Abstract
The present study compares diagnosis of avian
Introduction
Avian
Infection with ABV in birds is routinely detected by reverse transcription polymerase chain reaction (RT-PCR) and by serological procedures.6,13 Serological detection procedures most often rely on the detection of antibodies against the P40 protein, the most common ABV antigen in birds and mammals. 35 The problem with serological tests is that such tests give information with regard to the history of the infection and may not give information with regard to the current state. 34 In addition, immunological tolerance can interfere with the formation of antibodies, causing serological tests to be negative when the birds are infected.22,34 One of the difficulties with RT-PCR is that RNA is unstable because of hydrolysis 3 and, although methods of preservation have improved, long-term storage is still a problem. 24 Proteins are far more stable than either RNA (or DNA),1–3,29 but Western blotting and immunocytochemistry 13 for the routine detection of ABV have hardly been applied.
Freshly plucked feathers have been used before as a relatively noninvasive source of DNA and RNA for the identification of species and sex identification 33 as well as for the identification and/or cultivation of disease-causing viruses.5,11,18 Although cloacal swabs, throat swabs, and fecal matter are most often used for pathogen identification, freshly plucked feathers have been found to yield more reliable results. 18 The stability problem of nucleic acids on storage, however, still persists. In the current study, the usefulness of protein analysis by Western blot of proteins obtained from dried feather shafts has been explored and the results compared with those obtained with serological analysis for the presence of anti-ABV antibodies in plasma by enzyme-linked immunosorbent assay (ELISA) for the diagnosis of ABV.
Materials and methods
Study animals
The birds used in this study were maintained as described previously
6
in outdoor aviaries at the private residence of the principal investigator (SR de Kloet). The birds included sun conures (
Feather collection and preservation, and protein extraction
Contour and wing cover feathers were plucked, air dried for 30 min, and stored in plastic bags at room temperature (approximately 25°C). For the extraction of proteins from feather stems, approximately 5-mm long stems from 6–8 feathers were cut off into 100 μL of 1x protein loading bufferc and heated for 5 min in a boiling water bath. Ten to 20 μL of this solution was used in a Western blot. Protein extracts of tissues were made in a similar way. A 50-mg block of tissue was homogenized in 10 volumes of 1× protein loading buffer c and incubated in a boiling water bath for 5 min.
Antibodies and antigens used in Western blot and ELISAs
Blood (25 μL), as a source of antibodies, was obtained by brachial vein puncture and diluted with 250 µL of buffer A (0.15 M NaCl, 0.025 M ethylenediamine tetra-acetic acid [EDTA], pH 8.0). Diluted plasma was obtained after removal of the cells by 5 min of centrifugation at 5,000 ×
Western blot, using 10% polyacrylamide gels in tricine–sodium dodecyl sulfate
27
was carried out following standard procedures, using polyvinylidene fluoride membranes (PVDF),e,21 0.01 M
The ELISA for the detection of anti-ABV antibodies was carried out using recombinant P40, P29, P24, P16, P10, and a segment of the ABV RNA polymerase protein as a fusion product with maltose-binding protein (MBP). j The recombinant proteins were prepared as described previously. 6 The sequences of the genes of the proteins have been entered into the GenBank database with accession nos.: FJ792853 (P40), JF894121 (P29), HM369502 (P24), HM3369503 (P16), JF894120 (P10), and JF894122 (BPOL). In short, 1 μg of the recombinant fusion protein in 50 µL of 0.05 M sodium carbonate (pH 9.5) was added to the wells of a microtiter plate. k The plates were shaken for 1 hr on a rotary shaker at room temperature, washed with PBST using an automatic plate washer, l and blocked overnight with MPBST at 4°C. After washing with PBST, the plates were incubated at ambient temperature (approximately 25°C) for 1 hr with 2,000-fold diluted plasma or egg yolk IgY in MPBST containing 100 μg of MBP per mL, to suppress the nonspecific binding of plasma antibodies to MBP, the fusion partner of the recombinant proteins. Subsequently, the plates were washed with PBST and incubated for 1 hr with 2,000-fold diluted goat anti-bird secondary antibody conjugated with HRP. g After the plates were washed again with PBST, 40 μL of nonprecipitating, stabilized TMB h solution was applied to the wells, and the plates were incubated for 15 min at 25°C. The reaction was stopped with 40 μL of 0.5 N HCl, and the HRP activity was measured at 450 nm. Measurements were carried out using a microplate reader. m Antibody titers were determined as described previously 6 by endpoint titration from 2-fold serial dilutions of the plasma samples and represent the reciprocal of the highest dilution that still gave a color change as compared to plasma of noninfected birds. 4 As a cutoff value for identifying positive sera, a value of 3 standard deviations above the mean absorbance value of sera from a group of 8 ABV-negative African grey parrots was taken. The approximate relative titers of the antibodies in the IgY used (of sun conure 666f) measured by this ELISA were as follows: anti-P40 = 5,000; anti-P24 = 5,000; anti-P16 = 5,000; and anti-P10 = 1,000. The titers of the antibodies in the plasma of bird 666f were as follows: anti-P40 = 10,000; anti-P24 = 16,000; anti-P16 = 20,000; and anti-P10 = 1,500. The titers of plasma ABV antibodies of a sun conure (4864f) with a low level of anti-ABV antibodies were as follows: anti-P40 = 2,000; anti-P24 = 200; anti-P16 = 250; and anti-P10 = 200.
Detection of ABV infection by RT-PCR, and avian sex identification
Detection of ABV infection using a RT-PCR of RNA isolated from throat swabs of hatchlings and feather stems of older birds was carried out as described previously, 6 using the primer pair Nterm and BoxR, which amplify the P10 gene, and the primer pair Bol4 and Bol5, which amplify a segment of the RNA polymerase gene of ABV. Primer pairs amplifying a segment of the glyceraldehyde dehydrogenase gene and a segment of the ribosomal 18S gene 17 were used as internal amplification controls. The amplification products were analyzed by 1.7% agarose gel electrophoresis in Tris–borate–EDTA (pH 8.0) 20 and staining with ethidium bromide (0.5 μg per mL). The sex of the birds was determined by amplification of a segment of the chromo-helicase-DNA–binding protein (CHD) gene as described previously.10,33
Results
Antibodies
The results presented in this study demonstrate that plasma from sun conures infected with ABV contained antibodies to the ABV P40, P24, P16, and P10 proteins. However, antibodies to the bornaviral P29 segment of the capsid protein and the RNA polymerase could not be, or could hardly be, detected (Fig. 1). The relative antibody concentrations specific for P40, P24, P16, and P10 in plasma were found to be highly different across birds and were more or less specific for the individual bird. The plasma of some birds contained mostly anti-P40 antibodies (e.g., birds 015m and 1332f), while others had high antibody levels for 3 or 4 ABV proteins (e.g., birds 666f and 3355m; Fig. 1).The data showed also that the nature and the relative concentration of anti-ABV antibodies in the plasma was similar to that in the egg yolk and stayed relatively constant in plasma over a period of several years.
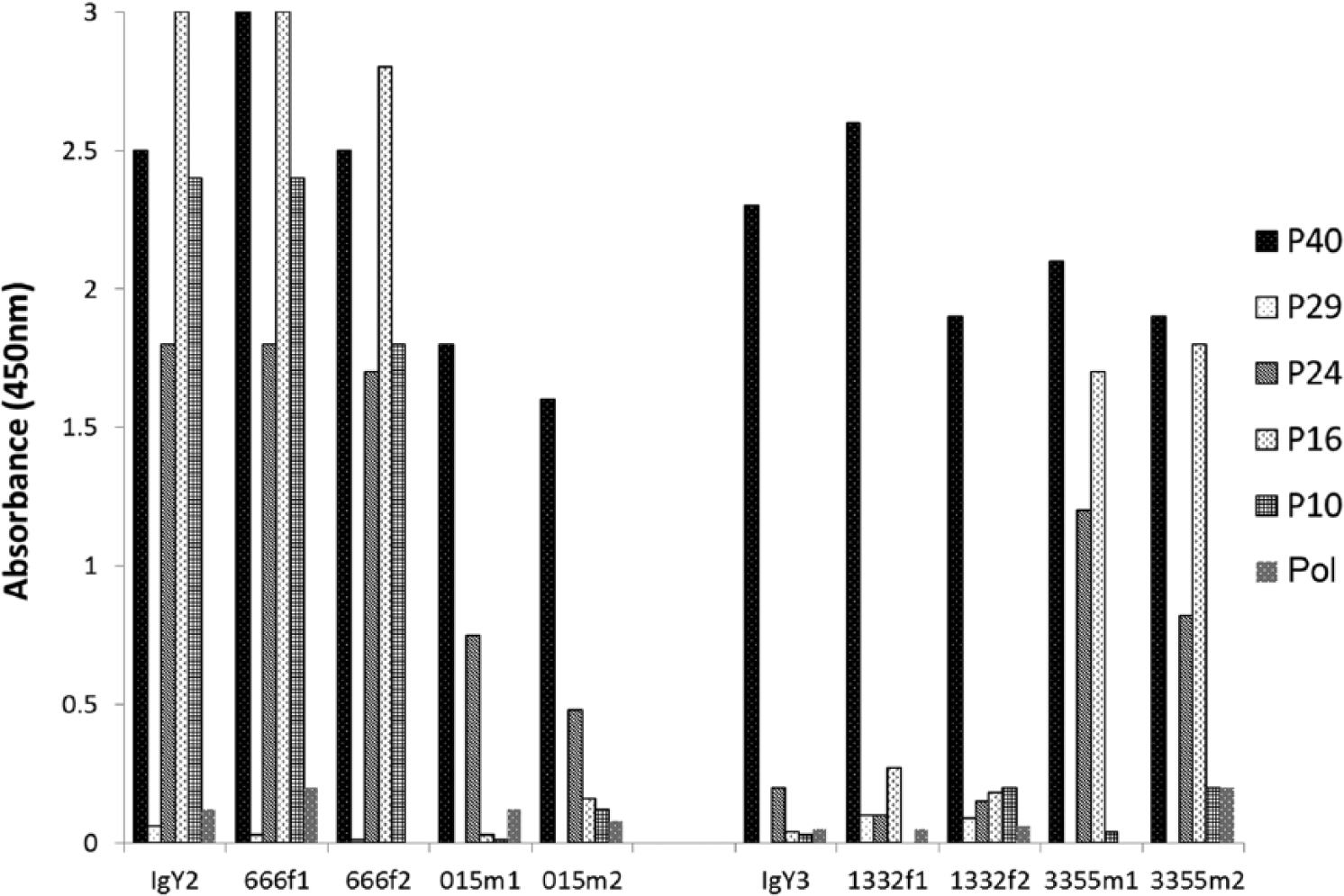
Anti–avian
Utility of feather calami
Western blots of protein extracts (Fig. 2) from feather stems from a number of psittaciformes that had been stored at room temperature for different lengths of time and probed with IgY from a sun conure (666f) infected with ABV (see Fig. 1) showed that 2 proteins with molecular sizes of approximately 40 kD and 24 kD could be detected. These bands are similar in size to the ABV P40 nucleoprotein and ABV P24 phosphoprotein, respectively. The infection of the birds with ABV used in Figure 2 had been determined previously through clinical signs and by RT-PCR at the time when the feathers were collected (between 2010 and present). These proteins could be detected in extracts from feathers of birds infected with ABV even after they had been stored for several years at ambient temperature (approximately 25°C). No material with the size of the matrix protein (P16, 16 kD) or the X protein (P10, 10 kD) could be identified, although antibodies to these proteins were found in plasma and IgY. Material of a very high molecular weight that could presumably be the RNA polymerase or capsid protein also could not be detected.
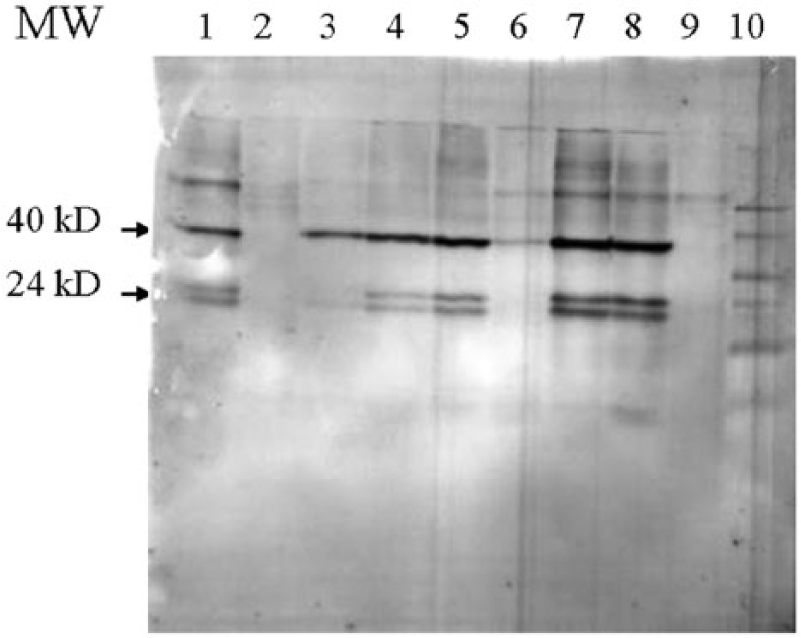
Western blot of feather calami extracts of different
Use of plasma from a sun conure or African grey parrot known to be not infected with ABV in similar blots (Fig. 3B), or use of the secondary antibody by itself (Fig. 3C), failed to show the P40 and P24 components, suggesting strongly that the P40- and P24-like material indeed represented these 2 bornaviral proteins. The data showed also that ABV protein could already be detected in developing feathers of birds of a young age (1 month). Almost all birds with PDD that were used in this investigation were infected with ABV genotype 2, as shown by sequence analysis of the P40 segment. The strain of ABV was apparently not of much consequence, as IgY that was isolated from an egg from a sun conure infected with ABV-2 detected ABV P40 and P24 protein in a sun conure infected with strain 4 (Fig. 2, lane 5) equally well. None of these components could be identified in birds that were not infected with ABV (Fig. 2, lanes 2 and 6), or when IgY from a sun conure not infected with ABV was used. The results showed also that the P24-like material was found to migrate as 2 components of a slightly different molecular size. Material with a molecular size of approximately 65 kD could be seen in almost all lanes (Fig. 2). A component of this size could also be detected in liver and brain tissue and in purified sun conure IgY (Fig. 3A). It also produced a signal with plasma of a bird not infected with ABV (Fig. 3B), or the secondary anti-bird antibody by itself (Fig. 3C), suggesting strongly that the 65 kD component is the heavy chain of IgY. 26
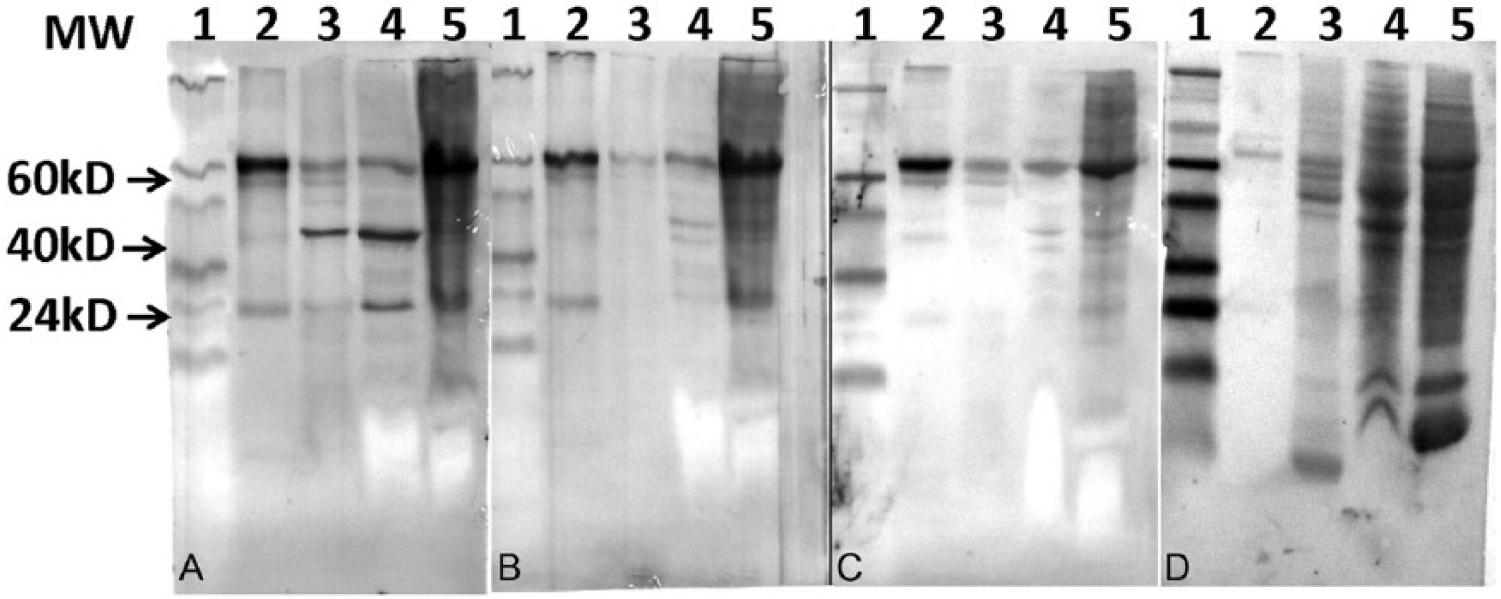
Identification of the 65-kD protein in protein extracts of tissues of sun conures (
Detection of anti-ABV antibodies in plasma of young sun conures infected with ABV
In 2013, 2 pairs of sun conures (pair I: 3355m and 1332f, and pair II: 189m and 4864f), both infected with ABV-2, each raised 2 female offspring. These 4 offspring were all infected with ABV-2 as determined by RT-PCR analysis of a throat swab at 2 weeks of age and analysis by Western blot of feather stem protein, when they were at least 1 month old (Fig. 4A). The 4 birds were still infected with ABV at 1 year of age (data not shown). The relative ELISA antibody levels for adults and their 1-month-old juveniles were determined (Fig. 4B) and clearly showed that plasma of all adults contained anti-P40 antibodies. Three of the 4 adults also contained anti-P24 and anti-P16 antibodies. Plasma of hatchlings nr7, nr9, and nr10 contained no detectable antibodies to recombinant ABV P40, P24, P16, or P10 proteins even at the age of 12 months (Fig. 5). Only hatchling nr8, the sister of nr7, produced anti-P40 antibodies at the age of approximately 1 month (Fig. 5). At 4 months, the anti-P40 antibody concentration in this bird reached a maximum, thereafter it gradually declined to a lower level at 10 months of age. In summary, in these experiments, only hatchling nr8 produced anti-bornaviral P40 antibodies. Antibodies against other bornaviral proteins could not be detected.
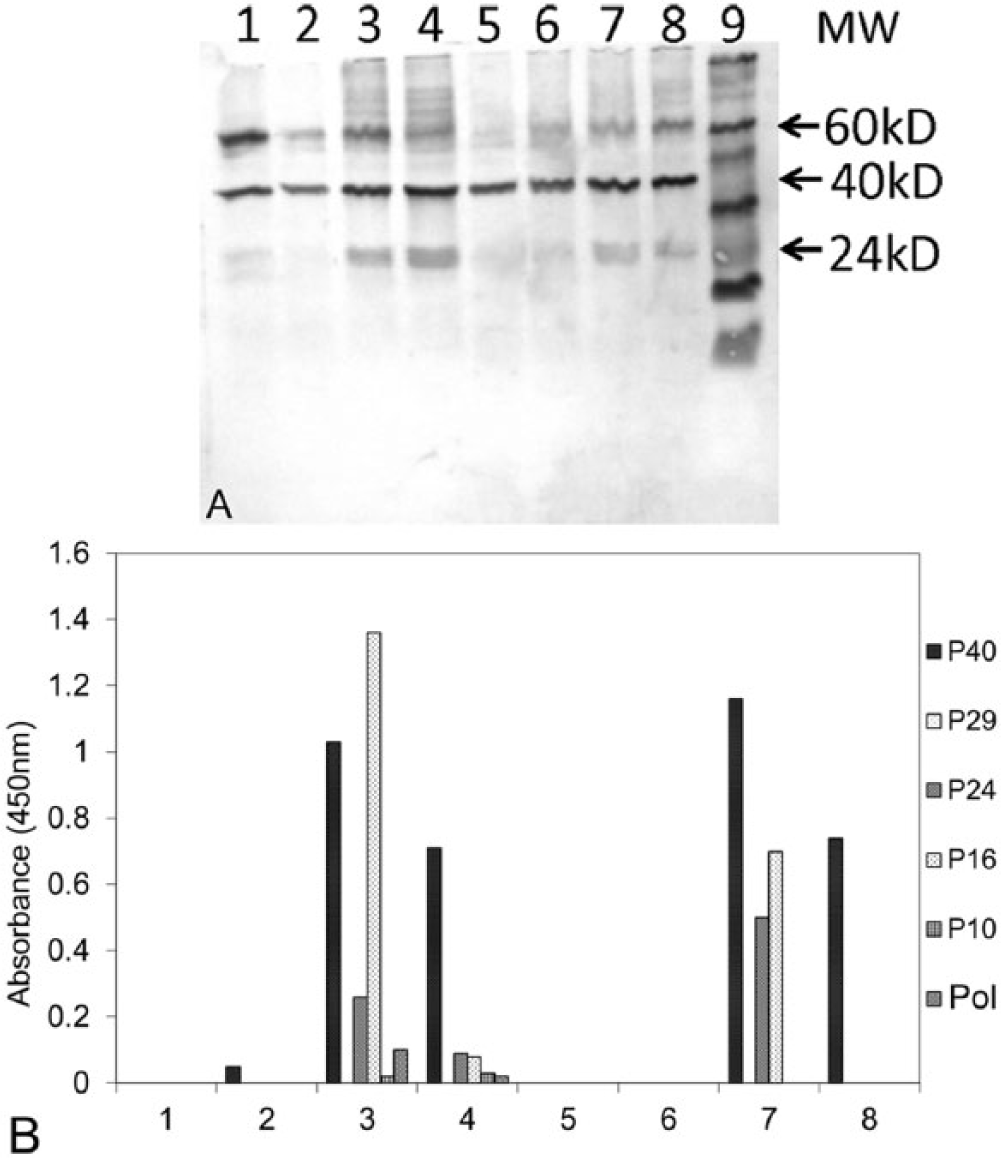
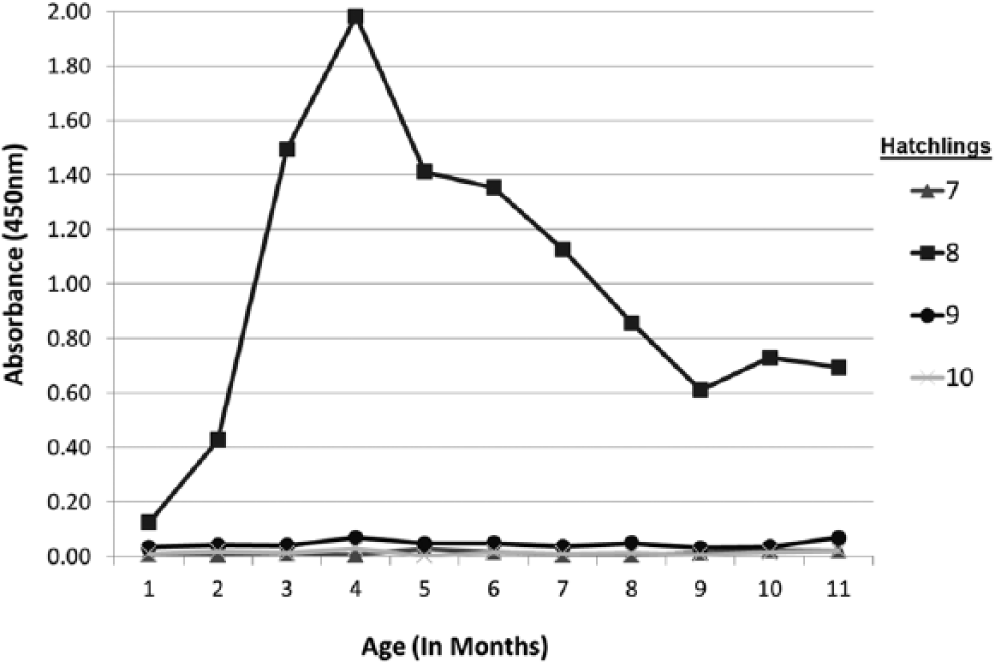
Time course of the formation of anti–avian
Avian Borna disease virus proteins and anti-ABV antibodies in older sun conures
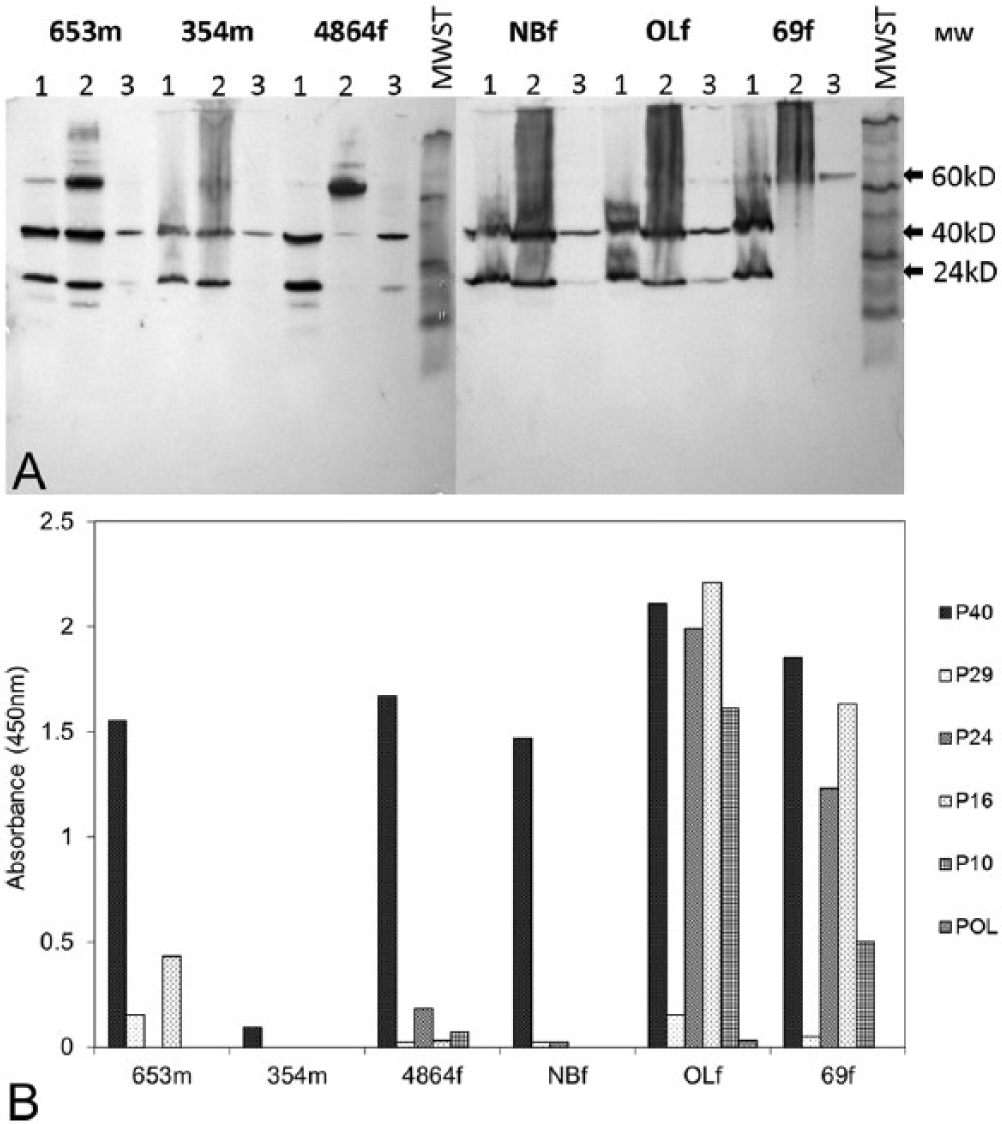
On testing of the older (>5 years) sun conures in the collection, 4 birds out of 42 were found to be free of ABV by RT-PCR and Western blot analysis of feather extracts. One of these birds was nr69f. When this bird suddenly died, the feather stem extracts were free of ABV protein, as were the liver, the kidneys, the ovaries, and the stomach. The brain was found to be infected with ABV (Fig. 6A). The ELISA results showed that the plasma of this bird contained a high level of anti-ABV antibodies (Fig. 6B). A similar result was obtained for another bird (666f) that suddenly died during egg laying. The plasma of this bird was also found to contain a high concentration of anti-bornaviral P40, P24, P16, and P10 antibodies (Fig. 1). Again, the feather stems and the liver did not contain bornaviral protein, but the brain tissue did (data not shown). Further studies showed that among older birds (>5 years), ABV detection results can be very different between individuals (Fig. 6A, 6B). Whereas birds nr653m, 354m, NBf, and OLf had bornaviral P40 and P24 in their brain, liver, and feathers, none of these proteins could be found in the liver of 4864f and 69f or in the feathers of 69f. The anti-bornaviral antibody levels were also found to be very different in the individual birds. While 653m, 354m, 4864f, and NBf were found to contain rather low levels, the plasma of 69f and OLf had very high levels of all 4 major antibodies.
Discussion
The results obtained in the present study demonstrate that dried feather calami can be used to detect ABV antigens by Western blot, even after the feathers had been stored for several years. In this investigation, IgY has been used primarily as a source of anti-ABV antibodies because of the noninvasive isolation of a relatively large amount of purified material. The availability of egg yolk is, however, seasonal. Plasma is available all year and was also used as a source of anti-ABV antibodies. In agreement with earlier studies obtained with chickens, 19 antibodies to the P40, P24, P16 and P10 proteins could be found in plasma or egg yolk of the same bird in the same relative concentration, but antibodies to the bornaviral P29 segment of the capsid protein and the RNA polymerase either could not be, or could hardly be, detected in these 2 sources. As shown previously, 6 the relative antibody concentrations specific for P40, P24, P16, and P10 in plasma and yolk could be markedly different between individuals and more or less specific for the individual bird. This is illustrated again in the experiments described in Figure 1 where it was shown that whereas some birds had mostly anti-P40 antibodies (e.g. birds 015m and 1332f), others had high antibody levels for 3 or 4 ABV pro-teins (e.g. birds 666f and 3355m). The nature and relative concentration of different anti-bornaviral antibodies in plasma and egg yolk for a given female were found to be comparable. The results showed also that the nature and the relative concentration of anti-ABV antibodies in the plasma of individual birds remains relatively constant over a period of several years and is similar to that in the egg yolk (Fig. 1).
Another reason for primarily using IgY was that the results obtained with IgY always produced Western blots with less background than when plasma was used. A problem may be that the manual Western blotting procedure is considered to be labor intensive, but recent advances have improved the method. The development of automated procedures involving capillary gel electrophoresis is rapidly progressing and has made a high throughput and quantitative assessment possible. 14
In this study, the sensitivity of the Western blot (3 ng) was found to be less than that reported for the RT-PCR (8 pg). 15 The sensitivity of the RT-PCR in these prior investigations had been measured previously using a concentration series of purified RNA transcribed from cloned ABV segments. 15 The sensitivity of the Western blot was determined in the current investigation using a dilution series of cloned recombinant P40 protein as the standard (data not shown). It is however difficult to compare these experiments with an RT-PCR of RNA found in feather extracts because of the uncertainties with respect to the presence of RT-PCR inhibitors and the occurrence of genomic as well as mRNA which both form a product in the RT-PCR. The sensitivity of the Western blot does not show these problems as it measures the concentration of the protein directly. Finally, the sensitivity in the Western procedure can be up to 100 times improved by replacing the use of TMB by staining with colloidal silver or chemiluminescence. 14
The results described in this investigation showed that the Western blot procedure in this study failed to identify material with the expected size of the ABV P16 matrix protein (16 kD), the P10 protein (10 kD), as well as material with a high molecular weight that could be the RNA polymerase or the capsid protein. The presence of antibodies against P16 and P10 and to a much lesser extent to the RNA polymerase in plasma and IgY of sun conures indicates however that these proteins are formed in a high enough concentration to stimulate the immune system, but in too low a concentration to be detected by the Western blot procedure. Analysis by ELISA showed that antibodies against the C-terminal P29 segment of the avian bornaviral capsid protein could not be detected. This might mean that the avian bornaviral capsid protein was not formed or that it never reached a concentration high enough to induce the formation of antibodies. It might also mean that the ABV capsid protein has a low immunogenicity. The absence of the capsid protein, if confirmed, would make ABV an exception to the importance of these proteins. Complete absence of the P29 protein in ABV would present difficulty in explaining why the viral gene of this protein does not contain any nonsense mutations, a situation that would be expected in the absence of a functional constraint requiring the presence of a gene product. In mammals, the bornaviral glycoprotein 7 is involved in viral entry into cells 9 and ABV glycoprotein may well have the same function in birds. A possible explanation of the apparent absence of the ABV capsid protein in the current investigation may be that there is so little of the capsid protein needed for cell entry in infected sun conures that it escapes detection in a Western blot and that more sensitive methods need to be used.
The current study shows that, at least in feather stems, material with the molecular size of the P24 component of ABV occurs in 2 forms of slightly different lengths. However, the relationship of these 2 proteins to each other or other bornaviral proteins remains to be established. It may be of relevance that in mammalian bornaviral N protein, the P38 form of this protein differs from the P39 form by lack of a 15 amino acid long NH2-terminal segment between the initiating methionine at AA 1 and the methionine at AA 16.28,30 This element includes the P40 nuclear localization signal. Whether a similar loss of a NH2- terminal segment occurs in the P24 protein remains to be investigated.
The data in this investigation showed also that analysis of plasma for the presence or absence of anti-bornaviral antibodies can be a poor indicator of a bornaviral infection, past or present. Although there were only 4 hatchlings available for the study of the time course of the formation of anti-bornaviral antibodies in newly hatched birds, these 4 birds showed that this course can be very different for individual birds, even when closely related. While 1 member from a pair of siblings (no. 8; Fig. 4A) began to form detectable anti-P40 bornaviral antibodies 1 month after hatching, its sister produced none at all at that age. The 2 offspring of a second pair also did not produce detectable anti-bornaviral antibodies even after 1 year, their age when this report was written (Fig. 5). All 4 birds were infected with ABV as was shown by protein analysis of feather stems (Fig. 4A). The data suggest therefore that immunological tolerance toward a particular avian bornaviral protein can be different between individual birds. As ABV is most likely vertically transmitted through the egg, 15 the consequent early exposure of the developing embryo to ABV antigens may well lead to immunotolerance to ABV.36,37 A comparison with anti-ABV antibody formation in birds that are horizontally infected, for example, by injection of ABV, is required.
In the older (>5 years) sun conures infected with ABV used in this study, some birds, characterized by high levels of antibodies against P40, P24, P16 and P10, often failed to demonstrate the presence of P40 and P24 antigens in feather stems and other peripheral tissues (Fig. 6A). Whether these results mean that, in older birds, the peripheral tissues become free of ABV as a result of the actions of the immune system is debatable, for some cases were found where high plasma anti-ABV antibody content was accompanied by the presence of ABV in these tissues. The continued presence of ABV in the brain of birds, whether these birds carry the virus in their peripheral tissues or not, is most likely the result of the immunologically privileged status of the central nervous system, which restricts the access by the organismal immune system. 32
The results presented in this investigation show that the accurate detection of ABV in birds is best done by a combined analysis of feather stems for avian bornaviral proteins and serological detection of anti-bornaviral antibodies. Protein analysis by Western blot of feather stem extracts for the diagnosis of an infection with ABV can be particularly useful as it may ease the problems encountered in transportation and preservation of diagnostic specimens under field conditions, because of the instability of RNA.
24
This is of importance because investigations on the occurrence of ABV in a natural habitat are lacking. The Western blot technique might also be useful in the analysis of museum specimens for the presence of ABV, for this might allow the determination of the approximate time that ABV first appeared in
Footnotes
Acknowledgements
The authors are indebted to Mrs. Janet Ryland for her help in the preparation of the article.
a.
Johnson Seed Co. Ashburn, GA.
b.
Provita Parakeet, OK Feeds, Miami, FL.
c.
Catalog no. DV4371, Promega Corp., Madison, WI.
d.
Pierce (catalog no. 29500), Thermo Fischer Scientific, Waltham, MA.
e.
Immunobilon, EMD Millipore, Billerica, MA.
f.
Owl, Thermo Fischer Scientific, Waltham, MA.
g.
Catalog no. A140-110P, Bethyl Laboratories Inc., Montgomery, TX.
h.
Catalog no. TM1500, ScyTek Laboratories Inc., Logan, UT.
i.
Life Technologies, Grand Island, NY.
j.
New England Biolabs Inc., Ipswich, MA.
k.
Nalgene (catalog no. 442404), Thermo Fischer Scientific, Waltham, MA.
l.
ELx405-selectCW, US BioTek Laboratories Inc., Seattle, WA.
m.
Tecan Group Ltd, Männedorf, Switzerland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
