Abstract
A 10-month-old, female African pygmy falcon (Polihierax semitorquatus) hatched and housed at the San Diego Zoo developed neurologic signs and died from a cerebral infection with the rat lungworm Angiostrongylus cantonensis. There was an associated mild nonsuppurative meningoencephalitis. This infection was diagnosed on histology and confirmed by detection of species-specific A. cantonensis DNA in formalin-fixed and frozen brain tissue by a polymerase chain reaction assay. To the authors’ knowledge, this infection has not previously been reported in a bird in the United States and has not been known to be naturally acquired in any species in this region of the world. The source of the infection was not definitively determined but was possibly feeder geckos (Hemidactylus frenatus) imported from Southeast Asia where the parasite is endemic.
The rat lungworm (Angiostrongylus cantonensis) is a metastrongyle nematode endemic to Southeast Asia, Australia, the Pacific Islands (including Hawaii), and the Caribbean Islands. In the mainland United States, this parasite has become established in Louisiana, and an isolated case was reported in a captive gibbon in Florida.1–3,5 Rats are the definitive host of A. cantonensis, with the adult nematodes found in the pulmonary arteries. First-stage larvae are shed in the feces and develop into infective third-stage larvae in a snail or slug intermediate host.7,14 Rats and aberrant hosts are infected on ingestion of the intermediate host, contaminated produce, or a paratenic host, which can include planarians, crustaceans, frogs, and lizards. 14 Within the rat, parasite migration involves the central nervous system (CNS), especially the rostral cerebrum; in aberrant hosts, this CNS migration can result in inflammation, primarily recognized as eosinophilic meningitis or meningoencephalitis in human beings.7,14
Angiostrongylus cantonensis infections in birds have previously only been reported in Australia. Cases have been described in tawny frogmouths (Podargus strigoides), captive gang-gang cockatoos (Callocephalon fimbriatum), and a captive yellow-tailed black cockatoo (Calyptorhynchus funereus).4,8,13 All birds presented with neurologic signs, including depression, paresis, or paralysis, and inability to perch or fly. Larval A. cantonensis were identified in sections of brain and spinal cord histologically and, in several cases, by direct examination of larvae retrieved from these tissues.4,8,13 The current report describes a single fatal case of neuroangiostrongyliasis in a captive falcon in California that was detected by histology and confirmed by polymerase chain reaction (PCR). The presence of A. cantonensis in a bird outside the known range of this parasite was concerning for the possible spread of A. cantonensis to this region. However, another potential source of infection in this case could have been feeder geckos imported from Southeast Asia.
A 10-month-old, female African pygmy falcon (Polihierax semitorquatus) hatched and raised at the San Diego Zoo in San Diego, California, was found on the floor of its enclosure unable to stand or fly. The bird was being housed in an outdoor enclosure, and the diet consisted of mice, gecko lizards, which were fed live, or freshly killed, commercial ground meat, a mealworms, and crickets. The geckos were common house geckos (Hemidactylus frenatus) imported live from Southeast Asia and obtained through a North American distributor. Their exact duration in the United States is unknown. On physical examination, the bird appeared depressed but was responsive to handling. It was unable to stand but was able to move all limbs and had normal cloacal tone. Head tremors and hind limb rigidity were seen intermittently. Other physical and neurologic examination findings were within normal limits. A complete blood cell count, serum chemistry panel, and radiographs were unremarkable, except for mild increases in aspartate transaminase (368 U/l) and creatine phosphokinase (2140 U/l), which were attributed to handling and intramuscular injections. Choanal and cloacal swabs were sent to a commercial diagnostic laboratory for West Nile virus testing by PCR assay; the test was negative. The bird was given supportive care with subcutaneous fluids and assisted feeding and was treated once per day with oral itraconazole b (10 mg/kg), enrofloxacin c (30 mg/kg), and ponazuril d (20 mg/kg). Intramuscular sodium phosphate dexamethasone e (2 mg/kg) was given twice the first day, then once a day for 2 days. The bird showed slight improvement neurologically but died 10 days after presentation.
A complete necropsy was performed within hours of death, and no significant lesions were seen grossly. Tissue sections collected at necropsy were fixed in 10% neutral buffered formalin, routinely processed and embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. Sections of brain examined histologically revealed multiple sections of a nematode parasite within the submeningeal parenchyma of the dorsal aspect of the right cerebrum. The parasite cross-sections were 150–200 µm in diameter and characterized by a thin smooth cuticle, polymyarian-coelomyarian musculature, basophilic accessory hypodermal cords, a pseudocoelom, a digestive tract composed of few, multinucleated cells, and 2 genital tubes (Fig. 1). This appearance was consistent with an immature female metastrongyle nematode. The adjacent leptomeninges were mildly thickened with hyperplasia, hemorrhage, and small numbers of lymphocytes and histiocytes. There was similar mild nonsuppurative inflammation and hemorrhage in the surrounding cerebral parenchyma and hemorrhage in the right lateral ventricle. Multifocally, in other areas of the brain, particularly the cerebellum, were poorly defined foci of meningitis, chronic malacia, gliosis, and Purkinje cell degeneration, mineralization, and loss. Other tissues, including spinal cord, were unremarkable.
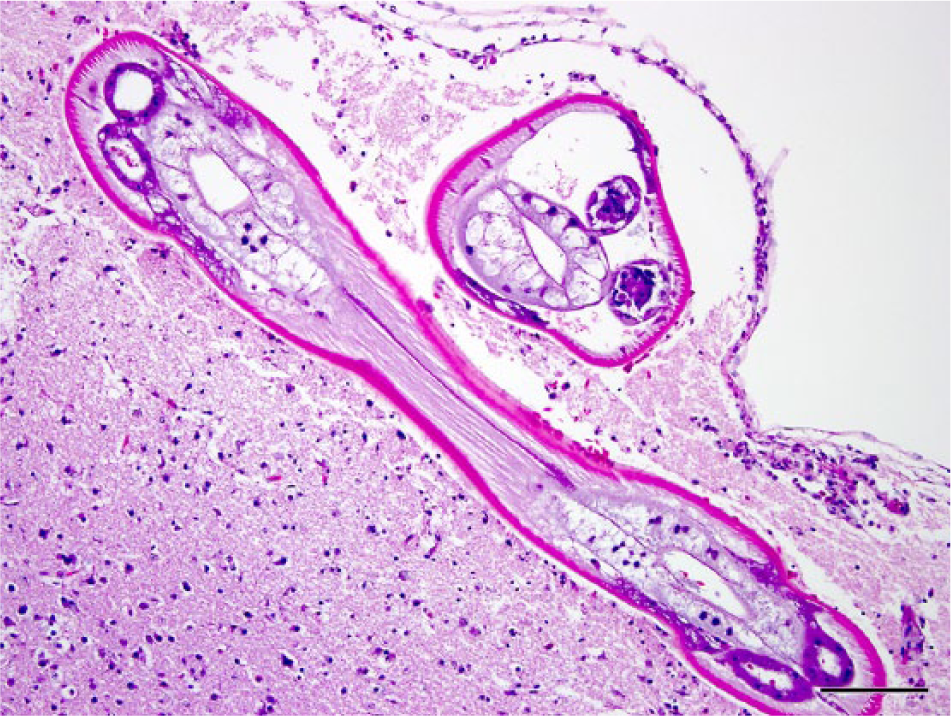
Brain; African pygmy falcon (Polihierax semitorquatus). Longitudinal and cross-sections of an immature metastrongyle nematode parasite are present in the submeningeal cerebral parenchyma. There is a mild nonsuppurative inflammatory infiltrate in the overlying leptomeninges. Hematoxylin and eosin. Bar = 100 µm.
At the time of necropsy, a small section of cerebrum was collected and frozen at −70°C. Three 10-µm-thick sections of formalin-fixed, paraffin-embedded brain containing sections of the nematode were collected in a semi-sterile manner to eliminate contamination with other sources of DNA. Using a commercial kit, f DNA was extracted from paraffin-embedded tissue sections and frozen brain. A real-time PCR assay developed to specifically detect A. cantonensis in host animals was performed as previously described. 10 Both the formalin-fixed and the frozen brain tissue samples tested positive for A. cantonensis DNA in this assay.
The clinical signs and death of the African pygmy falcon were due to cerebral infection with A. cantonensis as determined by characteristic histologic findings and identification of species-specific DNA sequences by PCR. The current case was similar to previous cases reported in birds, which have included tawny frogmouths and 2 species of cockatoos, all from Australia. As in the Australian cases, this African pygmy falcon presented with neurologic signs that included depression, weakness, ataxia, inability to fly, and hind limb paralysis.4,8,13 Differential diagnosis for the presentation includes head trauma, intoxications, and various infections. In human beings, a history of potential exposure, such as consumption of intermediate or paratenic hosts, analysis of cerebrospinal fluid, and results of serologic tests for A. cantonensis antigens or antibodies often lead to a diagnosis. 14 However, these methods may not be practical or available for birds, which makes antemortem diagnosis difficult. In 3 tawny frogmouths, presumptive antemortem diagnosis was only made following postmortem diagnosis of this condition in several other birds from the same area in the same time period. 4 Specific treatment of the 3 birds with different anthelmintics resulted in improvement in all, but full recovery only in 1 bird, which had also been given prednisolone. 4 Combined anthelmintic and corticosteroid administration is a common successful treatment modality in human beings; however, most human cases are not fatal and can resolve without treatment. 14 Additional investigation into antemortem diagnostic testing and treatment for avian angiostrongyliasis is therefore required.
Diagnosis of angiostrongyliasis in most birds has been postmortem. Immature metastrongyle nematodes are recognized histologically in sections of brain and spinal cord and are associated with variable degrees of nonsuppurative or granulomatous inflammation in the parenchyma and meninges.4,8,13 Nematodes or associated inflammatory or degenerative changes have been described in all areas of the brain. In gang-gang cockatoos, migrating larvae thought to be A. cantonensis were also seen in granulomas in the lung and wall of the gastrointestinal tract in association with arterioles. 13 In the falcon in the current report, the nematodes were only seen in the dorsal submeningeal parenchyma of the cerebrum, but there were also changes in the cerebellum and right lateral ventricle that were presumably caused by the parasite migration. Previously, in avian cases, the larvae and subadult nematodes have been definitively identified by examination of intact worms retrieved from formalin-fixed and unfixed neural tissue.4,8,13 The histologic features of the nematodes seen in the brain of the African pygmy falcon described herein were consistent with a metastrongyle nematode, such as A. cantonesis. 3 For definitive identification, DNA was extracted from sections of cerebrum that contained the parasite, and a species-specific real-time PCR assay was used to detect the DNA from A. cantonensis.
The source of infection in this falcon was not readily apparent. It had been hatched and raised in San Diego, and A. cantonensis is not known to be present endemically in southern California or most of the mainland United States, except in the region of Louisiana.1,5,14 Therefore, there was initial concern that the parasite could have spread to this region. Because it is a port city, San Diego is at risk for introduction of A. cantonensis in infected rats or intermediate hosts, such as the giant African snail (Achatina fulica), which can carry large numbers of A. cantonensis larvae. 6 Though numerous species of snails and slugs can be intermediate hosts of this organism, the dissemination of the giant African snail in particular has paralleled the establishment of A. cantonensis in new areas. 6 This invasive snail has previously been intercepted at U.S. ports, and small populations have been eradicated in several states. 6 The number of cases of angiostrongyliasis in tawny frogmouths is presumably related to their consumption of slugs and snails as part of their natural diet.4,8 The falcon in the present report was housed in an outdoor enclosure and could have encountered snails or slugs, although this had not been noted. As well, giant African snails have not been reported in the San Diego area. Opportunistically, over 20 wild Norway rats caught or found dead on zoo grounds were screened for the presence of metastrongyle larvae in feces using direct examination in saline or a modified Baermann technique. No metastrongyle larvae were detected (data not shown). In contrast, screening of rats in New Orleans after establishment of the parasite found that 20 of 94 rats were infected. 1 Therefore, the prevalence of the parasite, if present at all, does not appear to be high in the San Diego Zoo area.
Additional investigation into the diet of the falcon revealed that it had been fed live or freshly killed common house geckos that were collected in Southeast Asia where A. cantonensis is endemic. The geckos had been imported into the United States and were not bred prior to being obtained live from a distributor and fed to the falcon. Though geckos are not specifically known to be paratenic hosts of this parasite, raw consumption of monitor lizards has been implicated in several human infections.9,11 The yellow tree monitor (Varanus bengalensis) has been shown to be a viable paratenic host, and a high proportion of these snail-eating lizards were found to be infected in Thailand.11,12 Therefore, other lizard species could also potentially serve as paratenic hosts, though this has not been investigated. Unfortunately, by the time of diagnosis, none of the geckos from the batch fed to the falcon were available for testing. The current case illustrates the difficulty in determining the source of an infection outside the known geographical range, as well as the potential dangers of feeding wild-caught, exotic, or unscreened prey items to zoo animals.
Footnotes
Acknowledgements
The authors wish to thank Dr. Michael Schlegel for information on the diet, April Gorow for necropsy assistance, and Yvonne Cates for histology.
a.
Zoo Carnivore Diet 5, Dick Van Patten’s Natural Balance Pet Food Inc., Burbank, CA.
b.
Itraconazole, Centocur Ortho Biotech Products LP, Rantan, NJ.
c.
Enrofloxacin, Bayer HealthCare LLC, Animal Health Division, Shawnee Mission, KS.
d.
Ponazuril, Bayer HealthCare LLC, Animal Health Division, Shawnee Mission, KS.
e.
Sodium phosphate dexamethasone, Bimeda-MTC Animal Health Inc., Cambridge, Ontario, Canada.
f.
QIAamp DNA mini kit, Qiagen Inc., Valencia, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
