prompt treatment. 17
Serum amyloid A (SAA), a major feline acute-phase protein (APP), is reported to increase early during inflammation and in cases of tissue damage.11,12,18 Although its biological role is still unclear, SAA appears to have immunomodulatory activities and protective properties.16,18 SAA has been shown to be a significant prognostic marker in sick cats with various diseases 20 ; however, its potential to discriminate between a septic and a non-septic origin of SIRS is not well defined. 16
Diagnosis of feline sepsis is still challenging, given that veterinary reports that have investigated the diagnostic performance of selected clinicopathologic variables are limited. 3 A higher percentage of circulating band neutrophils and more severe hypoalbuminemia were associated with the presence of sepsis in a small prospective case-control study of cats with SIRS. 3 The presence of toxic neutrophils was shown to be a common finding in feline diseases of inflammatory and/or infectious origin. 19
We investigated the potential diagnostic utility of serum SAA concentrations and selected clinicopathologic variables measured at the time of hospital admission for discriminating between SIRS of infectious (sepsis) and non-infectious (trauma) origin in a population of critically ill cats. In addition, SAA data were also assessed for outcome prediction.
Critically ill cats with a clinical condition of SIRS related to trauma or sepsis, hospitalized at the ICU of the University of Bologna Veterinary Teaching Hospital between March 2012 and March 2014, were included retrospectively. The presence of SIRS was defined according to published criteria. 2 Additional inclusion criteria were hospitalization in ICU, complete medical records, and SAA evaluated upon admission, or presence of at least an aliquot of serum collected upon ICU admission and stored frozen at −80°C. The overall population of SIRS cats was divided into 2 subgroups according to the infectious or non-infectious SIRS origin (sepsis vs. trauma). Specifically, the trauma group included cats with blunt trauma associated with motor vehicle accident or high-rise syndrome; the sepsis group included cats with cytologic or bacteriologic evidence of bacterial infection. SIRS cats were also classified as survivors (alive to discharge) or non-survivors (died despite medical treatment or euthanized because of moribund conditions or end-stage disease). Cats euthanized for financial reasons or discharged against medical advice were excluded from the study. Controls were client-owned blood donor cats (n = 18) considered healthy according to history, physical examination, and clinicopathologic data.
Hematologic and biochemistry profiles performed upon hospital admission were reviewed in all of the cats enrolled. When SAA values were not available, frozen serum samples were used for analysis. Blood count was determined by an automated hematology system (ADVIA 2120 hematology system, Siemens Healthcare Diagnostics, Tarrytown, NY). May-Grünwald/Giemsa–stained (Merck, Darmstadt, Germany) blood smears were examined, and neutrophil morphology and toxic changes were recorded. The occurrence of Döhle bodies, cytoplasmic basophilia, vacuolation or foaminess, and toxic granulation of neutrophils were recorded. 19 SAA concentrations were measured on serum samples using a commercial immunoturbidimetric assay designed for human SAA (LZ Test Eiken SAA, Eiken Chemical, Tokyo, Japan), as validated previously for cats in our laboratory. 7 All biochemical analyses were performed with an automated chemistry analyzer (AU 400, Olympus/Beckman Coulter, Munich, Germany). All of the investigated variables were also measured in healthy controls.
Nonparametric statistics with post hoc analysis were used to compare variables between the different groups. Data were expressed by standard descriptive statistics and presented as median and range. Categorical variables were compared using Fisher exact test. Univariate logistic regression was used to assess the association between clinical and clinicopathologic variables at the time of hospital admission and the diagnosis of sepsis. Variables associated with a diagnosis of sepsis in the univariate analyses were entered into a multivariable model (stepwise selection). Binary logistic results were presented as odds ratio (OR) and 95% confidence interval (CI). Overall model fit was assessed by the percentage of outcome correctly classified by the receiver operating characteristic (ROC) curve analysis and by a significant Hosmer–Lemeshow test (p > 0.05). ROC curve analysis was used to find optimal cutoff values for variables predicting sepsis and to calculate the area under the ROC curve (AUC). Correlation between variables was assessed using the Spearman rank correlation coefficient. A p value <0.05 was considered significant. All analyses were performed using MedCalc statistical software (v.15.6.1, MedCalc Software, Ostend, Belgium).
A total of 56 cats with SIRS satisfied the inclusion criteria and were included in the study. The median age was 4.8 y (0.5–21), and the sex distribution was as follows: 18 of 56 male neutered, 15 of 56 spayed females, 13 of 56 intact males, and 10 of 56 intact females. Overall, 27 of 56 cats were included in the trauma group; 29 of 56 cats were included in the sepsis group. The origin of sepsis was related to pyothorax (10 of 29), septic peritonitis (7 of 29), pyelonephritis (4 of 29), septic arthritis (1 of 29), systemic toxoplasmosis (1 of 29), septic cholangitis (2 of 29), and suppurative cellulitis or abscesses (4 of 29). Data regarding lifestyle were available for 29 of 56 patients; among them, only 3 of 29 lived indoors exclusively; the others were outdoor or indoor and outdoor cats. Frequency of an outdoor lifestyle was not significantly different between the trauma (15 of 27) and the sepsis group (11 of 29). Overall, 38 of 56 (68%) cats were survivors; 18 of 56 (32%) were non-survivors; diagnosis of sepsis was significantly associated with a higher mortality rate (15 of 18, 83%).
Cats in the sepsis group had significantly increased serum SAA and total bilirubin concentration, and a significantly higher frequency of toxic neutrophil changes in blood smears compared to both the trauma group and controls (Table 1). SAA, total bilirubin concentrations, and the presence of toxic neutrophil changes were significantly associated with a diagnosis of sepsis (Table 2). When the multivariate logistic regression was performed, the only variables retained in the model were SAA concentration (OR = 1.01, CI = 1.0–1.02) and neutrophil toxic changes (OR = 10.4, CI = 2.2–49.8). However, according to the ROC curve analysis, no significant difference to correctly predict a diagnosis of sepsis was found among serum SAA, serum bilirubin, and neutrophil toxic changes: serum SAA >81 mg/L had a 79.3% sensitivity and a 77.7% specificity (AUC = 0.76); serum bilirubin >3.8 μmol/L had a 72.4% sensitivity and a 74% specificity (AUC = 0.79). The value of AUC for the neutrophil toxic changes was 0.74. No significant correlation was noticed between SAA concentration and the other investigated variables. No difference between survivors and non-survivors was detected for serum SAA concentration and neutrophil toxic changes upon admission. Conversely, serum bilirubin >3.6 μmol/L had a 94.4% sensitivity and a 68.4% specificity (AUC = 0.85) to predict outcome in cats included with a diagnosis of sepsis.
Clinicopathologic results of controls and cats with systemic inflammatory response syndrome (sepsis and trauma groups) analyzed with the Kruskal–Wallis analysis of variance test for comparisons. Only variables with p < 0.05 are presented.
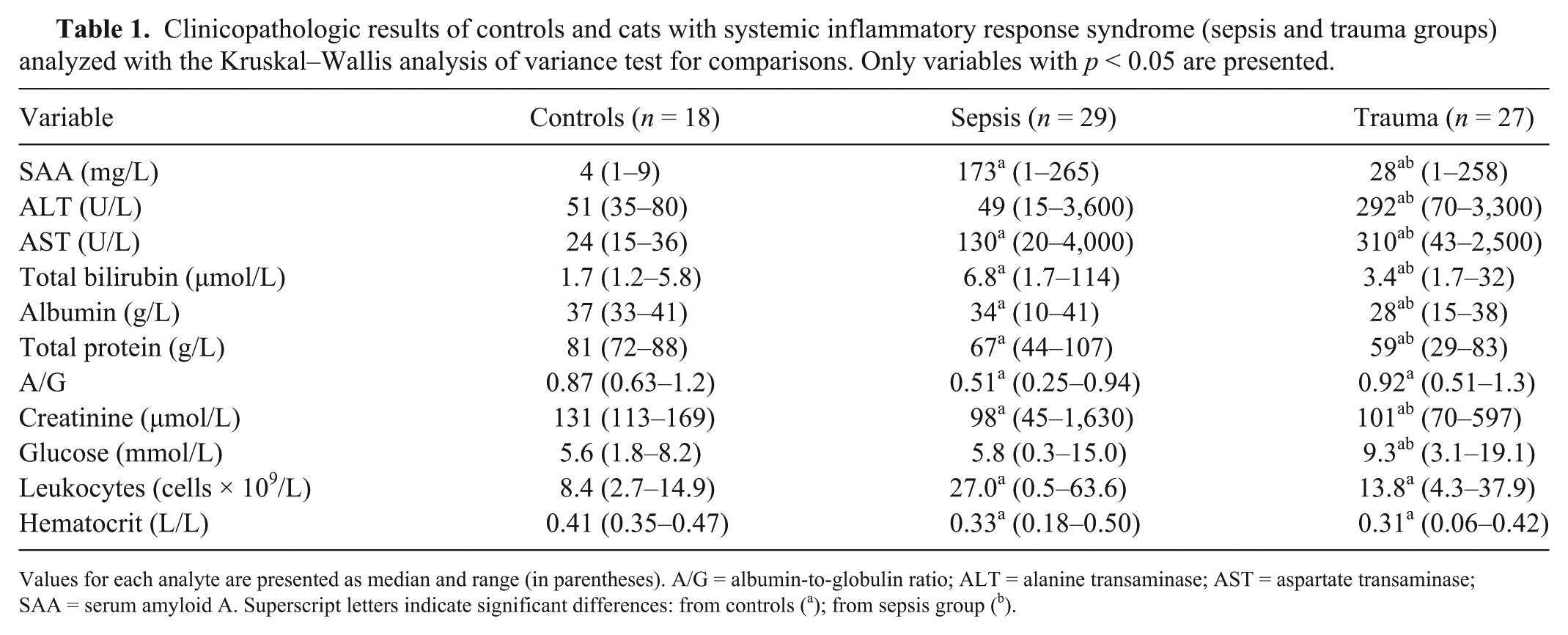
Values for each analyte are presented as median and range (in parentheses). A/G = albumin-to-globulin ratio; ALT = alanine transaminase; AST = aspartate transaminase; SAA = serum amyloid A. Superscript letters indicate significant differences: from controls (a); from sepsis group (b).
Univariate and multivariate binary logistic regression of clinicopathologic variables associated with sepsis diagnosis at the time of hospital admission in cats with systemic inflammatory response syndrome. Only variables with p < 0.05 are presented.
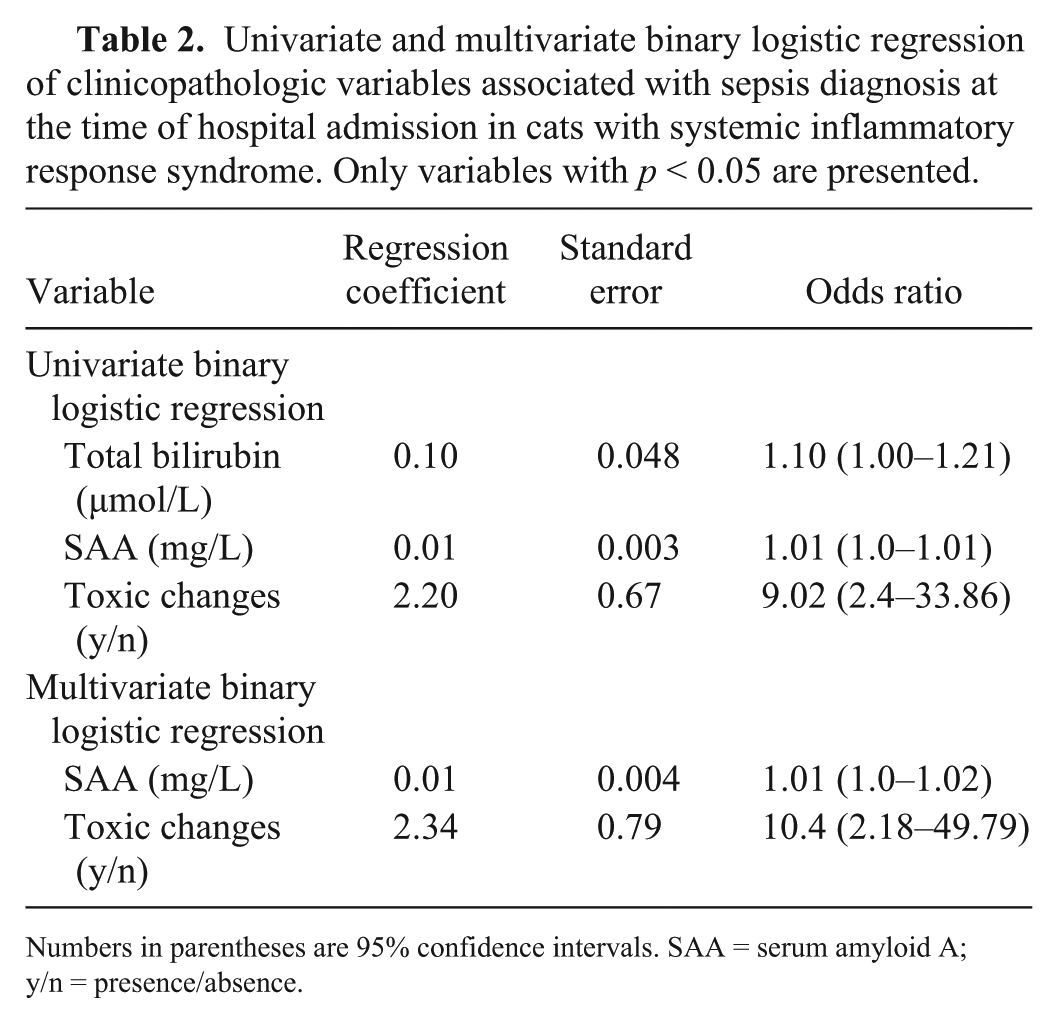
Numbers in parentheses are 95% confidence intervals. SAA = serum amyloid A; y/n = presence/absence.
Despite the limited value of the SIRS criteria in humans and dogs,8,10 diagnosis of sepsis still relies on the evaluation of these criteria in clinical settings. Similar criteria in cats derive from a retrospective autopsy-based study, and their specificity and sensitivity for the detection of feline sepsis have not been reported and evaluated extensively. Diagnosis of feline sepsis is additionally challenging because of the peculiar clinical manifestations of SIRS and the poor diagnostic value of leukocyte counts in cats.2,16
In our study, significantly higher SAA concentrations were documented in cats with SIRS of infectious and non-infectious origin compared to healthy controls. This result confirms the role of this APP for the detection of systemic inflammation in cats, and suggests its potential value as a biomarker of feline sepsis. The lack of statistical correlation between SAA concentration and leukocyte count in our population raises a concern regarding the traditional methods used to detect systemic inflammation in cats. Furthermore, significantly higher concentrations of SAA were documented in cats with sepsis compared to cats with trauma. Although higher values of SAA were previously reported in cats with inflammatory and/or infectious diseases compared to other sick cats, 18 we evaluated the potential of SAA in discriminating between septic and non-septic SIRS. However, according to the results of the ROC curve analysis, the performance of a SAA arbitrary cutoff (>81 mg/L) to correctly predict a diagnosis of sepsis in this cohort of animals was only moderate.
Cats included in the sepsis group had significantly increased serum bilirubin concentrations compared to those in the trauma group. Furthermore, this variable was able to predict outcome in the former group, although the accuracy was only moderate. Hyperbilirubinemia is a common finding in critically ill septic humans, being potentially related to sepsis-induced cholestasis or hepatic damage or dysfunction. 9 Increased bilirubin concentration and icterus have been reported previously as common abnormalities in cats with sepsis, 2 potentially indicating the presence of cholestasis in cats with this condition. Further studies evaluating the incidence of cholestasis and its prognostic significance in feline sepsis are warranted.
Leukocytosis with left shift has been reported to be more common in septic cats compared to cats with non-infectious SIRS, 3 and the presence of toxic neutrophils has been associated with various infectious feline diseases. 19 According to our findings, toxic neutrophil changes associated with elevated SAA values may aid clinicians in the diagnosis of feline sepsis.
No association between SAA concentration measured at the time of hospital admission and final outcome was identified in our population of SIRS cats. In humans, elevated SAA concentrations predicted outcome in people with neoplastic or immune-mediated diseases,5,15 but failed to reach this purpose in other conditions. 1 Similar controversies with respect to prognosis have been reported for SAA in horses,4,21 and for other APPs in veterinary medicine,6,14 suggesting that serial monitoring or combined APP profiles rather than single APP values may better predict outcomes. In a retrospective study including 175 cats with various diseases (neoplastic, inflammatory, and other diseases), cats with a SAA concentration above the reported reference interval at the time of the first evaluation had a significantly shorter median survival time compared to cats with non-elevated SAA concentration, regardless of the final diagnosis. 20 However, diagnostic criteria of these diseases were not uniform in the latter study and different treatments before and after diagnosis among the cats might have affected the prognosis. 20 Furthermore, mortality data of the cats with neoplasia were more abundant compared with the cats with other diseases. This might have led to a biased result.
The overall mortality rate for cats with SIRS was 32% in our study, with 83% of the non-survivors belonging to the sepsis group; the mortality rate in the latter group was 52%. These data are consistent with other reports of mortality of septic cats.3,13
Several limitations exist in our study. We included in the sepsis group only SIRS cats with bacteriologic or microbiologic confirmation of sepsis, being confident of their final diagnosis. However, we cannot exclude that some of the cats included in the trauma group may have indeed developed septic complications upon admission or during hospitalization. We included a relatively small number of patients, and this may have limited the statistical evaluation or the prognostic power of the investigated variables. Additionally, because the study was retrospective, we were not able to evaluate a clinical scoring system to stratify patients according to the severity of the clinical condition, preventing further statistical comparisons. Finally, blood sampling was performed at the time of hospital admission only; monitoring over time could have improved the prognostic value of the investigated variables.
Our results support the value of SAA as a potential diagnostic marker of feline SIRS and sepsis. The power of serum SAA in predicting the occurrence of sepsis was only moderate in this population; however, association with the measurement of serum bilirubin concentration and evaluation of the blood smear for the presence of neutrophil toxic changes may further aid the diagnosis of sepsis in cats. The prognostic potential of serum bilirubin in feline sepsis seems promising. Finally, further prospective large-cohort studies are needed to assess the prognostic significance of SAA, serum bilirubin, and neutrophil toxic changes in cats with SIRS, and to assess their value for the early detection of feline sepsis and the prompt recognition of critically ill patients.
Footnotes
Acknowledgements
Preliminary results were presented at the 15th EVECCS Congress, 2015, Lyon, France.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
