Abstract
Infection with bovine leukemia virus (BLV) results in economic loss because of reduced productivity, especially reduced milk production, and early culling. In the United States, studies in 1996, 1999, and 2007 showed BLV infection to be widespread, especially in dairy herds. We updated information herein on BLV seroprevalence in the United States, using samples submitted for testing and found negative for antibodies for
Species
Samples for the brucellosis surveillance program were submitted to 1 of 2 laboratories based on the geographic location of the 9 slaughter plants collecting samples. Samples used in this study were from 5 of the 7 slaughter plants submitting samples to the Kentucky Eastern Regional Federal Brucellosis Laboratory (Frankfort, Kentucky) and found negative on testing for
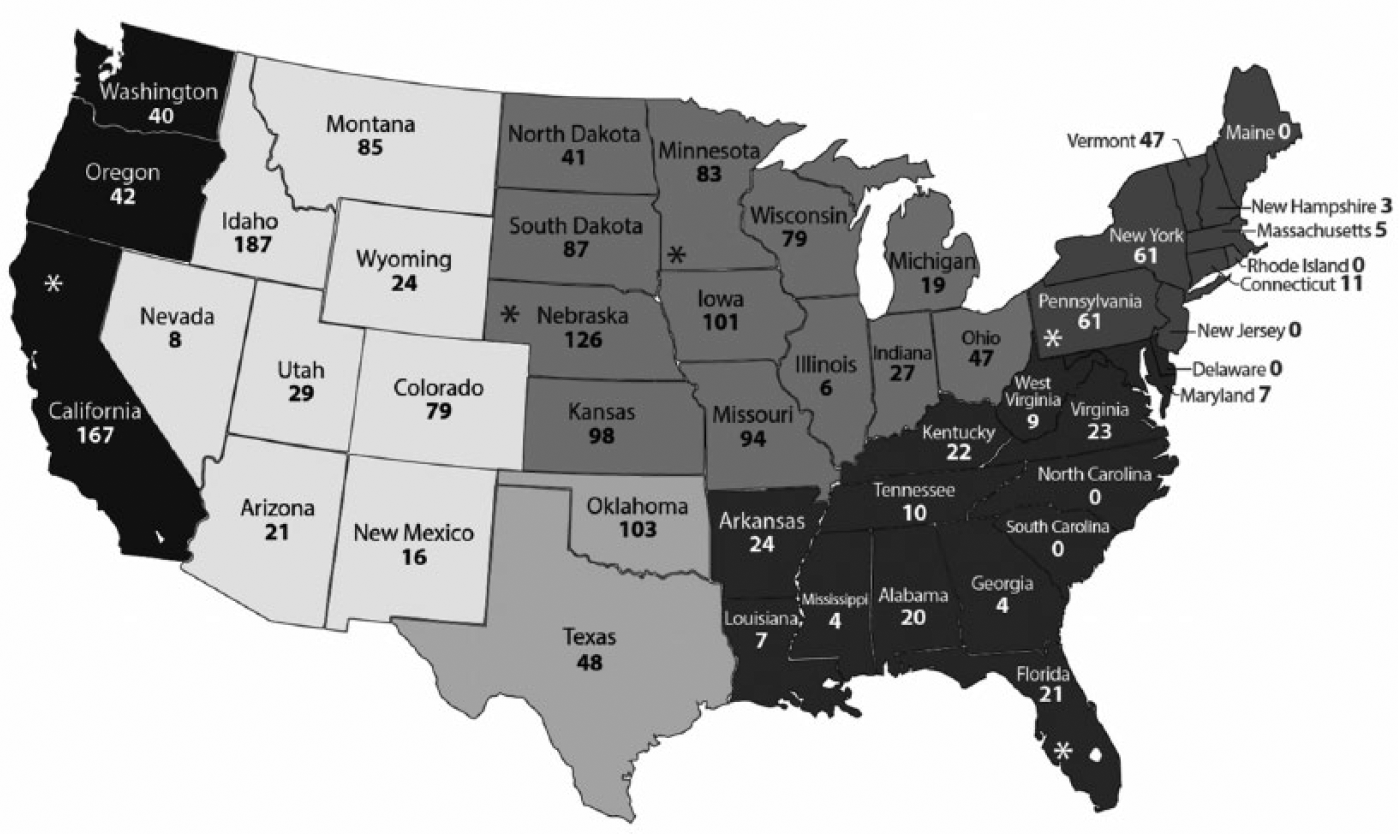
Number of samples for each state in the contiguous United States. Gray scale determines the 6 regions: Pacific West, Mountain West, Upper Midwest, South Central, Southeast, and Northeast. State of location for each processing plant is marked with an asterisk.
Samples were individually filtered using the 0.2-μm Millex-GV filter unit (Merck Millipore, Ireland). All of the samples were banked and held at −20°C until tested. Samples were tested individually by leukosis serum X2 Ab ELISA (IDEXX Laboratories, Hoofddorp, The Netherlands). Results were classified as positive, negative, or suspect based on the manufacturer’s recommendations. Samples with suspect results were re-tested to confirm their status. Samples classified as suspect after re-testing were considered positive for the analysis. The ELISA results along with information about the sampled animals including state of origin (based on backtag number) were entered into a spreadsheet and converted to a SAS (SAS Institute, Cary, NC) dataset for analysis.
Regions were defined based on the state of origin of the animal (Pacific West: CA, OR, WA; Mountain West: MT, ID, WY, NV, UT, CO, AZ, NM; Upper Midwest: NE, ND, MN, WI, SD, IA, KS, MO, IL, IN, MI, OH; South Central: OK, TX; Southeast: AR, LA, MS, AL, GA, FL, SC, NC, TN, KY, WV, VA, MD, DE; and Northeast: PA, NY, VT, CT, NJ, RI, MA, NH, ME). The slaughter plants from which the blood samples were collected were categorized into 3 types based on the annual mix of animals processed. Facilities in which ≥70% of the animals processed were beef cows were classified as “beef plants,” whereas facilities in which ≥70% of animals processed were dairy cows were classified as “dairy plants.” Facilities not fitting these criteria were classified as “mixed plants.” The prevalence of BLV antibodies was compared based on region as well as on plant type. Contingency tables were analyzed using software (SAS). The mean prevalence by region was compared using a Tukey correction for multiple comparisons. Statistical analyses with
Overall, 1,996 samples of the original 2,000 samples had sufficient serum and were available for testing (Table 1). The overall percentage of samples positive (positive and suspect results combined) for BLV antibody was 38.6%. Three samples were classified as suspect. The percentage of samples positive differed by region of origin overall (
Number and percentage of samples positive for bovine leukemia virus antibody by U.S. region.
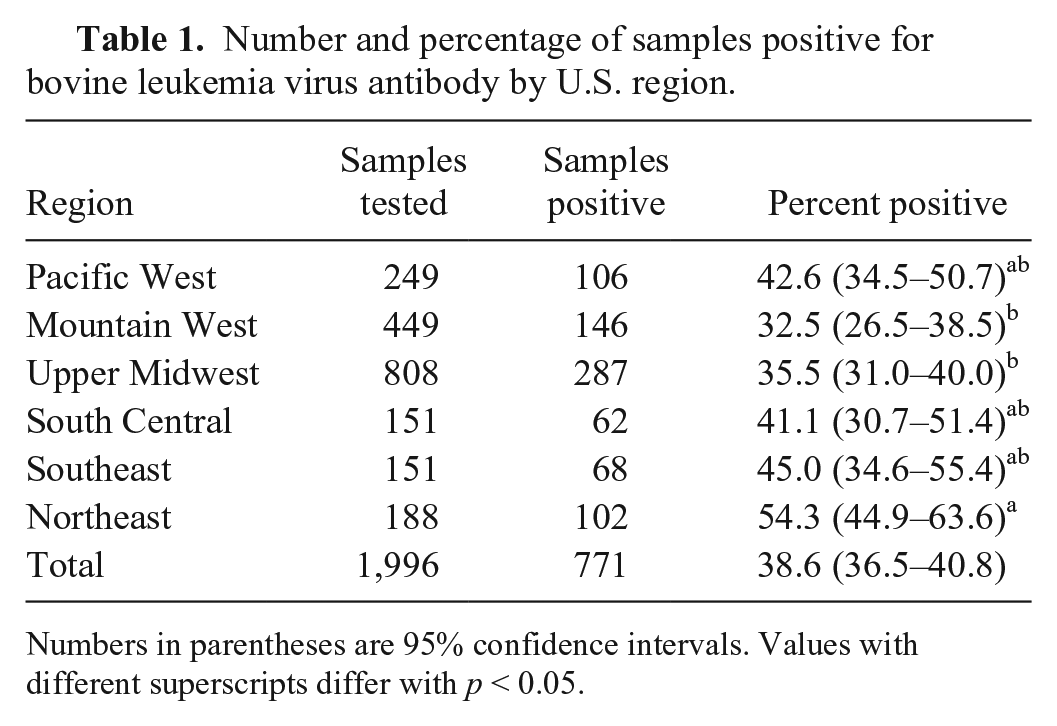
Numbers in parentheses are 95% confidence intervals. Values with different superscripts differ with
Number and percentage of samples positive for bovine leukemia virus antibody by type of slaughter plant.
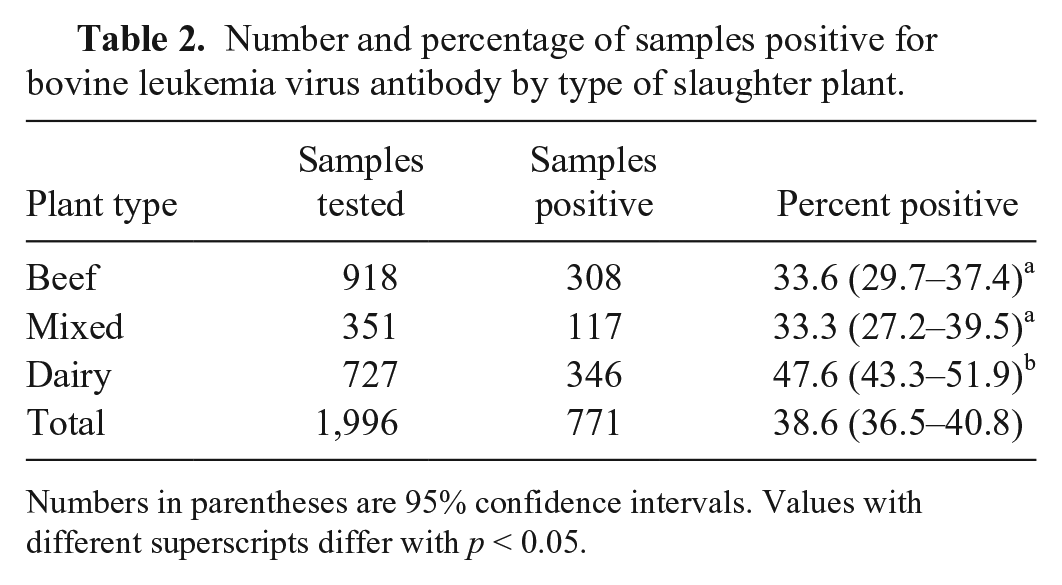
Numbers in parentheses are 95% confidence intervals. Values with different superscripts differ with
The results from our study suggest that BLV infection is widespread in the United States cattle population. The data are insufficient to determine infection rates separately for beef and dairy cattle. However, based on the difference in percentage of samples positive by plant type, these results are consistent with previous studies reporting that the infection rate is higher in dairy cattle than in beef cattle.8–10 Knowing the current status of the distribution of BLV infection may be useful in planning control of the disease in the United States.
Footnotes
Acknowledgements
We thank the staff at the Kentucky Eastern Regional Federal Brucellosis Laboratory including Christopher Cracraft, Shanna Wilburn, April Shannon, Lisa Burke, Megan Zinner, Phyllis Bays, and Jeffery Gifford for logistic support in identifying and shipping serum samples to the laboratory for testing. We also thank Dr. Mark Camacho for his technical assistance in setting up the collaboration between USDA:ARS and the laboratory, and Dr. Kathy Simmons and the National Cattlemen’s Beef Association for their interest in this project and their work to promote cattle health research projects at USDA-ARS.
Declaration of conflicting interests
Disclaimer: Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
