Abstract
Iodine excess and resultant hyperplastic goiter are well documented in neonatal ruminants, but little is reported on iodine excess in adult ruminants and associated histological changes of the thyroid gland. Two adult Holstein cows from a Michigan dairy herd that had lost several other animals had nonspecific clinical signs of illness and were submitted for necropsy. Thyroid glands of one of these 2 animals were grossly and markedly enlarged, and histologically, thyroid glands from both animals had regions of cystic nodular hyperplasia and follicular atrophy. Thyroid glands from both animals had markedly elevated iodine concentrations. Investigation into the potential source of excessive iodine on the farm revealed multiple sources of supplemental dietary iodine and probable uneven feed and mineral mixing. Based on the findings of this investigation, adult cattle could be susceptible to excessive doses of iodine. Possibility of previous iodine deficiency before supplementation period, with subsequent development and persistence of thyroid hyperplasia and cystic change, cannot be completely excluded. Current findings suggested that iodine excess in adult cattle can result in nodular hyperplastic goiter. Use of iodized salt in mineral supplements in adult dairy herds is common practice, and accidental excessive iodine supplement may be more common than reported. Recognizing gross and histological thyroid gland changes, consisting of concurrent cystic follicular hyperplasia, atrophy, and fibrosis should raise suspicion of iodine excess and/or prior deficiency in a cattle herd, and ancillary tests such as serum iodine measurements should be part of the diagnostic workup in suspected cases.
Iodine is an essential mineral obtained from the diet, and required for thyroid hormone synthesis. Thyroid hormones, triiodothyronine (T3) and thyroxine (T4), are involved in the growth, maturation, and cellular metabolism of animals. Thyroid hormone release is controlled by the pituitary gland that is under negative feedback by T3 and T4. The detrimental effects of iodine deficiency or excess have been well reported in the veterinary literature, and are characterized, classically, by thyroid hyperplasia, growth retardation, and fetal and neonatal death. 12 As such, prophylactic dietary iodine supplement is common on farms set in regions such as Michigan, where the soil is deficient in iodine content. 6 Thyroid hyperplasia, which can result in goiter, is relatively common in newborn calves, but is unusual in adult cattle.
In the present case, 2 adult Holstein cows from a Michigan dairy herd were submitted for necropsy to the Diagnostic Center for Population and Animal Health (DCPAH), Michigan State University (East Lansing, Michigan). Ten animals (out of a herd of 300) died on the farm in the 3 months prior to presentation of these 2 animals for necropsy. Clinical signs of affected animals included low-grade pyrexia, nasal discharge, respiratory distress, and watery stools. The first animal submitted, a 7-year-old, 725-kg Holstein cow, was received 1 week prior to submission of the second, a 4-year-old, 473-kg Holstein cow. Routine necropsy was performed on the animals on the respective days of their submission to DCPAH. At necropsy, the 7-year-old cow had marked bilateral multilobular cystic hyperplasia of the thyroid glands (Fig. 1). The glands each measured approximately 12 cm × 8 cm × 5 cm. Additional gross findings included bronchopneumonia with pleural adhesions, as well as septic peritonitis secondary to focal chronic perforating abomasal ulceration. There were no significant gross lesions found at necropsy in the 4-year-old cow.
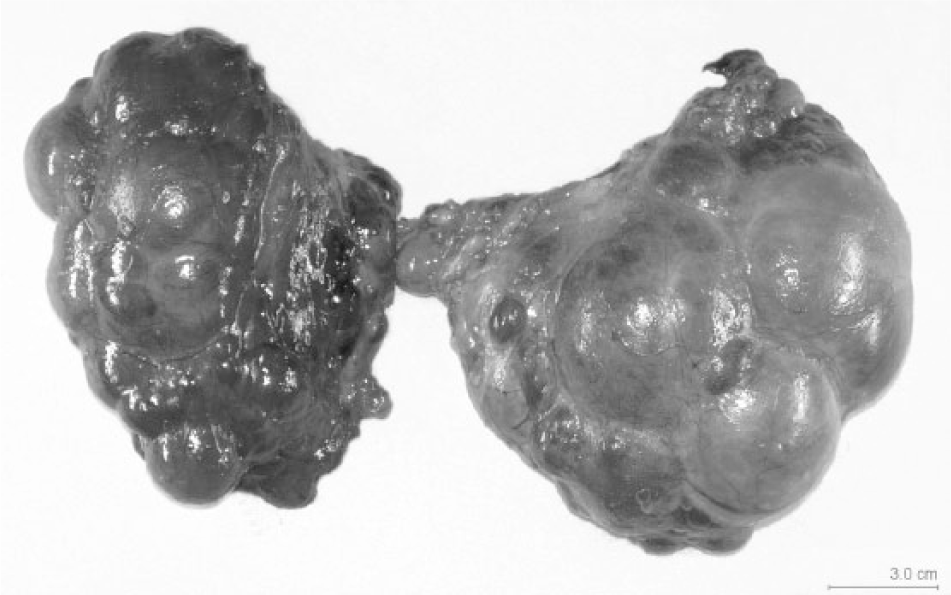
Left and right thyroid glands from the first cow with iodine toxicity. The glands are enlarged, asymmetrically sized and shaped, and consist of multinodular, variably sized fluid-filled cysts. Bar = 3.0 cm.
The thyroid glands and other major organs from the 2 animals were examined histologically. Thyroid glands of the 7-year-old cow had diffuse cystic hyperplasia mixed with follicular atrophy (Fig. 2). The cysts were filled with pale eosinophilic colloidal fluid. There was disruption of normal thyroid follicular architecture, characterized by multiple markedly expansile cystic follicles arranged in lobules, interspersed by adjacent atrophied thyroid follicles, and separation of atrophied and cystic follicles by moderate amount of fibrocollagenous stroma (Figs. 3, 4). The cystic follicles contained abundant amorphous eosinophilic material, consistent with colloid, and were lined by cuboidal to flattened atrophied follicular epithelium further supported by collagen and fibrocytes. Noncystic follicles were variably sized and largely devoid of follicular colloid. C-cells were indistinct throughout the thyroid section. Thyroid glands from the 4-year-old cow were grossly unremarkable, but were characterized histologically by regions of severe atrophy and multifocally extensive hyperplasia, as well as marked fibrocollagenous stromal proliferation. There was an overall reduction of thyroid parenchyma, and remnant thyroid parenchyma was characterized by hyperplastic cuboidal follicular epithelial that formed follicular structures that contained no or scant amounts of colloid. Multiple cystic follicles were also present in the thyroid gland of this cow, and the follicles were lined by flat epithelium and filled with abundant colloid. Fresh-frozen thyroid gland samples from each cow were submitted for iodine analysis. The wet-tissue iodine concentrations from the 7-year-old cow and the 4-year-old cow were 1,065 and 394 μg/g, respectively. The nonhomogeneous nature of the thyroid glands in these cases made the values difficult to interpret, and no attempt was made to differentiate the iodine concentrations in the cystic areas of the glands from the noncystic areas. Reference values for bovine thyroid iodine concentrations are not readily available. Based on available literature, Holstein bulls in one feeding trial that received diets with 4 mg/kg or 10 mg/kg iodine concentrations (well above dietary requirements) had thyroid iodine concentrations averaging 495 and 844 μg/g fresh tissue, respectively. 8 In pigs receiving diets ranging from zero to 5 mg/kg supplemental iodine, thyroid tissue iodine concentrations appeared to plateau at approximately 1,600 μg/g fresh tissue at a dietary iodine concentration of 2 mg/kg. 3 Unsupplemented pigs in that study had mean thyroid iodine concentrations averaging 620 μg/g fresh tissue.
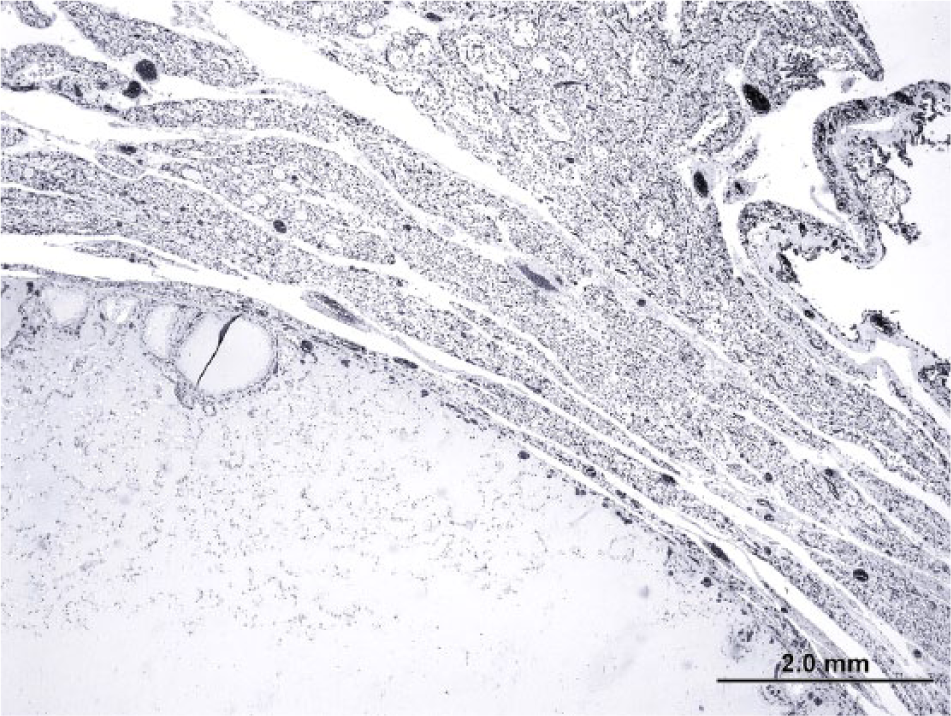
Thyroid gland from the first cow. There is cystic ectasia of a thyroid follicle that compressed surrounding variably atrophied thyroid follicular parenchyma. Ectatic follicles contain abundant colloid. Hematoxylin and eosin. Bar = 2.0 mm.
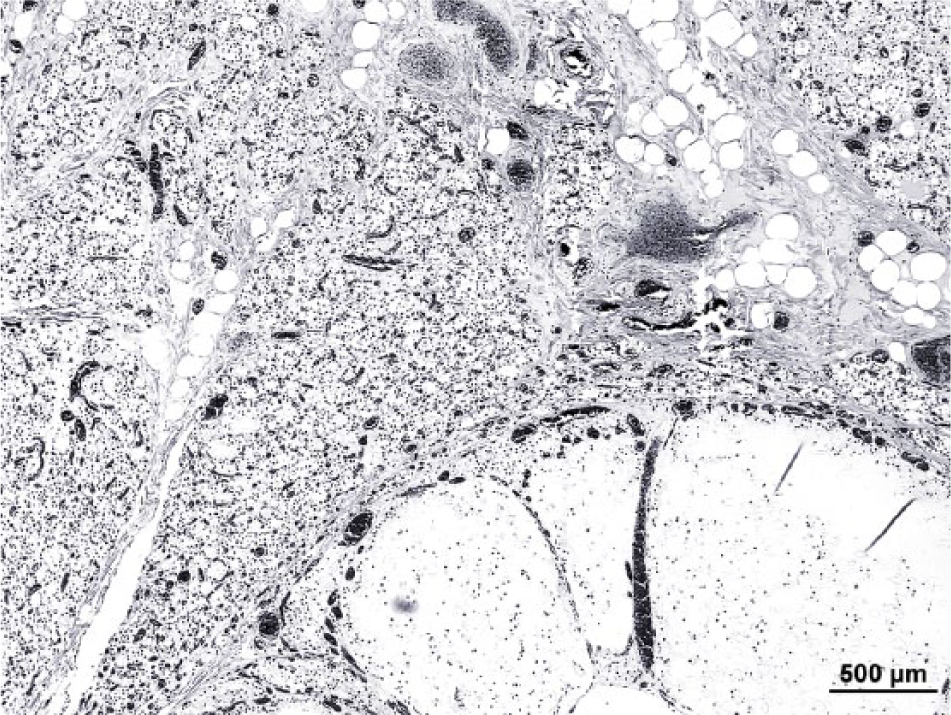
Higher magnification of thyroid gland from the first cow. Extensive fibrocollagenous tissue separated distorted thyroid lobules and individual atrophied thyroid follicles. Ectatic thyroid follicles at the bottom right are congregated, and contain abundant colloid. Hematoxylin and eosin. Bar = 500 μm.
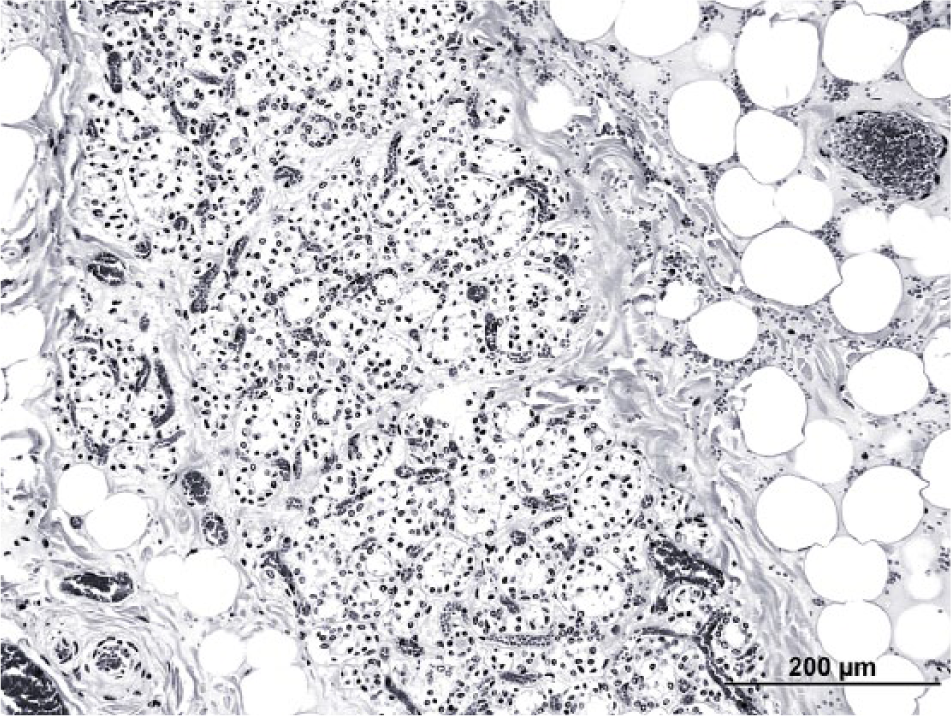
Thyroid gland from the first cow. Atrophied thyroid follicles are surrounded and separated by fibrocollagenous tissue and mature adipocytes. Hematoxylin and eosin. Bar = 200 μm.
Based on these findings, the iodine status of the herd was further investigated. Total mixed rations (TMR) for the dry cows, high-production cows, and low-production cows were analyzed at 1, 3, and 1.8 mg I/kg dry matter, respectively. Using estimated body weights of 600 kg and estimated dry matter intakes of 12 kg/day for dry cows and 25 kg/day for lactating cows, these concentrations would result in daily iodine intakes of 2 mg/100/kg body weight for dry cows and 12.5 and 7.5 mg/100 kg body weight for high- and low-producing cows, respectively. These intake rates are greater than the minimum daily requirement of 0.6 mg/100 kg for nonlactating cows and 1.4 mg/100 kg body weight for lactating cows. 9 However, these estimated iodine intake levels are substantially less than the intakes of 40–220 mg/100 kg that are of demonstrated toxicity in growing calves, 6 and 12–100 mg/100 kg that have been suggested to cause toxicity in adult dairy cattle under field conditions. 11 In these cited studies of iodine toxicity in cattle, however, no incidences of gross or microscopic thyroid gland lesions were mentioned.
In the herd under investigation, in addition to the formulated TMR diets, additional mineral supplement was top dressed on the dry cow ration and trace mineralized salt containing iodine was available to the cows for free choice consumption. Neither quantitative estimates of consumption rates for these supplements, nor their iodine concentrations were available. The dairyman did observe that trace mineralized salt consumption was, in his estimation, high.
Subsequent to the initial necropsy findings, serum samples from 3 clinically normal adult dry cows in the herd were collected at 2 separate occasions. The first set of serum analyses revealed elevated serum iodine concentrations, ranging from 322 to 913 ng/ml (reference range: 50–100 ng/ml), indicating excessive intake of iodine by these animals. The owner elected to stop feeding both the free choice trace mineralized salt and the top-dressed mineral supplement, based on the authors’ recommendations. A second set of serum samples from the same animals, collected approximately 1 week after the first set and after the change in mineral feeding, had serum iodine concentrations of 46, 48, and 71 ng/ml, which were all within the reference range. As milk iodine levels from the herd are of public health concern, 3 milk specimens from the farm were analyzed. Milk iodine concentrations were 359, 423, and 503 ng/ml (reference range: 100–500 ng/ml), which were deemed within acceptable limits by the Michigan State Veterinarian’s office (Lansing, Michigan).
Total thyroxine (TT4) from these 3 animals were 38, 59, and 63 nmol/l, while total triiodothyronine (TT3) were 0.7, 0.9, and 1.1 nmol/l. Free thyroxine (FT4) values ranged between 12–16 pmol/l, and free triiodothyronine (FT3) values ranged between 0.8–1.4 pmol/l from the 3 live animals sampled. In adult cattle, reported adequate serum concentration ranges for T3, T4, FT3, and FT4 are 1.5–2.1, 38–57, 2.0–3.0, and 6.5–11.0 nmol/l, respectively. 5 Based on these values, TT3 values of the 3 animals were below normal limits, 1 out of 3 animals had elevated TT4, and all 3 animals had elevation of FT4 and decreased FT3. The effect of excessive iodine intake on thyroid status varies from hyper- to hypothyroidism, with the variation appearing to be dependent on the functional status of the thyroid prior to the excessive iodine intake.7,13
The pathogenesis of thyroid gland hyperplasia has been well described. This disease can be the result of dietary iodine deficiency or excess, dietary goitrogenic substances that disrupt thyroid hormone synthesis, and genetic thyroid hormone synthesis defect. With excess iodine intake, elevated blood iodine concentrations ensue and the rate of transport of iodine into the thyroid follicles is increased. This increase is initially accompanied by a reduction in iodination of thyroid proteins, 7 which results in decreased circulating T3 and T4 levels, and subsequent increase in thyroid-stimulating hormone secretion by the pituitary gland. Thyroid-stimulating hormone binds to thyroid follicular cells to stimulate thyroid hyperplasia. Excess iodine also inhibits thyroid hormone release by preventing colloid proteolysis in the thyroid gland. Hence, thyroid gland hyperplasia and goiter occur in conditions of both iodine deficiency and excess. 1 Goiter is typically a clinically significant disease of young animals. In adult animals, goiter is seldom reported, and is usually of little clinical importance with limited health impact. 1 In the present case, the clinical and pathological manifestations of excessive iodine intake seen in these adult cows are unusual.
In addition, feed supplementation and factors such as milking management and dietary goitrogens can affect total iodine intake in cattle, which also influence milk iodine levels. A 2011 study showed that in surveyed farms, mineral mixes provide 83% of total iodine intake, and more than 86% of farms tested were supplementing iodine at higher than the recommended level (0.5 I/kg of dry matter). 2 As only a small amount of iodine is required by the body for thyroid hormone synthesis, iodine toxicity is highly plausible from excessive dietary iodine supplementation in cattle. Daily iodine requirement in cattle is very small, approximately 0.016 mg/kg of body weight for adult cattle and 0.02 mg/kg bodyweight for calves. 12 Prolonged ingestion of a diet with high iodine content will result in iodine toxicity with detrimental effect on thyroid gland function (Hillman D, Stowe H, Sleight SD, Whitehair CK: 1979, Iodine nutrition and toxicity in cattle. Extension Bulletin E-1335. Cooperative Extension Service, East Lansing, MI. Available at: http://www.lib.msu.edu/uri-res/N2L/?urn:x-msulib::digital:agext:e1335), although thyroid gland dysfunction appears to occur only after chronic, persistent exposure to high doses of iodine supplementation. 12 In the present case, while it is unclear from the history how long the cows had been exposed to excess iodine intake, histological changes indicative of chronicity include the extensive thyroid gland fibrosis, and variable cystic and hyperplastic glandular architecture distortion.
Toxic doses of iodine in excess of recommended levels of 20–500 times in calves and 7.5–63 times in adult cattle have been reported. 12 Such wide ranges in toxic doses of iodine could be in part due to type and absorbability of iodine supplement, concurrent disease, individual animal feeding behavior, and other factors. 12 In the present case, it is likely that cows in the herd that had access to both the ad libitum salt licks and top-fed mineral supplements became subjects of excessive iodine supplementation. In addition, the reported pica by several individual dry cows likely contributed to excessive consumption of iodized mineral supplements.
Bronchopneumonia in the first cow of the present case could be related to excessive intake of iodine that is associated with pathology of respiratory system, thyroid, and adrenal glands. Clinical signs of iodine toxicity in cattle include hyperthermia, alopecia, scaly and poor hair coats, naso-ocular discharge, conjunctivitis, anorexia, dermatitis, increased opportunistic infections, decreased feed conversion ratio, and decreased milk production. 11 Iodine toxicity is also associated with suppression of both cellular and humoral immunity in cattle, resulting in increased susceptibility to infections such as pneumonia (Hillman D, et al.: 1979, Iodine nutrition and toxicity in cattle).11,12,14 Phagocytosis, antibody response to bacterial agents, and T- and B-cell production can all be decreased in calves given excessive doses of iodine. 4 Clinical signs in calves experimentally fed excessive amount of iodine, at 100 or 200 ppm, include coughing and profuse nasal discharge, as well as decreased weight gain. 10 Lung changes in cattle with iodine toxicity are typically characterized by suppurative, histiocytic, and lymphocytic bronchopneumonia; tracheitis; and trachea squamous metaplasia.6,12 Interestingly, while early exposure to excessive iodine causes an initial period of susceptibility to pneumonia, experimentally exposed calves appeared to be able to compensate for the high levels of iodine and develop diminished signs of toxicity with time. 6 When iodine supplement levels were decreased to standard recommendation levels are corrected away from toxic doses, the signs of iodine toxicity are reversed, 11 further indicating that clinical signs of iodine toxicity are not permanent. While it is not believed that iodine toxicity is directly responsible for the deaths of either of the cattle in the current report, however, the excessive amount of iodine could have contributed to the pneumonia, rumen ulceration, and other conditions that actually led to the cows’ demise.
In the present case, concomitant features of thyroid gland cystic follicular hyperplasia and follicular atrophy with fibrosis are indicative of concurrent follicular stimulation as well as atrophy, likely the result of disrupted thyroid gland function from chronic excessive iodine intake. An alternative explanation and speculation for the thyroid hyperplasia is that the affected cows were in an iodine deficient state prior to period of excessive iodine supplementation. This iodine deficient state could have resulted in chronic, cystic thyroid gland hyperplasia that persisted despite iodine supplementation. In the absence of retrospective data from the affected cows, unfortunately, this remains a speculation.
Interestingly, serum iodine levels sampled from 3 cows within the herd returned to normal levels in a single week following the removal of all supplemental iodine sources. This suggests that serum iodine levels can exhibit rapid fluctuation following correction of dietary iodine content. A single measurement of serum iodine concentration has value in assessing the current state of iodine level in a herd at a single time point, but serial sampling will be even more useful in investigation of suspected iodine toxicity.
Serum levels of T4, FT4, T3, and FT3 in a cattle herd have been shown to be increased in hyperthyroidism and decreased in hypothyroidism. 15 The free (non–protein-bound) fractions are biologically active and a better measure of thyroid gland functions. Triiodothyronine is the active thyroid hormone and is derived from direct thyroid gland production and conversion from T4, 15 and via the action of 5′-deiodinase enzymes outside of the thyroid gland. In the present case, FT4 values of the 3 animals sampled from the herd were increased while FT3 values were decreased, suggesting reduced conversion of T4 to T3. Based on histological evidence of thyroid gland atrophy and distortion, thyroid gland dysfunction was at least in part responsible for disruption of thyroid hormone synthesis and release, and hence decreased FT3 levels in the sampled serum.
In summary, histological changes of the thyroid gland characterized by regions of cystic follicular hyperplasia, as well as follicular atrophy and fibrosis, could be associated with chronic iodine excess (and/or possibly past iodine deficiency) even in adult cows. Because feed iodine intake of the cattle affects iodine content of milk and meat destined for human consumption, 14 recognition of thyroid gland lesions in adult cattle should prompt investigation into possibly excessive iodine supplementation or deficiency in a herd. Such diagnostic workup should also include ancillary tests such as serum iodine measurements in the herd animals, as described in the current report.
Footnotes
Acknowledgements
The authors would like to thank Tom Wood and the Histology Section of the Diagnostic Center for Population and Animal Health (DCPAH) for technical assistance processing the slides of the cases in this report. The authors also thank Dr. Tim Lyons, Dr. Daron Jones, Dr. James Averill, and Ron VanAlstine for their contributions to this case.
Declaration of conflicting interests
The author(s) declare that they do not have any conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The author(s) declare they received no financial support for their research and/or authorship of this article.
