Abstract
A 6-year-old female Parma wallaby (Macropus parma) at a zoo in California developed acute ataxia and left-sided circling. Despite intensive care, clinical signs progressed to incoordination and prostration, and the animal was euthanized. At necropsy, the left tympanic cavity was filled with homogeneous suppurative exudate that extended into the cranium expanding the meninges and neuroparenchyma in the lateral and ventral aspect of the caudal ipsilateral brainstem and medulla oblongata. Microscopically, the brainstem showed regional severe suppurative meningoencephalitis with large numbers of neutrophils, fewer macrophages, and lymphocytes admixed with fibrin, necrotic cellular debris, hemorrhage, and mineralization, with numerous intralesional Gram-negative bacilli. Bacteroides spp. and Porphyromonas spp. were isolated on anaerobic culture from the meninges, and the bacteria were further characterized by partial 16S ribosomal RNA gene sequencing as Bacteroides tectus and Porphyromonas gulae. Bacterial aerobic culture from the meninges yielded very low numbers of mixed flora and Proteus spp., which were considered contaminants. Culture of Mycoplasma spp. from middle ear and meninges was negative. Additionally, Toxoplasma gondii cysts were detected by immunohistochemistry in the heart and brain, and anti-Toxoplasma antibodies were detected in serum. The genera Bacteroides and Porphyromonas have been associated with oral disease in marsupials; but not with otitis and meningoencephalitis. The results of the present work highlight the importance of performing anaerobic cultures in the diagnostic investigation of cases of suppurative otitis and meningoencephalitis in macropods.
Anaerobic bacteria comprise a large percentage of the normal flora of the digestive and genital tracts of human beings and animals; however, they can be opportunistic pathogens when the balance of normal flora is disrupted or when they colonize abnormal anatomic sites. 15 Bacteroides spp. are obligate anaerobic, non–spore-forming, Gram-negative rods, and have been isolated from a variety of tissues under normal and pathologic conditions in wild and domestic animals and human beings.1,2,5 In people, Bacteroides spp. have been associated with chronic suppurative otitis and intracerebral abscesses in children,5,10 periodontal disease and periodontitis,2,24,28 and infected dog and cat bite wounds.1,27 In cats, subcutaneous abscesses, pyothorax, and gingivitis are the most common pathologic conditions caused by this bacterium. 15 Bacteroides spp. can be isolated from the oral cavity in normal individuals of various animal species2,15; however, in an extensive study of oral flora in 11 species of Australian marsupials, these bacteria were not recovered on anaerobic culture from any of the 90 animals sampled, 3 suggesting either that this genus is not part of their normal oral flora or that these bacteria are present in such low numbers that they cannot be recovered by this technique.
Porphyromonas spp., strictly anaerobic, Gram-negative, non–spore-forming, and nonmotile asaccharolytic bacilli, have been associated with oral diseases in both human beings and animals,12,13,16 and have been isolated from children with chronic suppurative otitis media. 5 Porphyromonas spp. are normal anaerobic flora of the feline and human oral cavity,7,15 and are frequently isolated from infected dog and cat bite wounds in human beings.7,27 Porphyromonas gingivalis is the species found most frequently in the oral cavity of healthy people, and it is also recognized as an important periodontal pathogen that has been shown to produce virulence factors such as collagenase, protease, and cytotoxin. 11 Porphyromonas gulae has been isolated from various species of wild and domestic animals (cats, dogs, bears, nonhuman primates, wolves, and a coyote) held in captivity in Australia, the United States, Canada, and Japan, with some isolates being associated with gingivitis and periodontitis. 9 In Australian marsupials, black-pigmented anaerobic bacteria belonging to the genus Porphyromonas were demonstrated to be part of the normal microbiota of the oral cavity of macropods, koalas (Phascolarctos cinereus), brushtail possums (Trichosurus vulpecula), and bandicoots (Isoodon obesulus). 3 Additionally, the species P. gulae has been identified by 16S ribosomal RNA (rRNA) gene sequencing from wild and captive Australian marsupials with varying degrees of periodontal disease. 15
Bacterial otitis media is a rare condition in marsupials. A single published case report in a red kangaroo (Macropus rufus) described this condition associated with Pasteurella multocida and Klebsiella pneumoniae infection. 21 The aim of the present work is to report and describe a spontaneous case of suppurative otitis media and ascending meningoencephalitis caused by coinfection with Bacteroides tectus and Porphyromonas gulae in a captive Parma wallaby (Macropus parma) with neurologic disease and chronic toxoplasmosis, and the diagnostic investigations conducted to confirm the diagnosis.
In October 2010, a 6-year-old female Parma wallaby at a zoo in California developed clinical signs of a staggered gait, ataxia, proptosis of the left eye with slight conjunctival discharge and corneal opacity, and left eye nystagmus with no blink reflex, which progressed to bilateral nystagmus. Despite intensive care and treatment with meloxicam and azithromycin, clinical signs progressed to incoordination and prostration after 2 days. The animal was humanely euthanized and the carcass submitted to the Davis branch of the California Animal Health and Food Safety Laboratory System, of the University of California, Davis, for postmortem examination and diagnostic workup.
A complete necropsy was done, and samples of brain (cerebral cortex, brainstem, and cerebellum), heart, lung, liver, skin, trachea, esophagus, skeletal muscle, tongue, thyroid glands, kidneys, spleen, adrenal gland, small intestines, and colon were collected for histology. Tissues were immersion fixed in 10% neutral buffered formalin (pH 7.2) for 24 hr, routinely embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin for microscopic examination. Selected brain sections were also stained with tissue Gram stain. Immunohistochemistry (IHC) was performed as previously described 19 on sections of heart and brainstem with rabbit primary polyclonal antisera against Toxoplasma gondii, using a commercially available kit. a Appropriate positive and negative controls were used for the Gram stain and T. gondii IHC.
Aerobic cultures (5% sheep blood agar and chocolate agar at 35–39°C in 5–10% CO2 for 48 hr; MacConkey agar at 35–39°C for 48 hr) were performed on liver, lung, brain, and swabs from the middle ear and leptomeninges. Additionally, brain and middle ear swabs were cultured anaerobically on prereduced phenyl ethyl alcohol agar and Brucella blood agar at 35–39°C for 5 days) and for Mycoplasma spp. (plated directly onto pleuropneumonia-like organism [PPLO] agar with serum and antibiotics along with enrichment in PPLO broth followed by a second culture on PPLO). Anaerobic isolates were identified to genus level by standard biochemical methods (sensitivity to colistin, vancomycin, kanamycin, and bile, production of indole, lecithinase, and lipase), and further speciated by partial 16S rRNA gene sequencing using standard methods. Briefly, genomic DNA was extracted using phenol–chloroform as previously described. 20 A portion of the variable region of the 16S rRNA gene was amplified using the forward primer 5′-AGAGTTTGATCCTGGCTCAG-3′ and reverse primer 5′-GGTTACCTTGTTACGACTT-3′. Polymerase chain reaction (PCR) was performed in a 50-µl reaction mixture containing 2 µl of DNA, 1× PCR buffer, 2.5 mM of MgCl2, 150 µM of each deoxyribonucleotide triphosphate, 0.4 mM of each primer, and 2.5 U of DNA polymerase. The PCR setup included initial polymerase activation at 95°C for 10 min, followed by 35 cycles of denaturation at 95°C for 30 sec, annealing at 56°C for 30 sec, and extension 72°C for 1 min; with a final extension step at 72°C for 10 min. The PCR products were electrophoresed on 3% agarose gel in Tris–borate–ethylenediamine tetra-acetic acid–ethidium bromide buffer (pH 8.4) and photographed with an imaging system. b The amplicons from the 2 reactions, an 865–base pair (bp) amplicon for the Bacteroides organism and an 834-bp amplicon for the Porphyromonas organism, were purified and sequenced with the same primers used for the PCR. Lastly, acid-fast stains (Kinyoun carbol fuchsin and Ziehl–Neelsen) were performed on direct smears of brain, and Salmonella spp. The PCR was performed using a published assay validated on fecal samples. 6
A serum sample collected at necropsy was analyzed for the detection and semiquantification of antibodies against T. gondii by a latex agglutination test. 8 Fresh liver samples were digested with nitric acid and subsequently analyzed for lead, manganese, cadmium, copper, iron, zinc, molybdenum, arsenic, and mercury (heavy metal screen) by inductively coupled argon plasma–emission spectrometry, c as previously described. 17 Additionally, frozen brain tissue samples including right and left cerebral cortex, brainstem, hippocampus, and cerebellum were tested by the California Department of Public Health for Rabies virus antigen detection by a direct fluorescent antibody test (http://www.cdc.gov/rabies/pdf/RabiesDFASPv2.pdf).
At postmortem examination, the carcass was well-fleshed and had adequate body fat reserves. All tissues were in a fair state of postmortem preservation. The left tympanic cavity was filled with abundant homogeneous, greenish, creamy, suppurative exudate (Fig. 1) that extended internally into the cranium, expanding the meninges and neuroparenchyma in the lateral and ventral aspects of the caudal ipsilateral brainstem and medulla oblongata (Fig. 2). Microscopically, the brainstem showed severe suppurative and necrotizing meningoencephalitis with large numbers of neutrophils, fewer macrophages, and lymphocytes admixed with fibrin, necrotic cellular debris, hemorrhage and mineralization, with numerous dense intralesional bacterial aggregates composed of large numbers of closely packed monomorphic Gram-negative bacilli (Figs. 3, 4). The inflammatory exudate expanded the leptomeninges and extended into the underlying white matter, which was multifocally hemorrhagic, vacuolated (spongiosis), and infiltrated by moderate numbers of gitter cells, with mild to moderate perivascular histiocytic encephalitis. The meningeal lesions extended to the adjacent cerebrum and cerebellum, and there was mild early meningeal fibrosis. The subependymal connective tissue of a ventricular choroid plexus was focally infiltrated by moderate numbers of lymphocytes and macrophages. Incidentally, in the heart, occasional cardiomyocytes contained round to oval intrasarcoplasmic, thin-walled, basophilic protozoal cysts that ranged from 30 to 125 µm in major diameter and contained numerous elongated basophilic bradyzoites (Fig. 5) positive for T. gondii by IHC (Fig. 6). Similar cysts were rarely detected in the brain, in sites that were distant from the area of inflammation and necrosis. There was no inflammation associated with these cardiac and brain cysts, and T. gondii antigen was not detected intralesionally in sections of brainstem.
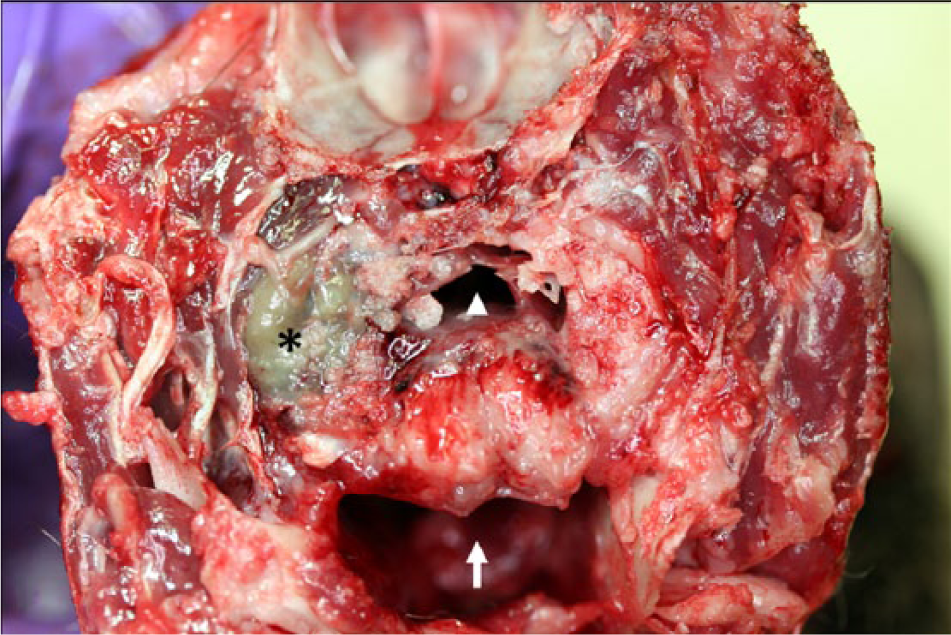
Parma wallaby (Macropus parma); head, transverse section at the level of the middle ear, caudal view. The left tympanic cavity is filled with abundant greenish suppurative exudate (asterisk), and there is associated osteomyelitis of the tympanic portion of the temporal bone. The arrow indicates the oral cavity and the arrowhead the pharynx.
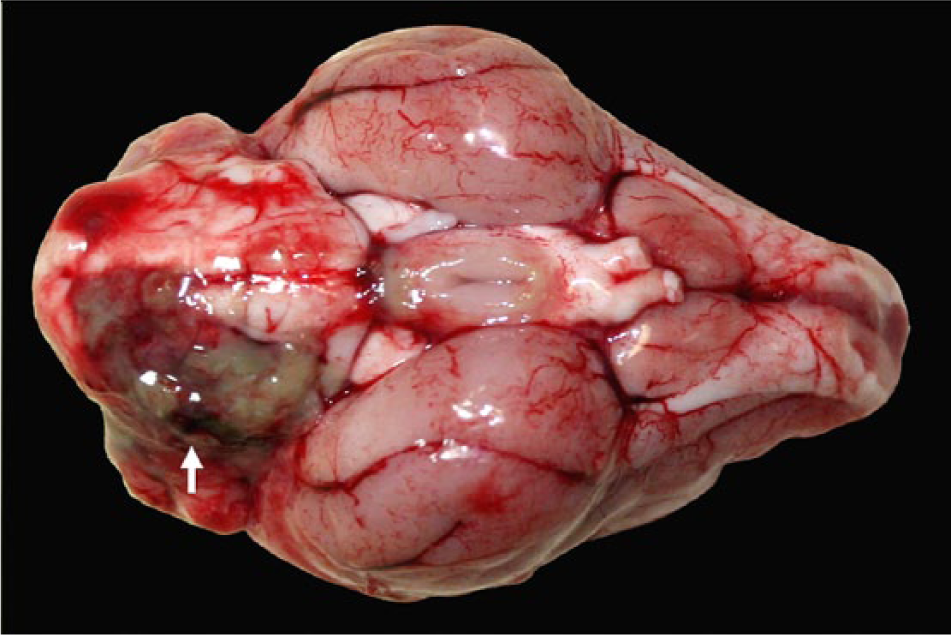
Parma wallaby (Macropus parma); brain, ventral view. Suppurative exudate expanding the leptomeninges in the left caudal brainstem, pons, and medulla oblongata (arrow). This exudate represented a continuum with the ear exudate shown in Figure 1.
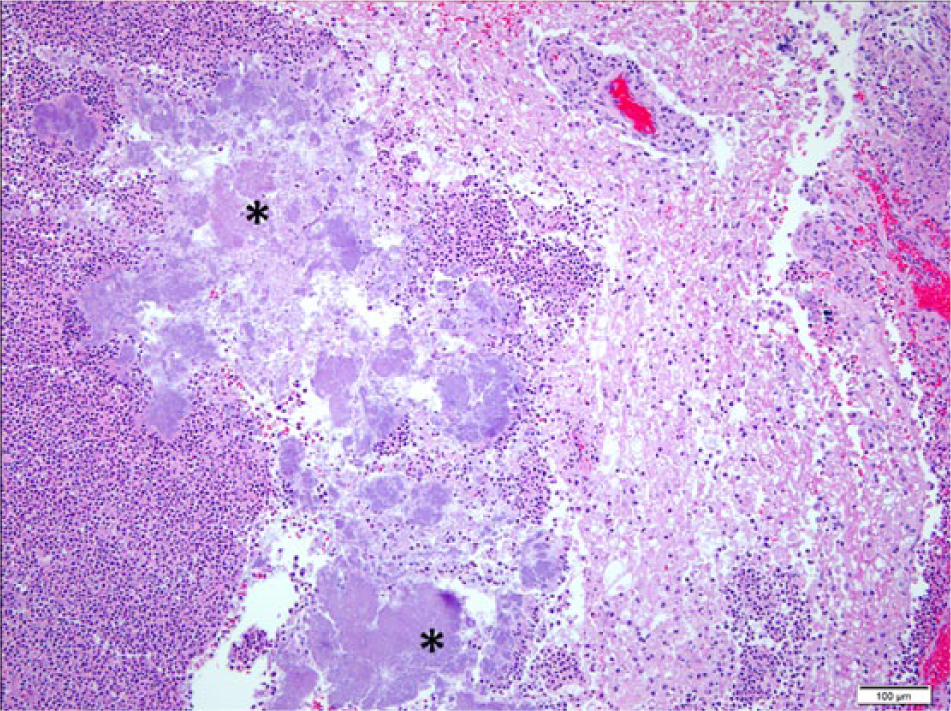
Parma wallaby (Macropus parma); histology of the brainstem. Extensive encephalomalacia with abundant neutrophilic infiltration and large numbers of intralesional dense basophilic bacterial aggregates (asterisks). Hematoxylin and eosin stain. Bar = 100 µm.
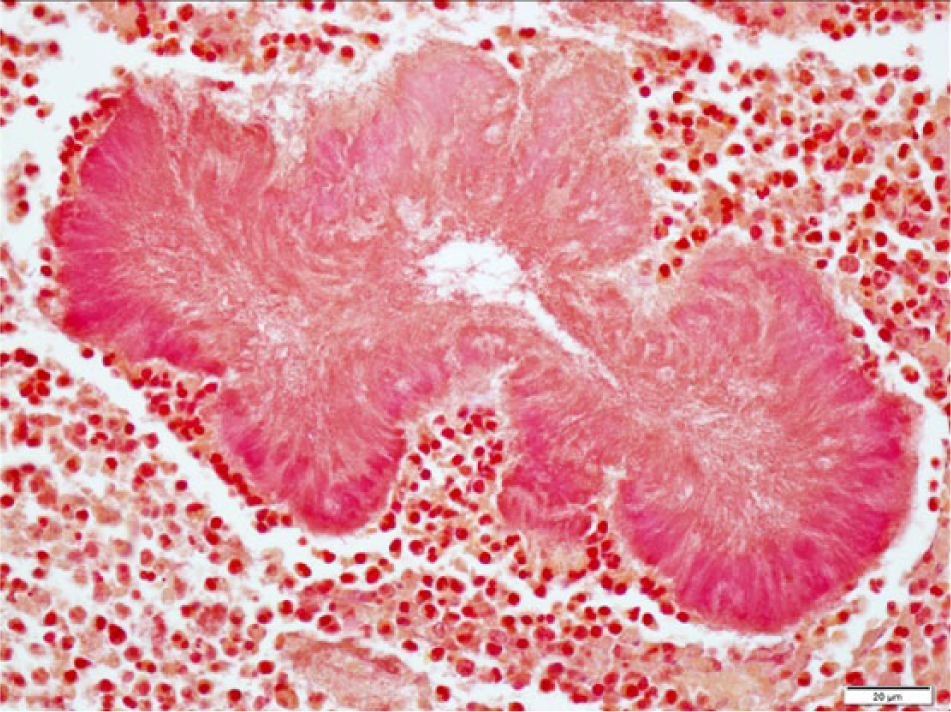
Parma wallaby (Macropus parma); histology of the brain. Well-demarcated, irregularly shaped dense bacterial aggregates (bacilli) depicted in Figure 3 stained fuchsia to pink with tissue Gram stain, characteristic of Gram-negative bacteria. Bar = 20 µm.
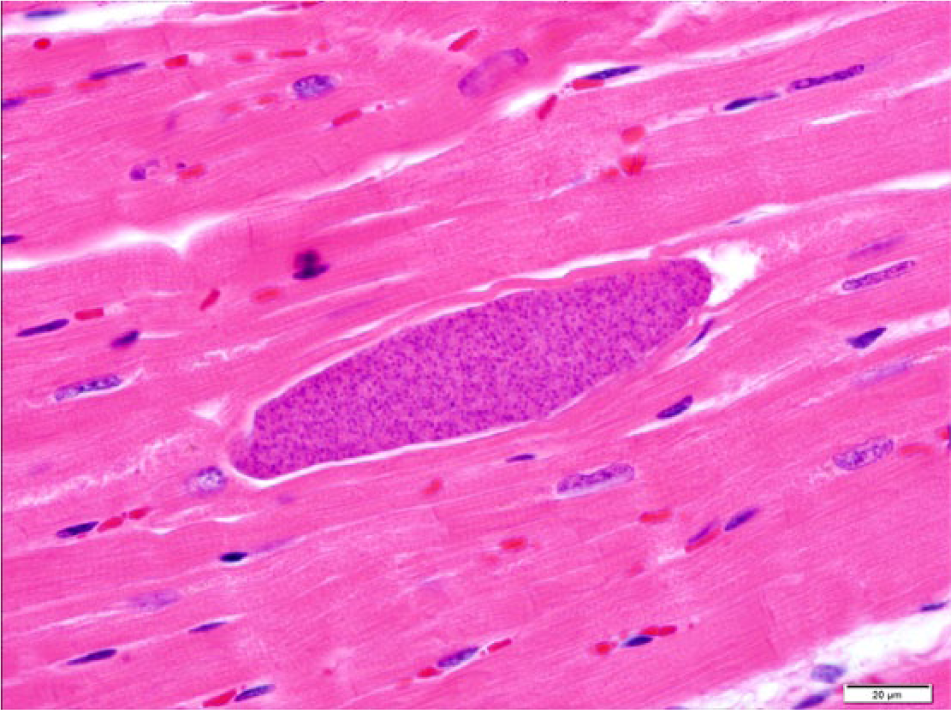
Parma wallaby (Macropus parma); histology of the heart. A cardiomyocyte contains an intrasarcoplasmic oval, 125.7 µm × 30.5 µm, thin-walled protozoal cyst containing numerous basophilic elongated bradyzoites and no associated inflammatory reaction. Hematoxylin and eosin stain. Bar = 20 µm.
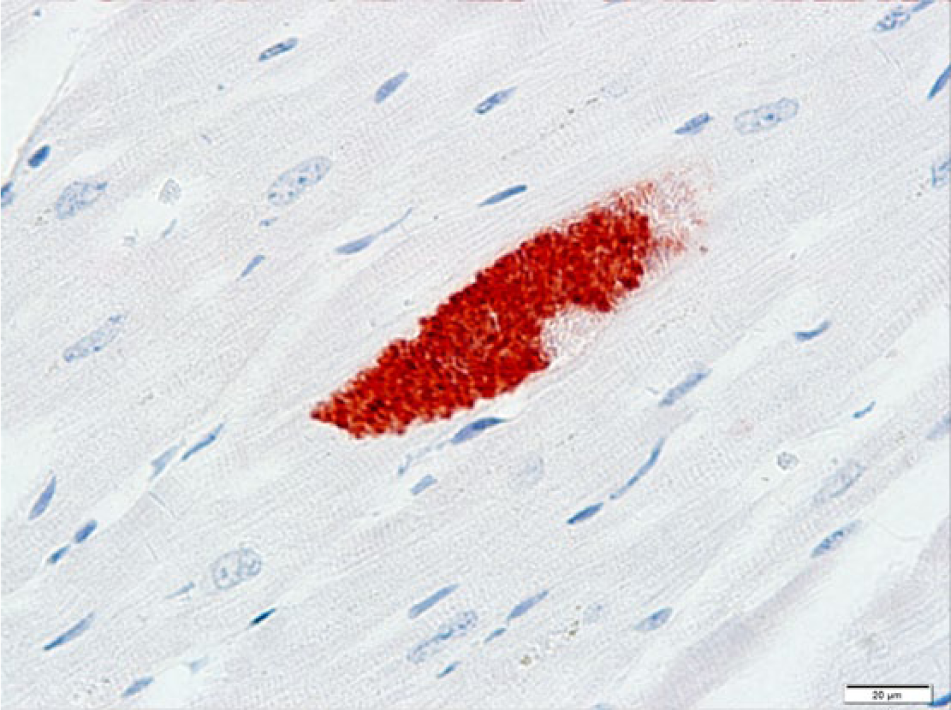
Parma wallaby (Macropus parma); heart. Bradyzoites within the protozoal cyst depicted in Figure 5 (serial section) are strongly immunoreactive with Toxoplasma gondii antisera. Immunohistochemistry for T. gondii antigen. Bar = 20 µm.
Bacteroides spp. and Porphyromonas spp. were isolated in moderate numbers on anaerobic culture from the meninges. By sequence analysis, these bacteria were speciated as Bacteroides tectus and Porphyromonas gulae. Bacterial aerobic cultures from the meninges yielded very low numbers of mixed flora and Proteus spp., which were considered contaminants. Mycoplasma spp. cultures from middle ear and meninges were negative, and no acid-fast organisms were detected on direct smears from brain. Salmonella spp. PCR on feces was negative.
The T. gondii antibody titer in serum was 1:256. The Rabies virus direct fluorescent antibody test was negative. The liver contained the tested heavy metals in concentrations not considered toxicologically significant.
The current work describes the occurrence of a spontaneous case of suppurative otitis and ascending meningoencephalitis caused by B. tectus and P. gulae in a captive Parma wallaby with neurologic disease. The diagnostic investigation ruled out other possible causes of neurologic disease such as lead and other heavy metal poisoning and rabies. Although infection with T. gondii was demonstrated by histology and IHC in brain and heart (encysted bradyzoites), as well as serology, this parasite was not detected intralesionally in brain tissue, and this protozoal infection was considered an incidental finding, probably unrelated with the lesions that lead to the clinical signs. Toxoplasma gondii encephalitis is common in captive macropods, with typical lesions being nonsuppurative to granulomatous in nature. 22 The fact that the brain lesions in this wallaby were suppurative and contained dense intralesional bacterial aggregates suggests that the bacteria played a primary causative role. However, clinical toxoplasmosis cannot be definitely ruled out, and it is possible that the protozoal infection contributed to the neurologic signs showed by the wallaby. It is worth mentioning that the demonstration of chronic T. gondii exposure in a zoo animal that had been in close contact with human beings may have importance from a public health perspective. Toxoplasmosis is well documented in macropods, and it has been reviewed previously. 22
Bacteria of the genera Bacteroides and Porphyromonas have been associated with jaw and periodontal disease in marsupials18,23,25; however, these anaerobic bacteria have not been associated with otitis and meningoencephalitis in macropods. Otitis media and/or interna in macropods have been associated with head trauma; infectious, fungal, or parasitic causes; or foreign-bodies.14,26 Bacterial otitis media is rarely described in macropods, although there is a report of this infection in a red kangaroo, 21 where the disease was attributed to P. multocida and K. pneumoniae infections; P. multocida was isolated premortem in a sample of purulent exudate from the ear canal 4 days after initial presentation, while K. pneumoniae was isolated on aerobic culture from a sample of petrosal bone collected at necropsy. There is no mention on whether anaerobic bacterial cultures were part of the diagnostic investigation in that report. 21 In the current case, the causative bacteria were only identified by anaerobic culture and 16S rRNA gene sequencing of the isolates, while aerobic and Mycoplasma spp. cultures were negative. This highlights the importance of including anaerobic bacterial cultures in the diagnostic workup of macropods with suppurative otitis and/or encephalitis.
Bacteria belonging to the genera Bacteroides and Porphyromonas have been isolated from children with chronic suppurative otitis media, 5 the intracranial complications of which are meningitis, focal encephalitis, intracranial (intracerebral, extradural, and/or subdural) abscesses, and otitic hydrocephalus. 4 Bacteroides spp. have also been associated with intracerebral abscess in children. 10 The anatomic distribution of the lesions in the wallaby of the present report suggests that the unilateral suppurative meningoencephalitis resulted as an extension from the middle and inner ear infection (ascending route). Given the prevalence of Porphyromonas spp. in the oral cavity of marsupials, 3 it is possible that this bacterium may have caused periodontal and/or root abscess with ascending otitis media and encephalitis. The dental arcade of this wallaby was not examined extensively, and the primary source of infection/contamination of the tympanic cavity was not identified. The current report indicates that anaerobic bacteria of the genera Bacteroides and Porphyromonas can cause suppurative otitis and ascending meningoencephalitis in macropods as described in human beings, and that anaerobic cultures should be included in the panel of diagnostic tests performed in cases of suppurative lesions in these species.
Footnotes
Acknowledgements
The authors thank all the bacteriology, histology, and pathology technicians from the California Animal Health and Food Safety Laboratory System for technical assistance.
a.
EnVision+ system HRP 3-amino-9-ethylcarbazole (AEC) rabbit kit, Dako North America Inc., Carpinteria, CA.
b.
FluorChem HD2 imaging system, Alpha Innotech Corp., Santa Clara, CA.
c.
FISONS, Accuris Model, Thermo Optek Corp., Franklin, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
