Abstract
The current article describes outbreaks of vesicular stomatitis (VS) in horses and cattle in Paraiba and Rio Grande do Norte states, northeastern Brazil, between June and August 2013. The reported cases affected 15–20 horses and 6 cattle distributed over 6 small farms in 4 municipalities, but additional data indicated the involvement of a large number of animals on several farms. The disease was characterized by blisters; eruptive lesions in coronary bands, lips, mouth, and muzzle; salivation; claudication and loss of condition. Swollen lower limbs and lips, and ulcerated and erosive areas in the lips and muzzle were observed in some horses. A necrotizing vesiculopustular dermatitis and stomatitis was observed histologically. Vesicular stomatitis virus was isolated from the vesicular fluid of a horse lesion and shown to be serologically related to the VS Indiana serogroup (VSIV) by virus neutralization. Convalescent sera of affected horses and cattle, and from healthy contacts, harbored high levels of neutralizing antibodies against the isolated virus (named VSIV-3 2013SaoBento/ParaibaE). Genomic sequences of VSIV subtype 3 (Vesicular stomatitis Alagoas virus) were amplified by reverse transcription polymerase chain reaction out of clinical specimens from a cow and a horse from different farms. Nucleotide sequencing and phylogenetic analysis of the phosphoprotein gene indicated that the 2 isolates were derived from the same virus and clustered them in VSIV-3, along with VS viruses identified in southeastern and northeastern Brazil in the last decades. Thus, the present report demonstrates the circulation of VSIV-3 in northeastern Brazil and urges for more effective diagnosis and surveillance.
Vesicular stomatitis (VS) is a viral disease of cattle, pigs, horses, and other mammals and may occasionally affect human beings. 13 In cattle and swine, VS is clinically very similar to foot-and-mouth disease (FMD) and, as such, should call for prompt differential diagnosis in FMD-free areas.6,13 The etiological agent of VS (Vesicular stomatitis virus [VSV]; order Mononegavirales, family Rhabdoviridae, genus Vesiculovirus) is classified into 2 main serogroups: New Jersey (VSNJV) and Indiana (VSIV).5,9 Vesicular stomatitis virus infection is endemic in northern South America, Central America, and southern Mexico, and sporadic outbreaks have been reported in central-northern Mexico and southwestern United States.1,7–10 In Brazil and, occasionally, in Argentina, outbreaks of VS have been associated with viruses serologically related to VSIV. These viruses have been tentatively classified as VSIV subtypes 2 or 3, from which Cocal virus (COCV) and Vesicular stomatitis Alagoas virus (VSAV), respectively, are the prototypes.3,11 Historically, VSIV-2 has been associated with cases or outbreaks of VS in several Brazilian regions (1966, 1979) and, in 1998, it was isolated in a VS outbreak in cattle in 2 southern states.2,12 A related virus was isolated in outbreaks in Argentina in 1963 and 1978.1,2 In contrast, VS outbreaks in northeastern Brazil (http://www.panaftosa.org.br/) are predominantly associated with VSIV-3.2,12
In the past 10 years, a number of cases of vesicular disease (i.e., VS or FMD suspect) were reported in South America, especially in northeastern and northern Brazil. On official investigation, however, many of these suspected cases turned out to be associated with other causes (http://www.panaftosa.org.br/). Regardless, a number of cases in horses and cattle in Paraíba (PB), Rio Grande do Norte (RN), and Ceará (CE) states were definitively associated with VSIV-like viruses (http://www.panaftosa.org.br/). In addition, data from 2005 to the present indicate that VSV infection is recurrent in Brazilian territory, with cases limited to 1 or more regions and affecting domestic and/or wild animals (http://www.oie.int/).
Thus, the objective of the current report is to describe outbreaks of vesicular disease associated with VSIV-3 infection in horses and cattle in northeastern Brazilian states in June–August 2013. Cases of vesicular disease were investigated in horses and cattle from 6 small farms in 4 counties of PB and RN (Fig. 1).
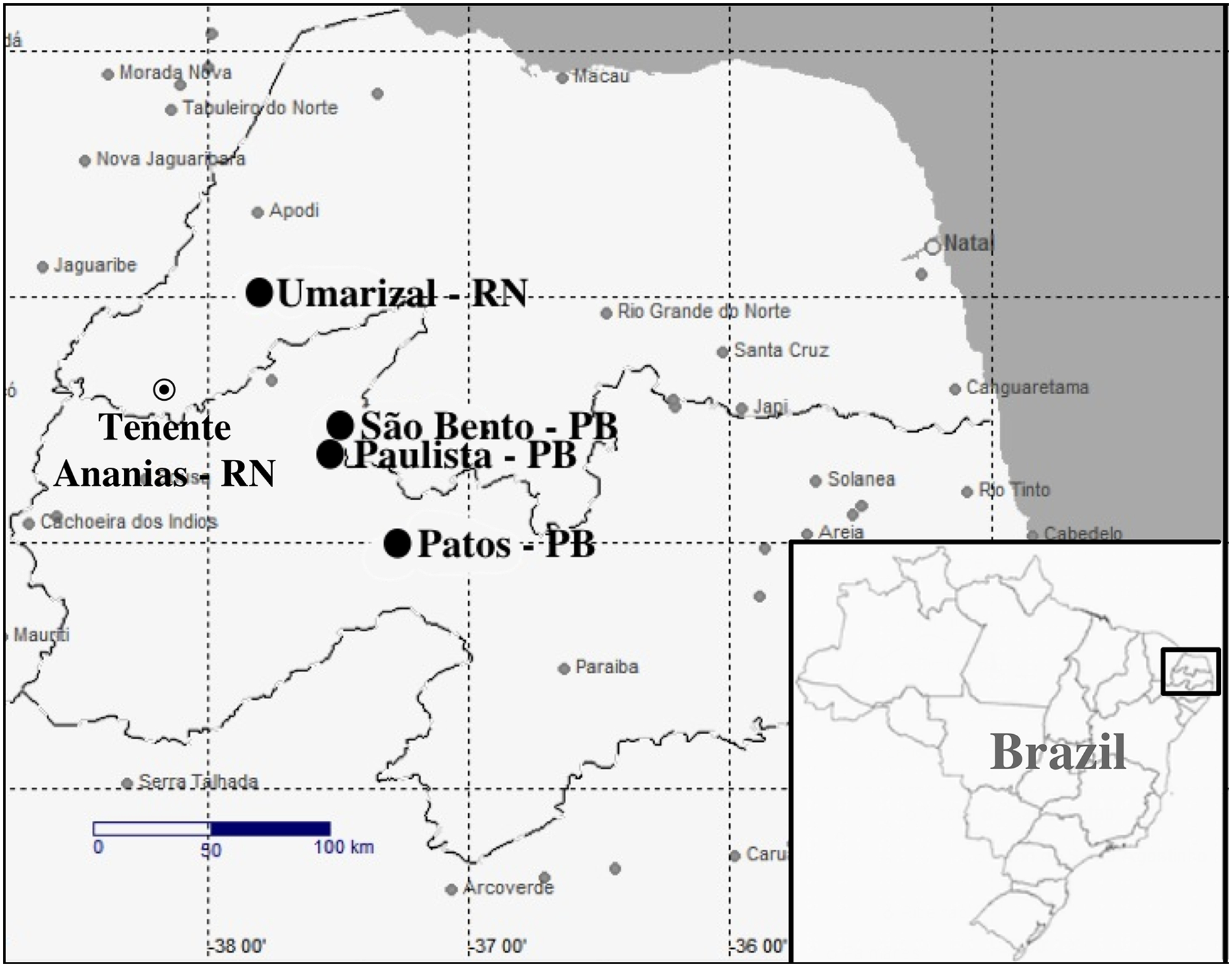
Location of outbreaks of Vesicular stomatitis virus (VSV) in northeastern Brazil. The highlighted figure shows the location of the herds where the VSV outbreaks occurred.
During June–August 2013, cases of vesicular disease affecting horses and cattle were reported to the Veterinary Hospital of the Federal University of Campina Grande, PB, Brazil. The reports and visits were performed according to the following timeline.
Outbreak 1 (Paulista, PB, visited on June 8; S 6°32′23.55″, W 37°30′16.21″; altitude of 154 m). The owner reported the occurrence of vesicular disease in 3 out of approximately 100 cattle raised in the herd. According to the owner, an adult cow, a 14-month-old heifer, and a 7-month old female calf presented claudication, vesicular and exudative lesions on the coronary bands, ptyalism, and blisters in the mouth, starting approximately 10–20 days prior to the visit. On examination, a healing lesion on the tongue, detachment of the tongue and lip epithelia, and exudative lesions in the coronary bands were observed in the 3 animals. Sera for serology were obtained from convalescent animals (bovines 1–3) and from a contact, healthy cow (bovine 4).
Outbreak 2 (São Bento, PB, visited on June 15; S 6°0′27.00″, W 37°49′4.60″; altitude of 141 m). The small farm housed 15 horses of different breeds and ages. Approximately 20 days earlier, some animals presented vesicular and exudative lesions in the coronary bands or mouth and claudication. On examination, the lesions in 2 horses were still evident, which consisted of blisters (0.3–0.5 cm in diameter) and ulcers distributed around the coronary band and gingivae. In 1 horse, the lesions were almost healed. Vesicular fluid, tissue fragments, and sera for serology were collected from the sick horses (equines 1 and 2).
Outbreak 3 (Umarizal, RN, visited on June 27; S 5°59′42.70″, W 37°48′49.40″; altitude of 155 m). The farm had only 2 horses, 1 of which (equine 3) presented swollen and hyperemic lips, blisters on the face (Fig. 2A), lips, and labial commissure, and a large (7 cm) yellowish and elevated area delimitated by a reddish margin in the internal face of the cheek. According to the owner, horse 4 presented similar lesions and clinical evolution, yet the lesions were noticeably milder. The 2 horses attended a regional rodeo (vaquejada) in Tenente Ananias County (close to the PB border) approximately 20 days before, in which a number of horses presented claudication and blistery lesions on their feet. Thus, it is reasonable to speculate that this event, which gathered a number of horses of different origins, was probably the origin of these reported cases.
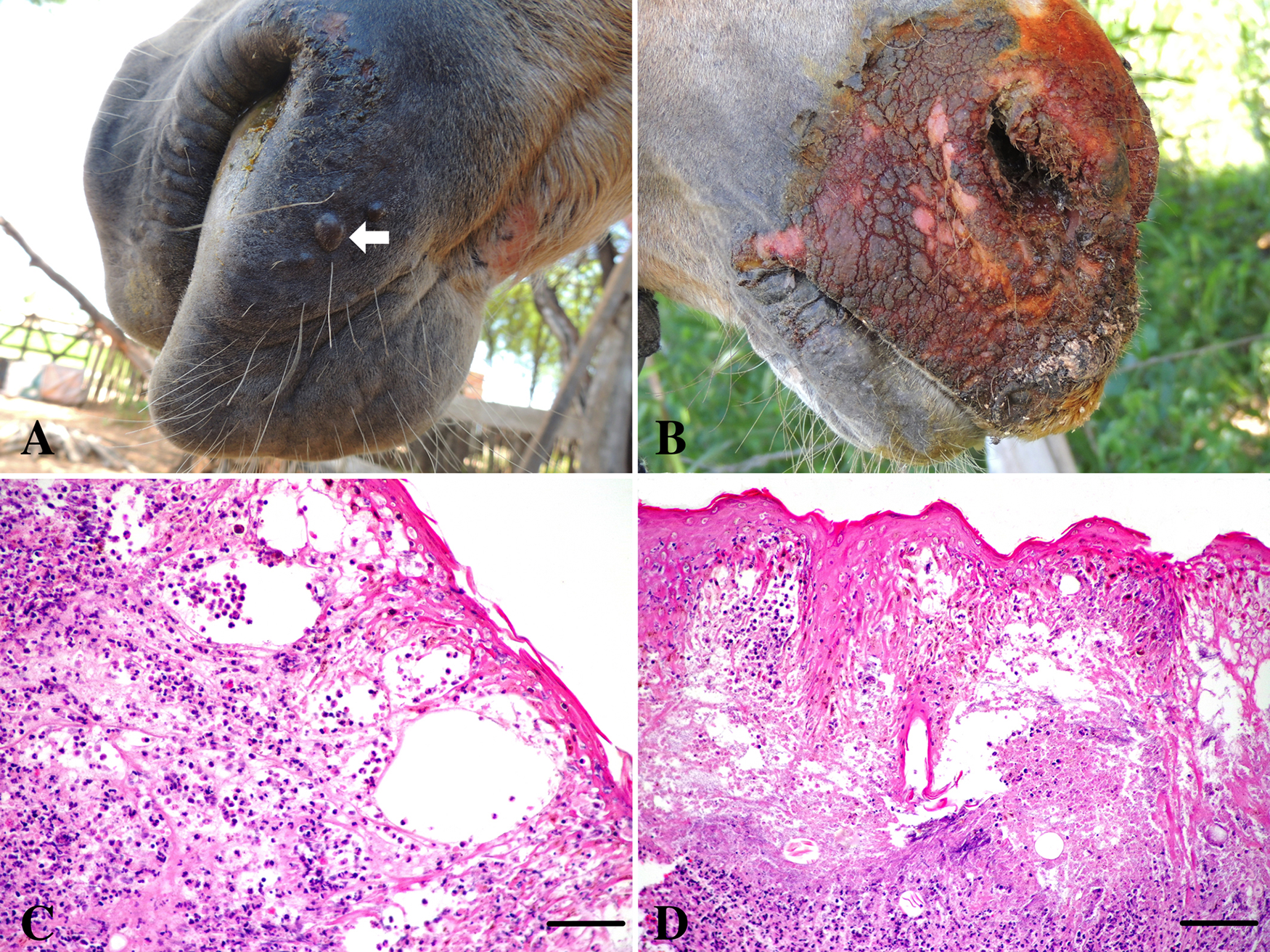
Vesicular stomatitis in horses.
Outbreak 4 (Umarizal, RN, visited on June 27; S 5°59′44.94″, W 37°48′50.34″; altitude of 155 m). One of 2 horses on the farm (equine 5) developed blisters in coronary bands of all limbs, with an evolution of approximately 24–48 hr. Examination revealed ruptured and healing blisters and pronounced swelling of the lower hind limbs. Donkeys and mules present on the farm were not affected. Sera for serology was collected from horses 5 (sick) and 6 (healthy contact).
Outbreak 5 (Umarizal, RN, visited on June 27; S 6°0′26.80″, W 37°49′4.60″; altitude of 155 m). The farm housed 14 horses and 22 cattle (8 cows, 8 calves, and 6 heifers), from which 8 horses and 3 cows were affected. The disease initially affected a cow, approximately 20 days prior to the visit, followed by the development of lesions on the foot and mouth of a horse approximately 5 days later. Two cows developed vesicles and blister lesions on their feet (coronary bands, interdigital space) and another cow presented vesicular and/or pustular lesions in the teats and udder. The affected horses presented claudication, blisters in the coronary bands, and vesicles and erosions in the mouth. Horse 7 presented severe erosive lesions in the skin of the superior lip and face, swollen lips, and palpebrae (Fig. 2B). On examination, horses 8–10 presented healing lesions and depigmented patches on the lips, labial commissures, and/or in the tongue. These animals underwent a clinical course of approximately 8 days. The index case (a mare) presented a healing lesion (depigmented patch) in the muzzle, near the nostril. Horse 12 was a healthy contact. Sera for serology were collected from the cows (bovines 5–7) and horses (equines 7–12).
Outbreak 6 (Patos, PB, animals submitted to Veterinary Hospital on August 2, 2013). On examination, a horse (male, 7 years old) and a mare (4 years old) from a farm (S 7°4′4.30″, W 37°16′49.90″, and altitude of 221 m) in the same municipality presented healing, ulcerative and scabby lesions in the oral and nasal cavities. According to the owner, the lesions appeared approximately 5–7 days earlier and affected other horses on the farm. Sera for serology were taken from the 2 examined horses (equines 13 and 14).
Tissue fragments obtained by incisional biopsy of the oral mucocutaneous junction (a cow and a horse) and coronary edge of the hull (a horse) were fixed in 10% buffered formalin, embedded in paraffin, cut in 4.0-μm sections, and stained by hematoxylin and eosin. Histologically, there was a necrotizing vesiculopustular stomatitis and dermatitis with multiple intraepidermal pustules, and multifocal to focally extensive areas of necrosis, extending from the basal cells to the granulosum extract (Fig. 2C, 2D). Neutrophils, many of which were degenerated, hemorrhages, and fibrin deposition between keratinocytes were observed in the necrotic areas. Spongiosis, acanthosis, and necrosis of keratinocytes of the granulosus and spinosus extracts were also observed in the epidermis. In the dermis, there was moderate to accentuated infiltration by neutrophils and macrophages (Fig. 2C). Necrosis of epithelial cells was observed in many hair follicles. In the horse, a severe fibroplasia and intense neovascularization characteristic of granulation tissue was observed.
Among the collected material, 4 specimens (vesicular fluid of 2 horses, outbreaks 2 and 3) and detached epithelium and scabs of bovines 2 and 3 (outbreak 1) were stored frozen and sent to the Virology Section, Federal University of Santa Maria, state of Rio Grande do Sul. Initially, the clinical specimens were submitted to virus isolation. For this, tissues were grinded with sterile sand, resuspended in minimal essential medium (10% w/v), and centrifuged; the supernatants were inoculated onto monolayers of Vero (African green monkey kidney) cells. Vesicular fluid diluted in phosphate buffered saline was submitted to low speed centrifugation to remove debris and then inoculated onto cell cultures. The material was submitted to 3 passages of 5 days each before considering it negative for virus. Cell monolayers inoculated with vesicular fluid of horse 1 (outbreak 2) developed cytopathic effect at the first passage, characterized by formation of foci of cell rounding, cell detachment and lysis, and rapid dissemination in culture. Monolayers were totally destroyed within 24–36 hr after the initial visualization of cytopathic effect. To confirm the presence of infectivity, 10-fold dilutions of the supernatant of positive cultures were prepared and inoculated onto fresh Vero monolayers, confirming the presence of an infectious, cytolytic agent. Virus identification was performed by submitting the isolate to virus neutralization (VN) assays, against VSIV-1 and VSNJV antisera produced in rabbits. Briefly, approximately 300–400 TCID50 of the isolate (grown out of the vesicular fluid of horse 1, outbreak 2) was incubated with 2-fold dilutions of the respective antisera for 1 hr. The plates were then added to a suspension of Vero cells and incubated for 48 hr at 37
To confirm the identity of the isolate, clinical specimens were submitted to a reverse transcription polymerase chain reaction (RT-PCR) able to differentiate between VSIV-2 and VSIV-3. 12 Total RNA was extracted from fluid or tissue samples (vesicular fluid, skin fragments) and from the supernatant of Vero cells inoculated with the isolate using TRIzol reagent. a Extracted RNA was submitted to a RT-PCR for VSV using the primers and conditions described previously. 12 For this, 2 different set of primers were used: one designed to amplify VSIV-2 and another that amplifies VSIV-3. This RT-PCR amplifies a fragment of the hypervariable region of phosphoprotein (P) gene. On RT-PCR amplification, a band of approximately 565 bp, corresponding to the expected amplicon for VSIV-3, was detected in DNA extracted from cells inoculated with the isolate in clinical specimens of cow 3 (outbreak 1) and horse 1 (outbreak 2). As this RT-PCR is designed to specifically amplify VSIV-3 sequences, and not VSIV-1, VSIV-2, or VSNJV, these results confirmed the identity of the isolate as VSIV-3. The isolated virus will be thereafter called VSIV-3 2013SaoBento/ParaibaE (“E” stands for equine), following the nomenclature used for VSV. The other virus, whose genomic sequences were amplified by RT-PCR, will be called VSIV-3 2013Paulista/ParaibaB (“B” stands for bovine).
To further characterize the isolateVSIV-3 2013SaoBento/ParaibaE (and VSIV-3 2013Paulista/ParaibaB), the amplicons were purified using a gel extraction and PCR purification combination kit b and submitted to nucleotide sequencing. The samples were sequenced in quadruplicates in an automatic sequencer. c The obtained sequences were analyzed by the program Staden 14 to obtain the consensus sequences. Alignment and identity matrices were performed using BioEdit version 7.0.5.3 (http://www.mbio.ncsu.edu/BioEdit/bioedit.html). The alignments of VSV sequences deposited in GenBank (VSIV-3 2013Paulista/ParaibaB: no. KJ746668; VSIV-3 2013SaoBento/ParaibaE: no. KJ746669) and the samples from the current study were used to construct a phylogenetic tree based on the P gene sequence, using the maximum likelihood method with 1,000 bootstrap replicates implemented by MEGA5.0, based on Tamura-3 parameters model. 15
The sequences obtained from VSIV-3 2013SaoBento/ParaibaE (horse 1, outbreak 2) and VSIV-3 2013Paulista/ParaibaB (bovine 3, outbreak 1) clustered together with VSIV-3 and other Brazilian VSIV-3 isolates from the 1970–1990s (Fig. 3). The nucleotide identity of the obtained sequences was 94% with Brazilian VSIV-3 strains (100% coverage), and was significantly lower with other VSV serogroups. The findings confirm that both viruses belong to VSIV-3 and are closely related to Brazilian VSIV-3 isolates identified over a long period of time. In addition, the 2 viruses (VSIV-3 2013SaoBento/ParaibaE and VSIV-3 2013Paulista/ParaibaB) presented an identity of 100% in the sequenced genome fragment, indicating they are likely (derived from) the same virus that was circulating in the region.
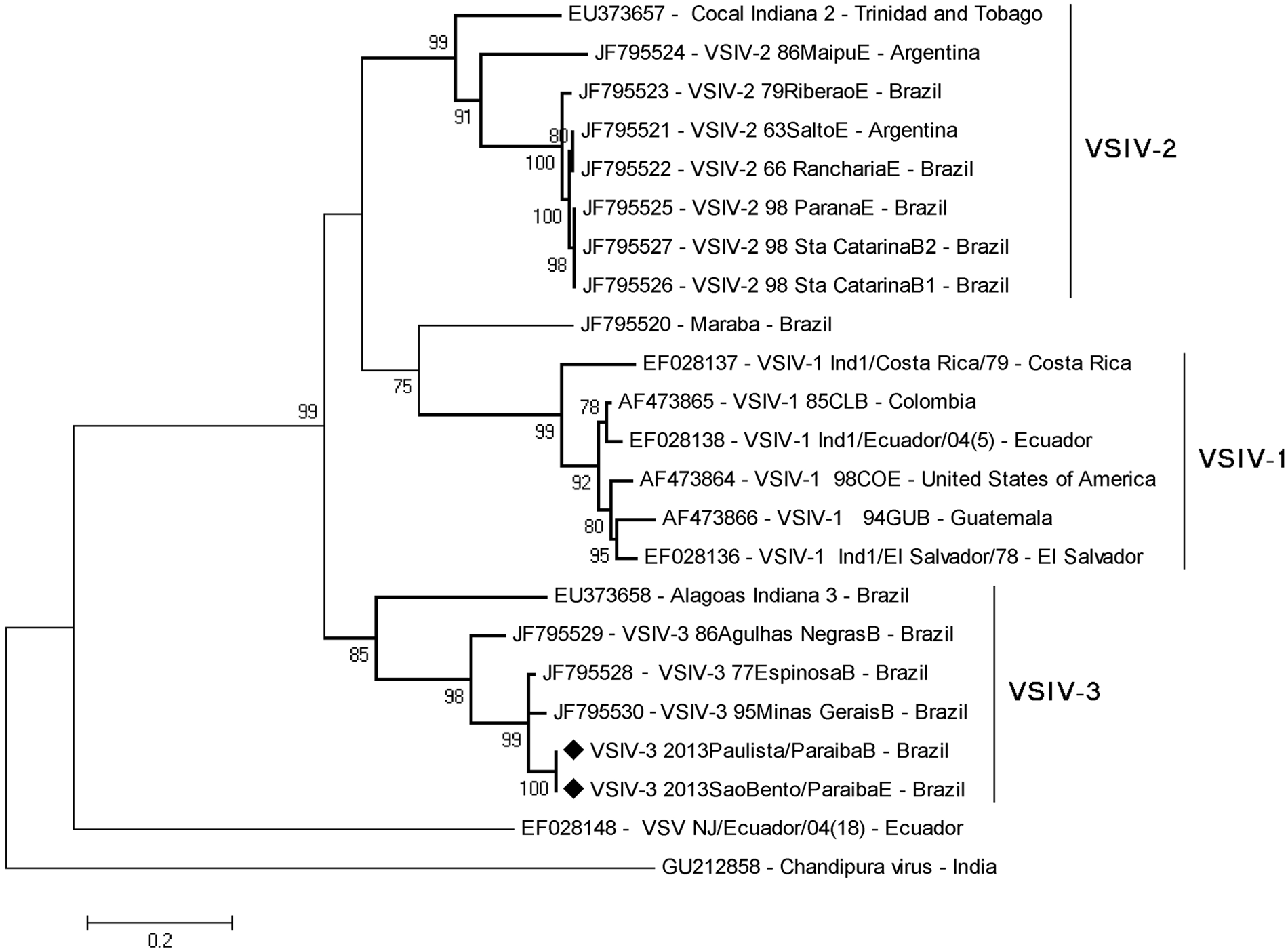
Phylogenetic tree based on the nucleotide sequences of phosphoprotein gene (P) of Vesicular stomatitis virus (VSV). The tree was constructed using the maximum likelihood method with 1,000 bootstrap replicates based on Tamura-3 parameters model and implemented by MEGA 5.0. Values >70% are shown. The Vesicular stomatitis Indiana virus subtype 3 (VSIV-3; Vesicular stomatitis Alagoas virus prototype) samples identified in the present report are identified with a black diamond.
As VSIV-3 was identified in specimens from 2 outbreaks, the participation of the same or related agent in the other described outbreaks was investigated. Unfortunately, specimens for direct diagnosis (tissues, scabs, fluid) from these cases were not collected, were insufficient, or were inappropriate for virus isolation and/or RT-PCR. Serum samples collected from convalescent horses and cattle, and from some healthy contact animals, in the affected herds were submitted to VN assays, as described above, against VSIV-1, VSNJV, and VSIV-3 2013SaoBento/ParaibaE. The results clearly showed a previous exposure to a virus closely related to VSIV-3 2013SaoBento/ParaibaE, reflected in VN titers, in all 6 affected farms (Table 1). Fifteen sera also reacted, in lower titers, with VSIV-1, reflecting the serological relatedness between VSIV subtypes 1 and 3. 11 Considering the clinical data, time and geographical clustering of cases, these results indicate that, even in the absence of direct diagnosis or paired sera testing, these other outbreaks investigated were also possibly associated with infection by VSIV-3 or by a closely related virus.
Neutralizing antibodies against different Vesicular stomatitis virus serotypes in serum samples collected from affected animals and contacts in outbreaks occurring in northeastern Brazil, June–August 2013.*
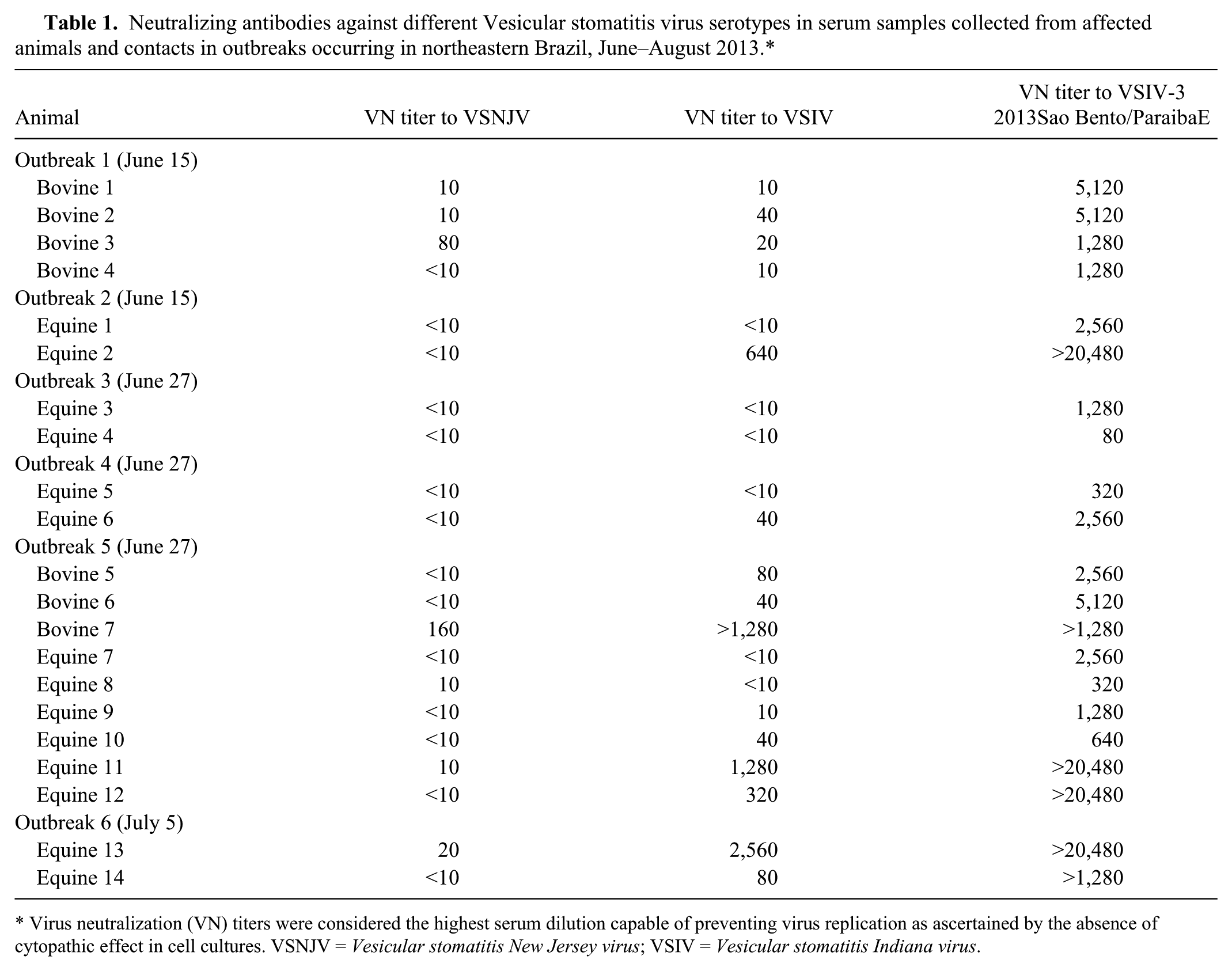
Virus neutralization (VN) titers were considered the highest serum dilution capable of preventing virus replication as ascertained by the absence of cytopathic effect in cell cultures. VSNJV = Vesicular stomatitis New Jersey virus; VSIV = Vesicular stomatitis Indiana virus.
Vesicular stomatitis virus infection is considered endemic in northeastern and northern Brazil (http://www.paho.org/panaftosa/). Nevertheless, reports of cases and/or outbreaks are practically absent from the scientific literature and, as such, this information is inaccessible to the general public. According to the official reports, a number of suspected and/or investigated cases end up being attributed to other infectious or noninfectious causes (http://www.paho.org/panaftosa/). On the other hand, undernotification of VSV probably also occurs, contributing to an incomplete knowledge of the real epidemiological situation. In this sense, the current report contributes to the overall knowledge on the VSV situation in northeastern Brazil, by providing compelling evidence of VSIV-3 circulation in horses and cattle in at least 2 states and calling attention to the need for more effective diagnosis and surveillance. It should be mentioned that from May 4 to August 2, 2013, several VS cases were officially reported in herds from PB and from other northeastern states (CE and Piauí) as well. However, it is reasonable to speculate that, due to the proximity and time frame, some of these cases and outbreaks could be related to the cases described in the present report. Finally, it is worth noting that virology diagnostics and conclusive etiological diagnosis (i.e., virology, molecular, and serology) of the present cases were only achieved a posteriori, months after the outbreaks clinical signs had ceased.
Footnotes
a.
TRIzol reagent (Ambion), Life Technologies, Grand Island, NY.
b.
PureLink quick gel extraction and PCR purification combo kit (Invitrogen), Life Technologies, Grand Island, NY.
c.
ABI-PRISM 3100 genetic analyzer (Applied Biosystems), Life Technologies, Grand Island, NY.
Declaration of conflicting interests
The author(s) declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JF Cargnelutti, EF Flores, R Weiblen, and F Riet-Correa are Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) research fellows. LA Maia and GMN de Aguiar are recipients of student fellowship from the Brazilian Federal Agency for the Support and Evaluation of Graduate Education (CAPES).
