Abstract
The diagnosis of neonatal and young calves persistently infected (PI) with Bovine viral diarrhea virus (BVDV) by antigen-capture enzyme-linked immunosorbent assay (ACE) may be complicated by interference from colostrum-derived specific antibodies. Ten calves, with 3 calves identified as PI and 7 as non-PI were used in the current study. All non-PI calves were shown to be seropositive for BVDV-specific antibodies by antibody enzyme-linked immunosorbent assay (Ab-ELISA) on serum. Serum samples, ear notch samples, and nasal and saliva swabs were collected from each calf from birth until 12 weeks of age and tested by ELISA for BVDV-specific antigen and antibodies. Following colostrum ingestion, Ab-ELISA sample-to-positive (S/P) ratios rose by a mean of 0.95 (95% confidence interval [CI] = 0.64–1.25) and 1.72 (95% CI = 1.55–1.89) in seropositive, non-PI calves and in PI calves, respectively. The mean S/P ratios then declined to approximately 1.1 in non-PI calves and 0.5 in PI calves at between 60 and 80 days of age. In PI calves, testing for antigen in serum and nasal and saliva swabs was subject to interference by colostrum-derived antibodies in calves up to 3 weeks of age. Nasal swabs were less affected than serum and saliva swabs. Ear notches maintained positive ACE corrected optical densities at all sample times, despite a drop in the signal following the ingestion of colostrum.
Acute infection of a cow or heifer with Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) during gestation may lead to fetal infection with varying outcomes. 3 When infection occurs prior to the development of fetal immunocompetence at approximately 120 days, the resultant calf (if it survives) is likely to be born persistently infected (PI) with BVDV. 1 Within hours of birth, the calf usually suckles and ingests BVDV-specific immunoglobulin-rich colostrum if the dam has been previously infected with or vaccinated against BVDV. 7 Colostrum-derived antibodies in the serum have been shown to produce false-negative results in PI calves when using the antigen-capture enzyme-linked immunosorbent assay (ACE). 2 While reverse transcription polymerase chain reaction (RT-PCR) is not subject to the same interference,2,11 it is also more expensive and requires more specialized equipment than ACE. Ear notch samples have been heralded as a solution to the problem of colostrum-derived antibody interference with ACE 5 ; however, a 2012 study 2 provides conflicting results, dependent on the specific ELISA used. Therefore, the aims of the current study were to measure the decline over time of colostrum-derived BVDV-specific antibodies in PI and seropositive, non-PI calves and to compare the impact of serum antibodies on the ability of a commercial ACE to detect BVDV antigen in different sample types collected from PI calves.
Three BVDV PI calves and 7 seropositive, non-PI calves born following natural infection of their previously BVDV naïve (antibody-negative, antigen-negative by ELISA a ) dams, through exposure to a BVDV PI cow beginning on days 69–90 of gestation were used in the current study. The PI cow remained in contact with the dams for 28 days, until seroconversion had been observed in all dams. The calves were suckled by their dams throughout the study period. Quantitative RT-PCR was performed, as previously reported, 4 on serum samples collected from each calf on day 14 of age to confirm the BVDV status of the calves. Calves were considered PI with BVDV when: either, precolostral samples returned a positive corrected optical density (OD) by ACE, or serum collected at 14 days of age returned a positive result by qRT-PCR; and, Ab-ELISA returned a negative sample-to-positive (S/P) ratio on precolostral serum (where available). All calves returning negative ACE and PCR results were considered non-PI (Table 1). All non-PI calves were shown to be seropositive for BVDV-specific antibodies by ELISA on serum, either collected prior to colostrum ingestion or collected at conclusion of the trial following waning of maternal antibodies.
Bovine viral diarrhea virus (BVDV) status (persistently infected [PI] or non-PI and seropositive) of each of 10 calves born following acute infection of the dam during early gestation, as determined by commercial antibody and antigen-capture enzyme-linked immunosorbent assays (ELISAs) on serum collected prior to colostrum ingestion and quantitative reverse transcription polymerase chain reaction (qRT-PCR) results on serum collected at 14 days of age.
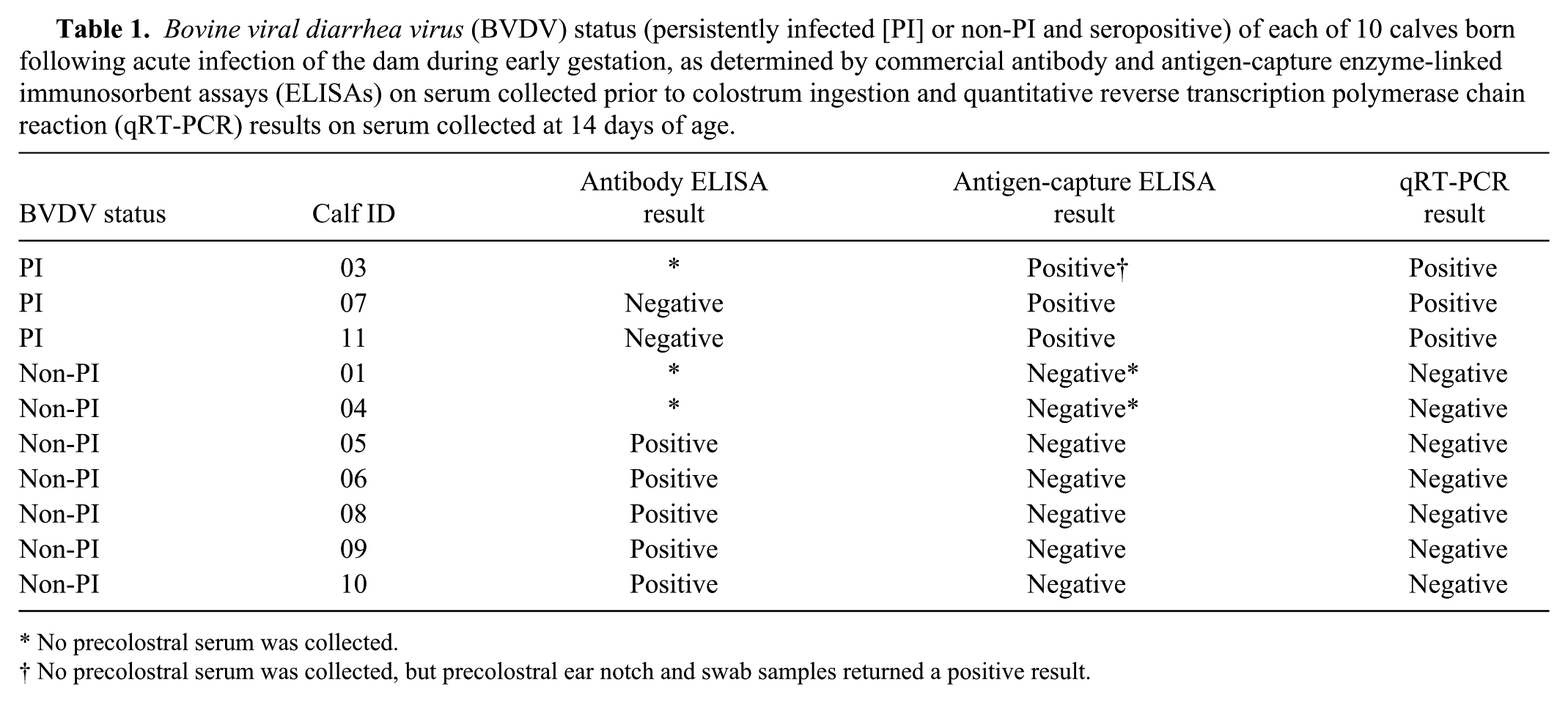
No precolostral serum was collected.
No precolostral serum was collected, but precolostral ear notch and swab samples returned a positive result.
Genotyping of the persisting strain of BVDV in each PI calf and the PI cow used for infection of the dams revealed homologous sequences consistent with BVDV type 1c, showing 98% homology with the Manasi reference strain (GenBank accession no. EU159702). 10 This is consistent with type 1c being the most prevalent strain of BVDV in Australia. 9
Each dam was fitted with a custom made udder cover (Fig. 1) when calving appeared imminent to prevent the calf from suckling. Serum, ear notch tissue, and nasal and saliva swabs were collected from all calves within 8 hr of birth, prior to the removal of the udder cover and subsequent suckling and colostrum ingestion. This procedure was unsuccessful in 2 of the dams resulting in 2 calves (nos. 1 and 4) suckling prior to first sample collection. One additional calf (3) had ear notch and swab samples collected prior to suckling; however, serum was not collected until several hours after first colostrum ingestion. Further samples (serum, ear notch, and nasal and saliva swab) were collected every 2 days from birth until 14 days of age. Serum and nasal and saliva swabs were then collected weekly until 12 weeks of age. A final serum sample was collected from each surviving calf at approximately 6 months of age.

A heifer fitted with a custom-made udder cover prior to calving to allow sample collection from the calf prior to colostrum ingestion.
All the samples were stored at −80°C between collection and preparation. All ear notch and swab samples were prepared for testing by soaking in ear notch tissue soaking buffer a (ear notches 250 µl; swab tips 1 ml) for 24 ± 1 hr at room temperature. After soaking, samples were separated from the supernatant. Supernatants and samples were stored separately at −80°C until testing.
Serum samples were tested for the presence of BVDV-specific antibodies by commercially available Ab-ELISA, a with an S/P ratio >0.3 considered positive as per manufacturer’s recommendation and previous validation. 6 Of those calves (n = 7) from which serum samples were collected prior to colostrum ingestion, 2 were PI and 5 were seropositive and non-PI. The PI calves (nos. 7 and 11) demonstrated negative Ab-ELISA S/P ratios in serum prior to colostrum ingestion of 0.09 and 0.29, respectively. All non-PI calves tested positive for BVDV-specific antibodies prior to colostrum ingestion, with Ab-ELISA S/P ratios ranging from 0.39 to 1.24. Following colostrum ingestion, Ab-ELISA S/P ratios rose by a mean of 0.95 (95% confidence interval [CI] = 0.64–1.25) in the seropositive, non-PI calves from which samples were collected prior to colostrum ingestion and by a mean of 1.72 (95% CI = 1.55–1.89) in PI calves. The mean Ab-ELISA S/P ratios were 1.67 (95% CI = 1.50–1.84) in all the seropositive, non-PI calves and 1.95 (95% CI = 1.88–2.02) in all the PI calves, at 2 days of age (Fig. 2).
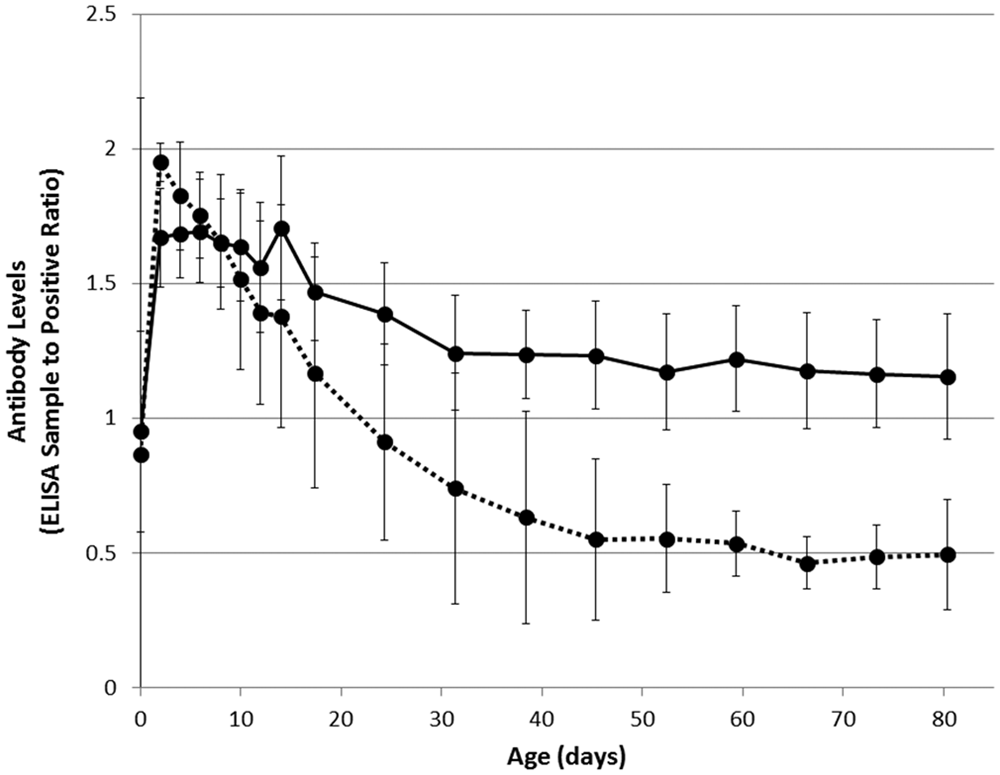
Declining Bovine viral diarrhea virus (BVDV)-specific antibodies in calves that are persistently infected (PI; dotted line) or non-PI (solid line) with BVDV following colostrum ingestion, as measured by commercial antibody enzyme-linked immunosorbent assay (ELISA). a Antibody ELISA results are expressed as sample-to-positive (S/P) ratios, with an S/P ratio >0.3 considered positive. Error bars show 95% confidence intervals. Note: day-of-birth (day 0) mean includes some sample collected after colostrum ingestion.
Antibody ELISA S/P ratios declined from 2 days of age until 12 weeks of age (Fig. 2). The decline in Ab-ELISA S/P ratios was more rapid in PI calves than seropositive, non-PI calves, with PI calves approaching an S/P ratio of approximately 0.5 and seropositive, non-PI calves approaching an S/P ratio of 1.1 at 12 weeks of age. This is in contrast to a previous study 8 that observed that colostrum-derived antibody titers in PI calves did not persist beyond 4 weeks of age when tested by virus neutralization and suggests caution when using Ab-ELISA on calves less than 6 months of age: seropositivity and PI are not necessarily mutually exclusive.
At approximately 6 months of age, the PI calves had a mean Ab-ELISA S/P ratio of 0.20 (95% CI = 0.09–0.32; negative at the manufacturer’s positivity threshold), and the seropositive, non-PI calves retained a mean S/P ratio of 1.42 (95% CI = 1.17–1.68). This maintenance of seropositivity confirms that the non-PI, seropositive-at-birth calves in the present study were subject to acute fetal infection following development of immunocompetence.
All samples were tested by ACE, a following the manufacturer’s protocol recommended for serum. That is, sample incubation consists of a 50-µl sample (serum or supernatant) incubated with 50 µl of detection antibodies for 2 hr at 37°C, with a corrected OD >0.3 considered positive. Following colostrum ingestion, serum samples from all PI calves produced negative ACE corrected ODs (Fig. 3). Positive ACE corrected ODs were obtained at 8 days, 10 days, and 4 weeks of age in calves 7, 11, and 3, respectively. A strong signal in the ACE (equivalent to the corrected OD observed prior to colostrum ingestion) was recovered at 12 days, 14 days, and 5 weeks of age in calves 7, 11, and 3, respectively. This is comparable with previous findings 2 where sera were observed to return a negative result until 5–14 days of age.
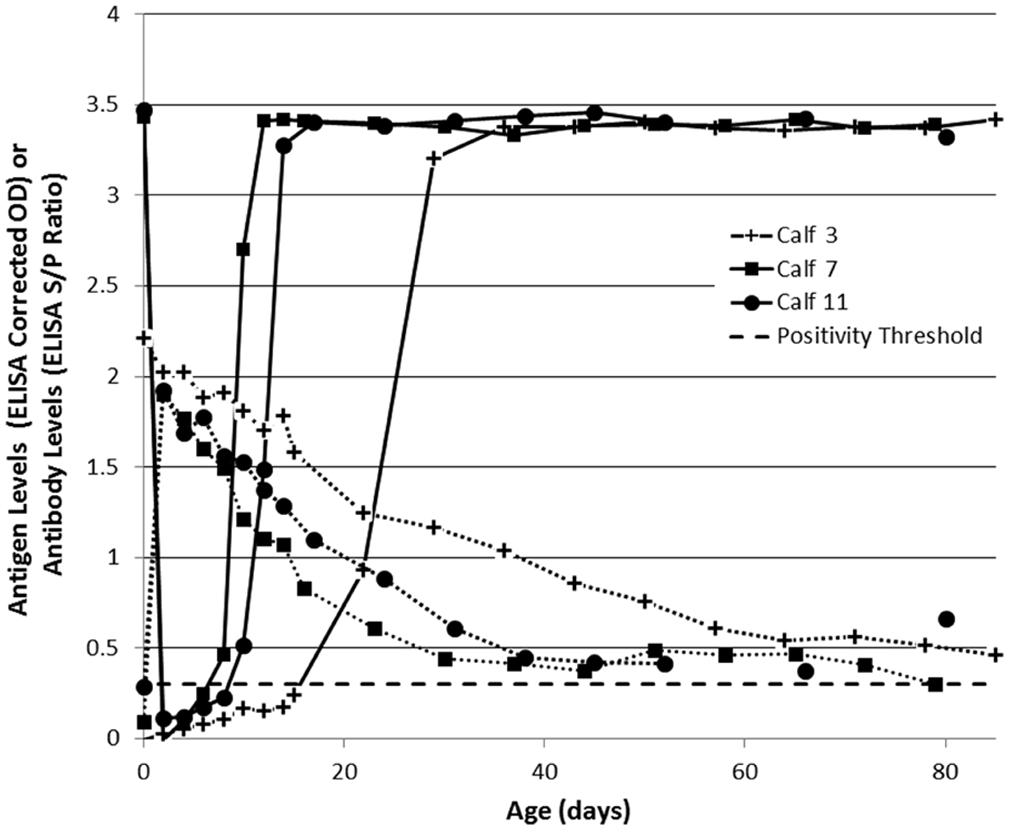
Bovine viral diarrhea virus (BVDV)-specific antigen (solid lines) and antibodies (dotted lines) in serum of calves (n = 3) persistently infected (PI) with BVDV, as measured by commercial antigen-capture or antibody enzyme-linked immunosorbent assay (ELISA) a and expressed as either corrected optical density (OD) or sample-to-positive (S/P) ratio. A corrected OD or S/P ratio >0.3 was considered positive. Day-of-birth (day 0) samples from calves 7 and 11 were collected prior to colostrum ingestion.
The ACE corrected ODs for ear notch supernatants from PI calves also declined following colostrum ingestion (Fig. 4). Calves 7 and 11 were less affected, with ACE corrected ODs in ear notch supernatants never dropping below 1.5. Antigen ELISA corrected ODs in ear notch supernatants from calf 3 reduced to a low of 0.41 and 0.42 corrected OD on days 10 and 12, respectively. All PI calves returned high positive ACE corrected ODs (>2.0) in ear notch supernatants by day 14. This is similar to reports in previous studies2,5 where ear notch supernatants appeared to be the least affected of all sample types when tested by ACE in the presence of colostrum-derived antibodies. A previous study 2 showed that ear notch supernatants returned a positive result in this same ELISA at all time points, despite greatly reduced signal.
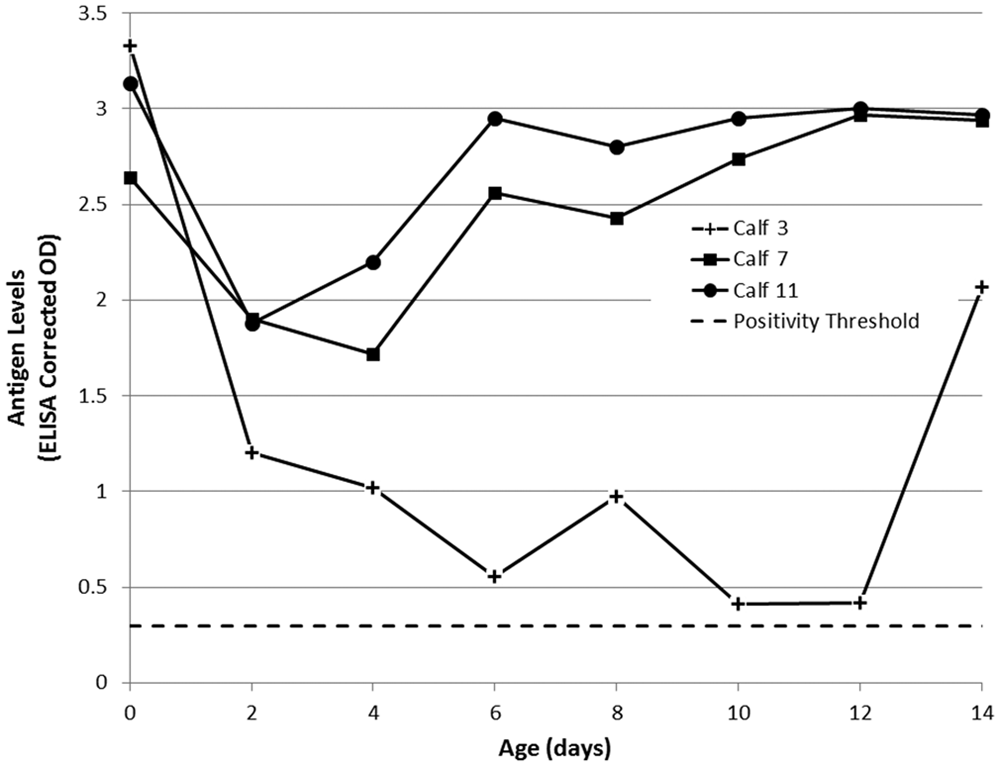
Bovine viral diarrhea virus (BVDV)-specific antigen in ear notch supernatants of calves persistently infected with BVDV, as measured by commercial antigen-capture enzyme-linked immunosorbent assay (ACE) a and expressed as corrected optical density (OD). A corrected OD >0.3 was considered positive. All day-of-birth (day 0) samples were collected prior to colostrum ingestion.
When swab supernatants were tested by ACE, the corrected ODs declined following colostrum ingestion (Figs. 5, 6). All nasal swab supernatants recovered to produce positive corrected ODs at approximately 3 weeks of age, with all calves returning consistent high positive corrected ODs (>2.5) from approximately 40 days of age onward (Fig. 6). The timing and magnitude of recovery of ACE corrected ODs in nasal swabs was similar that observed in serum. This provides some indication that the off-label use of this ELISA for testing nasal swab supernatant samples may have similar performance to that observed for serum samples, and may suggest that further investigation of nasal swabs as an alternative sample for detection of adult PI individuals is warranted. By comparison, saliva swab supernatants recovered substantially in all calves by approximately 3 weeks of age, but continued to produce somewhat erratic corrected ODs throughout the study period (Fig. 5). This is most likely an effect of varying amounts of saliva collected by the swabbing procedure. Saliva swab collection protocols that allow for standardization of saliva volume collected would need further investigation if this sample type were to be pursued. As the calves were not weaned until after the conclusion of the study, the inconsistency of saliva swab results is also potentially affected by the time since suckling, with milk-derived antibodies possibly retained in the oral environment and contaminating the swab sample, causing additional interference with the ACE.
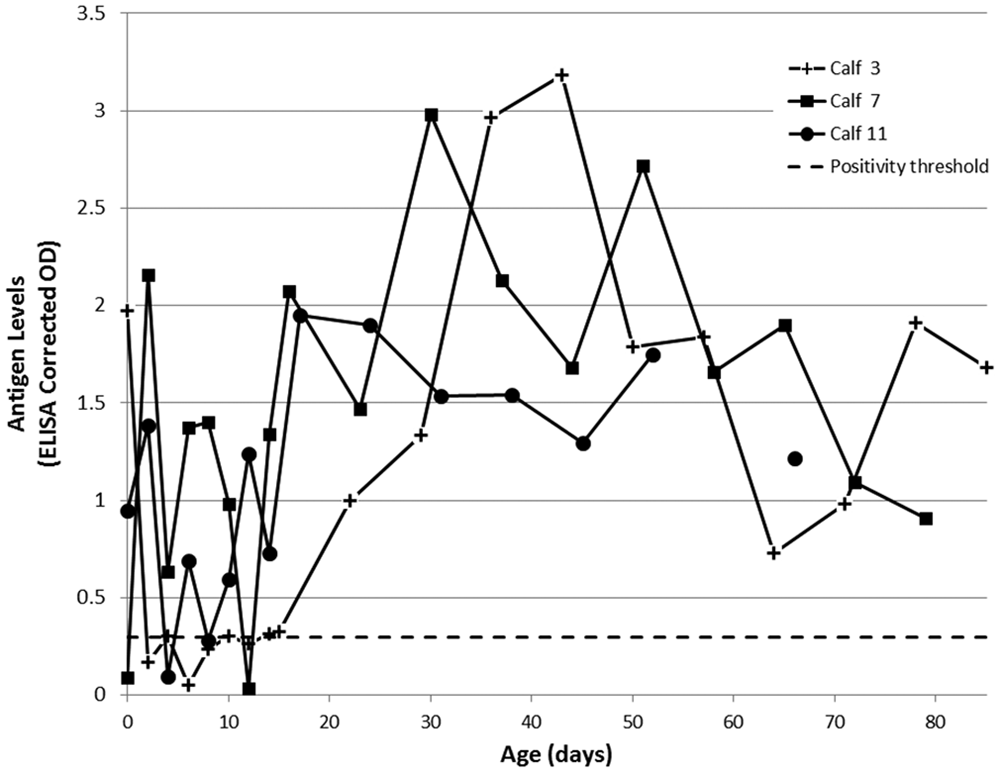
Bovine viral diarrhea virus (BVDV)-specific antigen in saliva swab supernatants of calves persistently infected with BVDV, as measured by commercial antigen-capture enzyme-linked immunosorbent assay a and expressed as corrected optical density (OD). A corrected OD >0.3 was considered positive. All day-of-birth (day 0) samples were collected prior to colostrum ingestion.
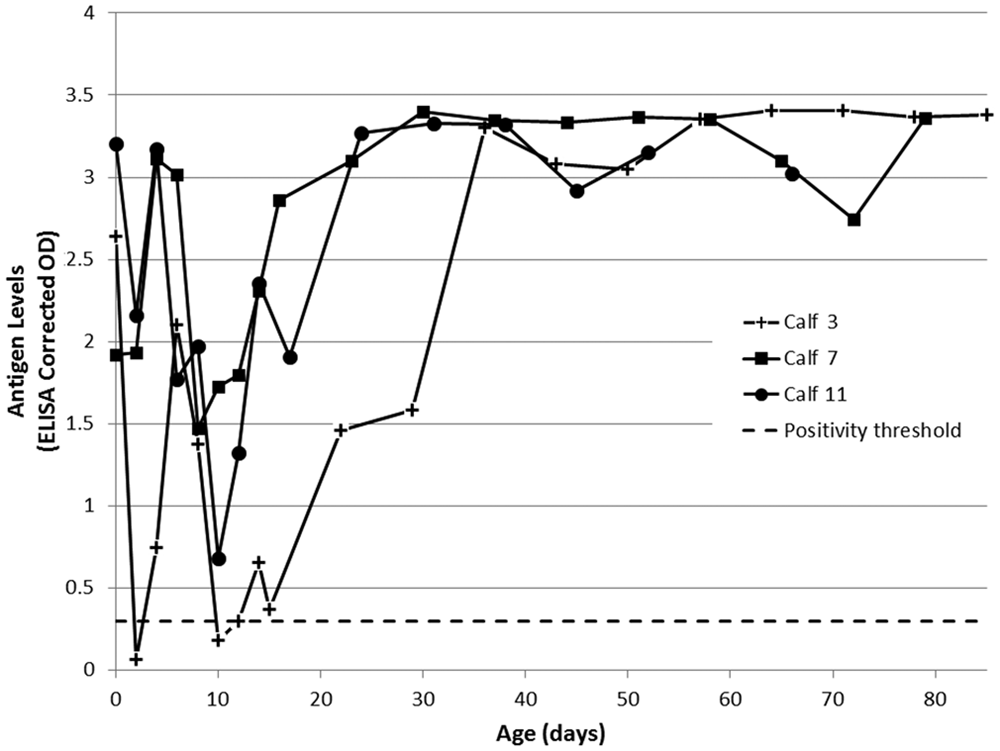
Bovine viral diarrhea virus (BVDV)-specific antigen in nasal swab supernatants of calves persistently infected with BVDV, as measured by commercial antigen-capture enzyme-linked immunosorbent assay a and expressed as corrected optical density (OD). A corrected OD >0.3 was considered positive. All day-of-birth (day 0) samples were collected prior to colostrum ingestion.
It is useful to note that calf 3 was the last to recover signal in all sample types. Closer inspection of the antibody profiles in the 3 PI calves revealed that while Ab-ELISA S/P ratios were similar at 2 days of age, calf 3 maintained an S/P ratio >1.5 until 3 weeks of age, while S/P ratios in calves 7 and 11 dropped below this level at 10 and 6 days, respectively. This highlights that the diagnostic window during which interference from colostrum-derived antibodies may occur is likely to vary between calves. Such variation may be dependent on both the quality and the quantity of colostrum and the efficiency of absorption of immunoglobulins. It appears, from the results of the current study, that recovery of a positive ACE corrected OD on serum corresponds with the Ab-ELISA S/P ratio in any given PI calf declining below approximately 1.5.
In conclusion, the results of the present study suggest that ear notch samples remain the preferable sample for detection of young, colostrum-fed PI calves using the ACE, although a substantial drop in signal suggests that caution in interpretation of results may be warranted. Both nasal and saliva swab supernatants exhibited similar testing limitations to serum in the presence of colostrum-derived antibodies. Nasal swabs may be worth further investigation as swabs are simple and inexpensive to collect and provide consistent high positive results after the effects of colostrum-derived antibody interference have waned.
Footnotes
Acknowledgements
The authors thank the staff of Martindale Holdings, particularly Grant Jarvis, Neil Stanley, and John Matheson; the veterinarians of the University of Adelaide Production Animal Clinic; Kate Cantlon and Rick McCoy of Gribbles Veterinary for technical assistance; Milton McAllister, Ian Wilkie, and Adrian Hines; and Malcolm Crossman, Caitlin Evans, Caitlin Jenvey, Brenden Johansson, Claire Castles, Damien Hunter, and Shaun Whitington for their support and tireless assistance.
a.
Ear notch tissue soaking buffer, BVDV Total Ab ELISA, BVDV Serum/Ag Plus ELISA; IDEXX Laboratories Inc., Rydalmere, New South Wales, Australia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by Meat and Livestock Australia (project code B.AHE.2014) and the University of Adelaide, with all ELISA kits provided by IDEXX Laboratories Inc.
