Abstract
Bovine viral diarrhea virus (BVDV) is divided into 2 different species within the Pestivirus genus, BVDV type 1 (BVDV-1) and BVDV type 2 (BVDV-2). Further phylogenetic analysis has revealed subgenotype groupings within the 2 types. Thus far, 12 BVDV-1 subgenotypes (a–l) and 2 BVDV-2 subgenotypes (a and b) have been identified. The purpose of the current study was to determine the prevalence of BVDV subgenotypes in the United States and Australia and to determine if there are detectable antigenic differences between the prevalent subgenotypes. To determine prevalence, phylogenetic analysis was performed on 2 blinded panels of isolates consisting of 351 viral isolates provided by the Elizabeth Macarthur Laboratory, New South Wales, and 514 viral isolates provided by Oklahoma State University. Differences were observed in the prevalence of BVDV subgenotypes between the United States (BVDV-1b most prevalent subgenotype) and Australia (BVDV-1c most prevalent subgenotype). To examine antigenic differences between the subgenotypes identified in samples from the United States and Australia, polyclonal antisera was produced in goats by exposing them at 3-week intervals to 2 noncytopathic and 1 cytopathic strain of either BVDV-1a, BVDV-1b, BVDV-1c, BVDV-2a, or Border disease virus (BDV). Virus neutralization (VN) assays were then performed against 3 viruses from each of the 5 subgenotypes. Comparison of VN results suggests that there are antigenic differences between BVDV strains belonging to different subgenotypes. The present study establishes a foundation for further studies examining whether vaccine protection can be improved by basing vaccines on the BVDV subgenotypes prevalent in the region in which the vaccine is to be used.
Introduction
Bovine viral diarrhea virus (BVDV; family Flavivir-idae, genus Pestivirus) is segregated into 2 different species, type 1 (BVDV-1) and type 2 (BVDV-2). 44 Although this segregation was first based on phylo-genetic analysis, 34,37 subsequent characterization of viral strains from the 2 species has demonstrated antigenic differences. 39 The clinical significance of the observed antigenic differences was evidenced by the failure of vaccines and diagnostics based on BVDV-1 strains to control and detect, respectively, BVDV-2 strains. 13 The recognition of genomic and, particularly, antigenic difference between the 2 BVDV species led to the redesign of vaccines and diagnostics. 36
Further phylogenetic analysis has revealed subgeno-type groupings within the BVDV-1 and BVDV-2 species. Thus far, 12 BVDV-1 subgenotypes (a–l) 48 and 2 BVDV-2 subgenotypes (a and b) 17 have been identified. Novel subgenotypes of BVDV-1 have been identified in isolations of BVDV from cattle in South Africa and Switzerland but have not yet been assigned names. 5,28,29 Different BVDV subgenotypes predominate in different geographic locations. 4,5,14,15,21,23,25–28,30–35,42,43,45–47 The clinical significance of segregation into subgenotypes is still a matter of discussion and is not officially recognized by the International Committee on Taxonomy of Viruses. In addition to genomic differences, studies have shown antigenic differences between subgenotypes as demonstrated by differences in cross-neutralization, 5,35 monoclonal antibody binding, 11 and response of persistently infected animals to vaccination. 22 It is not known if variations among subgenotypes is significant enough to impact detection or the protection afforded by vaccination. It is possible that regional control programs would be improved by taking into consideration the BVDV subgenotypes present in the design of diagnostics and vaccines.
Viral isolates used in generation of polyclonal antisera. *

Goats were sequentially inoculated with viral isolates in the order listed. BVDV = Bovine viral diarrhea virus; BDV = Border disease virus.
Materials and methods
Subgenotype prevalence survey
Phylogenetic analysis was performed on sequences derived from 2 blinded panels consisting of 351 viral isolates provided by the Elizabeth Macarthur Laboratory (EMAI; New South Wales, Australia) and 514 viral isolates provided by Oklahoma State University (OSU; Stillwater, OK). The EMAI panel was collected over a 25-year period from cattle in Australia and was selected to represent a diversity of clinical presentations and geographical locations. The majority of these isolates were made from samples collected from persistently infected (PI) animals. The OSU panel was collected between June 2007 and June 2008 from PI animals identified in southwestern U.S. feedlots.
The basis of the phylogenetic analysis was a comparison of 5'-untranslated region sequences generated by cycle sequencing of polymerase chain reaction amplicons as described previously. 41 Segregation into subgenotypes was based on comparisons to BDDV-1 47,48 and BVDV-2 17 subgenotype type sequences previously reported.
Virus propagation for use in inoculum
Viruses were propagated in either bovine turbinate (BT) cells (BVDV-1a–c, BVDV-2a) or ovine fetal turbinate (OFT) cells (Border disease virus [BDV]). Bovine turbinate and OFT cells were grown in complete cell culture medium that was composed of minimal essential medium (F15 Eagle medium a supplemented with 10% fetal bovine serum, 1-glutamine [final concentration, 1.4 mM], and gentamicin [final concentration, 50 mg/l]). The fetal bovine serum contained no BVDV and no antibodies against BVDV. 7 Viral titers were determined via limiting dilution in BT cells (BVDV-1a–c, BVDV-2a) or OFT cells (BDV). 38 Endpoints were based on cytopathic effect (cytopathic strains) or binding of monoclonal antibodies to the E2 structural protein (noncytopathic strains) as detected by immunoper-oxidase staining as described below. 6
Production of caprine polyclonal antisera
Handling and treatment of goats used to generate polyclonal antisera complied with the Animal Welfare Act. 2 Polyclonal antisera was produced in goats that tested free of BVDV based on virus isolation and free of BVDV antibodies based on serum neutralization, using the strains BVDV1a-Singer and BVDV2a-296c, as described previ-ously. 38 Goats were housed in individual, climate-controlled pens under biosafety laboratory 2 containment. Goats were inoculated at 3-week intervals with 1 cytopathic and 2 noncytopathic strains of either BVDV-1a, BVDV-1b, BVDV-1c, BVDV-2a, or BDV (Table 1). Because pestivirus strains cannot be purified, 44 the inoculum consisted of 5 ml of freeze-thawed lysate of infected cells (1.0 × 106 tissue culture infective dose [TCID]/ml) delivered by the nasal route. 40 Polyclonal antisera were harvested 3 weeks after the third inoculation.
Virus neutralization tests
Titers of viral neutralizing antibodies in serum were determined against 3 noncytopathic isolates from each of the 5 genotypic groups (BVDV-1a–c, BVDV-2, and BDV; Table 2). Prior to comparing cross-neutralizing titers, all sera were initially diluted to a virus-neutralizing titer of 1/256 against the cytopathic virus used to generate the antisera. A microtiter plate assay was then used to determine titers with endpoints based on the binding of monoclonal antibodies to the E2 structural protein. 6 Briefly, following that initial dilution, serial 2-fold dilutions of sera in complete cell culture medium were prepared. A 100-μl aliquot of diluted serum and a 50-μl aliquot of virus containing 1,000 TCID were added to each well and incubated for 1 hr at 37°C. At the end of this incubation period, 20,000 BT cells (BVDV-1a–c and BVDV-2a isolates) or OFT cells (BDV isolates) in a 100-μl aliquot of complete cell culture medium were added to each well. Inoculated cultures were incubated for 4 days at 37°C. Cell monolayers were rinsed in deionized, double-distilled water and fixed by adding 50 μl per well of fixation buffer (60% w/v bovine serum albumin, 40% v/v acetone in phosphate buffered saline [PBS]) and incubating for 10 min at room temperature (RMT). The fixation buffer was then removed, and plates were dried at 37°C for 1 hr. After drying, a 50-μl aliquot of primary monoclonal antibody (N2 hybridoma supernatant 37 ) in PBSTN binding buffer (0.05% v/v Tween-20, 2.95% w/v sodium chloride in PBS) was added followed by incubation at RMT for 1 hr. The primary monoclonal antibody solution was removed, and wells were washed with 50 μl of PBST wash buffer (0.05% v/v Tween-20 in PBS). A 50-μl aliquot of goat antimouse immunoglobulin G (IgG) b (secondary antibody) prepared in PBSTN binding buffer was then added followed by incubation at RMT for 1 hr. After incubation, the secondary antibody was removed, cells were washed with PBST, and a 50-μl aliquot of protein G conjugated to horseradish peroxidase c prepared in PBSTN was added. After a 1-hr incubation at RMT, substrate in the form of 3-amino-9-ethylcarbazole was added, as described previously, 1 and color allowed to develop for 10 min. Reaction was stopped by removing substrate solution and adding water to each well. Stained cells were observed with a microscope.
Genotypes of isolates used in viral neutralization tests. *
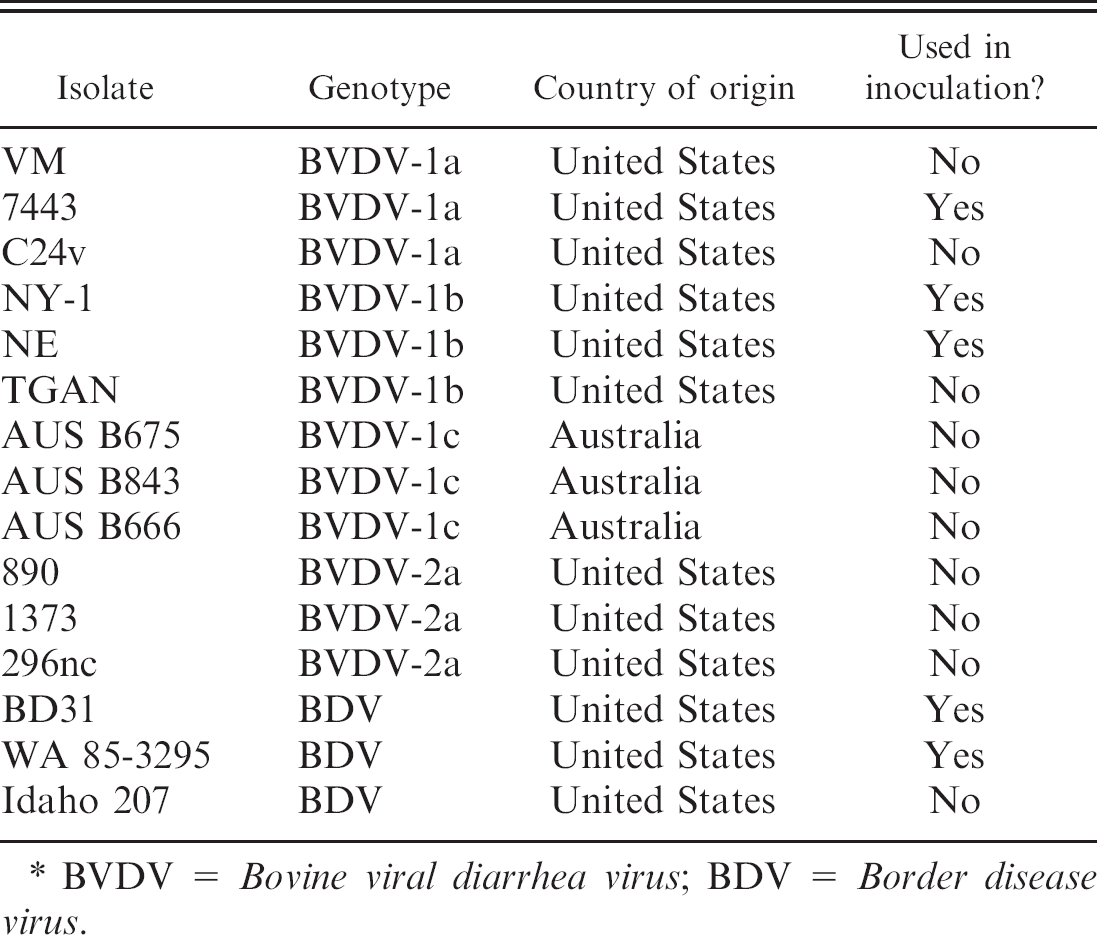
BVDV = Bovine viral diarrhea virus; BDV = Border disease virus.
Each neutralization assay was done in a replicate of 5, and titers were calculated using the Spearman–Kärber method. 16 The endpoint dilutions reflected the highest dilution of serum that inhibited the growth of virus. The serologic relatedness was expressed by 2 different methods. The simpler method involved calculating the ratio (P, stated as percentage) of the heterologous virus neutralization (VN) value as compared with the homologous VN value using the following formula:
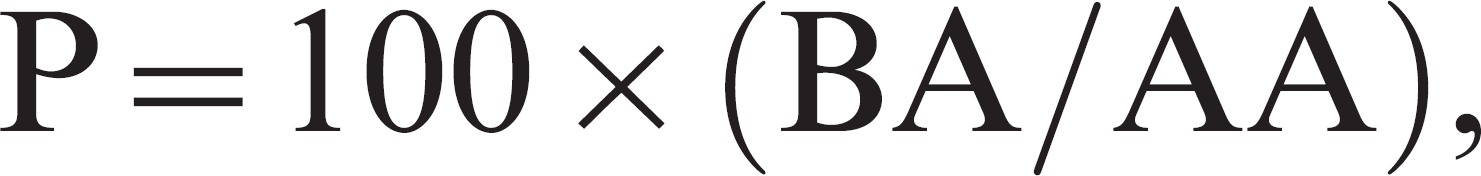
where BA is the VN titer against strain B using antiserum A, and AA is the VN titer against strain A using antiserum A.
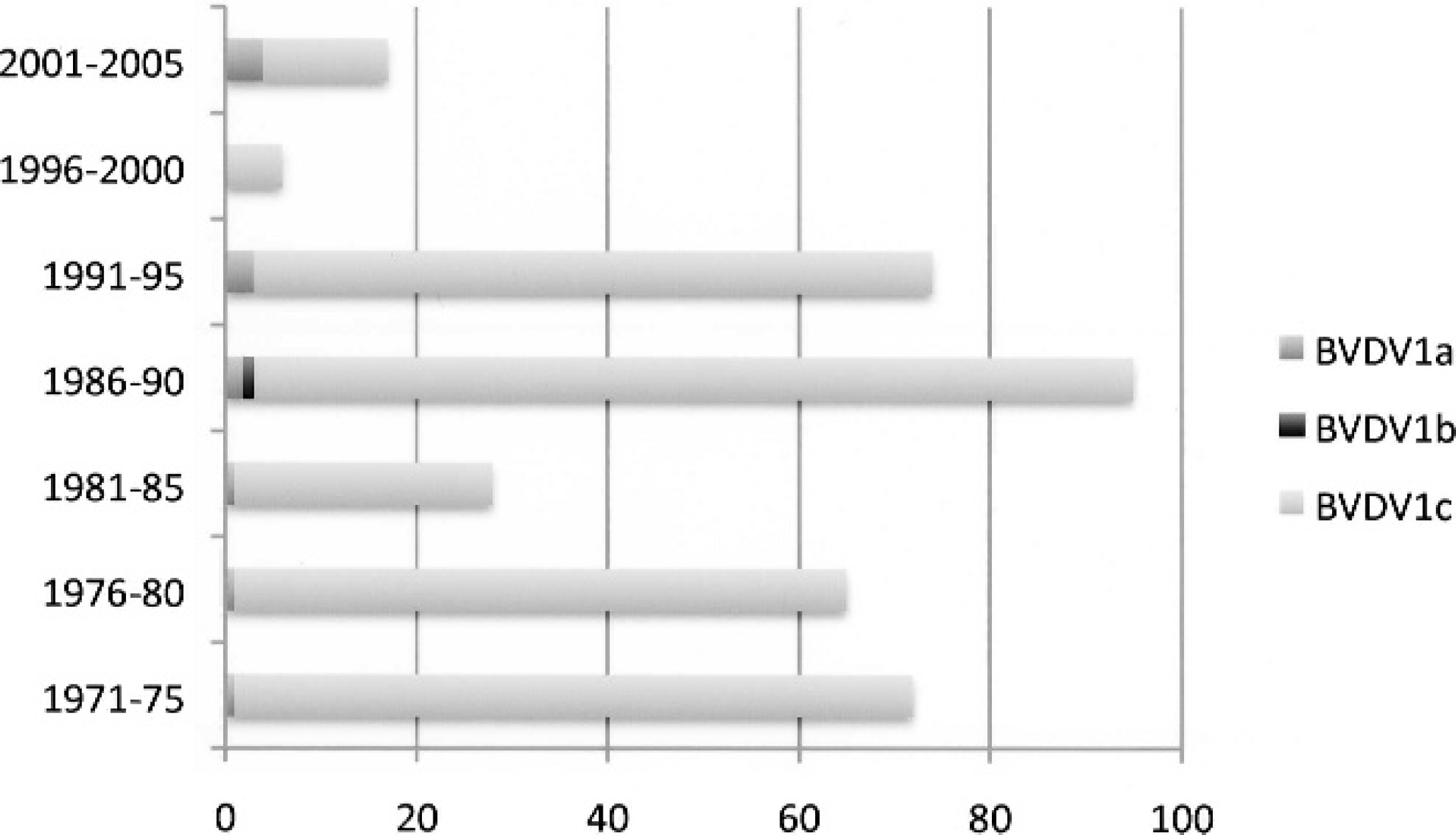
Temporal distribution of Australian Bovine viral diarrhea virus (BVDV) subgenotypes collected over a 25-year time span. The numbers of isolates characterized as BVDV-1a, BVDV-1b, or BVDV-1c are listed for 5-year time blocks.
A second method determined the coefficient of antigenic similarity (R) between each subgenotype and between each subgenotype and BDV by a method previously described 3,5 using the following formula
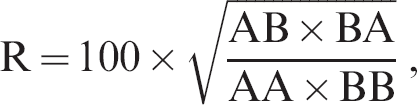
where R is the coefficient of antigenic similarity, AB is the VN titer against strain A using antiserum B, and BB is the VN titer against strain B using antiserum B.
Results
Three subgenotypes of BVDV-1 and 1 subgenotype of BVDV-2 were identified among the BVDV isolates included in the 2 panels. Among the 351 isolates of the EMAI panel were 11 (3.1%) BVDV-1a strains, 1 (0.3%) BVDV-1b strain, 338 (96.3%) BVDV-1c strains, 4 (1.1%) BVDV-2a strains, and 1 (0.3%) BDV isolate. The temporal distribution of subgeno-types over the 25-year span is shown in Figure 1. The subgenotype of the BDV isolate was not determined for the current study. In contrast, phylogenetic analysis of the 514 isolates in the OSU panel revealed 62 (12.1%) BVDV-1a strains, 387 (75.3%) BVDV-1b strains, no BVDV-1c strains, 65 (12.6%) BVDV-2a strains, and no BDV strains.
The average VN value for each serum against strains from each of the 4 subgenotypes plus BDV is shown in Figure 2. Error bars represent the standard error of the mean. In all cases, the highest average VN titers against each subgenotype were observed when strains from 1 subgenotype were neutralized using antiserum produced in goats against strains from the same subgenotype (homologous neutralization). Virus neutralization titers were higher against viruses from different subgenotypes within the same species as those strains used to produce the antisera than to viruses from other species (Table 3). The VN values were between 36% and 52% of homologous neutralization for the 3 BVDV-1 subgenotypes. In contrast, VN values were less than 10% of homologous neutralization for viruses from different species.
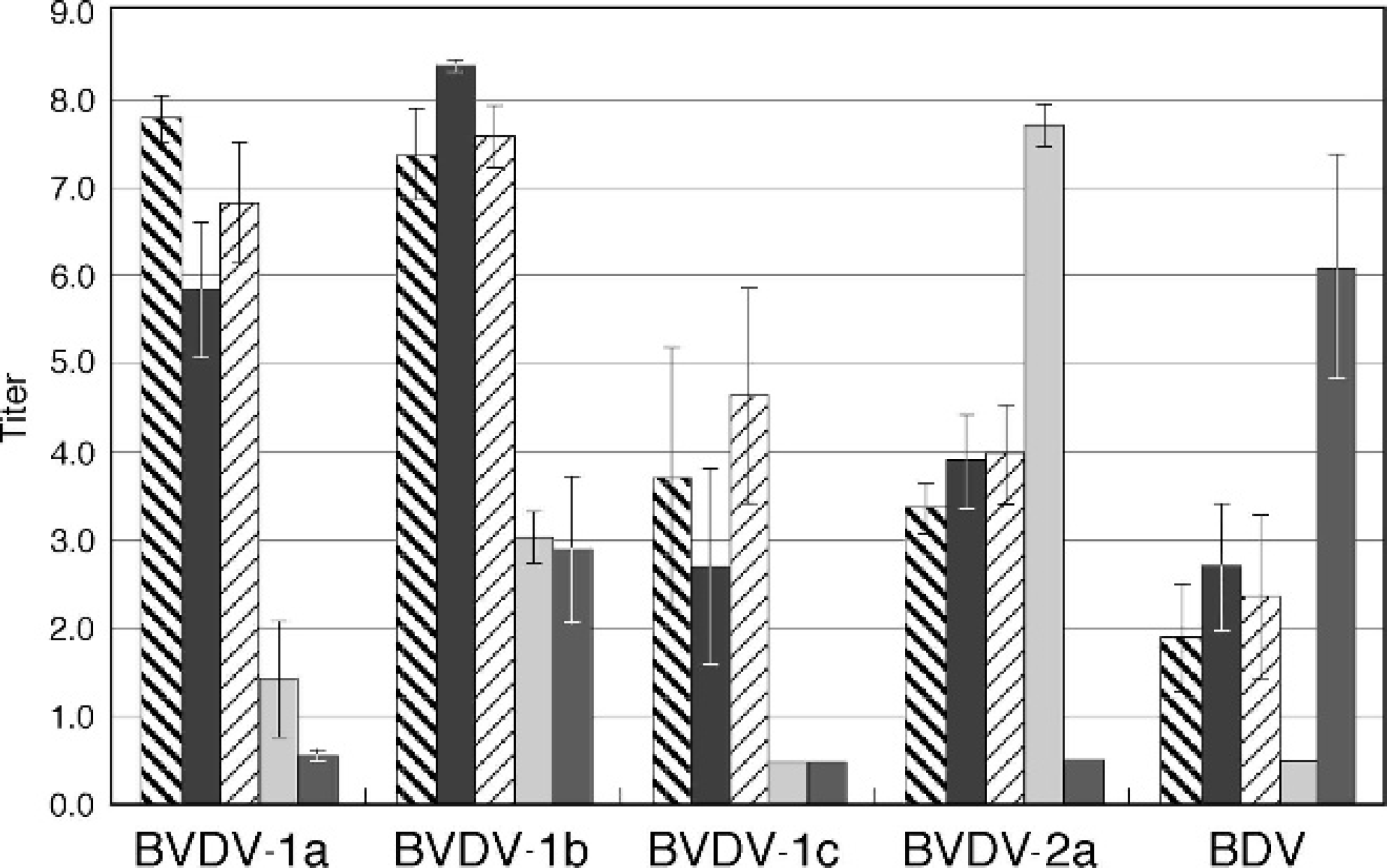
Virus neutralization (VN) titers of goat polyclonal sera produced against different pestivirus species and subgeno-types. Average VN titers of goat polyclonal sera produced against Bovine viral diarrhea virus (BVDV)-1a, BVDV-1b, BVDV-1c, BVDV-2a, and Border disease virus (BDV) strains when used to neutralize BVDV-1a, BVDV-1b, BVDV-1c, BVDV-2a, and BDV strains. The Y-axis is the log base 2 of the reciprocal of the endpoint dilution. Error bars represent the standard error of the mean.
The coefficients of antigenic similarity (R-values) between BVDV-1 (all 3 genotypes), BVDV-2, and BDV were less than 10, indicating significant anti-genic differences (Table 4). The R-values among the 3 BVDV-1 subgenotypes ranged between 36 and 52 (Table 4). The R-value of 36 between BVDV-1a and BVDV-1b strains was similar to the R-value of 35 between BVDV-1a and BVDV-1b determined in a previous study. 5
Discussion
In the current study, a marked difference was observed in the prevalence of BVDV subgenotypes in panels generated from Australian and U.S. isolates. The predominant species in each panel was BVDV-1; however, in the EMAI panel, the predominant subgenotype was BVDV-1c (>96%), while the predominant subgenotype in the OSU panel was BVDV-1b (>75%). Previous studies have shown variations in the presence and prevalence of BVDV species and subgenotypes in different continents and countries. A summary of those BVDV subgenotypes surveys based on isolates from 18 different countries is shown in Table 5. The number of subgenotypes identified in each country varies from 2 to 9, and the number of isolates characterized ranged from 8 to 475. Similar to the results of the current study, the most frequently reported species was BVDV-1. The most frequently reported subgenotype was BVDV-1b with 16 countries isolating BVDV-1b among cattle populations. Of these 16 countries, 6 countries identified it as a predominant strain as was observed in the OSU panel characterized in the present report. Although the presence of BVDV-1c strains was reported in 6 countries, Australia is unique in having a predominance of BVDV-1c strains. Further, the high percentage of isolates in 1 subgenotype (>96%) is also unique to the Australian samples, despite the fact that they were obtained from a wide range of locations over a 20-year period. The lack of diversity may be related to the fact that there are no ungulates native to the continent and that BVDV may have been introduced to Australia with the importation of cattle, goats, or sheep. In contrast to the United States, modified live BVDV vaccines have never been used in Australia. Although it is tempting to speculate that this has limited the genetic diversity in Australia, the diversity of observed BVDV-1 subgenotypes in European countries does not correlate with the use of modified live vaccines (Table 5). The geographic isolation and import restrictions in Australia have also probably limited the genetic variability of BVDV isolates. The single isolate of the type 1b subgenotype is of interest. Although the isolate cannot be linked directly to the known importation of a PI animal, it is likely that this virus is linked to importations of genetic materials from North America or England, the sources of most imported bovine genetics, prior to the introduction of more stringent import requirements (P.D. Kirkland, unpublished data, 2009).
Cross-neutralization as a percentage of homologous neutralization. *

BVDV = Bovine viral diarrhea virus; BDV = Border disease virus.
Coefficients of antigenic similarity (R) between different pestivirus species and subgenotypes. *

BVDV = Bovine viral diarrhea virus; BDV = Border disease virus.
The number of isolates characterized in the current study is larger than the majority of previously published surveys. Although there is no accepted definition of the number of isolates that must be examined to determine subgenotype prevalence, surveys based on less than 50 isolates are probably not as reliable for prevalence determination as larger surveys. This is particularly true if care is not taken to compensate for multiple isolates from the same outbreak.
Summary of studies characterizing Bovine viral diarrhea virus (BVDV) subgenotypes isolated from 19 different countries. *
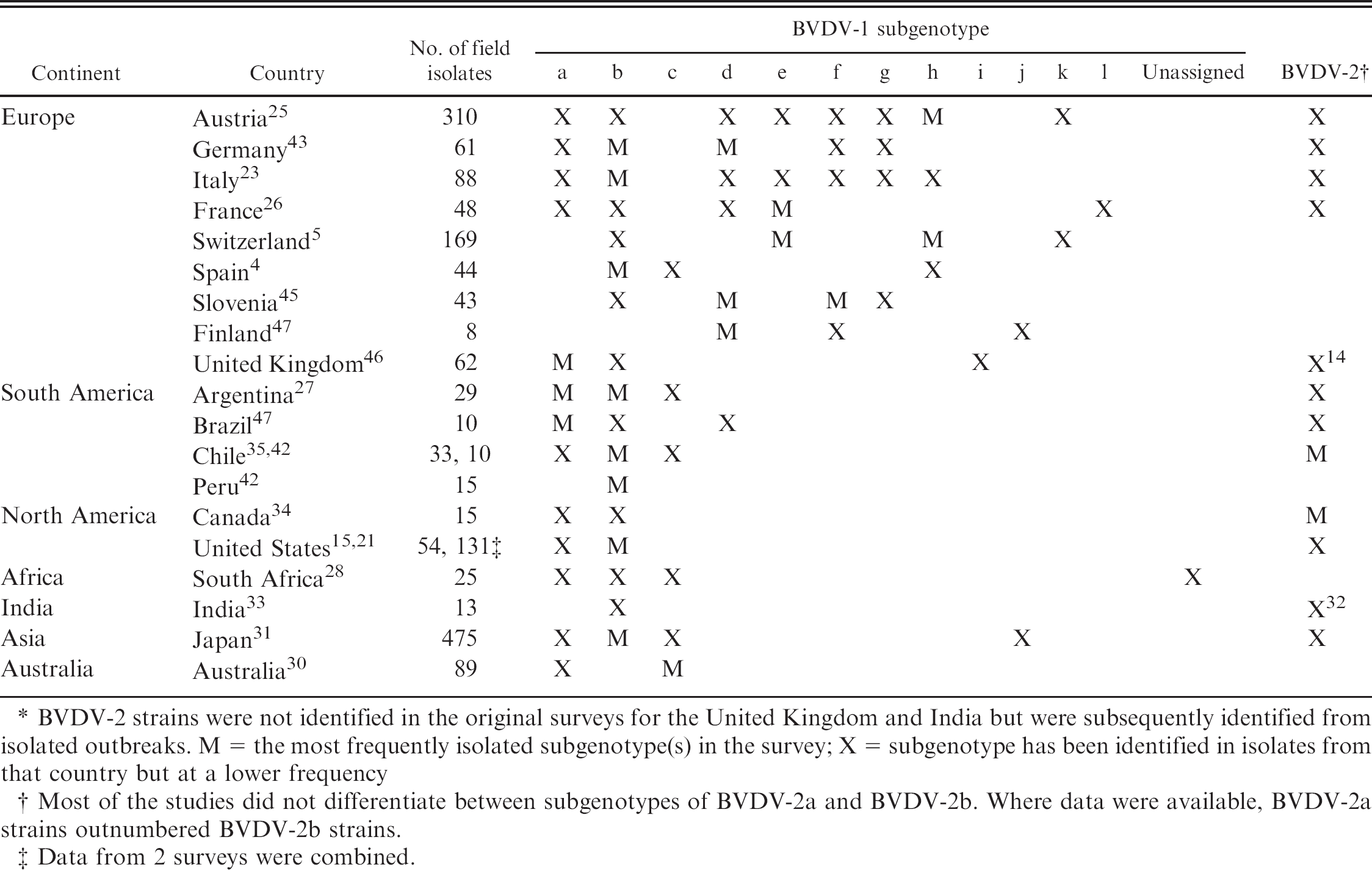
BVDV-2 strains were not identified in the original surveys for the United Kingdom and India but were subsequently identified from isolated outbreaks. M = the most frequently isolated subgenotype(s) in the survey; X = subgenotype has been identified in isolates from that country but at a lower frequency
Most of the studies did not differentiate between subgenotypes of BVDV-2a and BVDV-2b. Where data were available, BVDV-2a strains outnumbered BVDV-2b strains.
Data from 2 surveys were combined.
Neutralizing antibodies observed following vaccination with commercial Bovine viral diarrhea virus (BVDV) vaccines.
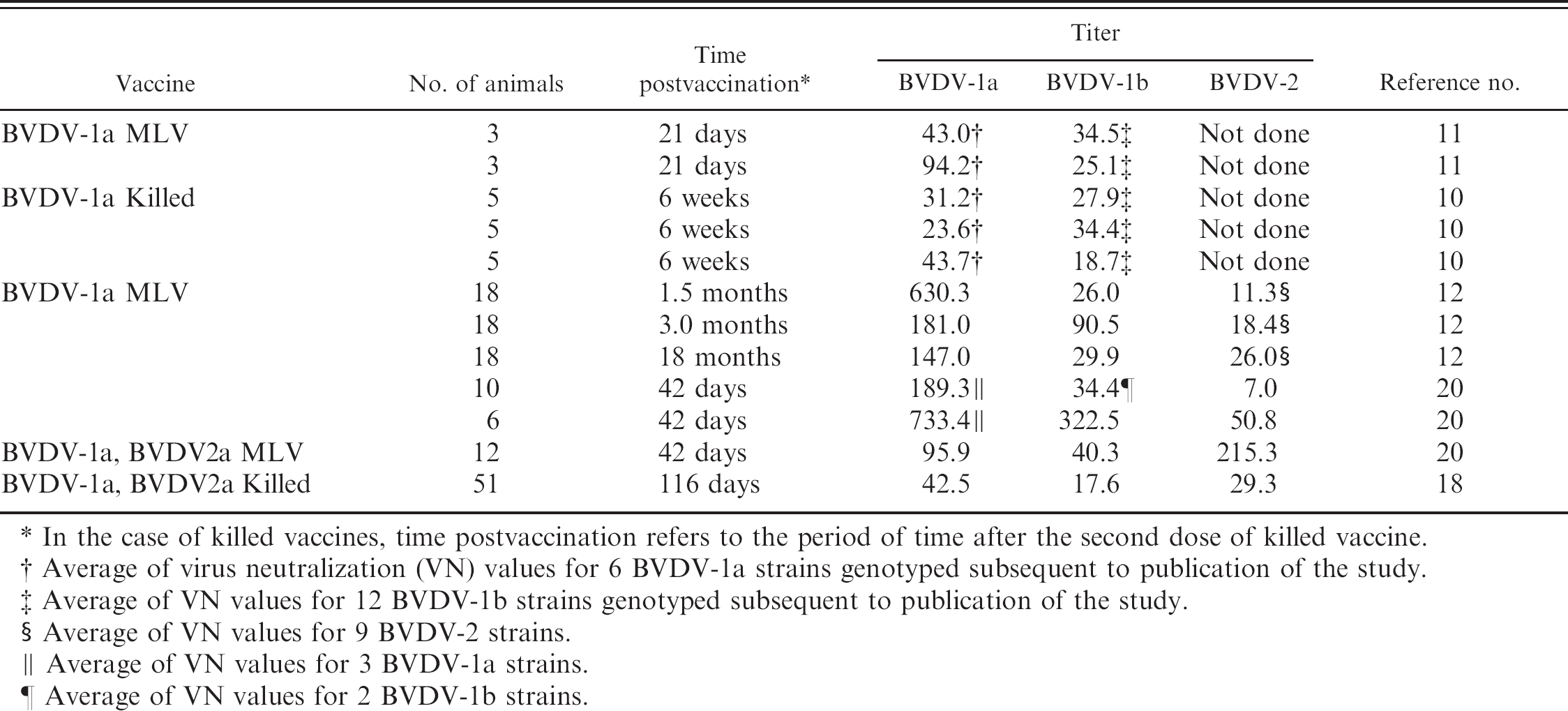
In the case of killed vaccines, time postvaccination refers to the period of time after the second dose of killed vaccine.
Average of virus neutralization (VN) values for 6 BVDV-1a strains genotyped subsequent to publication of the study.
Average of VN values for 12 BVDV-1b strains genotyped subsequent to publication of the study.
Average of VN values for 9 BVDV-2 strains.
Average of VN values for 3 BVDV-1a strains.
Average of VN values for 2 BVDV-1b strains.
In the present study, antigenic variation based on differences in calculated R-values was observed between pestivirus species (BDV, BVDV-1, and BVDV-2) and between BVDV-1 subgenotypes (BVDV-1a–c). The variation between species was greater than the variation between subgenotypes. The antigenic variation between the BVDV-1 and BVDV-2 species has led to the inclusion of BVDV-2 strains in some vaccines. 36 It is not known if antigenic variations among subgenotypes are great enough to influence detection or the protection afforded by vaccination. Previous studies have shown antigenic differences based on cross-neutralization, 5,35 monoclonal antibody binding, 11 and response of PI animals to vaccination. 22 In a study 5 comparing 7 different BVDV-1 subgenotypes isolated from cattle in Switzerland, a variation was noted in the coefficient of antigenic similarity among subgenotypes similar to that seen in the present study. In the Swiss study, antisera collected from animals vaccinated using a BVDV-1a killed vaccine neutralized all 7 subgeno-types. However, neutralization of viruses from BVDV-1 subgenotypes different from the BVDV-1 subgenotype of the vaccine virus (heterologous neutralization) varied from 34% to 51% of the VN values of homologous neutralization. A summary of reports detailing antibody responses observed in cattle following vaccination with commercial vaccines is shown in Table 6. Although the practical significance of these antigenic differences among subgeno-types is undetermined, lower VN values for BVDV-1b strains were observed following vaccination with both killed and modified live vaccines containing only BVDV-1a strains or both BVDV-1a and BVDV-2a strains for 11 of the 12 vaccine studies listed in Table 6. It is interesting to note that inclusion of both a BVDV-1a strain and a BVDV-2a strain, which theoretically should increase the antigenic range of the vaccine protection, did not alleviate the lower VN values for BVDV-1b strains. Is the reduced neutralization of BVDV-1b strains of practical significance? Although there is currently no definitive answer to this question, there are published studies regarding the level of neutralizing antibody associated with prevention of viremia and clinical disease and the transmission of virus by PI animals in the face of vaccination. In a study assessing protection afforded by passively acquired neutralizing antibodies, 8 it was found that fever and systemic spread of virus were detected in calves that had viral neutralizing titers of 256 or lower following experimental exposure to a highly virulent BVDV-2 strain. Calves that had viral neutralizing titers lower than 16 developed clinically severe disease. In a study of transmission of virus to vaccinated calves exposed to cattle persistently infected with BVDV-1b, 19 it was shown that titers up to 64 did not prevent BVDV-1b from being isolated after exposure. In a study assessing fetal protection in cattle, 24 both nonvaccinated and vaccinated pregnant cattle were exposed to 4 PI animals of which 2 carried a BVDV-1b strain and 2 carried a BVDV-2a strain. Vaccinated animals received 2 doses of a killed vaccine containing a BVDV-1a strain and a BVDV-2a strain. Titers in vaccinated animals averaged 724 against a BVDV-1a strain and 351 against a BVDV-2a strain prior to exposure to PI animals. Fetuses were harvested at approximately 150 days gestation. BVDV could be isolated from brain tissue of 14 out of 14 fetuses of nonvaccinated animals and from 4 out of 14 fetuses of vaccinated animals. Eight of the fetuses, from nonvaccinated cattle, carried the BVDV-2 strain (57%) and 6 carried the BVDV-1b strain (43%), based on virus isolation from brain tissue. In contrast, the BVDV-2 strain could be isolated from brain tissue of only 1 of the 4 infected fetuses of vaccinated animals (25%), while the BVDV-1b strain was isolated from brain tissue of all 4 (100%). These studies suggest that the cross-neutralization between BVDV-1a or BVDV-2a strains may not be great enough to prevent infection with BVDV-1b strains.
In the last decade, there has been a consolidation of veterinary biologics companies accompanied by a reduction in the range of strains used in BVDV vaccines. In the United States, these strains belong predominately to the BVDV-1a and BVDV-2a genotypes. 20,36 In particular, the assimilation of regional companies has resulted in a removal from the market of BVDV vaccines based on strains endemic to the region. Large international companies offer BVDV vaccines based on a limited number of strains. Although further research is necessary, variation in prevalence of BVDV subgenotypes and variation in cross-protection between BVDV subgenotypes suggest vaccine protection may be improved by basing vaccines on the BVDV subgenotypes prevalent in the region in which the vaccine is to be used.
Acknowledgements
The authors thank Patricia Federico and Kathryn Fulk for technical support and Harold Ridpath for statistical analysis. Disclaimer: Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Footnotes
a.
GIBCO®, Life Technologies Corp., Carlsbad, CA.
b.
Cappel Laboratories Inc., Cochranville, PA.
c.
Zymed Laboratories Inc., San Francisco, CA.
