Abstract
An Amies agar gel swab transport system was evaluated for its ability to maintain bacterial viability and relative quantity after freezing at −80°C. Nine American Type Culture Collection (ATCC) bacterial strains were used: 3 anaerobic strains (Propionibacterium acnes, Peptostreptococcus anaerobius, and Clostridium sporogenes) and 6 facultative or strict aerobic bacterial strains (Stenotrophomonas maltophilia, Escherichia coli ([ATCC 25922 and ATCC 11775], Salmonella enterica subsp. enterica serovar Typhimurium, Staphylococcus saprophyticus, and Lactobacillus casei). The bacterial species were chosen because they corresponded to bacteria identified in psittacine feces and cloacal samples. There were no significant differences between growth scores at baseline and after storage at −80°C for 40 days for any of the bacteria examined after 48 and 72 hr of incubation, with the exception of P. anaerobius. For P. anaerobius, there was a significant reduction (P < 0.001) in the growth score after storage at −80°C for 40 days from that of the baseline; however, the bacteria were still viable. The tested swab transport system may be useful when lengthy storage and transport times necessitate freezing samples prior to culture.
Swab transport systems are routinely used in clinical and research settings. For most diagnostic and research studies, samples are cultured immediately, cultured after brief refrigeration, or stored in swab transport systems for short periods prior to culturing. However, with microbiologic research expanding into remote locations, such as the Amazonian River Basin, storage time of samples from collection to culture may be days to weeks. Viability of organisms after storage for varying periods at ambient or refrigerated temperatures is dependent on the organism, transport system, temperature, and storage period (Van Horn K, Warren K, Tóth C: 1999, Comparison of four swab systems for recovery of anaerobic bacteria, abstract C-32. In: Proceedings of the 99th general meeting of the American Society for Microbiology, Chicago, IL). 4,5,9,11–14,16–21 Frozen semisolid or liquid transport media can maintain bacterial viability for days. 2,6,7,15 The viability of Bacteroides sp. in Amies medium is preserved longer, from 2 to 12 weeks, when storage temperature decreases from −10 to −20°C, respectively, 8 and the viability of Gram-negative enteropathogenic bacteria in feces is maintained for up to 12 months when the feces is stored at −70°C.3 However, studies on the efficacy of swab transport systems stored at −80°C could not be found.
In some situations, samples may undergo extended storage time and transport, spanning weeks and thousands of miles, prior to culturing. Hence, a critical question is whether or not bacterial viability is maintained when a swab transport system is frozen for an extended time. The objective of the current study was to evaluate a commercially available anaerobic swab transport system a for its ability to maintain bacterial viability and relative abundance after storage at −80°C for 40 days.
Nine bacterial reference strains, b reflecting those that may be found in the cloaca and feces of psittacine birds, 21 were examined: 3 anaerobic organisms (Propionibacterium acnes American Type Culture Collection [ATCC] 6919, Peptostreptococcus anaerobius ATCC 27337, Clostridium sporogenes ATCC 3584) and 6 facultative or strict aerobic bacterial organisms (Stenotrophomonas maltophilia ATCC 51331, Escherichia coli ATCC 25922 [a hemolytic strain] and ATCC 11775 [a nonhemolytic strain], Salmonella enterica subsp. enterica serovar Typhimurium ATCC 14028, Staphylococcus saprophyticus ATCC 15305, and Lactobacillus casei ATCC 393). A suspension of each bacterial strain was prepared in 0.9% sterile saline solution to a turbidity equivalent to a 0.5 McFarland standard. The anaerobic transport swab a was immersed in the standardized bacterial suspension for 10 sec, pressed firmly on the inside wall of the tube to remove excess fluid, and 1) plated immediately (baseline); 2) placed immediately into the transport media system, held at room temperature (23 ± 2°C) for 1 hr, and then plated; or 3) placed immediately into the transport media system, stored at −80°C for 40 days, removed, allowed to thaw at room temperature for 1 hr, and then plated. For each bacterial species, handling and/or storage conditions were performed in triplicate.
Each swab was rolled 4 times over a 1-cm 2 area of the agar, and then a 4-quadrant streaking method was utilized to streak the plates. Anaerobic bacteria (P. acnes, P. anaerobius, and C. sporogenes) were cultured on Brucella agar with 5% horse blood c and incubated at 37°C in an anaerobic environment. d Aerobic bacteria (S. maltophilia, E. coli ATCC 25922 and ATCC 11775, Salmonella serovar Typhimurium, S. saprophyticus, and L. casei) were cultured on trypticase soy agar with 5% sheep blood c (TSA agar) and incubated in a 5% CO2 environment at 37°C; the Gram-negative aerobic bacteria (S. maltophilia, Escherichia coli ATCC 25922 and ATCC 11775, and Salmonella serovar Typhimurium) were cultured on MacConkey agar c and incubated in air at 37°C; and, L. casei was cultured on Lactobacilli de Man, Rogosa, and Sharpe agar e and incubated in a 5% CO2 environment at 37°C.
At 24, 48, and 72 hr of incubation, growth was assessed and scored from 0 to 5 using a scoring system adapted and modified from previous studies (Beall AR, Pringle T, Underwood M: 2004, Evaluation of four swab transport systems: recovery of clinically relevant organisms after variable incubation times, abstract C-160. In: Proceedings of the 104th general meeting of the American Society for Microbiology, New Orleans, LA). 9,12,19 Scoring was as follows: 0 = no growth; 1 = growth of 1–20 colonies in the first quadrant; 2 = growth of >20 colonies in the first quadrant; 3 = growth in the first and second quadrants; 4 = growth in the first, second, and third quadrants; and, 5 = growth in all 4 quadrants. Differences in growth scores between baseline (i.e., swabs plated immediately after inoculation) and the 2 storage conditions (room temperature for 1 hr and −80°C for 40 days) were analyzed using Friedman repeated measures analysis of variance on ranks; if a significant difference was found, the individual storage conditions were compared with the baseline using the Holm–Sidak method for pairwise comparisons. A P value of less than 0.05 was considered significant.
Data are summarized in Table 1. At 48 and 72 hr of incubation, the growth scores of P. anaerobius after storage at −80°C for 40 days were significantly less (P < 0.001) than baseline growth scores. At 48 and 72 hr of incubation, no significant differences were found between P. acnes growth scores at baseline and either of the storage conditions. However at 24 hr of incubation, P. acnes growth scores for the storage conditions of room temperature for 1 hr and of −80°C for 40 days were significantly less (P < 0.004) than baseline growth scores. No significant differences were found between C. sporogenes growth scores at baseline and either of the storage conditions.
There were no significant differences in the growth scores between baseline and the different storage conditions for any of the Gram-negative aerobic bacteria (S. maltophilia, E. coli [ATCC 25922 and ATCC 11775], and Salmonella serovar Typhimurium). There were no significant differences in the growth scores between baseline and the different storage conditions for the Gram-positive aerobic bacteria (S. saprophyticus and L. casei). However, for L. casei, no growth was evident at 24 hr of incubation on TSA agar for baseline and the 2 storage conditions, but growth was apparent at 48 and 72 hr of incubation; growth scores for L. casei at 24 hr of incubation were significantly less (P < 0.001) than those at 48 and 72 hr of incubation.
After storage at −80°C for 40 days, the anaerobic swab transport system tested was effective in maintaining anaerobic and aerobic bacteria viability and relative abundance; however, some variations were noted among the organisms. The growth scores of P. anaerobius were significantly reduced from baseline when the swab transport system was stored at −80°C for 40 days. Recovery of P. anaerobius from a variety of swab transport systems drops to zero when stored for periods of more than 24 hr at temperatures above freezing (Van Horn K, Warren K, Tóth C: 1999, Comparison of four swab systems for recovery of anaerobic bacteria. In: Proceedings of the American Society for Microbiology. Abstract C-32) 11,14,16 ; however, viability is better maintained when the swab transport system is stored at 4°C rather than at 24°C. 13,18 The results of the present study support previous findings indicating that P. anaerobius viability is better maintained when swab transport systems are stored at near- or subzero temperatures. However, reduction in the number of colony forming units with storage seems to occur.
Propionibacterium acnes is a slow-growing anaerobic bacterium, 10 a factor that may have contributed to its delayed growth and lower growth scores at 24 hr of incubation. The cause of the significantly lower growth scores for P. acnes when stored at room temperature for 1 hr and for −80°C for 40 days is uncertain. Still, the growth scores between baseline and either storage condition, when examined at 48 and 72 hr of incubation, were not significantly different, demonstrating the swab transport system's ability to maintain P. acnes viability. These findings support previous research on P. acnes at temperatures above freezing and for durations of up to 48 hr (Van Horn K, et al.: 1999, Comparison of four swab systems for recovery of anaerobic bacteria).
Mean growth scores of bacteria (n = 3) at baseline and after the 2 different storage conditions. *
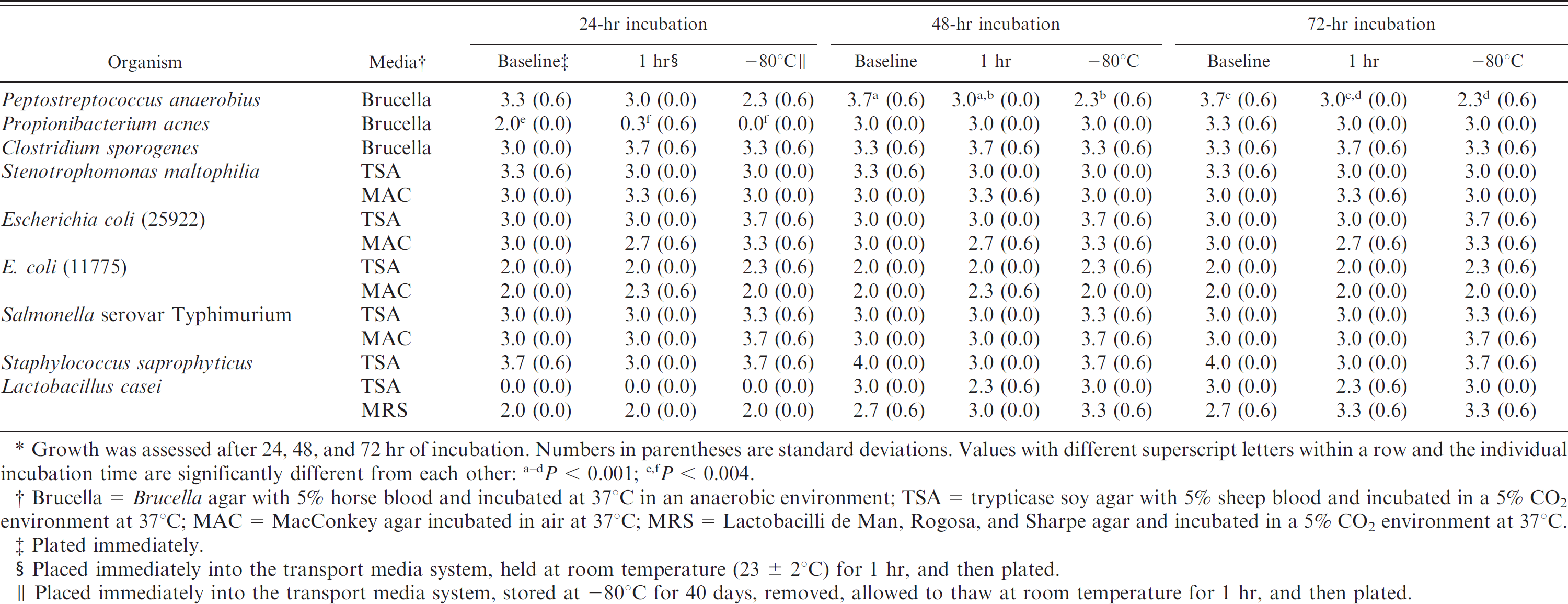
Growth was assessed after 24, 48, and 72 hr of incubation. Numbers in parentheses are standard deviations. Values with different superscript letters within a row and the individual incubation time are significantly different from each other: a , d P < 0.001; e ,f P < 0.004.
Brucella = Brucella agar with 5% horse blood and incubated at 37°C in an anaerobic environment; TSA = trypticase soy agar with 5% sheep blood and incubated in a 5% CO2 environment at 37°C; MAC = MacConkey agar incubated in air at 37°C; MRS = Lactobacilli de Man, Rogosa, and Sharpe agar and incubated in a 5% CO2 environment at 37°C
Plated immediately.
Placed immediately into the transport media system, held at room temperature (23 ± 2°C) for 1 hr, and then plated.
Placed immediately into the transport media system, stored at −80°C for 40 days, removed, allowed to thaw at room temperature for 1 hr, and then plated.
In the current study, anaerobic and aerobic bacteria viability was maintained with storage at −80°C for 40 days, supporting earlier findings that anaerobic transport systems are effective for preserving the viability and relative quantity of anaerobic, facultative anaerobic, and aerobic bacteria. 11,17 The organisms used in the current study were representative of a bacterial diversity previously sequenced and phylogenetically analyzed with bacterial 16S ribosomal DNA from cloacal samples of 8 wild and 8 captive macaws. 21 In cloacal swabs and feces of healthy captive psittacine birds, E. coli is an abundant and commonly identified organism 1,21 ; thus, the current study used 2 different E. coli strains: the hemolytic strain ATCC 25922 and the nonhemolytic strain ATCC 11775. The authors recognize that actual cloacal swab samples may contain more than 1 species of bacteria; however, results from previous work with psittacines has shown a low species diversity of 1–3 different species per sample. 21 Utilizing pure cultures of bacteria in the current study simplified the semiquantification of growth, and it reduced possible bacterial interactions during storage or culture, as well as possible organism interactions during sampling, storage. Culturing should be investigated further.
Because the gastrointestinal tract and feces of birds contains obligate and facultative anaerobic and aerobic bacteria, sample collection, storage, and transport systems should be able to maintain both anaerobe and aerobe viability. Even though a transport system has not been developed that will maintain the viability of all groups of bacteria under all situations, the anaerobic swab transport system tested in the current study was effective in maintaining anaerobic and aerobic bacteria viability and relative abundance when stored at −80°C for 40 days and should be a satisfactory tool for preserving samples from remote locations where periods of extend storage and transport times are likely to be encountered.
Acknowledgements. The work was supported in part by The Schubot Exotic Bird Health Center at Texas A&M University, College Station, Texas. The authors recognize and thank Debra Turner for her technical assistance and critical review of the manuscript.
Footnotes
a.
BD Culture Swab™ MaxV(+), Becton, Dickinson and Company, Sparks, MD.
b.
American Type Culture Collection, Manassas, VA.
c.
Becton, Dickinson and Company, Sparks, MD.
d.
AnaeroGen Pack, Oxoid Company, Nepean, Ontario, Canada.
e.
Northeast Laboratory Services, Winslow, ME.
