Abstract
Acute phase proteins (APPs) have been studied in many companion and large animals and have been reported to have a differential sensitivity to traditional markers of inflammation. Studies have been performed indicating the conservation of these proteins as well as the application and cross-reactivity of automated assays among different species, but few reports have detailed APPs in marine mammal species. In the present study, automated assays were utilized to generate reference intervals for C-reactive protein, haptoglobin, and serum amyloid A using 44 serum samples from healthy Atlantic bottlenose dolphins (Tursiops truncatus). A total of 25 samples were obtained from dolphins under human care and 19 samples were obtained from free-ranging dolphins. Mild yet statistically significant differences were observed in levels of haptoglobin and serum amyloid A between these groups. The reference intervals from the combined groups were as follows: C-reactive protein 3.1–19.7 mg/l, haptoglobin 0–0.37mg/ml, and serum amyloid A 17.5–42.9 mg/l. These baseline data should provide an important foundation for future studies of the application of APP quantitation in monitoring the health and stressors of dolphins under human care and with live capture of free-ranging dolphins.
Keywords
Atlantic bottlenose dolphins (Tursiops truncatus) are common animals under human care at zoological institutions and aquaria. Live capture of free-ranging dolphins has also been the subject of population health assessments with the goals of monitoring and understanding the impact of natural and anthropogenic stressors.4,5,36 It has been reported that clinical signs of ill health are often masked in dolphins; thus, diagnostic testing inclusive of traditional hematologic and biochemistry testing has been important for clinical investigations and animal care.6,27
Acute phase proteins (APPs) have been well documented in companion animals and large animals.7-9,12,24,28,29 As markers of the acute phase response, APPs have been shown to be sensitive indicators of infection, inflammation, neoplasia, trauma, and stress. These responders are part of the innate immune system with the goal of promoting healing and reestablishing homeostasis. While the acute phase response is thought to promote changes in over 200 proteins, several APPs have been identified as those with more pronounced changes. 23 Major APPs are the first responders within 24–48 hr of stimulus and have greater than 10-fold increases in magnitude. Such APPs as well as those proteins with moderate and minor classifications have been shown to provide a differential sensitivity to the inflammatory pathway versus traditional markers such as total white blood cell count, band neutrophils, and fibrinogen. 8 These differences are related not only to the magnitude of change but also to the early time line of expression. While these nonspecific markers cannot aid in providing a specific diagnosis, they are excellent markers of prognosis and have also been shown to have utility in detecting subclinical disease, stress, and chronic inflammatory processes.8,11,12
APPs have been studied in some marine mammal species. C-reactive protein (CRP) has been described in harbor seals (Phoca vitulina) with up to 50-fold increases in animals with severe inflammatory based disease. 13 Haptoglobin (HP) has also been identified in harbor seals; elevated levels were associated with an epidemic of Phocine distemper virus.22,32 Serum amyloid A (SAA) was demonstrated to be a sensitive diagnostic tool for inflammatory disease in manatees (Trichechus manatus latirostris).15,16 Haptoglobin also had a high diagnostic specificity, but no reactivity was observed for CRP. 16 In total, the studies support a role for APP quantitation as a monitor of marine mammal health. The present study was conducted to determine reference intervals for APP in healthy bottlenose dolphins.
A total of 44 serum samples was obtained from 2 sources. Samples from animals under human care were obtained from the Georgia Aquarium facilities (n = 25; Atlanta, Georgia and St. Augustine, Florida) and were from individual animals with normal clinical history and physical examination. Routine diagnostic testing, including hematology and serum biochemistry, also showed no abnormalities. Within this group, there were 11 males (age: 0.5–44 years) and 14 females (age: 2–57 years). Samples from free-ranging animals were obtained from the Harbor Branch Oceanographic Institute at Florida Atlantic University (n = 19; Fort Pierce, FL) and collected as part of a health assessment study of dolphins in the Indian River Lagoon. Free-ranging animals had a normal physical examination, and all routine blood work was within normal limits. 14 Within this group, there were 15 males (estimated age: 5–18 years) and 4 females (estimated age: 5–16 years). Serum samples were aliquoted and frozen at −80°C until transport to the University of Miami Acute Phase Protein Laboratory (Miami, FL) for analysis.
All assays were implemented on a clinical chemistry analyzer. a C-reactive protein was quantitated using purified canine CRP b to obtain a standard curve and a CRP reagent a as previously described.25,26 Using dolphin sera, the assay coefficient of variation (CV) was 8.4%. Haptoglobin was quantitated using a commercial kit inclusive of reagents and calibrators. c The CV was 8.3%. SAA was quantitated using a commercial kit d inclusive of reagents and calibrators as previously described. 21 The CV was 6.6%. Statistical analyses were conducted using commercial software.e,f Data were examined for outliers using the Kolmogorov–Smirnov test and found to have normal distribution. Per current recommendations on reference interval generation from the American Society for Veterinary Clinical Pathology, data are presented as mean, standard deviation, median, and minimum–maximum values (http://www.asvcp.org/pubs/qas/index.cfm). Significance was considered at P ≤ 0.05 after use of the t-test. Results are reported as mean, median, min–max, and standard deviation, and the 2-sided P-value of the test. Reference intervals were generated using the robust method and reported with 2-sided 90% confidence intervals.
No significant differences were observed in CRP levels in samples from dolphins under human care and those that were free-ranging (Table 1). A mild decrease in the mean value of SAA was observed in samples from free-ranging dolphins, and this difference was found to be significant although there was a clear overlap in minimum–maximum values for the 2 groups. Free-ranging dolphins were observed to have slightly higher levels of HP, and this difference was also found to be significant. As the combined data set met the minimum recommendation of n = 40, the robust method was used to generate reference intervals for the 3 APPs (data presented in Table 2).
Mean, median, standard deviation, and minimum–maximum for acute phase proteins from serum samples from bottlenose dolphins (Tursiops truncatus) that are under human care versus free-ranging.*
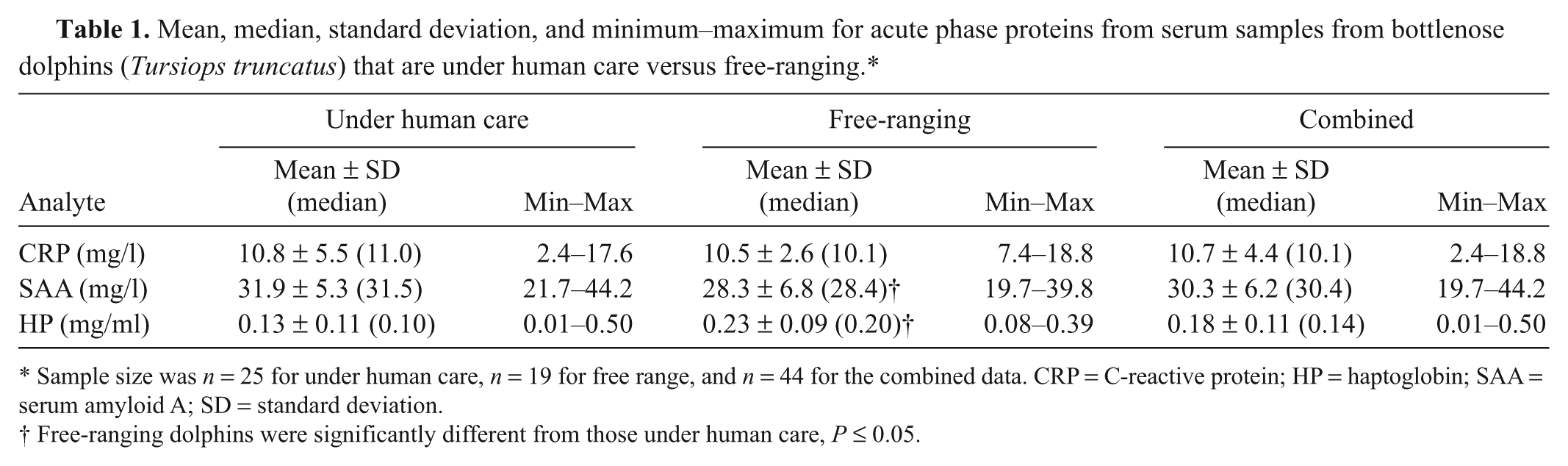
Sample size was n = 25 for under human care, n = 19 for free range, and n = 44 for the combined data. CRP = C-reactive protein; HP = haptoglobin; SAA = serum amyloid A; SD = standard deviation.
Free-ranging dolphins were significantly different from those under human care, P ≤ 0.05.
Reference intervals (RI) using the combined data and the robust method of RI generation.*
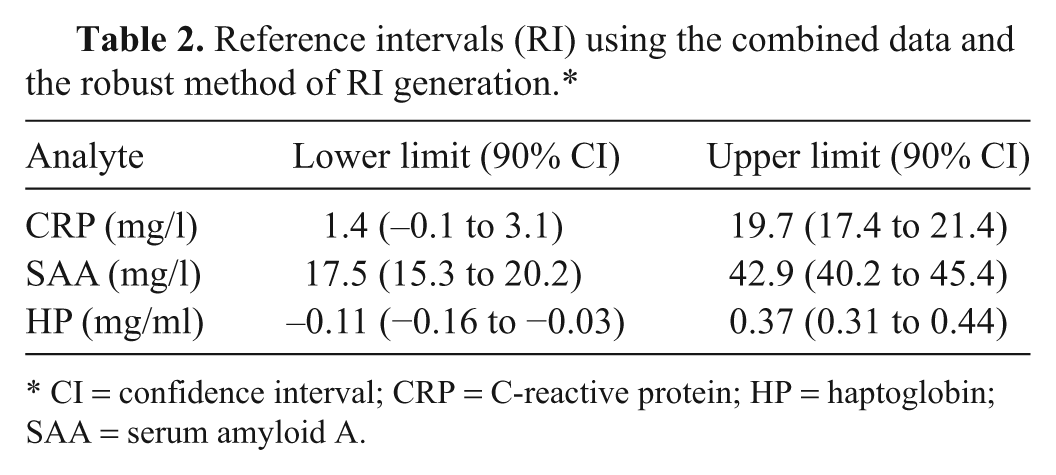
CI = confidence interval; CRP = C-reactive protein; HP = haptoglobin; SAA = serum amyloid A.
C-reactive protein does not appear to be expressed in all species. In human beings, it is a major APP and has been used as a risk marker or biomarker of cardiovascular disease. 8 In dogs, where CRP is also a major APP, an upper limit of 20 mg/l has been reported. 25 These levels are also similar to those reported with a species-specific assay for harbor seal CRP 13 and appear to be consistent with initial findings in dolphin sera in the current study.
SAA is believed to be one of the most conserved APP across mammalian and nonmammalian species and most often is characterized to have a role of a major APP.8,35 A reference interval of 0.5–20 mg/l has been reported for horses.1,20 This contrasts with the lower limit of 17.5 mg/l found in the current study. In most mammals, near zero levels are often observed in clinically healthy animals.8,24 The current findings may suggest the presence of a mild constitutive expression of SAA in dolphins similar to fibrinogen, which also has a higher normal resting expression. The mild yet statistically significant higher value for SAA found in dolphins under human care is interesting but the clinical significance is not known. Further studies using a larger sample set as well as dolphins from different facilities is warranted. Of note, no statistical difference in SAA was observed in free-ranging versus captive manatees. 17
In most species, HP has been described as a minor APP, and expression is very low in clinically healthy animals.8,10 The current findings using the dolphin sera are consistent with reports of intervals of less than 1 or 2 mg/ml in other mammals. 8 Previous reports on samples from free-ranging harbor seals, using the same assay as the current study, indicated a reference interval of 0.11–1.24 mg/ml. 22 The lower upper limit found with dolphin samples may reflect an absence of significant basal inflammatory processes. While mild, there was a statistically significant 1.8-fold increase observed in HP levels in free-ranging dolphins versus those under human care. In some species, HP has been potentiated as a superior marker of chronic inflammatory disease.8,18 Thus, the increased HP in free-ranging dolphins may reflect the effects of underlying stressors and infectious disease. Possible etiologies including subclinical morbillivirus infection and gastritis have been noted in this particular population of free-ranging dolphins.4,5,14 Importantly, the increased HP was observed in the absence of abnormal blood hematology and biochemistry findings indicating, as have other APP investigations, that these biomarkers may be more sensitive for the detection of subclinical inflammation. 8 Of note, increased HP was observed in harbor seals subject to a distemper epidemic. 22 In addition, a 3-fold increase in HP over a 4-week period was observed in free-ranging Steller sea lions (Eumetopias jubatus) that were temporarily held captive. 34 Last, a 1.6-fold mean increase in HP was observed in free-ranging versus captive manatees. 17
The conservation of APP among different species forms the basis for the potential cross-species use of automated assays. Reagents used in the current study for SAA and CRP were developed for use in human beings but have been demonstrated to have high reactivity for many species including horses and dogs.21,24,25 These assays and others were also described in a 2009 study to have reactivity with several species of mammals including impala (Aepyceros melampus), Asian elephant (Elephas maximus), musk ox (Ovibos moschatus), and chimpanzee (Pan troglodytes). 3 Another assay has previously been described to have reactivity for manatee SAA. 15 In unpublished data, the current authors have subsequently found that this assay is not equivalent to the assay described in the present study when manatee samples were analyzed (Cray, personal observation, 2011). These data and other reports indicate that both reagent and species-specific reference intervals should be utilized when employing these types of biomarker assays. The assay for HP has been widely used in species ranging from the harbor seal to cattle, horses, and capybara (Hydrochoerus hydrochaeris).2,8,19,22,30,31,33 In contrast to the SAA and CRP assays, the HP method is colorimetric rather than antibody based, and species cross-reactivity is not an issue. 8
The functions of APP in opsonization, chemotaxis, tissue repair, and cytokine induction are a few examples of their keystone role in the innate immune response. There is a wealth of literature demonstrating changing levels of APP with infection, inflammation, neoplasia, trauma, and stress in both experimental and natural models in many different species. 8 In many reports, the changes in APP levels have been exploited to provide diagnostic and prognostic information. The current study provides valuable baseline data for clinically healthy dolphins. Additional work is needed to define possible age- and sex-related differences and to develop full reference intervals for APP present in dolphins under human care and free-ranging dolphins. Future studies will elucidate the use of these biomarkers in health screens, prognostication, and population studies and further define the roles of these specific APPs in acute and chronic inflammatory processes. As additional tools are developed and new proteins are identified in the growing field of proteomics, the use of biomarker testing in marine mammal species should be significantly enhanced.
Footnotes
Acknowledgements
The free-ranging bottlenose dolphin Health and Environmental Risk Assessment (HERA) research was conducted under National Marine Fisheries Service Permit No. 998-1678-02 and issued Florida Atlantic University IACUC Protocol No. A10-13. The authors thank the entire HERA project staff and Georgia Aquarium and Dolphin Quest veterinary and husbandry staff for their efforts in this project. The authors thank Marineland Dolphin Adventure staff members Jessica Aditays, Melissa Berdine, Donna Peaslee, Cat Rust, Dani Salvatore, Stacey Spong, Jamie Stanley-Bahnsen, Kevin Stephens, and Lindsay Tafs, and Georgia Aquarium staff members Elizabeth Beasley, Lloyd Dodge III, Rachel Friedman, Jeff Fogle, Ann Hoedt, Ashley Gerhart, Dru Jojola, Kristen Knowles, Kevin Krueger, and Lisa Mignogna. The authors also thank Wayne McFee for age analysis on the free-range dolphins. Disclaimer: This publication does not constitute an endorsement of any commercial product or intend to be an opinion beyond scientific or other results obtained by the National Oceanic and Atmospheric Administration (NOAA). No reference shall be made to NOAA, or this publication furnished by NOAA, to any advertising or sales promotion which would indicate or imply that NOAA recommends or endorses any proprietary product mentioned herein, or which has as its purpose an interest to cause the advertised product to be used or purchased because of this publication.
a.
RX Daytona analyzer, Randox Laboratories-US Ltd., Kearneysville, WV.
b.
Life Diagnostics Inc., West Chester, PA.
c.
Phase Haptoglobin kit, Tridelta Tri-DD, Boonton, NJ.
d.
Eiken SAA kit, Mast Group Ltd., Merseyside, United Kingdom.
e.
SAS version 9.3, SAS Institute Inc., Cary, NC.
f.
MedCalc version 10.4.5.0, MedCalc Software, Mariakerke, Belgium.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided, in part, by the State of Florida Protect Wild Dolphins specialty license plate program and the Georgia Aquarium.
