Abstract
Liquid-phase blocking enzyme-linked immunosorbent assay (LPBE) using the neutralizing monoclonal antibody (mAb) sandwich method (M-LPBE) for detection of Foot-and-mouth disease virus (FMDV) type O antibodies was developed. Two neutralizing mAbs, 72C1 and 65H6, were raised against the FMDV O/JPN/2000 strain, and used as trapping and peroxidase-labeled detecting antibodies, respectively. Sera from animals experimentally infected with FMDV showed specific positive results by M-LPBE, which were correlated with the results of the virus neutralization test (VNT). When 303 negative bovine and 302 negative swine sera were tested, the specificity of M-LPBE was 100% and 99.7%, respectively. In addition, nine samples that had been collected in 2000 in Japan and regarded as evidently false positives by LPBE (supplied by the World Reference Laboratory for Foot-and-Mouth Disease) were uniformly negative by M-LPBE, just like VNT. Therefore, M-LPBE seems to have sufficient specificity for FMDV type O antibody screening and diagnosis.
Foot-and-mouth disease (FMD) is caused by the FMD virus (FMDV), which belongs to the Aphthovirus genus of the family Picornaviridae. There are 7 immunologically distinct serotypes: O, A, C, Asia1, and South African Territories types 1, 2, and 3. Foot-and-mouth disease is one of the most economically important diseases of clovenhoofed animals. Foot-and-mouth disease virus can cause blistering, vesicles, and ulcers in the epithelia of the mouth, nose, and feet. When FMD outbreaks spread to FMD-free countries, the economic losses are devastating; therefore, rapid eradication is important to protect the global livestock industry. Large-scale serologic surveillance is necessary to perform an eradication program after outbreaks of FMD and to restore FMD-free country status as soon as possible. 1,2,7
The first FMD outbreak in Japan occurred in 2000 after 92 years of disease freedom, and the O/JPN/2000 strain was isolated from Japanese black cattle. In experimental infections using O/JPN/2000, pigs showed typical symptoms, but Japanese black cattle showed no foot lesions, and Holstein cattle, goats, and sheep showed no clear symptoms such as blistering, vesicles, or ulcers in the mouth, nose, and feet (Sakamoto K, Yamakawa M, Kanno T, Yoshida K: 2001, Susceptibility and pathogenicity of cloven-hoofed animals inoculated with foot-and-mouth disease virus O/JPN/2000 isolate, appendix, pp. 3–8. Office International des Epizooties, Report of the Second OIE Meeting of Foot and Mouth Disease Control in East Asia, Seoul, Korea). 5,12 The clinical signs in cattle, goats and sheep with this outbreak were not consistent with what was expected.
In addition, persistent infections occur in some ruminants (especially in water buffalo and vaccinated infected cattle), which can maintain FMDV in their esophagus for a few weeks up to several years. 10 Because of the atypical cases and persistent infection, laboratory detection of antibodies seems to be even more important to identify subclinically infected animals and to eradicate FMDV.
The liquid-phase blocking enzyme-linked immunosorbent assay (LPBE) supplied by the World Reference Laboratory for Foot-and-Mouth Disease (WRL-LPBE) has been reported as a method that produces a significant number of false-positive reactions. 3,6 WRL-LPBE yielded false-positive results during the diagnosis and surveillance of the 2000 FMD outbreak in Japan, as well. There was a 16.1% rate of false positives in Japanese black cattle sera. WRL-LPBE and the virus neutralization test (VNT) had to be performed repeatedly against false-positive animals for a week or more after the first test.
In the present study, monoclonal antibodies (mAbs) against the FMDV O/JPN/2000 strain were produced as previously described 8 and characterized by VNT and enzyme-linked immunosorbent assay (ELISA). Neutralizing mAbs were used to develop a mAb-based sandwich LPBE (M-LPBE) for detection of FMDV type O antibodies.
Baby hamster kidney (BHK-21) and porcine kidney (IB-RS-2) cells were maintained in Eagle's minimum essential medium a with 0.3% tryptose phosphate broth b supplemented with 0.3 mg/ml of L-glutamine, 1.5% of 7.5% NaHCO3, and 5% fetal bovine serum. c Foot-and-mouth disease virus strains O/JPN/2000, O1 BFS 1860, and O/ TAW/97 grown on monolayers of IB-RS-2 and/or BHK-21 cells were used as ELISA antigens for characterizing mAbs and/or as VNT antigens. Inactivated reference antigen O1 Manisa was also used as an ELISA antigen for characterizing mAbs. The O/JPN/2000 strain was grown in IB-RS-2 cell monolayers, and the purification of the antigen for M-LPBE was performed essentially as described previously. 4
Characteristics of monoclonal antibodies (mAbs).*

Hybridoma culture fluids were used for each test. VNT = virus neutralization test (numbers in parentheses indicate neutralization titers); ELISA = enzyme-linked immunosorbent assay.
The VNT was carried out according to World Organization for Animal Health (OIE) protocol, 11 which defines the result of VNT ≤1:8 as negative, between 1:11 and <1:32 as retest, and ≤1:45 as positive. In the current study, sera were diluted 2-fold (starting from 1:4 dilution), and titers 1:32 (>1.51 log10) were scored as positive. Neutralizing titers of hybridoma supernatant fluids were determined by the same method with 2-fold dilution from nondiluted fluid. To confirm the reactivity of mAbs against each type O virus, direct ELISA was performed using the mAbs as peroxidase-labeled detecting antibodies. Conjugation of peroxidase with mAbs was performed using a commercial peroxidase-labeling kit d according to the manufacturer's instructions. The peroxidase-labeled mAbs were stocked at −20°Cas a 50% suspension with glycerol and used in each ELISA at a 600-fold dilution with phosphate buffered saline (PBS) containing 0.05% Tween 20 and 8% skim milk e adjusted to pH 7.6 by adding 0.8 M NaOH (PBSTM).
In the present study, mAbs that neutralized the O/JPN/2000 strain were analyzed in terms of their reactivity against other type O virus strains (O1 Manisa [ME-SA topotype], O1 BFS 1860 [EURO-SA topotype], and O/TAW/97 [Cathay topotype]) using VNT and ELISA. Though mAb 63A11 showed neutralization reactivity to the O/JPN/2000 strain, other tested type O virus strains were not neutralized by mAb 63A11. Monoclonal antibody 63A11 reacted with the O/JPN/2000 and O1 Manisa strains in ELISA. Monoclonal antibody 68F9 reacted with the O/JPN/2000 strain but did not react with other type O virus strains by VNT and ELISA. Monoclonal antibodies 65H6 and 72C1 showed neutralization ability and reactivity with ELISA against all tested virus strains: O/JPN/2000, O1 Manisa, O1 BFS 1860, and O/TAW/97 (Table 1). Given these findings, mAbs 72C1 and 65H6 were selected for the trapping mAb and peroxidase-conjugated detecting mAb to be used in M-LPBE, respectively.
M-LPBE was performed as follows: mAb 72C1 was diluted with 0.05 M carbonate buffer (pH 9.6), f and 50 μl/ well of diluted mAb was coated in 96-well flat-bottom plates g at 4°C overnight or for a few weeks using a commercial ELISA coating stabilizer. h The plates were washed 4 times with 350 μl of 0.005 M PBS. Serum samples were diluted 1:5 with PBSTM. Viruses suitably diluted with PBSTM, and serum samples diluted with PBSTM were mixed 1:1 and incubated for 1 hr at room temperature. After incubation, 50 μl of incubated serum and virus mixture were added to mAb-coated flat-bottom plates that had been washed with PBS 4 times. After incubation for 1 hr at 37°C, the plates were washed with PBS 4 times, and 50 μl of 1:600 diluted peroxidase-labeled mAb (see above) with PBSTM was added to the wells and incubated for 45 min at 37°C. After washing the plate 8 times, 50 μl of o-phenylenediamine/H2O2 substrate was added to the plates, which were then incubated in a dark place for 15 min at room temperature. The reaction was stopped with 50 μl of 1.25 M H2SO4, and the optical density (OD) was measured with an ELISA reader i at 492 nm and 690 nm.
Percentage inhibition (PI) was calculated as follows:
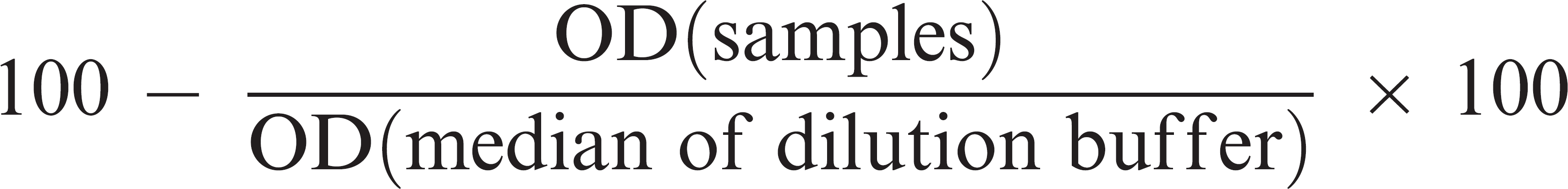
To obtain the sera from animals experimentally infected with FMDV, 2 Japanese black cattle and pigs were inoculated with the O/JPN/2000 strain. Their rectal temperatures were measured, and clinical symptoms were observed. Blood samples were collected by venipuncture for 0–7, 9, 11, and 13 days after FMDV inoculation. Pigs were inoculated with O/TAW/97 strains. Blood samples were collected by venipuncture for 0, 2, 4, 6, 8, 10, 12, and 14 days after virus inoculation. To obtain the O1 Manisa-vaccinated sera, Japanese black cattle and pigs were inoculated with O1 Manisa monovalent vaccine j at six 50% protection doses. Sera were collected by venipuncture for several months. False-positive sera had been collected in the screening for the 2000 FMD outbreak in Japan and were included among the FMD diagnoses after the outbreak in 2000. These false-positive sera showed various high inhibition rates with WRL-LPBE. All sera had titers >1:90 by WRL-LPBE and <1:4 by VNT using the O/JPN/2000 strain. Negative swine serum samples (n = 302) were collected between 1992 and 1993 for porcine epidemic diarrhea and porcine reproductive respiratory syndrome surveys in Japan. Negative bovine samples (n = 303) were also collected in 2000 in Japan.
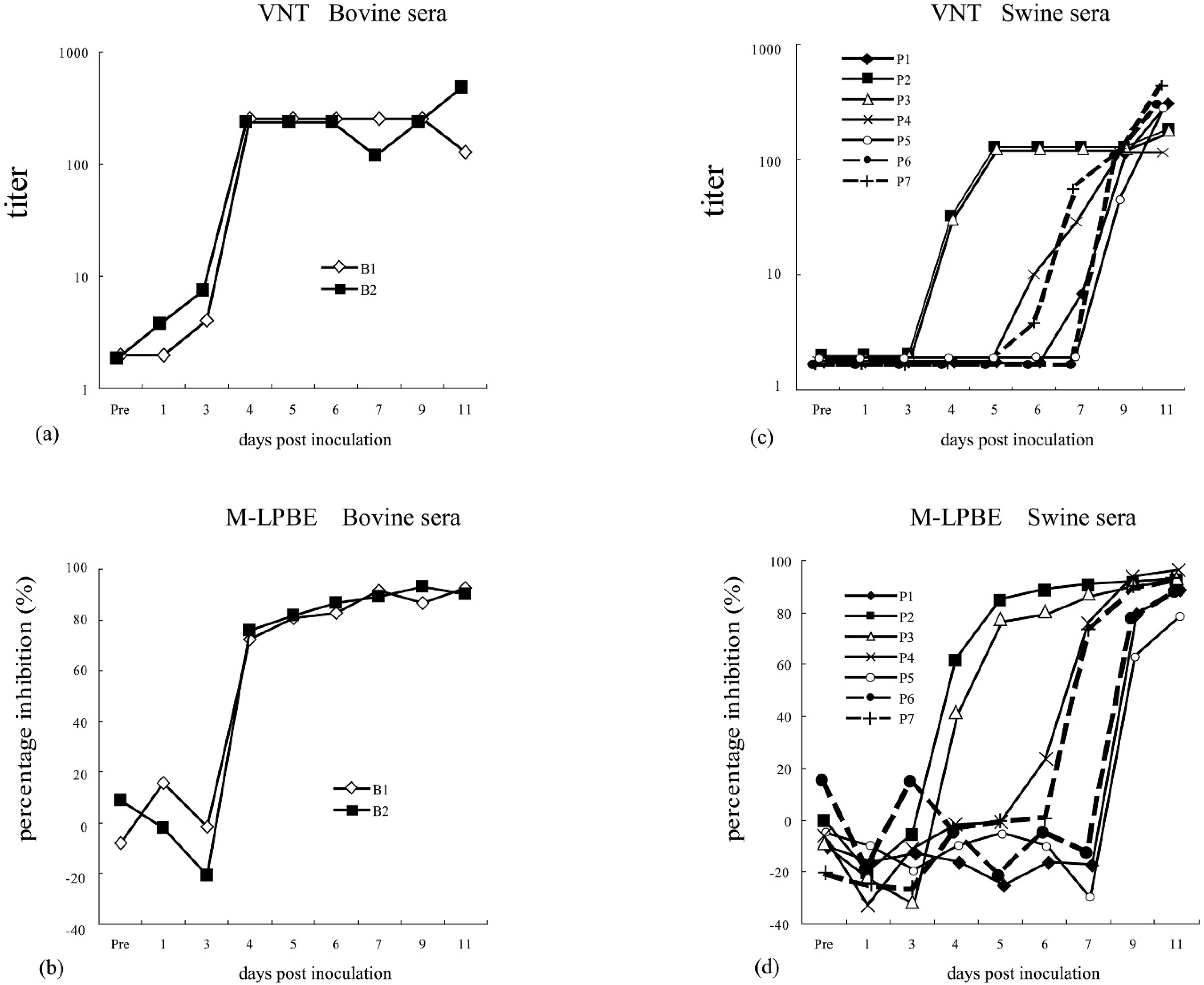
Results of virus neutralization test (VNT) and monoclonal antibody–based liquid-phase blocking enzyme-linked immunosorbent assay (M-LPBE) using O/JPN/2000 strain–infected bovine and swine sera. Bovine 1 and 2 (B1, B2) were inoculated with the O/JPN/2000 strain; sera were collected sequentially after inoculation. a and b, results of VNT and M-LPBE of O/JPN/2000 strain–infected bovine sera, respectively. A low titer (104 TCID50) of the O/JPN/2000 strain was inoculated to pigs 1–5 (P1–P5). Pigs 6 and 7 (P6 and P7) were contacted 1-day postinoculation (dpi). P2 and P3 showed clinical signs and symptoms immediately, but P1 and P6, and P4 and P7 showed clinical signs and symptoms after 6 dpi and 5 dpi, respectively. P5 showed no clinical signs or symptoms for 11 dpi. c and d, results of VNT and M-LPBE of infected swine sera, respectively.
The M-LPBE PIs and the VNT titers were compared using experimentally infected sera from O/JPN/2000 strain–inoculated cattle and pigs. Both VNT and M-LPBE could detect the antibody from 4 days after inoculation (Fig. 1). The dynamics of the VNT titers and PIs of M-LPBE were well correlated throughout the experiment.
Negative field samples of bovine (n = 303) and swine (n = 302) sera were examined by M-LPBE. Statistical results were as follows: The average PIs of negative bovine and negative swine sera were 9.72% (standard deviation [SD]: 9.59) and −3.77% (SD: 13.64), respectively (Table 2). The specificities of M-LPBE for negative bovine sera and negative swine sera at 40% PI were 100% and 99.67%, respectively. The total specificity of 605 sera was 99.83% (95% confidential interval: 98.76–100; average PI: 2.99; SD: 13.58; Table 2). Both 50% PI and 40% PI showed the same specificity. Therefore, a level over 40% PI (more than 3X SD) was used as a positive limit in the current study.
Specificity of neutralizing monoclonal antibody–based sandwich method against negative bovine and swine sera.*
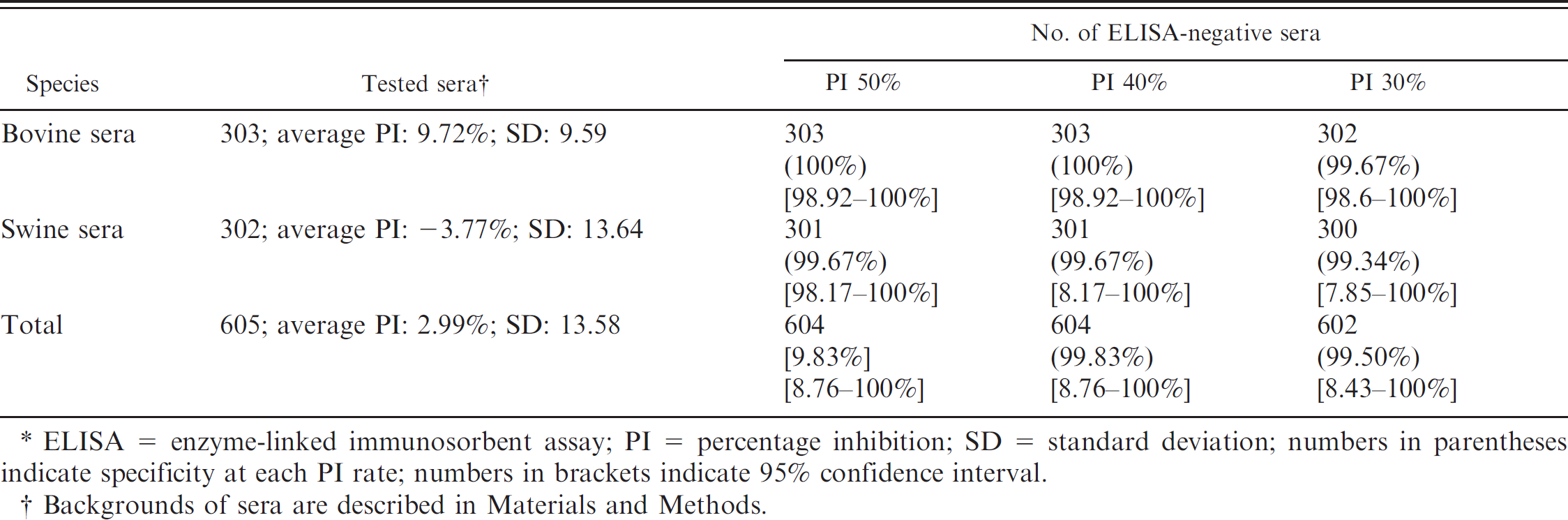
ELISA = enzyme-linked immunosorbent assay; PI = percentage inhibition; SD = standard deviation; numbers in parentheses indicate specificity at each PI rate; numbers in brackets indicate 95% confidence interval.
Backgrounds of sera are described in Materials and Methods.
The heterologous type O–positive sera were examined. Bovine and swine sera inoculated with the O1 Manisa vaccine were examined by M-LPBE and VNT. M-LPBE could detect the antibodies of O1 Manisa–vaccinated bovine and swine sera from 1 week to 3 months after vaccination, without error (Table 3). Sera from O/TAW/97 strain–inoculated pigs were examined by M-LPBE and VNT using homologous (O/TAW/97) and heterologous (O/JPN/2000) virus strains. On the fourth day after inoculation, PIs were slightly high for the homologous virus, but after the sixth day, there were no differences between the 2 PIs (Table 4). These results suggest that the M-LPBE could be applicable to the Cathay topotype and probably to other type O topotypes such as EURO-SA. Recently, FMDV isolates belonging to the Cathay topotype have been found in Vietnam, The Philippines, Thailand, and Hong Kong. 12,13 The sensitivity of M-LPBE to the Cathay topotype is thus quite important.
During the outbreak of FMD in Japan in 2000, many samples showed a nonspecific reaction in WRL-LPBE. Because of the false-positive results, the serologic tests had to be repeated numerous times. To examine the specificity of M-LPBE, the samples that were regarded as positive by WRL-LPBE and were regarded as negative (<4) with VNT (O/JPN/2000 strain) during the Japanese outbreak in 2000 were retested by WRL-LPBE (antigen for competition was O1 Manisa), M-LPBE, and VNT (O/JPN/2000 strain). WRL-LPBE was performed as described in the OIE manual. 11 All sample sera showed more than 1:90 dilutions and were regarded as positive by WRL-LPBE in the current study. On the other hand, these sera had PIs <40% (more than 3X SD) at 1:5 dilutions with M-LPBE and less than 1:4 dilutions with VNT (data not shown). Hence, the problem of nonspecific reaction with WRL-LPBE was essentially eliminated with M-LPBE. Samples showing a nonspecific reaction by high titer in WRL-LPBE were selected; one such sample had a titer over 1:362. A previous study reported that one of the reasons for a nonspecific reaction was animal stress (Haas B: 1994, Application of the FMD liquid-phase blocking sandwich ELISA. Problems encountered in import/export serology and possible solutions. Report of the session of the research group of the standing technical committee of the European Commission for the Control of Foot-and-Mouth Disease, pp. 124–127, Vienna, 19–22 September 1994), but the causes of nonspecific reactions are still unclear.
Detection of antibodies of vaccinated sera.
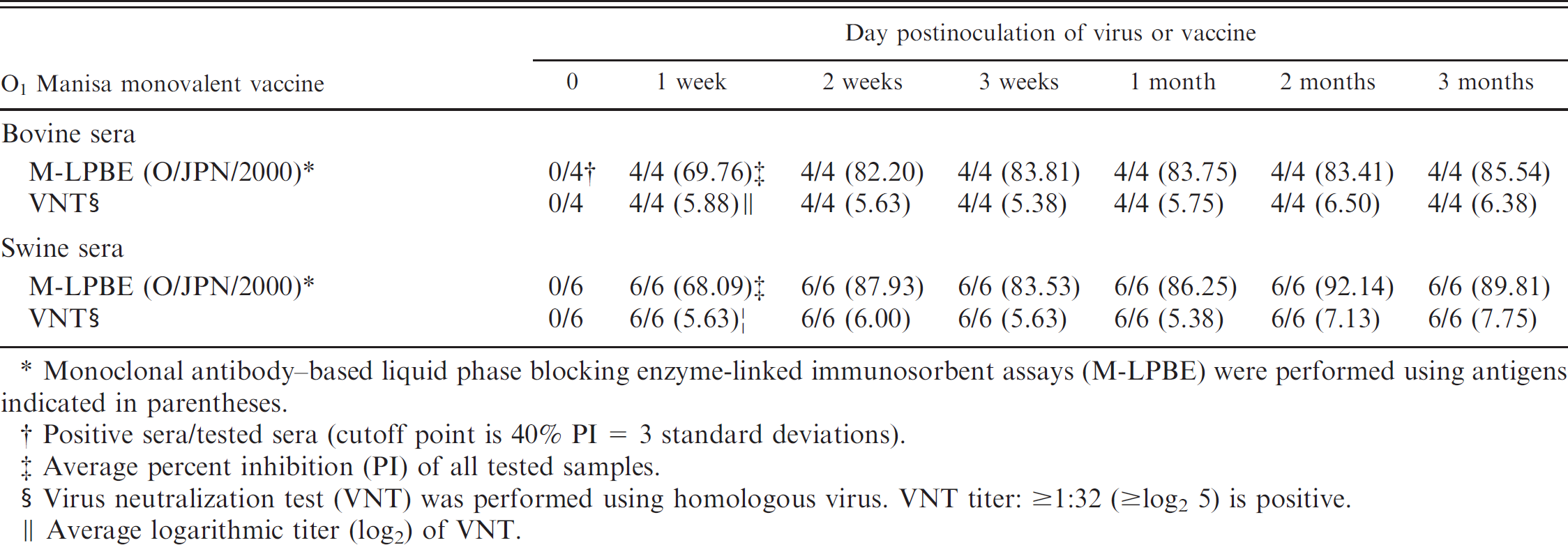
Monoclonal antibody–based liquid phase blocking enzyme-linked immunosorbent assays (M-LPBE) were performed using antigens indicated in parentheses.
Positive sera/tested sera (cutoff point is 40% PI = 3 standard deviations).
Average percent inhibition (PI) of all tested samples.
Virus neutralization test (VNT) was performed using homologous virus. VNT titer: ≥1:32 (≥log2 5) is positive.
Average logarithmic titer (log2) of VNT.
Comparison of the results of virus neutralization test (VNT) and monoclonal antibody-based liquid phase blocking enzyme-linked immunosorbent assay (M-LPBE) using animal experimental test swine sera of heterologous virus.
M-LPBE and VNT were performed using antigens indicated in parentheses.
Positive sera/tested sera (cutoff point is 40% percentage inhibition = 3 standard deviations).
Numbers in parentheses are the average percent inhibition of all tested samples.
VNT titer: ≥1:32 (≥log2 5) is positive.
Average logarithmic titer (log2) of VNT.
In the current study, M-LPBE using neutralizing mAbs was developed. M-LPBE is a sandwich ELISA using 2 different neutralizing mAbs as a trapping and peroxidase-labeled detecting antibodies, respectively. The specificity of M-LPBE had already been verified against many samples, including the Japanese black cattle sera in 2000 in Japan that caused a high nonspecific reaction in WRL-LPBE.
Consequently, M-LPBE could be useful for diagnosis and eradication during serologic surveillance of FMD outbreaks and could help restore countries' FMD-free status in a shorter time. Although VNT has excellent specificity, it has the disadvantages of being time-consuming and unsuitable for large numbers of samples, and requiring the matching of the VNT antigen with prevalent virus. By contrast, M-LPBE can obtain specific data within a few hours. In future work, M-LPBE needs to be validated using a larger number of field-positive sera against other topotypes of type O virus strains and using the sera from other FMDV-susceptible animals.
Footnotes
a.
Nissui Pharmaceutical Co. Ltd., Tokyo, Japan.
b.
Difco Laboratories Inc., Detroit, MI.
c.
GIBCO®, Invitrogen Corp., Carlsbad, CA.
d.
Peroxidase Labeling Kit-SH, Dojindo Laboratories, Kumamoto, Japan.
e.
Wako Pure Chemical Industries Ltd., Osaka, Japan.
f.
Sigma-Aldrich Co. Ltd., Ayrshire, UK.
g.
Immulon™ 2 HB, Thermo Scientific, Milford, MA.
h.
Antigenix America Inc., Huntington Station, NY.
i.
iEMS-Reader, Labsystems, Helsinki, Finland.
j.
Aftopor®, MERIAL Limited, Duluth, GA.
