Abstract
The raccoon (Procyon lotor) is almost ubiquitous in North America. In recent times, it was introduced in many parts of the world where it has now become largely feral. Since the outbreak of raccoon rabies epizootic in eastern United States and Canada, most diagnostic laboratories have had increased numbers of raccoon carcasses or raccoon brain submissions for diagnosis of rabies. However, since a number of other diseases that affect the central nervous system and have similar clinical signs as rabies have been documented in this species, the current review attempts to bring together the published information on neurologic disorders of raccoons.
Introduction
In 1981, the article “Neurologic disorders in the raccoon from northeastern United States” was published, 91 which described viral, toxic, and neoplastic causes that affected the central nervous system (CNS) of raccoons (Procyon lotor). At that time, the authors were able to describe a total of 8 individual conditions in this species that affected the CNS. Since then, there has been ever-growing new information published, predominantly single case reports, on this subject. Therefore, the aim of the current review is to provide documentation of diseases and pathological conditions that have influenced the nervous system of North American raccoons since the 1980s.
Geographic distribution
Raccoons belong to the family Procyonidae, in which there are 9 genera and a total of 18 species. 28 The genus Procyon has 7 species. Procyon lotor is found over a large geographical area of North America. It ranges from southern Canada through most of the continental United States and into Central America. Raccoons have been introduced in many other parts of the world from North America. Between 1931 and 1958, over a thousand raccoons were released in widely scattered areas of the former USSR for their fur, 1 and the 1964 figures estimated that there were between 40,000 and 45,000 in that region. 1 According to a study published in 1966, the introduced animals have lost most of their North American parasites, but have acquired new species of endoparasites and ectoparasites. 1 Raccoons were introduced into German forests in the 1930s and today, as many as 1 million raccoons are estimated to be in that country. 24 Since their introduction, raccoons have moved across the border to infest most of Germany’s neighbors. Also, starting in the 1970s, raccoons were imported into Japan as pets. 83 Since then, the number of escaped and abandoned raccoons in that country has increased, and the animal has become largely feral. 83
Raccoons are primarily nocturnal and are most active at dawn and dusk (unless they suffer from neurologic disorders). They prefer to make their dens in trees near water. They are omnivorous and will flourish on a wide variety of feeds but prefer frogs, mussels, fish, small mammals, invertebrates, and a wide selection of seeds. 28
The breeding season of the raccoon is long (January –June), and it is not uncommon for females in the southern latitudes to have 2 litters per year. 28 The gestation period averages 63 days in length, but may vary from 60 to 73 days. In each litter, there are 1–7 kits (average 3–4) that weigh approximately 70 g at birth. The females begin to bear young at 1 year of age. Although the life expectancy of the raccoon can exceed 10 years in captivity, free-ranging raccoons usually do not survive beyond 2 years 78 because most raccoons do not survive predation and diseases in the wild. 78
Raccoons are highly susceptible to a variety of infectious diseases, both in free-ranging and under experimental conditions (Fig. 1). Many of these diseases are common to domestic dogs and cats. Raccoons can also transmit many diseases to human beings and animals. Since raccoons share our immediate habitat, they have been used as environmental sentinels.6,40

Three experimental raccoon (Procyon lotor) kits in a plastic tunnel (provided for enrichment of their environment) at the National Animal Disease Center, Ames, Iowa.
Diseases
The following brief description of diseases and lesions consisting of various infectious agents (predominantly viral causes), and other noninfectious conditions such as toxic, neoplastic, and other incidental but abnormal conditions have been documented in raccoons. Although many viral diseases of raccoons have been documented, only a handful of bacterial diseases, namely leptospirosis, 44 tuberculosis, 8 campylobacteriosis, 69 Tyzzer disease, 103 nocardiosis, 65 listeriosis, 51 and Staphylococcus aureus 36 have been reported in this species. However, none of these bacterial agents appeared to have caused any neurologic problems in raccoons. Similarly, only a few fungal infections, namely oral candidiasis 51 and pulmonary adiaspiromycosis, 35 have been reported in raccoons but none have involved the CNS.
Rabies
Rabies followed by canine distemper are the 2 most important diseases that affect the CNS of raccoons. Rabies virus (order Mononegavirales, family Rhabdoviridae, genus Lyssavirus) causes an acute, progressive polioencephalomyelitis, with a high mortality. Once the clinical signs of the disease appear, it is usually considered to be invariably fatal, and only a handful of clinical cases of rabies have survived the disease.57,102 The epizootic rabies among raccoons was first identified in Florida in the 1940s, and the area affected gradually expanded into other southeastern states. In the late 1970s, a second focus of rabies among raccoons emerged on the border of West Virginia and Virginia.12,25 Epizootiologic and virologic investigations indicated that this new focus resulted from the translocation of raccoons incubating rabies from the southeastern United States and, that for the first time in 1990, raccoons outnumbered skunks as the primary rabies reservoir in the United States. In 2008, there were 6,841 cases of rabies in animals, of which 2,389 (34.9%) were in raccoons. 7 Since 2000, at least 1 case of human rabies has been attributed to the raccoon Rabies virus variant. 7
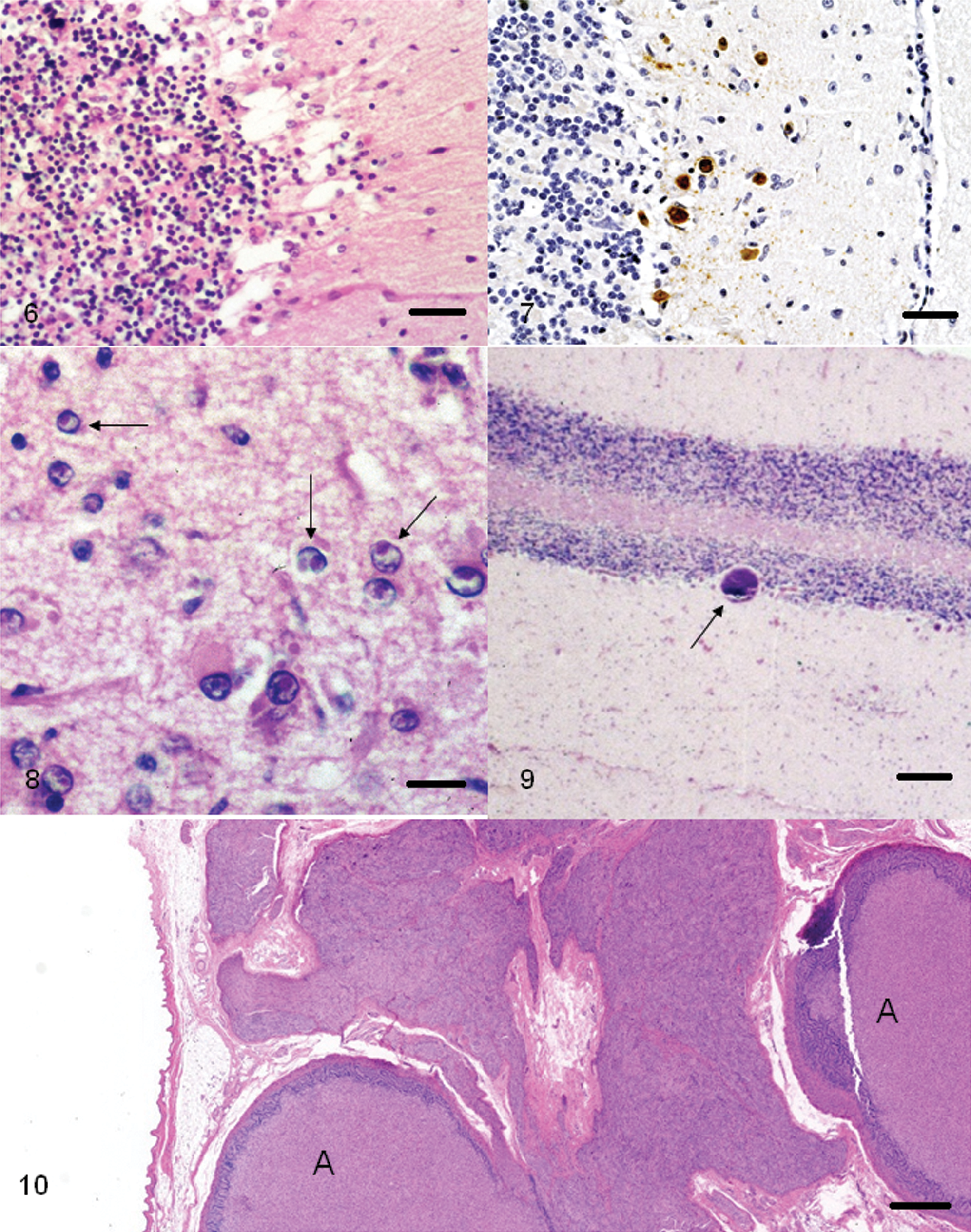
Rabid raccoons, like other infected animals, exhibit dramatic neurologic signs. At necropsy, gross lesions are not seen in the CNS.55,56 For the diagnosis of rabies, a fluorescent antibody test is used on fresh brain tissue, which is the gold standard for the confirmation of the disease. Histologically, severe and extensive histopathologic lesions of nonsuppurative polioencephalomyelitis with marked perivascular cuffing with mononuclear cells (Fig. 2) are commonly seen in the cerebrum, brainstem, and spinal cord sections of raccoons that were experimentally infected with the raccoon Rabies virus strain (Fig. 3). Those infected with a canine strain of Rabies virus had lesions confined to the brainstem and spinal cord (Fig. 3), and these raccoons died suddenly without showing marked neurologic signs. 56 Therefore, it appears that the distribution of lesions in the brain of a raccoon would depend on the source of the infection. Raccoons infected with the raccoon isolate of Rabies virus show numerous neuronal inclusions (Negri bodies) in the CNS (Fig. 4) as well as in non-CNS tissues such as tongue, salivary glands, pancreas, para-adrenal glands (Fig. 5), and intestines. In the non-CNS organs, the viral inclusions are present within ganglion cells. 55 In raccoons naturally infected in the raccoon rabies endemic areas, the immunohistochemical labeling for the antigen was widely dispersed. 99
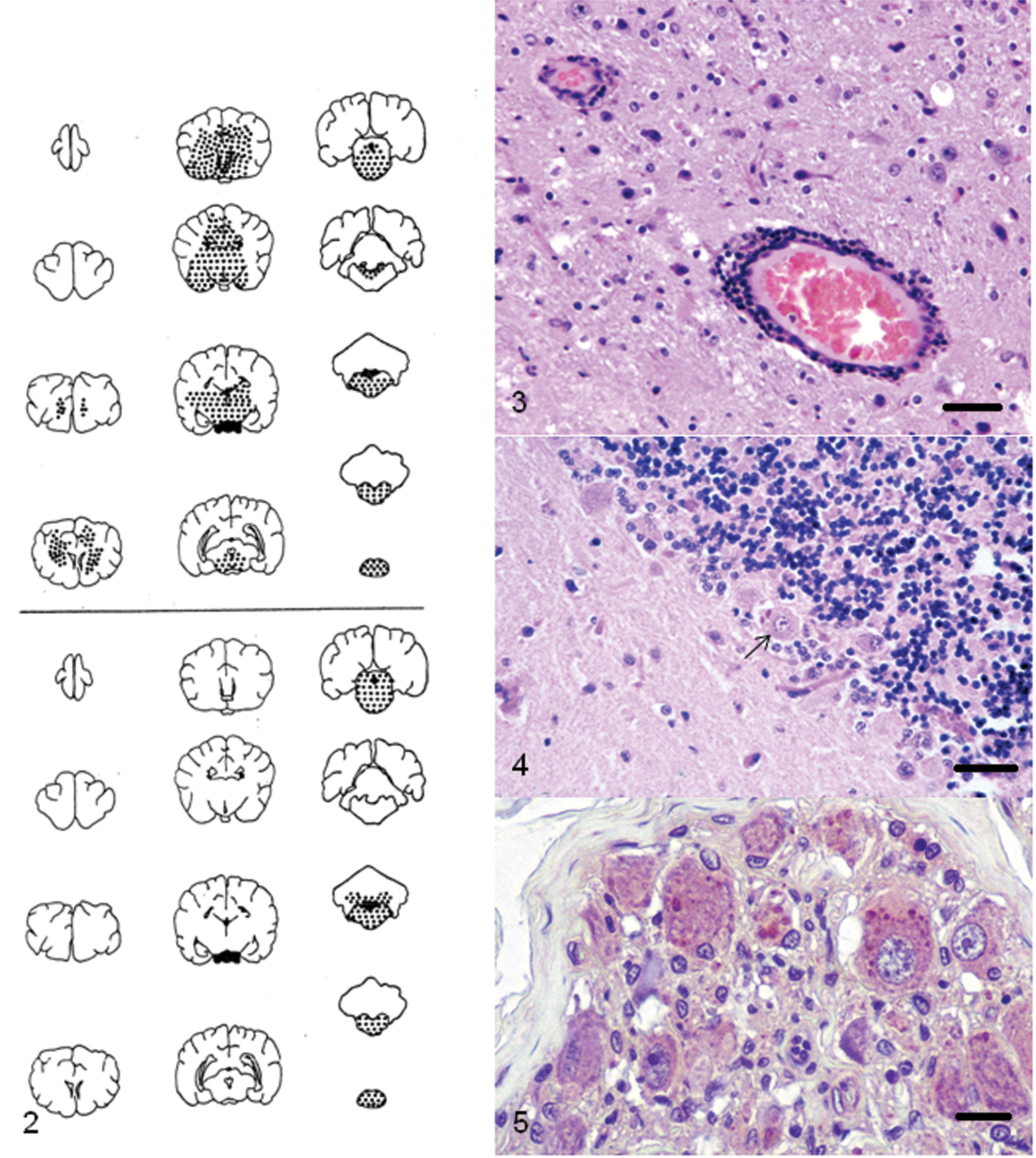
Top, location of microscopic lesions in brain and anterior cervical spinal cord of raccoon (Procyon lotor) experimentally infected with a raccoon isolate of street Rabies virus. Bottom, location of microscopic lesions in brain and anterior cervical spinal cord of raccoon experimentally infected with a canine isolate of street Rabies virus. ● = presence of nonsuppurative encephalitis.
Canine distemper
In addition to the dog, Canine distemper virus (CDV; order Mononegavirales, family Paramyxoviridae, subfamily Paramyxovirinae, genus Morbillivirus) is seen in a variety of other carnivores and is considered a major disease problem of raccoons. Natural outbreaks and lesions of the disease have been documented in raccoons.75,80,86 In most cases, there is generalized depletion of cells in lymphoid organs (lymph nodes, spleen, bone marrow, etc.), diffuse interstitial pneumonia, and multifocal areas of cerebellar necrosis (Fig. 6) with demyelination of the cerebellar white matter. 94 Viral inclusions are present in many tissues (Fig. 8). A previous study 31 documented the incidence and anatomic locations of distemper inclusions, which include the brain, bronchi, salivary glands, bile ducts, lymph nodes, spleen, urinary bladder, stomach, colon, and adrenal glands. Testicular degeneration and mineralization of seminiferous tubules has also been described in raccoons with this infection. 59 Since CDV infection produces immunosuppression in the host, it is susceptible to a variety of secondary opportunistic infections, and occasional cases of concurrent infections with rabies, toxoplasmosis, Tyzzer disease, and neosporosis have been documented.16,32,61,70,87,103 Morphologically, because of the immunosuppression caused by canine distemper, there is usually a paucity of host cellular inflammatory infiltrate that is directed at the opportunistic pathogen. The CDV antigen (Fig. 7), however, can be demonstrated in the brains of these raccoons.61,70
Raccoon (Procyon lotor) with Canine distemper virus infection; cerebellum. There is a focal area of cerebellar necrosis involving granular and Purkinje cell layers. Hematoxylin and eosin. Bar = 200 µm.
Pseudorabies and herpesvirus encephalitis
Although since 2003 no commercially produced pig herds have been found to be infected with Pseudorabies virus, sporadic infections have been found in feral pigs. Prior to 1981, there had been 2 reports of herpesviral infection in raccoons.91,97 Although the affected raccoon tissues were examined by electron microscopy, the tissues were not tested for the presence of pseudorabies. The disease can infect many mammalian species and, on the basis of experimental evidence, the raccoon is considered a major reservoir of Pseudorabies virus. 104 Microscopically, pseudorabies cases involve the brain and show herpes-like viral lesions in neuronal and non-neuronal tissues. 30 A non-pseudorabies herpesviral disease has also been described in a raccoon with lesions in non-neuronal tissues. 60 The only documented case of herpesvirus encephalitis was described in 1981. 91 The encephalitis was characterized by perivascular cuffing, choroiditis, and presence of magenta-colored intranuclear inclusions in choroid plexus and adrenal medulla. 91 As indicated previously, the case was not differentiated from pseudorabies.
Other viral diseases
Encephalomyocarditis virus (order Picornavirales, family Picornaviridae, genus Cardiovirus) was isolated from a raccoon in Florida, 26 and an experimental transmission study has been documented. 105 However, although some of the raccoons in the study seroconverted, no clinical signs or lesions were observed in the experimental raccoons. 105 In addition to the above-mentioned diseases that affect the nervous system, raccoons have been shown to have evidence (serology, polymerase chain reaction, Western blot, etc.) of other viruses such as Venezuelan equine encephalitis virus 5 and West Nile virus. 29 However, clinical cases of these diseases have not been documented in raccoons.
Prion diseases
Although naturally occurring transmissible spongiform encephalopathies (TSEs) have not been reported in the raccoon, reports of successful experimental transmission of similar spongiform diseases, transmissible encephalopathy of mink (TME) and sheep scrapie, have been documented in this species21,49,52,53 by intracerebral (IC) and oral inoculations. 21 In the IC-infected route, the raccoons were administered TME and developed a rapidly progressive disease within 190 days, whereas the orally infected raccoon developed neurologic signs after 306 days, which included weakness, behavior change, and incoordination. Scrapie-infected raccoons required a 2-year period to develop the disease when inoculated by the IC route.49,52,53 Microscopically, the lesions were identical in both groups (TME and scrapie) and consisted of widespread vacuolar changes, neuronal degeneration, and astrocytosis in the brain (except in cerebellum). Neuronal vacuolar changes, although described in scrapie and related diseases including naturally occurring TME, were rare.49,52,53
According to some researchers, Spiroplasma mirum is the causative agent of animal TSEs.3,4 However, when this organism was inoculated intracerebrally into raccoons, they did not succumb to any form of disease, and neither lesions nor abnormal prion protein of TSE was detected in the brains of these animals when necropsied at 30 months postinoculation. 42
Toxoplasmosis
Various surveys indicate a high prevalence of Toxoplasma gondii antibodies in U.S. raccoon populatiotns,9,16,77 and, in 1992, a case of clinical toxoplasmosis with presence of the organism in liver and brain was reported. 16 In the 1992 case, the raccoon was also found to have co-infection with CDV. 16 Experimental transmission of the organism has also been shown in the raccoon. 18
Neospora caninum
Antibodies to Neospora caninum were demonstrated in approximately 10% of raccoons from the eastern United States 88 and, to date, only 1 naturally occurring case in a juvenile raccoon has been documented. 87 This raccoon also had concurrent CDV infection. Thus, the findings of necrotizing encephalitis in the raccoon were largely due to the viral infection. 87
Sarcocystis neurona encephalitis
Sarcocystis neurona is the etiological agent of equine protozoal myeloencephalitis (EPM), which has resulted in the deaths of large numbers of horses in North America. Sarcocystis neurona has also been reported in the raccoon, skunk, mink, and cat.15,17,20 In a retrospective survey of 84 adult raccoons, 2 raccoons were found with S. neurona–associated granulomatous encephalitis. 38 Both raccoons also had extensive nonsuppurative myocarditis and 1 raccoon revealed S. neurona in the myocardium by immunohistochemistry. The CNS lesions were characterized by perivascular infiltration with mononuclear cells and presence of randomly distributed glial nodules. The organisms were observed in the inflammatory foci. 38 The life cycle of S. neurona is not fully understood. The opossum is recognized as a definite host, and horses and other susceptible animals are considered as aberrant hosts. Susceptible animals become infected by ingesting S. neurona sporocysts, which get excreted in feces of infected opossums. 19
Other Sarcocystis species in brain
Four species of Sarcocystis have been reported in striated muscles of raccoons.81,98 However, in North America, only Sarcocystis kirkpatricki has been documented in the muscles of raccoons. Although the prevalence of infection of this parasite appears to be variable, up to 50% of raccoons in a study from the northeastern United States were found to be infected in the heart, tongue, diaphragm, masseter muscle, or the esophagus. 81 A previous study 98 designated this organism S. kirkpatricki and reported a prevalence of 66% in raccoons from Illinois. Neither of these studies reported Sarcocystis in the brain of these raccoons. Subsequently, there has been a single report of S. kirkpatricki in the cerebellum (Fig. 9) of a raccoon with dual infections of T. gondii and CDV. 16 Also, a retrospective survey of 760 raccoons revealed 9 animals with sarcocysts of S. kirkpatricki in the brains. 39 Seven of the raccoons had concurrent viral diseases (canine distemper or rabies), suggesting that concurrent viral infections in raccoons may facilitate infection of brain tissue with S. kirkpatricki. 39
Eosinophilic meningitis and encephalitis
In normal free-ranging raccoons, eosinophilia of cerebrospinal fluid appears to be a common finding. 71 A case of generalized eosinophilic myositis with eosinophilia of blood and cerebrospinal fluid has been described in a raccoon, 66 which was presumed to be due to immune-mediated causes. Also, a raccoon with experimental canine Rabies virus infection has been documented with eosinophilic polioencephalomyelitis. 62 However, that raccoon did not show eosinophilia of the blood.
Neural and visceral larva migrans
Neural and visceral larva migrans have been documented in a large number of domestic and wild animal species (Snyder DE: 1983, The prevalence, cross-transmissibility to domestic animals and adult structure of Baylisascaris procyonis (Nematoda) from Illinois raccoons (Procyon lotor). PhD diss. University of Illinois, Urbana, IL). 79 The common round worm, Baylisascaris procyonis, found in raccoons, appears to be a frequent etiologic agent that is responsible for severe CNS disease in these parantenic hosts. However, visceral larva migrans due to B. procyonis has not been documented in raccoons. Other nematode species known to cause neural and visceral larva migrans include Toxocara spp., Angiostrongylus spp., and Strongyloides spp.
In raccoons, a form of visceral larva migrans associated with a trematode (Phagicola sp.) has been described. 67 However, these parasites did not migrate into neuronal tissues. A case of verminous encephalitis associated with an ascarid larval nematode in a raccoon has been described. 68 Although the morphologic characteristics indicated that the nematode was an ascarid larva, it was smaller than the larva of Baylisascaris sp. This appears to be the first documented case of cerebral larva migrans in a raccoon. 68
Neoplasms
Neoplastic lesions have infrequently been documented in the family Procyonidae. With the exception of thyroid adenomas76,85 and adrenal gland adenomas, 64 neoplasia appears to be rather rare in raccoons, and only infrequent case reports have been documented. In a retrospective survey of over 400 raccoon necropsies, 13 only 1 astrocytoma of the brain was found. Since that time, 1 additional astrocytoma involving the brain, cervical spinal cord, and spinal nerves has been documented. 58 In both cases, the characteristics of the neoplasm were similar to that published in other species. Also, a ganglioneuroma involving an adrenal gland has been seen (Fig. 10; Hamir AN, personal observation).
Environmental toxins
Lead
Lead is an environmental toxin with multisystemic toxic effects.31,34 A clinical case showing neurologic abnormalities in a free-ranging raccoon has been documented, 14 and also high levels of tissue lead levels have been reported from different areas of the United States.14,39 In the reported case, 14 the CNS lesions were subtle and consisted of mild endothelial hyperplasia and scattered neuronophagia. In experimental animals, administration of inorganic lead may lead to the impairment of immune functions, resulting in increased susceptibility to infectious agents.74,84,90Although neurologic manifestations of lead toxicosis has been reported in a raccoon, 14 experimental administration of oral lead acetate did not succeed in producing neurologic signs when the compound was administered over an 8-week period. 50
Ethylene glycol
Neurologic signs of ethylene glycol (antifreeze) toxicity include depression, ataxia, seizures, and coma, 22 and since these signs can be confused with other disorders in this species, clinical differential diagnosis of this condition is rather difficult. A diagnosis of ethylene glycol toxicosis is rarely reported in free-ranging wildlife; 2 however, morphologic lesions of this toxicity are often seen in diagnostic cases. There has been only 1 reported case of ethylene glycol poisoning in a free-ranging raccoon. 22 Since antifreeze is commonly found in the environment, and cases of this toxicity are frequently seen in domestic animals, it may be assumed that cases of ethylene glycol toxicity are being overlooked in free-ranging wildlife.
Trauma
It has been the author’s experience that during routine necropsy of free-ranging raccoons (>500) from the eastern and northwestern United States, 55 minor abrasions and/or trauma to the skin was a common finding in these animals. Fighting among males appeared to be the main reason for these skin lesions (Hamir AN, personal observation). Physical trauma due to gunshot wounds, collisions with automobiles, and falls from heights (in raccoons with neurologic abnormalities) were frequently observed in field specimens. In the latter cases, the animals had usually some preexisting neurological abnormalities. One raccoon with neurologic signs was observed to have a piece of porcupine quill logged in the brain tissue (Hamir AN, personal observation).
Miscellaneous conditions of unknown etiology
Ischemic encephalopathy
One acute and 1 chronic case of cerebral infarction syndrome has been described in a free-ranging and a laboratory-confined raccoon, respectively. 63 Both were mature adults. The raccoon with the acute form of the condition had abnormal neurologic signs whereas the raccoon with the chronic lesion did not. In the chronic case, the lesion was present unilaterally (Fig. 11) and since only one-half of the brain was available for examination in the acute case, it was not possible to determine if there were multiple lesions. In both cases, the nature and distribution of the lesions was compatible with an ischemic etiology, although vascular lesions were not observed. 63
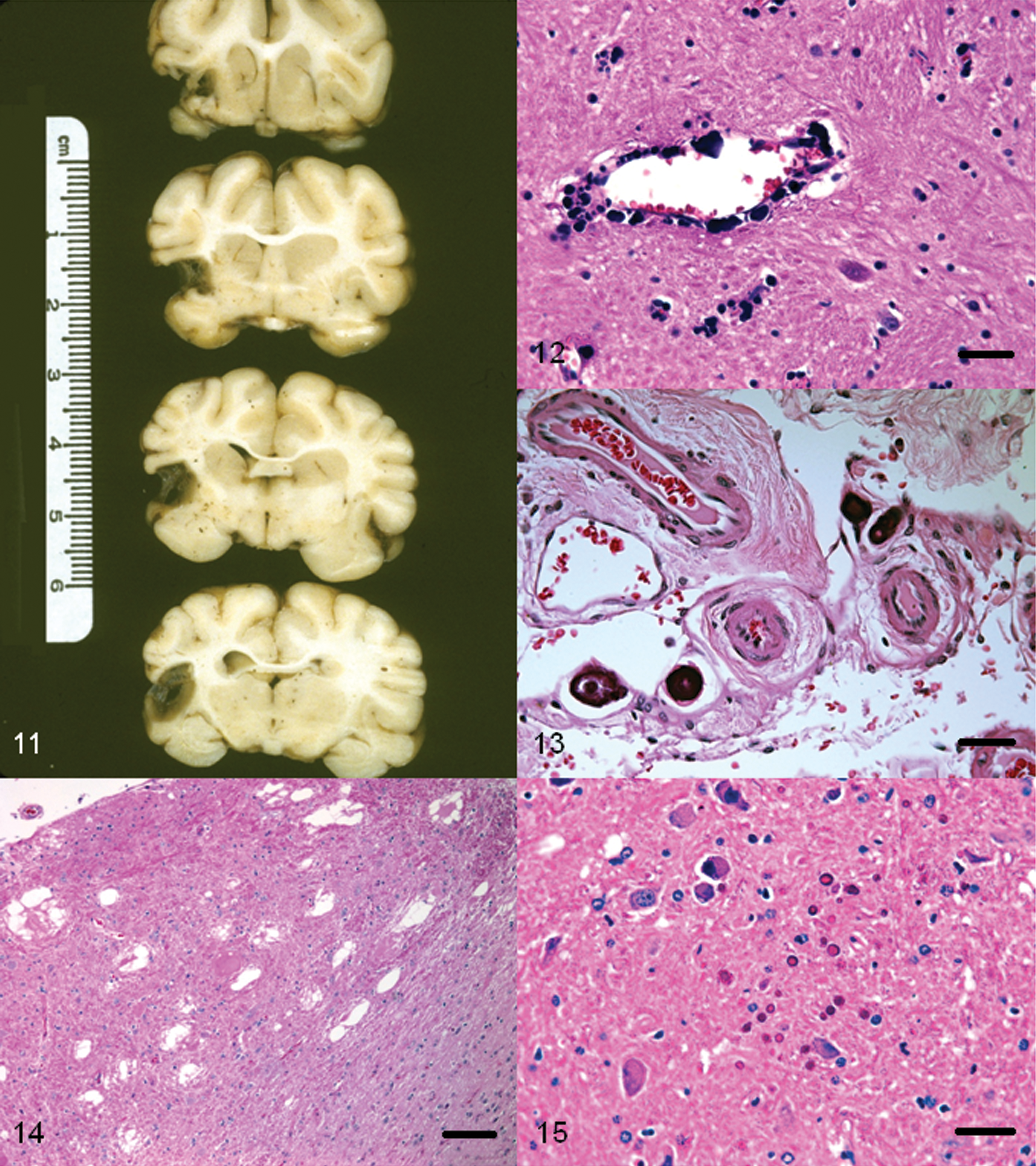
Raccoon (Procyon lotor) brain. Coronal sections of cerebrum showing a unilateral area devoid of brain parenchyma due to chronic ischemia.
Cerebrovascular mineralization
A case of cerebrovascular mineralization in an aged (>10 years old) female with uremia associated with polycystic kidney disease has been reported in a raccoon. 47 The vascular lesions were bilateral and consisted of multifocal mineralized foci in the walls of blood vessels in globus pallidus (Fig. 12). The affected vessels were patent, and there was no inflammatory cellular response to the mineralized foci. Morphologically, this lesion appeared to be similar to a nonsymptomatic condition that has been documented in other species. 27
Neuronal lipid vacuolation
Spongiform changes affecting the neuropil as well as neuronal cell bodies have been described in a newborn calf, 43 in experimental and natural cases of rabies in skunks, 11 foxes 11 and in a heifer. 23 Neuronal perikaryonic vacuolation is also seen in scrapie and related spongiform encephalopathes, 33 and in lysosomal and acquired storage diseases of various animals. 100 Naturally occurring TSE and storage diseases have not been documented in raccoons. Rabies, on the other hand, is commonly encountered in raccoons of the eastern United States. However, vacuolation of either the neurons or the neuropil has not been reported in raccoons with naturally acquired rabies, or in raccoons experimentally infected with either a dog or a raccoon isolate of Rabies virus. 55
During a 2-year period, clear vacuoles were morphologically detected in neurons of 42% of raccoon brains examined in Oregon.41,46 Neither age nor sex predisposition was apparent in this population. Microscopically, the vacuoles were variable in size, were in the perikarya, and were consistently present in pontine nuclei. Brain tissues were negative for Rabies virus antigen by fluorescent antibody test and for the protease-resistant protein (PrP) prion by immunohistochemistry. Electron microscopic examination of the brainstem of selected cases revealed accumulation of electron-dense material within neuronal perikarya. 41 Based on light and electron microscopic findings, the accumulated intracellular material appeared to have a high lipid content. These lesions indicate a form of neuronal storage condition.
Mineralized foci (psammoma bodies) in meninges and choroid plexus
With advancing age, the human brain shows increased presence of focal mineralized foci. 10 Often referred to as psammoma bodies, such mineralized foci are small, hardened bodies composed of concentric rings of calcium carbonate and calcium and magnesium phosphate. 10
Microscopic evidence of multifocal mineralizations (psammoma bodies) were seen in the brain of 62% of raccoons necropsied on Parramore Island, Virginia. 45 The mineralized foci revealed concentric laminations and were present in small capillaries of meninges of the brain, choroid plexus (Fig. 13), or at both these sites. In 2 raccoons, the lesions were confined to the meninges of the proximal cervical spinal cord. In most cases, the affected vessels appeared to have been completely occluded. However, no evidence of ischemic changes in the brain parenchyma was seen, and none of the raccoons showed abnormal neurologic signs prior to euthanasia. The condition appears to be a common incidental histopathological finding in raccoons from the eastern United States. 45 It is speculated that mineralization and subsequent occlusion of larger blood vessels may result in cerebral ischemia. Although the exact cause of this condition is not known, a primary vascular insult with resultant dystrophic mineralization of the affected vessels is suspected. 45
Neuroaxonal dystrophy
During a 12-month period, microscopic evidence of neuroaxonal dystrophy was seen in 12 out of 29 raccoons from Iowa. 54 In the affected animals, clinical signs were not apparent. Three of the animals were kits (<3 months), and none revealed lesions. The brain did not show any gross lesions. Microscopically, the lesions were confined to the dorsal caudal medulla where certain nuclei (predominantly gracilis and cuneate) were affected. The lesions consisted of eosinophilic axonal spheroids (Fig. 14) with presence of amphophilic periodic acid–Schiff-positive granular material within some of the degenerate neurons and axones. In a few cases, the latter had completely disappeared leaving behind clear areas in the neuropil (vacuolar degeneration). None of the affected cases revealed any appreciable numbers of inflammatory cells within the areas of degeneration. Neuroaxonal dystrophy has been observed in human beings and in various animals, and these changes have been associated with advancing age. Although none of the 3 raccoons examined in the study were old, it was presumed that since the lesions were not present in kits, the findings may be age-related. Subsequently, it was shown that severe neuroaxonal dystrophy was present in old (>7 years of age) raccoons. 37
Lafora bodies (polyglucosan inclusions)
Polyglucosan inclusions or Lafora bodies are inclusions of complex glycoprotein polymers in tissues. 100 Although not reported often in wild animals, Lafora bodies have frequently been reported in dogs and in a few other domestic animals. These inclusions are age-related and may or may not be associated with abnormal neurologic signs. 100 There has been only 1 reported occurrence of Lafora bodies in the brain (Fig. 15) of a raccoon, which did not show any overt abnormal neurologic signs. 37
Amyloidosis
Amyloidosis has been documented in both free-ranging and captive raccoons. 48 It is predominantly seen in the pancreas where it is deposited in the islets of Langerhans. Although not reported in the brain, a single case of amyloidosis in choroid plexus of an aged raccoon has been observed (Hamir AN et al., unpublished data). Amyloid accumulation was associated with multifocal lymphocytic cell infiltrations. The raccoon did not show any neurologic signs.
Congenital conditions
Although albinism is not uncommon in raccoons, 89 only a few other congenital lesions have been reported in this species. Individual case reports of congenital amelia of forelimbs, 73 lack of tail, 73 raccoon with 2 tails, 101 congenital diaphragmatic hernia, 96 renal hypoplasia, 92 dental anomalities, 82 and other skeletal anomalies 93 have been reported.
Congenital abnormalities affecting the nervous system or other body systems that can cause adverse effect on the nervous system are rare, and only 1 documentation of a report of ocular and nasomaxillary anomalies with neural defects has been published. 95 However, during a field safety trial of the vaccinia-rabies glycoprotein (V-RG) vaccine, 72 a raccoon with a unilateral micro-ophthalmia was observed (Hamir AN et al., unpublished data).
Conclusion
Raccoons are almost ubiquitous in North America. Since the 1980s, a large number of diseases and lesions, many involving the nervous system, have been documented in this species. As a fair number of these have been individual case reports, it is difficult to access the general importance of these conditions on the local populations of raccoons. Also, some of the described conditions appear not to have been of immediate severe consequence to the raccoons. However, such reports are scientifically useful in recognizing pathological conditions that exist at a given time and are also useful for future disease investigations. Furthermore, since raccoons are no longer confined to North America, it would be of interest to compare disease prevalence and pathology in different raccoon populations of the world with conditions that are reported in the current review.
Footnotes
Acknowledgements
The author would like to thank Dr. Charles E. Rupprecht, now at Centers for Disease Control and Prevention, Atlanta, GA, for the opportunity to examine and learn from a large number of raccoon necropsies from his project of vaccinia-rabies glycoprotein (V-RG) vaccine development during the 1980s.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
