Abstract
Acute phase protein (APP) immunoassays and serum protein electrophoresis (SPEP) are assays for evaluating the inflammatory response and have use as diagnostic tools in a variety of species. Acute phase proteins are markers of inflammation that are highly conserved across different species while SPEP separates and quantifies serum protein fractions based on their physical properties. In the current study, serum samples from 35 clinically healthy Asian elephants (Elephas maximus) were analyzed using automated assays for C-reactive protein, serum amyloid A, and haptoglobin and SPEP. Robust methods were used to generate reference intervals for the APPs: C-reactive protein (1.3–12.8 mg/l), serum amyloid A (0–47.5 mg/l), and haptoglobin (0–1.10 mg/ml). In addition, SPEP was performed on these samples to establish reference intervals for each protein fraction. A combination of APPs and SPEP measurements are valuable adjunctive diagnostic tools in elephant health care.
Keywords
Acute phase proteins (APPs) are components of the innate immune system that have demonstrated clinical value as indicators of infection, inflammation, neoplasia, and tissue injury in human beings and many domestic species.1,4,5,9,11,12,17,18,21,23 More recently, there have been descriptions of usage of APP in a wide range of nondomestic species.2,6–8,14,16,26 During the acute phase response, APPs are produced by the liver in response to cytokines released from leukocytes. The proteins are initially released into serum 24–96 hr following an acute inflammatory stimulus, where they function to promote healing, reestablish homeostasis, and inhibit microbial growth; the time course of expression varies with the specific APP and the resolution of the initial insult. 9 Although the full spectrum of APP response involves increases or decreases in the serum concentrations of more than 200 proteins, the major APPs are those proteins with greater than 10-fold increases in magnitude. 10 In human beings and several other mammals, the clinically useful APPs include C-reactive protein (CRP), serum amyloid A (SAA), and haptoglobin (HP).
Similarly, total protein (TP) and serum protein electrophoresis (SPEP) offer other analytes for evaluating changes in the proteins associated with the inflammatory response. Specifically, in human beings and several nondomesticated mammalian species, SPEP is used to quantify albumin concentrations and globulin fractions giving a broader assessment of the underlying inflammatory processes. 10 In human beings and some animal species, SPEP has been particularly useful in the diagnosis of neoplastic diseases characterized by abnormal serum protein production such as multiple myeloma and lymphoma. 10 Serum protein electrophoresis can be used in conjunction with traditional markers of inflammation used in assessing mammals, including total white blood cell count and fibrinogen levels.
Concurrent measurement of major APPs and SPEP provides early markers of inflammation and possible prognostic indicators. Although APPs and SPEP have been minimally evaluated in elephants, they have already shown some clinical value in monitoring elephant health. 26 Published elephant studies include SPEP performed on a group of deceased wild African savannah, or bush, elephants (Loxodonta africana) following a cull, on a small survey of living, healthy Asian elephants (Elephas maximus), and in a sick Asian elephant calf.3,20,22 A case series also describes APP measurement in 2 Asian elephants. 2 In Florida manatees (Trichechus manatus latirostris), another member of the superorder Afrotheria, SAA was demonstrated to be a sensitive diagnostic tool for inflammatory disease.8,14 Acute phase proteins are impressively conserved among different species, and reagents made for human APPs have been demonstrated to be cross-reactive in many animal species.1,2,4–9,14,15,17–19,26 Validation of these reagents has been based on standard methods in clinical pathology (coefficient of variation, linearity under dilution, and recovery analyses) and examination of samples from clinically abnormal animals with known inflammatory processes.1,2,4,5,7,8,14,18,26 In captive nondomestic species, concurrent measurement of APPs and SPEP fractions are noninvasive assays that can supplement and extend the baseline information obtained from the complete blood cell count, fibrinogen, and standard serum chemistry panels.
The objective of the current study was to provide preliminary reference intervals for both APP and SPEP fractions from healthy, captive, Asian elephants housed in North America. A secondary objective was to compare APPs and SPEP protein measurements and determine their potential clinical value from an opportunistic sampling of Asian elephants, categorized either as having observable clinical problems or as clinically healthy.
Serum samples were obtained from 35 healthy Asian elephants (group A) ranging in age from 3 to 66 years. Within group A, there were 6 intact males, 1 castrated male, and 28 females. The clinical health of the animals was verified by a review of the medical history and visual physical examination. Elephants were kept in an indoor–outdoor enclosure during daylight hours at a private facility in central Florida. The animals were fed ad libitum grass and orchard hay, various browse species, supplemental bread, fresh fruit, and elephant supplement
Using standard phlebotomy techniques, approximately 6–9 ml of venous blood was collected from an ear vein of each elephant into a plain glass 10-ml red-top Vacutainer b tube. After visible clotting occurred and within 1 hr of collection, the blood samples were centrifuged (10 min; 2,479 × g). The serum was drawn off with a disposable pipette, aliquoted into cryovials, and frozen at −80°C until transport to the University of Miami Acute Phase Protein Laboratory (Miami, Florida) for analysis. Serum was thawed on the day of testing.
All APP assays were performed on a clinical chemistry analyzer. c C-reactive protein was quantitated using purified canine CRP d to obtain a standard curve and a high linearity CRP reagent c as previously described.18,19 Using elephant sera, the assay coefficient of variation (CV) for CRP ranged from 5.5% to 13.5%. Serum amyloid A was quantitated using a commercial kit e inclusive of reagents and calibrators as previously described.1,15,26 Using elephant sera, the CV for SAA was 5.0–10.9%. Haptoglobin was quantitated using a commercial kit f inclusive of reagents and calibrators. 26 Using elephant sera, the CV for HP was 4.1–9.1%. Total protein was determined by refractometry. Serum protein electrophoresis was conducted using split beta gels on an automated system g using the manufacturer’s recommended methodology. Reproducible protein fractions observed on the electrophoretogram consisted of albumin, alpha-1 globulins (alpha 1), alpha-2 globulins (alpha 2), beta globulins (beta), and gamma globulins (gamma). Absolute individual SPEP fractions were determined by calculation using electrophoretogram-generated percentages.
Statistical analyses for the reference interval estimations were conducted using SAS version 9.3 h and MedCalc 10.4.5.0 i statistical software packages. The data sets for each analyte were found to have normal distributions with the Kolmogorov–Smirnov test. Using recommendations for reference interval generation from the American Society for Veterinary Clinical Pathology, data are presented as mean, standard deviation, median, and minimum–maximum values. Reference intervals were generated using the robust method and reported with 2-sided 90% confidence intervals. A summary of the results from the clinically healthy elephants in group A is presented in Table 1, which provides the estimated reference intervals for CRP, SAA, HP, TP, and all the SPEP protein fractions.
Reference intervals for acute phase proteins and serum protein electrophoresis fractions from 35 clinically healthy Asian elephants (Elephas maximus) in group A.*
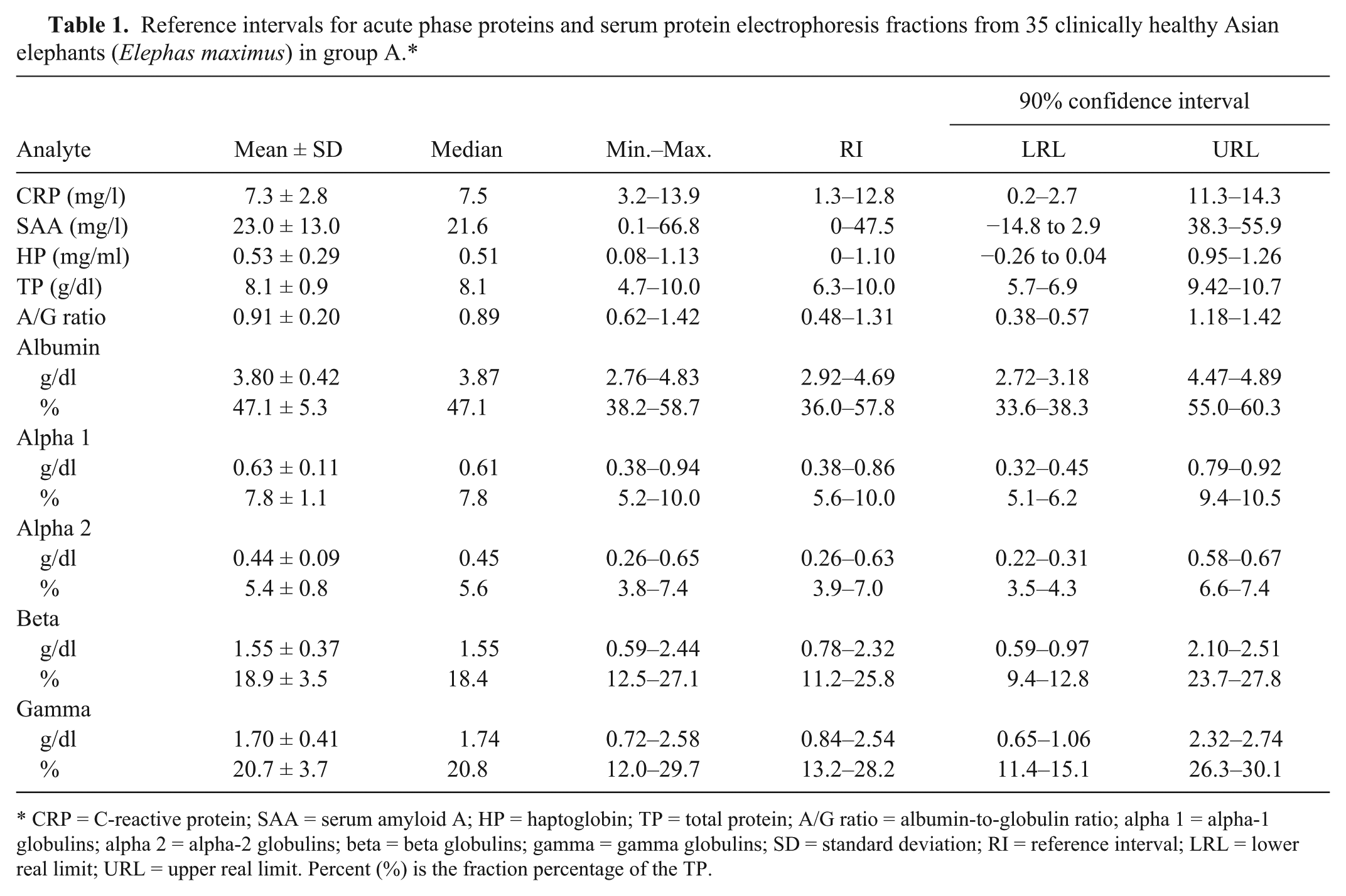
CRP = C-reactive protein; SAA = serum amyloid A; HP = haptoglobin; TP = total protein; A/G ratio = albumin-to-globulin ratio; alpha 1 = alpha-1 globulins; alpha 2 = alpha-2 globulins; beta = beta globulins; gamma = gamma globulins; SD = standard deviation; RI = reference interval; LRL = lower real limit; URL = upper real limit. Percent (%) is the fraction percentage of the TP.
During the study period, an additional 17 serum samples from 17 individuals (group B) were opportunistically collected from Asian elephants originating from the same population of elephants as group A. For this secondary analysis, these new samples were divided into 2 groups based on physical examination findings. Nine of the group B elephants had evidence of active pododermatitis, active dental disease, or orthopedic trauma and were labeled as having active clinical problems (group B1). The remaining 8 elephants (group B2) were considered to be clinically healthy based on physical examination and medical history review. All the clinically healthy group B2 elephants were also members of group A, but although the individual elephants were the same, the group B2 samples were collected at different dates than the group A samples and thus their samples were not used twice in group A. After diagnostic review of the resulting data set from all group B elephants, the group B1 elephants with active clinical problems were compared to the group B2 clinically healthy elephants using an exact Kolmogorov–Smirnov 2-sample test for CRP, SAA, HP, TP, and all the SPEP proteins. A nonparametric test was selected to detect differences in the medians because the group B1 animals tended to have nonnormal distributions of the protein measurements. Significant P values were considered less than P = 0.05.
Table 2 provides a listing of the clinical problems, ages, sex, APP, and SPEP results for all group B elephants. Of the measured APP, only SAA had both a significant difference (P < 0.001) between the 2 groupings and a median value (178 mg/l) in the abnormal clinical problems subset (group B1) that was above the reference intervals. Figure 1 is a pair of box plot graphs that summarizes the SAA results from the group B elephants.
Clinical problems, age, sex, acute phase proteins, and serum plasma electrophoresis results from 17 Asian elephants (Elephas maximus) in group B.*
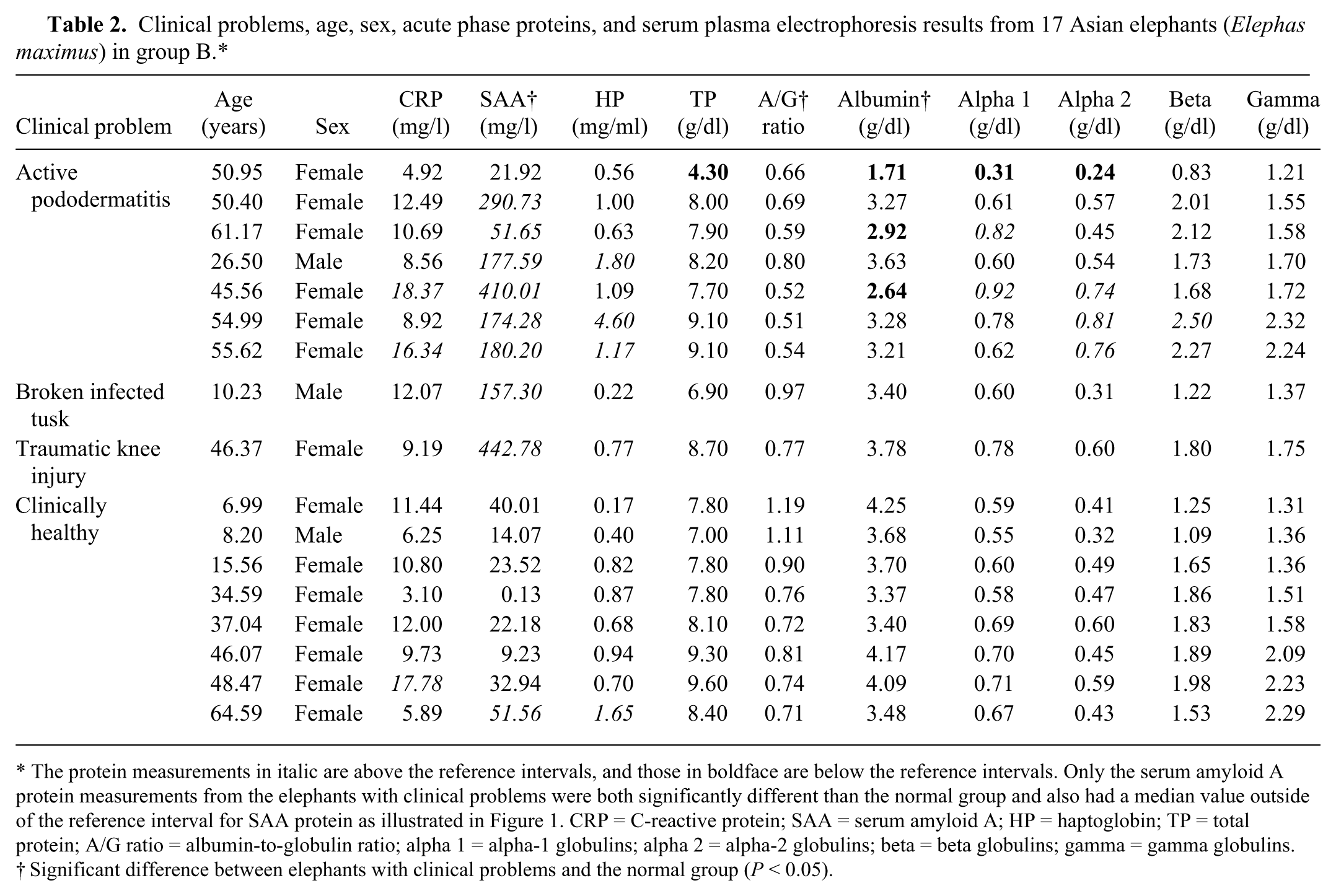
The protein measurements in italic are above the reference intervals, and those in boldface are below the reference intervals. Only the serum amyloid A protein measurements from the elephants with clinical problems were both significantly different than the normal group and also had a median value outside of the reference interval for SAA protein as illustrated in Figure 1. CRP = C-reactive protein; SAA = serum amyloid A; HP = haptoglobin; TP = total protein; A/G ratio = albumin-to-globulin ratio; alpha 1 = alpha-1 globulins; alpha 2 = alpha-2 globulins; beta = beta globulins; gamma = gamma globulins.
Significant difference between elephants with clinical problems and the normal group (P < 0.05).
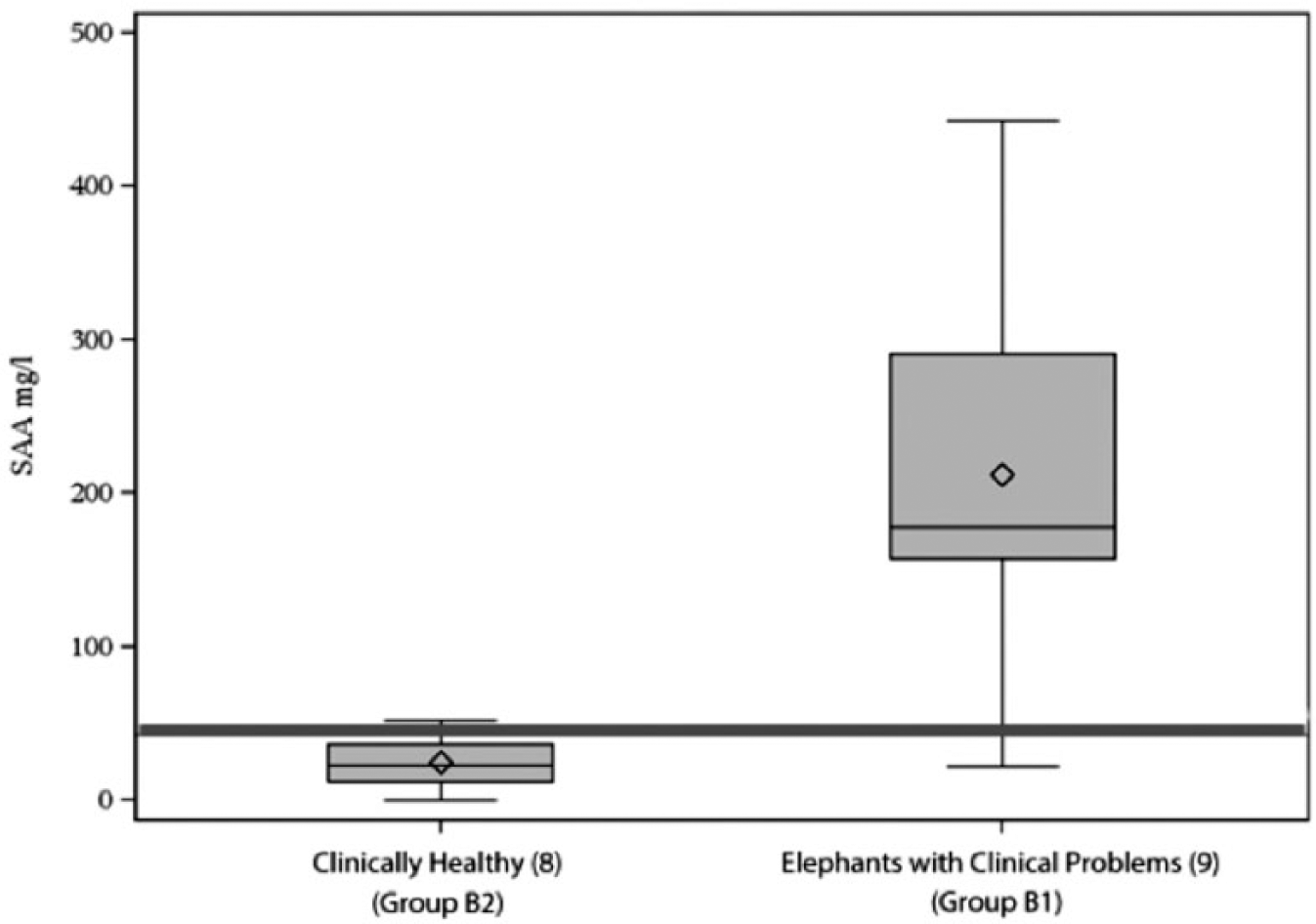
Paired box plot graph summarizes the serum amyloid A (SAA; mg/l) values from the 17 group B Asian elephants (Elephas maximus) that were either clinically healthy (group B2) or had observable clinical problems (group B1). The red line represents the upper reference interval for SAA values (47. 5 mg/l). There is a significant relationship (P < 0.001) between elevated SAA protein values and the presence or absence of clinical problems.
For all the SPEP fractions measured in group B, the median values for both groups B1 and B2 were all within the reference intervals. Notably however, albumin and the related albumin-to-globulin (A/G) ratio levels for group B1 tended to be lower than the group B2 clinically healthy elephants and were significantly different when compared with group B2 (P = 0.016 and P = 0.032). In many other vertebrate species, albumin is considered a negative APP. 8 While marked albumin changes can be observed during severe inflammatory processes, little albumin change has been reported in manatees undergoing rehabilitation where sensitivity for inflammation was reported as only 33%. 5 The lack of albumin change and A/G ratios below the reference intervals in group B1 suggests that although a detectible difference may exist, they may not be clinically relevant differences.
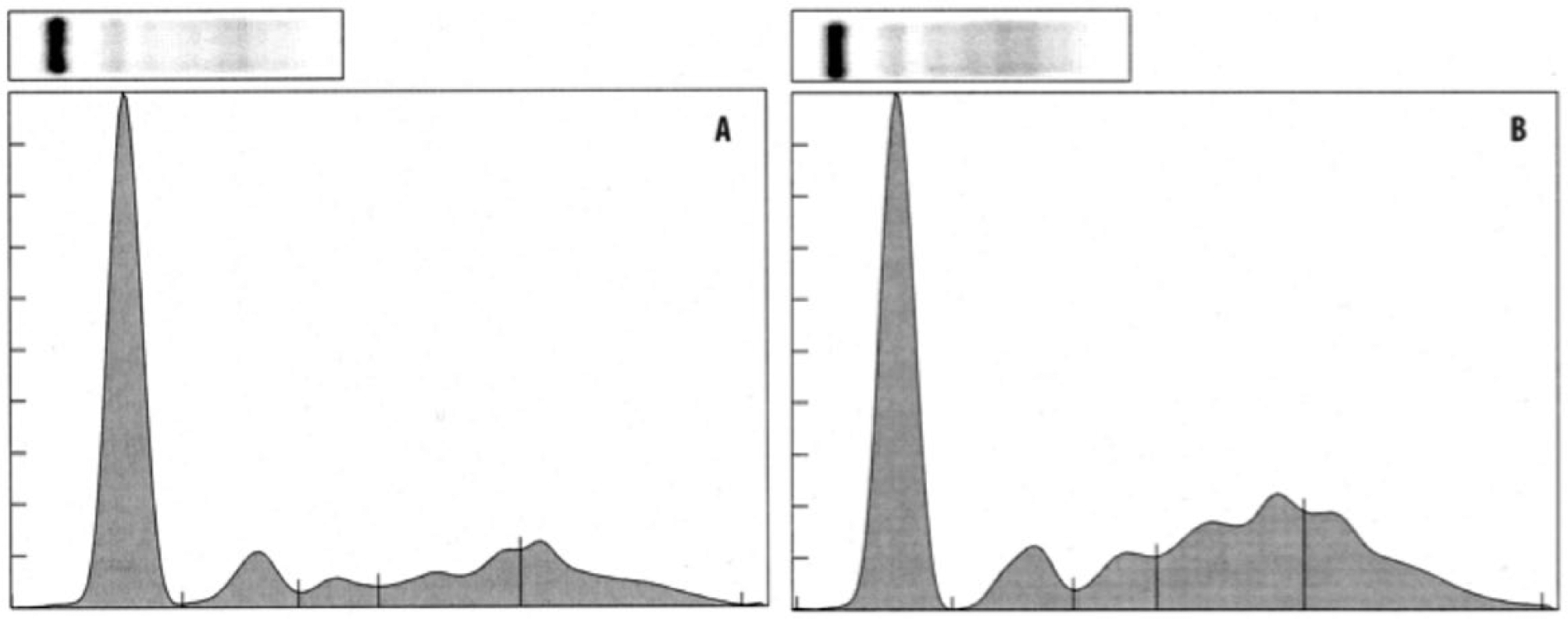
As clinical examples, the SPEP results from 2 Asian elephants are presented and compared in Figure 2. The SPEP graph from a clinically healthy 4-year-old male (elephant 1) with an A/G ratio of 1.29 is compared with an abnormal 50-year-old female elephant (elephant 2) with active pododermatitis and an A/G ratio of 0.69. Of the 3 APPs and all the SPEP proteins measured in the clinically affected elephant 2, only SAA (291 mg/l) was elevated above the reference intervals.
Electrophoretogram
The conservation of APPs among different species allows cross-species use of automated assays. Although the reagents used in the current study were developed for use in human beings, the reagents have shown appropriate reactivity in a variety of wild and domestic species. However, species- and method-specific reference intervals should always be used when using these assays. It is important to note that none of the APPs are specific for particular disease processes, and that their interpretation must be assessed in conjunction with other diagnostic procedures.
The reference intervals for CRP, SAA, HP, TP, and SPEP components were similar to reference intervals reported for other domestic mammals.1,4,5,9,11,12,17,18,21,23 In the present study, SAA is demonstrated to be the most responsive major APP in elephants. This is in agreement with 2 previous reports where SAA elevations were noted consistently in elephants with Elephant endotheliotropic herpesvirus (EEHV) and in 2 captive elephants with inflammatory lesions.2,26 As an extremely conserved protein across numerous species, SAA serves multiple functions that include cell adhesion, cell migration, and cell aggregation. In clinically healthy animals, SAA is usually close to zero, but can increase 1,000-fold during inflammatory processes related to surgery, trauma, infectious agents, neoplasia, and systemic inflammatory diseases.1,4,5,9,11,12,17,21,23 The reference interval for SAA (0–47.5 mg/l) in clinically healthy Asian elephants suggests that this species has low-level constitutive expression. This reference range is similar to horses, where calculated reference intervals for SAA are 0–20 mg/l, but a higher upper range of 47.8 mg/l have been reported in horses with noninflammatory disease.1,15 In clinically healthy manatees, an Afrotherian species closely related to elephants, the reported SAA reference interval ranged between 0 and 46 mg/l. 8
Haptoglobin expression was very low in the current study, as is the case in several other species.4,5,9,11,12,17,21,23 Haptoglobin appears to be associated with chronic disease in some species and is often considered a minor or moderate APP. 9 In Asian elephant serum, the reference interval for HP (0–1.10 mg/ml) is consistent with reports of reference intervals of less than 1 or 2 mg/ml in other mammals.4,5,9,11,12,17,21,23 In a small study of Asian elephants negative for EEHV, HP ranged from 0.96 to 2.96 mg/ml. 26
C-reactive protein expression in Asian elephant serum was also low in the present study, which is also typical of several other species.4,5,9,11,12,17,18,21 However, CRP is not expressed equally in all species. For example, in human beings, CRP is a major APP as well as a biomarker for cardiovascular disease. In dogs, another species where CRP is a major APP, the reported normal reference intervals are 0–20 mg/l, which is higher than was found for Asian elephants in the current study.9,18,19 The reference interval for CRP reported herein for Asian elephants (1.3–12.8 mg/l) is similar to CRP intervals reported in harbor seals (Phoca vitulina) and bottlenose dolphins (Tursiops truncatus).6,13 Further studies are needed to address the reactivity of the CRP reagents with elephant proteins and to consider the use of either elephant-specific reagents or non–antibody-based assays.
The clinical use of SPEP also necessitates use of species- and method-specific reference intervals for the SPEP fractions. 10 Reports of SPEP reference values in nondomestic mammalian species are limited to a few marsupial species and a few marine mammals.24,25,27 The SPEP protein fractions from African and Asian elephants have been previously published and are similar to those reported in the current study.3,20 In those earlier elephant studies, it should be noted that older, non-automated methods for sample analysis and graphing SPEP electrophoretograms were used, and protein fraction delimitations were not consistently indicated or defined. In the current study, each electrophoretogram is marked to delineate the protein fractions (Fig. 2), and corresponding values for the protein fractions are provided so that elephant SPEP can be compared among different laboratories running the same automated methods.
In summary, the reference intervals for APP and SPEP reported in the present study extend the toolbox of diagnostic and monitoring tools available to clinicians involved in the clinical evaluation of captive Asian elephants. The current study is limited by the relatively small sample size and sampling of a single herd of captive Asian elephants. In the future, when larger sample sets are available, the development of reference intervals for elephants associated with age and sex may be possible. Additionally, as new proteins are identified in the growing field of proteomics, the use of novel protein biomarker testing of elephants may significantly enhance elephant diagnostic capabilities.
Footnotes
Acknowledgements
The authors gratefully acknowledge the staff of the Ringling Brothers and Barnum and Bailey, Center for Elephant Conservation, for their assistance in conducting this study. The current study was approved under UF IACUC Study #201106469.
a.
PMI Nutrition International LLC, Brentwood, MO.
b.
Vacutainer, Becton, Dickinson & Company, Franklin Lakes, NJ.
c.
RX Daytona analyzer, Randox Laboratories-US Ltd., Kearneysville, WV.
d.
Life Diagnostics Inc., West Chester, PA.
e.
SAA kit, Eiken Chemical Co. Ltd, Tokyo, Japan.
f.
Phase haptoglobin kit, Tridelta Tri-DD, Boonton, NJ.
g.
SPIFE 3000 system, Helena Laboratories, Beaumont, TX.
h.
SAS version 9.3, SAS Institute Inc., Cary, NC.
i.
MedCalc version 10.4.5.0, MedCalc Software, Ostend, Belgium.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that there was no financial support for the research, authorship, and/or publication of this article.
