Abstract
Vesicular diseases are of high importance for livestock, primarily because of foot-and-mouth disease (FMD), which is a high-morbidity disease that generates direct losses caused by low milk production, weight loss, and indirect losses because of the need for sanitary barriers. Other vesicular diseases are also of importance for livestock because of direct impacts or because their clinical signs may be confused with those of FMD. We report herein the detection of multiple infections in cattle with suspected vesicular disease in the Brazilian states of Amazonas (AM), Mato Grosso (MT), and Roraima. Thirty-seven epithelial samples from cattle and 1 sample from a buffalo were sent to the laboratory for testing for FMDV and similar disease agents. All samples from MT were positive for parapoxvirus (Pseudocowpox virus and Bovine papular stomatitis virus). In addition, 3 samples were positive for Bluetongue virus, and 5 samples were positive for Bovine herpesvirus 1. Among these samples, 1 was positive for all of these 3 agents. Only 2 samples from AM were negative for parapoxvirus. The molecular tests conducted in this study detected multiple infections, with a high prevalence of parapoxvirus.
Introduction
Cattle vesicular diseases are of high importance for livestock, primarily because of foot-and-mouth disease caused by Foot-and-mouth disease virus (FMDV; order Picornavirales, family Picornaviridae, genus Aphthovirus). FMD is a high-morbidity disease that generates direct losses caused by low milk production, weight loss, and indirect losses caused by the need for sanitary barriers. 14 Strategies are needed to prevent the emergence of outbreaks, such as active surveillance, vaccination, and strict sanitary control. 21 For these reasons, FMD is on the notifiable diseases list of the World Organization for Animal Health. 35
Other vesicular diseases are also of importance for livestock because of direct impacts or because their clinical signs may be confused with those of FMD. Among the diseases with viral etiologic agents, the following RNA viruses can generate vesicular signs: vesicular stomatitis viruses (VSVs; Vesicular stomatitis Alagoas virus, Vesicular stomatitis Indiana virus, Vesicular stomatitis New Jersey virus, and Cocal virus), Bovine viral diarrhea virus 1 and 2 (BVDV-1, -2), and Bluetongue virus (BTV). 17 Some DNA viruses, such as Vaccinia virus (VACV), Bovine papular stomatitis virus (BPSV), Pseudocowpox virus (PCPV), Bovine herpesvirus 1 and 2 (BoHV-1, -2), and Ovine herpesvirus 2 (OvHV-2), are also of importance for differential diagnosis.27,34 In 1992, the Brazilian Ministry of Agriculture, Livestock and Supply (MAPA) established the National Program for the Eradication of Foot and Mouth Disease (http://goo.gl/XdJ8bl). Since that time, the laboratory structure has been expanded to protect animals from FMD and diseases with similar clinical signs. Any vesicular disease suspects are currently sent to one of the official laboratories of MAPA (National Agricultural Laboratory of Minas Gerais [Lanagro/MG]) for appropriate testing.
Disease control is directly associated with government actions, so MAPA works closely with all Brazilian states to detect any suspect FMD. Speed and efficiency is important to eradicate any disease, for example the eradication of African swine fever in Brazil in 1978. 23 Molecular tests are routinely used in Brazil to detect FMD and to quickly, safely, and correctly determine which agent caused the vesicular disease.
We report herein the detection of multiple infections in cattle with suspected vesicular disease in the Brazilian states of Amazonas (AM), Mato Grosso (MT), and Roraima (RR). Molecular tests were conducted to detect various viral agents and multiple infections, revealing a high prevalence of parapoxvirus (PPV), including the report of a virus from this genus in a buffalo in Brazil.
Materials and methods
Samples
MT samples originated from a property that was located in Mirassol d’Oeste and had 289 cattle; of these cattle, 21 had lesions suggestive of vesicular disease. The 21 cattle were 5–12 months of age. This property acquired 303 cattle from the state of MG; 18 of these animals died during transit. The lesions were characterized by ulcers and desquamated epithelium with a rounded shape and sharp edges. Some animals exhibited widespread peeling of the tongue epithelium, and 6 of the animals had a body temperature >41°C. Twenty-one serum samples and 21 epithelial samples were sent to Lanagro/MG. Seventeen cattle serum samples, 7 cattle epithelium samples, and a serum sample and an epithelium sample from a buffalo were received from the AM municipalities of Parintins, Barreirinha, and Urucurituba, and the RR municipality of Alto Alegre.
Virus isolation, ELISA for antigen detection, and serologic tests
Approximately 2 g of epithelium were macerated with a mortar and pestle, then clarified and subjected to virus isolation. 36 One milliliter of the clarified suspension was inoculated into cultures of Vero (African green monkey kidney) and BHK-21 (baby hamster kidney) cells, respectively. The cultures were examined for 48–72 h to observe the development of cytopathic effects. If no such effects were observed, the cells were frozen, thawed, and used to obtain passage 2 cells, as described above. Clarified suspensions of the samples were used to test for antigens of FMDV and VSV by sandwich indirect enzyme-linked immunosorbent assay (ELISA). 4 The serum samples were analyzed for the presence of antibodies to VSV (specifically, Alagoas, Indiana, New Jersey, and Cocal) 36 and for the detection of nonstructural virus antibodies against FMDV, according to the manufacturer’s recommendations. a
Molecular analysis
RNA was extracted using TRIzol according to the manufacturer’s instructions. b DNA extract was produced with a commercial extraction kit. c Both processes were accompanied by a control that contained no genetic material for the evaluation of contamination.
The preliminary test for detecting pathogens was a test for the presence of FMDV, 8 and the genetic material extraction was evaluated by detecting the gene or messenger RNA of beta-actin. 7 Negative results allowed us to continue the work with lower biosecurity when handling samples during searches for other agents. The differential diagnosis process continued with the following molecular tests: BVDV,6,25 VSV (protocol used for detection subtypes Alagoas, Indiana, New Jersey, and Cocal), 29 OvHV-2 and BTV,18,30 orthopoxvirus (OPV), 13 PPV, 37 and BoHV-1. 33 All PCR assays were accompanied by blank controls to evaluate contamination.
Samples positive for PPV after PCR were subjected to nested PCR, 2 and the amplicons were sent for Sanger sequencing using an automatic sequencer. d Electropherograms were analyzed using the BioEdit software. 15 The sequences were submitted to BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) for identification. 5 Phylogenetic relationships to other viral sequences in GenBank3,9,10,16 were analyzed using MEGA 6.02. 32
Results
Virus isolation and serologic tests
Virus isolation was negative for all samples. All samples were also negative for the presence of antigens or antibodies to FMDV and VSV, as indicated by ELISA.
Molecular analysis
Nine samples from MT and all samples from RR states were positive for PPV; in addition, 3 samples from MT were positive for BTV, and 5 samples were positive for BoHV-1. Among these samples, 1 sample was positive for all 3 agents. Only 2 samples from AM were negative for PPV.
The results for BTV were confirmed using 2 different quantitative real-time PCRs; however, low amounts of virus did not allow for the isolation or identification of serotypes. Positive results for PPV were confirmed by sequencing, and BLAST was performed to determine the species. The results indicated the presence of PCPV, with the following exceptions: 3 samples from Mirassol d’Oeste in MT and 1 sample from Barreirinha in AM were positive for BPSV.
The phylogenetic tree showed a clear separation between PCPV and BPSV (Fig. 1). The PCPV described in our work clustered with other samples from Brazil detected in the state of Rondônia (RO) in 2012. BPSV did not cluster with other Brazilian samples but did cluster with a sample from Bangladesh.
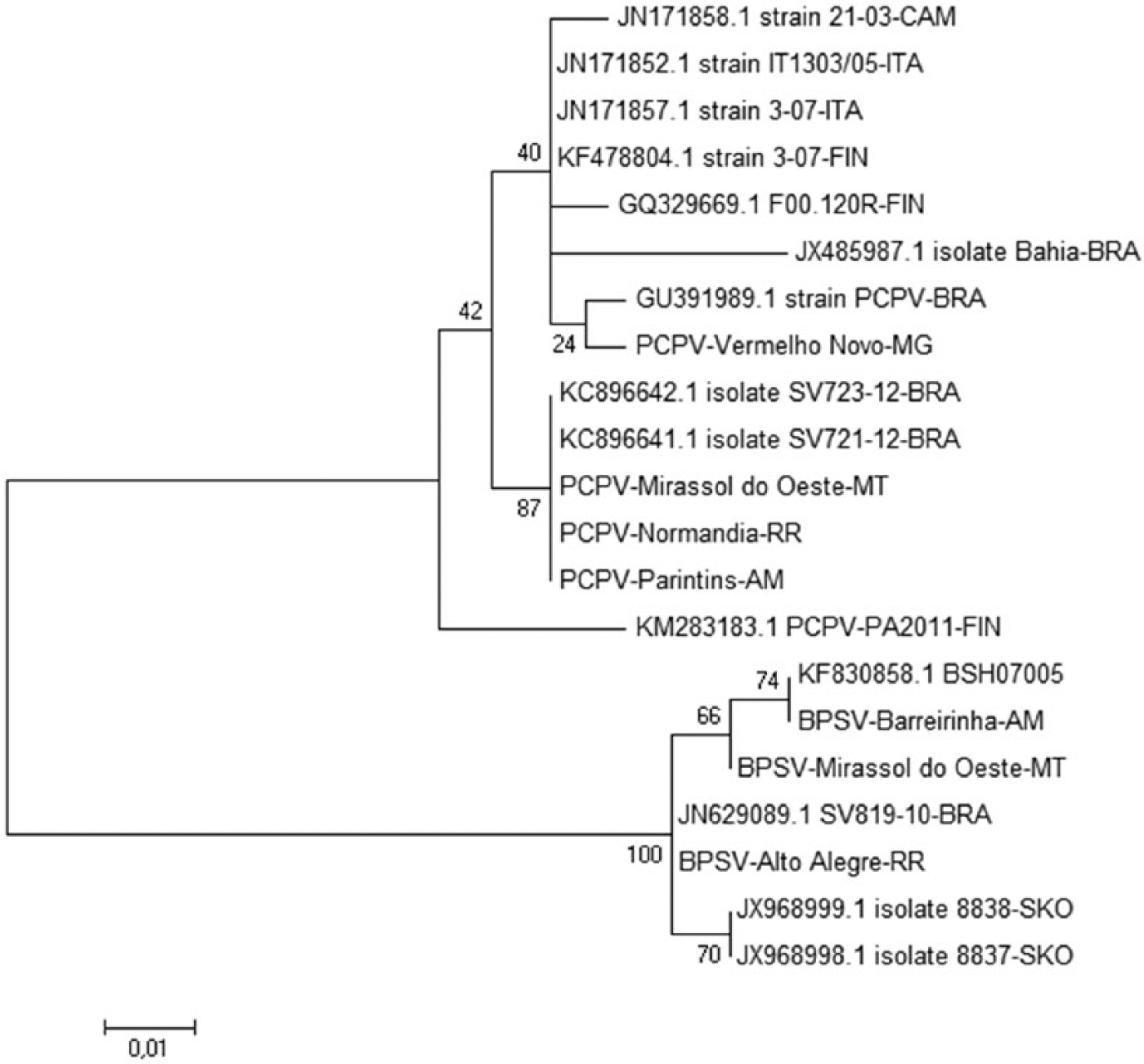
Phylogenetic tree generated using neighbor-joining method after alignment of nucleotide sequences. There is a clear separation between Pseudocowpox virus (PCPV) and Bovine papular stomatitis virus (BPSV) sequences. Identical samples found in our study were grouped by species and origin. PCPV sequences grouped with other Brazilian sequences from the Brazilian state of Rondônia. BPSV sequences clustered in a clade with a sequence from Bangladesh.
Discussion
The last outbreak of FMD in Brazil occurred in 2006 in the states of Mato Grosso do Sul and Paraná. 24 Since that time, no additional cases of the disease have been reported; however, Brazil continues to carry out a monitoring program to detect suspected vesicular disease, and vaccination for FMDV still occurs in several states. The diagnosis of FMD is important; however, it is also essential to continue research to determine the microorganisms that are present in similar injuries.
The cases studied in our work revealed a few samples that were positive for BTV and BoHV-1 in animals from MT. Such animals were transported from the state of Minas Gerais to MT for the purpose of fattening. The stress generated during transport may have contributed to depression of the immune system and increased viremia 28 ; therefore, viremia is an incidental finding in lesions rather than a cause. The infection may have occurred in the state of MG, as BTV causes an extended viremia 22 and BoHV-1 can enter latency 28 ; both of those viruses can be found in that state, often without overt clinical signs. 19
The presence of PPV was the most common finding, more precisely PCPV and BPSV. Both of these viruses have been described in nearby regions, such as RO and Goiás, 12 including cases of coinfection. In addition, both viruses were found in animals from the same farm. 9 Data obtained in this study demonstrate the wide circulation of these viruses in states in the north and midwestern regions of Brazil, where animal traffic occurs frequently, especially from MT to the states of RR, RO, and AM. A study conducted in northern Brazil showed the importance of the marketing and movement of animals in the spread of VACV in this region.1,26
PCPV, which was sequenced in our study, clustered with other samples from Brazil, particularly samples from an outbreak that occurred in RO in 2012. 9 This state usually buys cattle from MT, which may explain the genetic proximity in this case. BPSV did not cluster with samples from other Brazilian states but did cluster with a sequence from Bangladesh. 20 The information in the phylogenetic tree is important to identify correctly the viruses detected, because it is not possible to use the gene sequence to study any link between geographic location and clustering in the genetic tree for BPSV and PCPV.
Among the results of our study, we found the presence of PCPV in a sample derived from a buffalo with vesicular disease. This sequence was identical to other PCPV sequences found in this study. Buffalo are subject to various vesicular diseases 31 ; however, among poxviruses, records in Brazil are restricted to OPV. 11 In other countries, attempts to detect outbreaks of PPV based on characteristic clinical signs were successful in cattle but not in buffalo. 20 Cattle and buffalo are prominent in AM and can occur concurrently, which may pose a risk to herds of buffalo. Further studies are needed to detect the presence of PPV and determine the importance of this infection in this species and if there is any genetic modification in other parts of the virus genome.
Our study describes the presence of multiple infections in animals transported in Brazil, as well as the presence of more than one species of PPV in the same herd. The detection of PCPV infection in a buffalo warrants further studies of the susceptibility of these animals to this group of viruses.
Footnotes
Authors’ contributions
M Laguardia-Nascimento contributed to conception and design of the study. ÉB Sales and GG Souza contributed to acquisition, analysis, and interpretation of data. MR Gasparini and AF dos Santos contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. JAG da Silva contributed to conception and design of the study, and critically revised the manuscript. NM de Souza and FR Carani contributed to acquisition, analysis, and interpretation of data, and critically revised the manuscript. AV Rivetti Jr contributed to conception and design of the study; critically revised the manuscript; and gave final approval. MF Camargos contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; and gave final approval. AA Fonseca Jr contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and gave final approval. AV Rivetti Jr, MF Camargos, and AA Fonseca Jr agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
PrioCHECK FMDV NS antibody ELISA kit, Thermo Fisher Scientific Inc., Waltham, MA.
b.
TRIzol products, Thermo Fisher Scientific Inc., Waltham, MA.
c.
DNeasy blood and tissue kit, Qiagen GmbH, Hilden, Germany.
d.
Applied Biosystems 3500 genetic analyzers, Thermo Fisher Scientific Inc., Waltham, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to National Agricultural Laboratory of Minas Gerais (Lanagro-MG), National Science and Technology Institutes (INCT Pecuária), and National Council of Scientific and Technological Development (CNPq) SAGRES Project 457417/2012-9 for financial support and fellowships.
