Abstract
Coxiella burnetii and certain members of the Chlamydiales order are zoonotic, intracellular, Gram-negative bacteria, with abortigenic potential in ruminants. These pathogens have a broad host range and worldwide geographical distribution. The current study aimed to reveal the importance of C. burnetii and Chlamydiales spp. in abortions in domestic ruminants and their occurrence in wild ruminants with real-time polymerase chain reaction (qPCR) assays, histology, and immunohistochemical staining (IHC). From the 111 abortion cases of domestic ruminants examined, C. burnetii was detected in 33 placenta samples (cattle, n = 22; sheep, n = 10; goat, n = 1), and members of the Chlamydiales order were detected in 32 placenta samples (cattle, n = 14; sheep, n = 16; goat, n = 2) using qPCR. Coinfection with both C. burnetii and Chlamydiales spp. were identified in 12 cases (cattle, n = 3; sheep, n = 8; goat, n = 1) out of the qPCR-positive samples. The presence of the relevant antigen was confirmed by IHC in 20 cases (C. burnetii, n = 2, in sheep; Chlamydiaceae, n = 17, in sheep [n = 15] and goat [n = 2]; and both pathogens in 1 sheep). Coxiella burnetii was identified in 2.2% (2/91) of the wild ruminants, but the samples were negative by IHC. Uncultured Chlamydiales spp. were detected in 4.4% (4/91) of the placenta samples by qPCR. In conclusion, Q fever is widespread among domestic ruminants in Hungary, and, in several cases, C. burnetii was implicated as the primary cause of abortions. Waddlia chondrophila, Parachlamydia spp., and uncultured Chlamydiales spp. were present only sporadically in samples from cattle and wild ruminants.
Keywords
Coxiella burnetii is a small, intracellular, Gram-negative bacterium that is widespread worldwide, infecting a broad range of host species and causing Q fever. In ruminants, Q fever generally remains asymptomatic with shedding of the bacteria by feces, urine, milk, and vaginal discharges; however, when abortions occur, metritis and placentitis are observed. 1 The pathogen can be transmitted to human beings by direct contact or through inhalation of contaminated dust particles, and can cause a wide variety of clinical symptoms including influenza-like symptoms, pneumonia, hepatitis, abortion, and endocarditis. 1 The apparent prevalence of C. burnetii at the European level is 1.0–44.9%, 3.1–30.0%, and 6.5–75.0% in cattle, sheep, and goats, respectively. 8 Although C. burnetii has been detected in wild ungulates as well, limited information is available about the pathogenic potential of the organism in these species. 2
Similarly to C. burnetii, members of the Chlamydiales order are intracellular, Gram-negative bacteria that are prevalent worldwide and have a broad host spectrum. 16 Contaminated uterine discharges of abortions are the main sources of infection for susceptible animals or human beings through ingestion or inhalation. 21 Members of family Chlamydiaceae (e.g., Chlamydia abortus, the causative agent of ovine enzootic abortion), 6 and the Parachlamydiaceae and Waddliaceae families have been described as being involved in abortions in ruminants,5,19 and in respiratory diseases or abortions in human beings.3,4 Chlamydia abortus is one of the major abortigenic agents in most sheep-producing countries, 6 while endemic but subclinical chlamydial infections have been recognized in cattle herds. 18 Wild ungulates were found to be reservoirs for Chlamydiaceae in Spain and for Parachlamydia spp. in Switzerland, but their pathogenicity in wild host species remains unknown.17,21
Given the complex etiology of abortion in ruminants, the causative agent often remains undetermined. Previous studies in Hungary described infections with Neospora caninum, Bovine herpesvirus 4, Bovine viral diarrhea virus, fungi (Aspergillus and Mucor sp.), and Chlamydia sp. in aborted cattle,7,11 and a recent survey revealed 38.0% (57/150) seroprevalence of C. burnetii in this species. 10 In small ruminants, ovine enzootic abortion was diagnosed as the most frequent cause of abortions with infectious origin in the country. 22 Coxiella burnetii–induced abortion was diagnosed in 2% (5/246) of ovine and in 1% (1/75) of caprine abortion cases using immunohistochemical staining (IHC), 22 and a 6.0% (3/50) seroprevalence of the pathogen has been reported in sheep in Hungary. 10 Information on abortion cases of wild ruminants in the country is scarce, but Bovine herpesvirus 4 was detected in these animals as well. 12
Since the first human Q fever infection in Hungary in 1950, major outbreaks have occurred in the country in 1976–1980 and in 2013, with 70 human cases confirmed and a sheep flock identified as the source of infections in the latest epidemic. 10 Chlamydia abortus–related human infections are rare and have not been reported in Hungary since 2004.
The aim of the current study was to estimate the occurrence and significance of C. burnetii and members of the Chlamydiales order as the cause of abortions in domestic and wild ruminants in Hungary. Cotyledons from aborted placenta samples from 111 domestic ruminants (cattle, n = 85; sheep, n = 21; goat, n = 5) and cotyledons of 91 wild ruminants (roe deer [Capreolus capreolus], n = 33; fallow deer [Dama dama], n = 22; red deer [Cervus elaphus], n = 36) were examined between 2011 and 2013. Abortion samples from domestic ruminants were submitted for routine diagnostic procedures to the Hungarian Veterinary Diagnostic Directorate from various regions of the country (Fig. 1), and all available samples were included in the study. Samples from wild ruminants were collected during 3 hunting seasons from the southwestern part of Hungary. Ethical approval was not required for the study as all samples were collected during routine diagnostic examinations or necropsies. Cotyledons were sliced up and mixed with 10 mL of phosphate buffered saline, then homogenized with a laboratory blender a (5 min at blending speed of 10 strokes/sec). The sediments of centrifuged homogenates (5,000 × g for 5 min) were subjected to DNA extraction with a commercial kit b according to the manufacturer’s instructions.
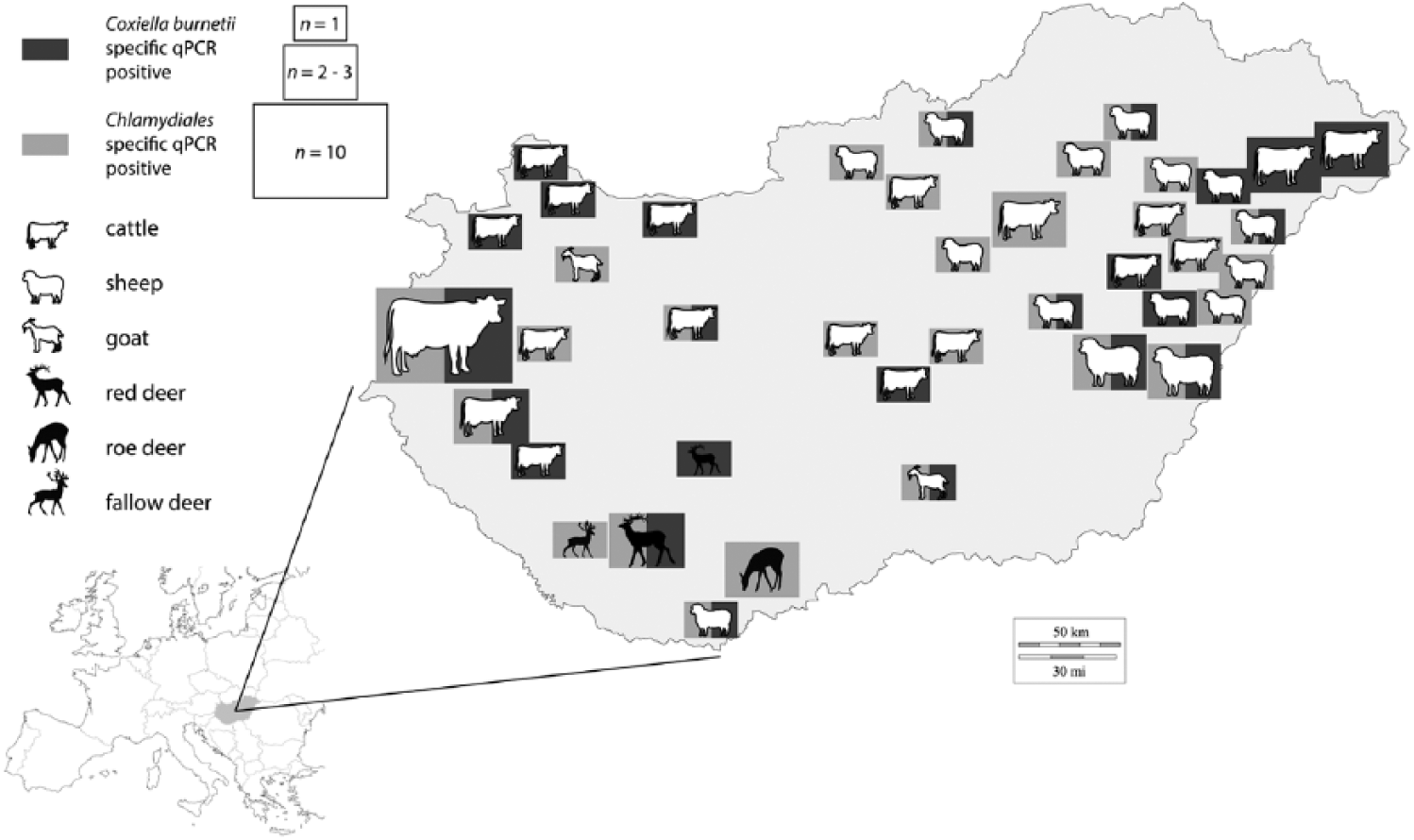
Geographic distribution of the samples that tested positive by Coxiella burnetii– and/or Chlamydiales-specific real-time polymerase chain reaction assay. Animal icons represent the samples’ hosts of origin. Rectangles represent the presence of C. burnetii (dark gray) and/or members of the Chlamydiales order (light gray). The size of the rectangles represents the number of tested animal hosts originating from the same area.
For the detection of C. burnetii, a real-time polymerase chain reaction (qPCR) assay targeting the IS1111 insertion element was performed as described previously. 15 A commercially available positive control c was used in each assay. The detection threshold of the PCR system is approximately 1 colony-forming unit (CFU) with a threshold cycle (Ct) of 36.95 according to the instruction manual supplied with the positive control. c As the examined IS1111 element is present in multiple copies in the bacteria, and the number of copies varies between strains, accurate copy numbers could not be determined by this assay, 13 but an approximate quantity of the bacteria could be estimated from the results. Members of the Chlamydiales order were detected with the 16S ribosomal RNA gene–based Chlamydiales-specific qPCR assay as previously described. 14 Genomic DNA from W. chondrophila was used as positive control in the PCR assays. The detection threshold of the system is 5 genome copies (Ct: 36.3). 14 In order to identify the detected Chlamydiales spp., the sequences of the PCR products were determined d and submitted for BLAST search in GenBank (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi).
Histological and IHC examinations were performed on samples shown to be positive with either of the PCR assays. Formalin-fixed, paraffin-embedded cotyledons were stained with hematoxylin and eosin, and pathological changes were detected by light microscopy. For the detection of bacteria and fungi, Giemsa staining and Gomori methenamine silver staining were used. The IHC examinations were performed as described previously, 22 using a Chlamydiaceae family–specific mouse monoclonal antibody e directed against the chlamydial lipopolysaccharide at a dilution of 1:400, and a rabbit antiserum against C. burnetii f used at a dilution of 1:10,000 as primary antibodies. Tissue sections infected with the corresponding agent were used as positive controls.
Coxiella burnetii DNA was detected in 26% (22/85) of the abortion samples of cattle, which is consistent with the seroprevalence rates (38%, 57/150) reported previously in Hungary. 10 A high DNA load (Ct: 14.12–17.06) was detected in 3.5% (3/85) of the animals, but placentitis was not found in these cases, and the infections were not confirmed by IHC. Bacterial placentitis with necrosis and purulent infiltration was diagnosed in 6 samples, and mycotic placentitis was observed in 1 sample by histological examinations (Table 1). Necrotic placentitis accompanied by vasculitis but without evidence of bacteria or fungi in tissue sections was found in 1 case, while Ct values for C. burnetii were below the detection threshold (Ct: 39.68). In 3 cases, coinfections of C. burnetii with W. chondrophila (n = 1) or uncultured Chlamydiales spp. (n = 2) were diagnosed with the qPCR assays. The abortive characteristics of C. burnetii have not been confirmed experimentally in cattle, and the pathogen is suggested to be an infrequent cause of abortions in this animal species even in endemic areas. 1 In the present study, the regularly high Ct values (22.3%, 19/85), and the absence of antigen detection suggest subclinical infections in the hosts and that C. burnetii should not be considered as a significant abortive agent in cattle in the country.
Real-time polymerase chain reaction (qPCR), histology, and immunohistochemical staining (IHC) results of placenta samples from domestic ruminants and cervids.*
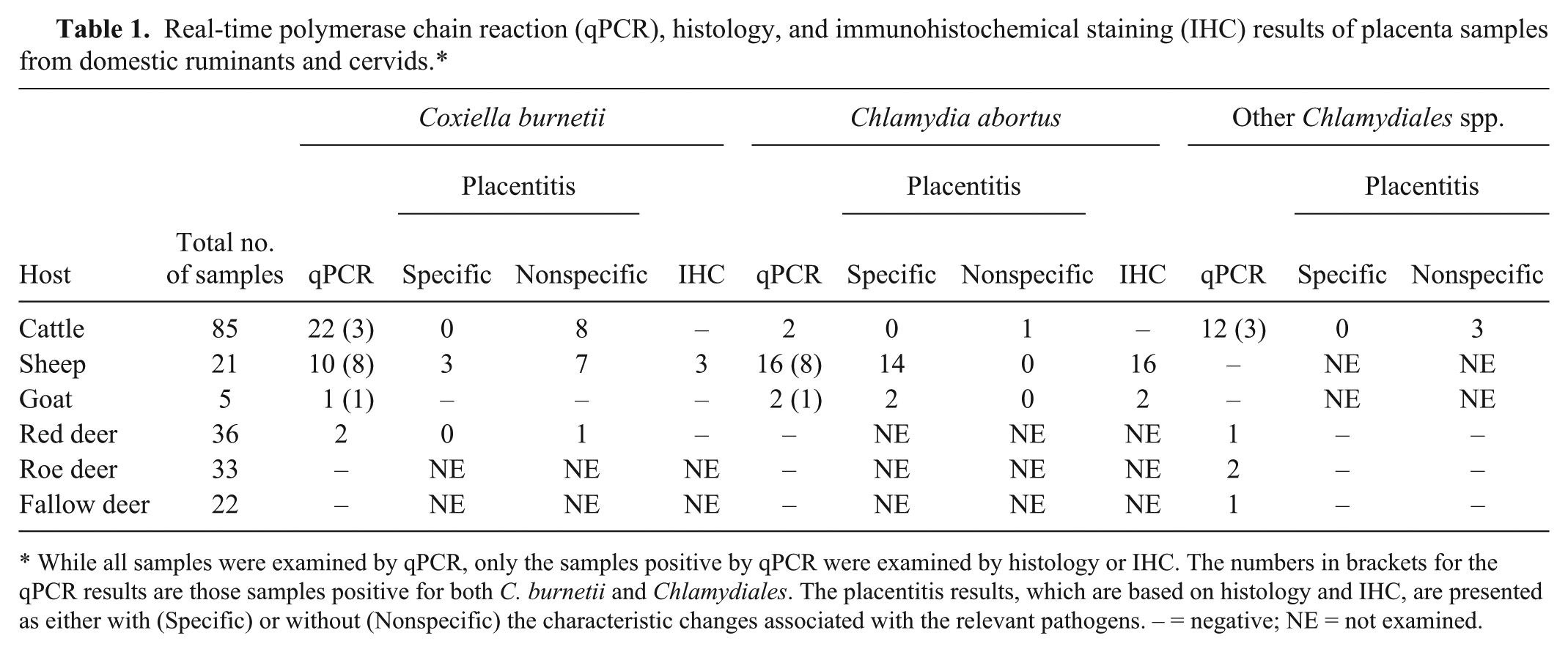
While all samples were examined by qPCR, only the samples positive by qPCR were examined by histology or IHC. The numbers in brackets for the qPCR results are those samples positive for both C. burnetii and Chlamydiales. The placentitis results, which are based on histology and IHC, are presented as either with (Specific) or without (Nonspecific) the characteristic changes associated with the relevant pathogens. – = negative; NE = not examined.
Chlamydia abortus (n = 2), W. chondrophila (n = 4), Parachlamydia sp. (n = 1), and uncultured Chlamydiales spp. (n = 7) were detected with qPCR assay and identified by sequencing in 14 samples originating from cattle. The amount of bacteria (Ct: 42.37–47.71) was less than 5 genome copies in every sample. Bacterial placentitis with necrosis and purulent infiltration was described in 2 samples and, in 2 cases, mycotic placentitis was diagnosed. Based on these results, C. abortus has limited importance in bovine abortions in Hungary, which is in accordance with previous studies on bovine chlamydiosis. 18 In the present study, the detection rates for other Chlamydiales spp. (14.1%, 12/85) in cattle were similar to those reported in Switzerland (14.3%, 49/343). 5 However, Parachlamydia sp., W. chondrophila, and uncultured Chlamydiales spp. were detected only with qPCR assays and with high Ct values in the current study, thus these species may not represent a significant threat for Hungarian cattle herds.
The DNA of C. burnetii was detected in 47.6% (n = 10/21) of the ovine abortion samples, a value much higher than the seroprevalence data reported previously in Hungary (6%, n = 3/50). 10 Placentitis was found in all qPCR-positive cases in the present study (Table 1). The infection was confirmed in 3 cases (Ct: 8.51–13.07) by IHC and histology, which indicates that C. burnetii was the causative agent of these abortions. The rate of infection detected by IHC (14.3%, 3/21) in the present study and previous data from Hungary (2%, 5/246) 22 exceed the rate of C. burnetii–caused abortions reported in other European studies (approximately 1% or less). 1 In 8 of the 10 C. burnetii–positive ovine cases, C. abortus was also detected by PCR and IHC techniques in the current study. Serous or necrotic placentitis suggestive of C. abortus infection was observed in 7 Coxiella-specific qPCR-positive samples (Ct: 30.59–37.23). The frequent occurrence of subclinical C. burnetii infections in ovine abortion cases (33%, 7/21) and a recent Q fever outbreak in Hungary 10 highlight the importance of improvement of hygiene practices in small ruminant flocks in the country. Although limited numbers of goat abortion samples were submitted for examinations (n = 5), the finding of 1 positive sample, detected by qPCR (Ct: 35.3, a value close to the detection limit) in coinfection with C. abortus, is similar to previous results where Q fever was diagnosed in low occurrence (1%, n = 1/75) in caprine abortion cases by IHC in Hungary. 22 Previous observations in other countries showed that Q fever–related abortions are more frequent in goats than in sheep or cattle. 1 In contrast with intensive goat farming countries (such as The Netherlands), the relatively small national goat flock in Hungary (55,000 goats compared to the sheep stock of 1,160,000 animals in 2010; http://mjksz.hu/, in Hungarian) is usually kept in flocks of 1–50 heads per farm, which might explain the low occurrence of C. burnetii in caprine abortion samples in the country.
Chlamydia abortus was detected by qPCR (Ct: 26.61–35.89) and identified by sequencing in 76.2% (n = 16/21) of the ovine samples. Serous and necrotic placentitis were found in 1 and 13 cases, respectively. Chlamydiaceae-specific immunolabeling was detected in all 16 samples. In 1 of the 3 cases where C. burnetii antigens were also found with IHC, several hypertrophic trophoblast cells characteristic for C. burnetii infection were found with histology. From the examined 5 caprine abortion samples, C. abortus was detected in 2 cases (40%, 2/5) with qPCR (Ct: 34.73 and 41.72) and IHC. The high rate of infection with C. abortus in ovine and caprine abortion samples exceeds the previously reported data in the country (46% [113/246] and 17% [17/75] of sheep and goats, respectively), 22 and are similar to observations in Ireland (70% [32/46] were qPCR positive in the ovine aborted placenta samples). 9 Members of the Chlamydiales order, apart from C. abortus, were not detected in ovine or caprine abortion samples in the current study.
In 2 placenta samples from red deer, C. burnetii was detected with qPCR assays (Ct: 34.73 and 35.98), and multifocal necrotic placentitis without the presence of bacteria or fungi was found in 1 of the cases. Uncultured Chlamydiales spp. were identified from 2 roe deer, 1 fallow deer, and 1 red deer, with less than 5 genome copies detected (Ct: 45.88–49.25; Table 1). Previously in Spain, the DNA of C. burnetii was detected in roe deer (5.1%, 4/78), 2 while 15.4% (6/39) and 29.3% (34/116) seropositivity was detected in roe deer and red deer, respectively. 20 According to Chlamydiales spp., in other regions of the continent, 3–20.4% seroprevalences of C. abortus were observed, and 31.9% (31/99) occurrence of Parachlamydia spp. was detected by qPCR in wild ungulates.17,21
In conclusion, the results of the present study show that Q fever is widespread among domestic ruminants in Hungary and, in some cases, this bacterium might be the primary cause of abortions. High rates of infections with both C. burnetii and C. abortus were detected in sheep. Only a few genome copies of W. chondrophila, Parachlamydia spp., and uncultured Chlamydiales spp. were identified in bovine abortion cases, thus their role in the induction of abortions might be negligible. Coxiella burnetii and members of the Chlamydiales order were present sporadically in samples from wild ruminants.
Footnotes
a.
BagMixer 400 VW, InterScience, Saint Nom, France.
b.
QIAamp DNA mini kit, Qiagen GmbH, Hilden, Germany.
c.
Adiavet Cox, AesChemunex Inc., Cranbury, NJ.
d.
ABI 3730xl DNA analyzer, Applied Biosystems Inc., Foster City, CA.
e.
Clone AC-1, Progen GmbH, Heidelberg, Germany.
f.
Kindly provided by Wolfgang Baumgärtner, University of Veterinary Medicine Hanover, Foundation, Hanover, Lower Saxony, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Lendület (Momentum) program (LP2012-22) of the Hungarian Academy of Sciences.
