Abstract
Intranuclear coccidiosis in 3 calves is described. Two calves with no genetic relationship were kept in the same pen, and the third calf was on another farm. The animals suffered from watery diarrhea or severe emaciation. Histologic examination showed epithelial desquamation (cases 1 and 2) or atrophy (case 3) of the jejunal villi. Coccidial meronts or merozoites were present in the nuclei of the majority of villus epithelial cells. There were rare intranuclear macrogametocytes, macrogametes, microgametocytes, microgametes, and oocysts in cases 1 and 2, but these were more easily observed in case 3. Parasite 18S ribosomal RNA sequences from case 1 showed 99.5% sequence identity with Cyclospora sp. Guangzhou 1, which has been found in fecal samples of cattle from China. In addition to the molecular results in one of the cases, the fact that the microgametocytes and oocysts were noticeably smaller in size than those of Eimeria alabamensis in all cases indicates the close association between Cyclospora and the 3 cases described herein.
Keywords
Intranuclear coccidiosis, defined as parasite development within host cell nuclei, is more frequently observed in lower vertebrate hosts such as fish, 15 amphibians, and reptiles 16 than in mammals.4,13,18 Although capable of causing death in tortoises,6,9 intranuclear coccidia are usually nonpathogenic. Merogonic and gametogonic stages of Eimeria hermani and Eimeria stigmosa are present within the nucleus of enterocytes of geese,5,17 but appear to be symbionts. Only 3 species are known as intranuclear coccidia in mammals. Cyclospora caryolytica 4 and Cyclospora talpae 13 develop intranuclearly in the small intestine and liver of moles, respectively. Eimeria alabamensis is marginally pathogenic and causes watery diarrhea in calves. 18 However, Cyclospora-like oocysts have been detected in fecal specimens from cattle in China and belong to the genus Cyclospora, along with primate-derived Cyclospora species. 11 The current study reports 3 cases of bovine intranuclear coccidiosis associated with Cyclospora spp. The parasites were genetically similar to Cyclospora in one of the cases, and morphologically distinct from E. alabamensis in all cases.
In Oita Prefecture in the southern part of Japan, intranuclear coccidiosis was diagnosed in 3 Japanese Black castrated male calves from 2 farms in 2010–2011. The clinical and macroscopic findings are presented in Table 1. On the first farm, a calf (case 1) was euthanized because of watery diarrhea. On the second farm, calves with growth retardation (cases 2 and 3) were kept in a pen with other 3 healthy calves, but had no genetic relationship.
Clinical data and gross pathology.

The fecal flotation method with saturated sucrose solution for Eimeria oocysts was employed in all cases, with negative results. After formalin fixation, however, feces from the large intestine were examined with the sucrose centrifugal-flotation method, 1 and a few oocysts, 10 μm in diameter, were found in case 3.
Tissue samples were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). Sections were also stained with phosphate buffered Giemsa solution (pH 6.4), and were differentiated and dehydrated with isopropyl alcohol. Many tissue sections from a previously published case of E. alabamensis infection 14 were also made and used as a control to compare parasite morphology.
Total DNA was extracted from paraffin sections by the phenol–chloroform–isoamyl alcohol method. 8 Polymerase chain reaction amplification of the 18S ribosomal RNA coding region in the parasite genome followed by nucleotide sequence analyses were performed as described previously. 14 The primers used were Ei18Sf1 forward (5′-AGCTCGTAGTTGGATTTCTG-3′) and Ei18Sr1 reverse (5′-CTAGTCGGCATAGTTTATGG-3′). The nucleotide sequences obtained were compared to homologous sequences of previously characterized Cyclospora spp. and Eimeria spp. using the basic local alignment search tool (BLAST; http://blast.ncbi.nlm.nih.gov/Blast.cgi). Phylogenetic analysis of nucleotide sequences was by the neighbor-joining method 20 using the ClustalW multiple sequence alignment program (version 2.1; www.ddbj.nig.ac.jp/), and the phylogenetic tree was constructed using Tree View (version 1.6.6; http://taxonomy.zoology.gla.ac.uk/rod/treeview.html).
On histologic examination, Giemsa stain was more successful than HE stain in detecting parasites. In the jejunum of cases 1 and 2, most villus epithelial cells had nuclei with parasites, and showed a strong tendency to desquamate. The majority of the villus epithelium appeared exfoliating into the intestinal lumen, but some desquamated epithelial cells were found in the lamina propria. Nuclei with several parasites were frequently seen, and had sometimes disintegrated. The parasite stages seen were predominantly meronts or merozoites, and rarely macrogametocytes, macrogametes (Fig. 1A), microgametocytes, microgametes (Fig. 1B), or oocysts. The upper part of the villus lamina propria was edematous and devoid of cellular elements and collagen fibers, while the lower part contained a significant number of histiocytes and epithelioid cells. Phagocytized parasites were observed within the latter. The number of intraepithelial lymphocytes in case 2 was less than in case 1.
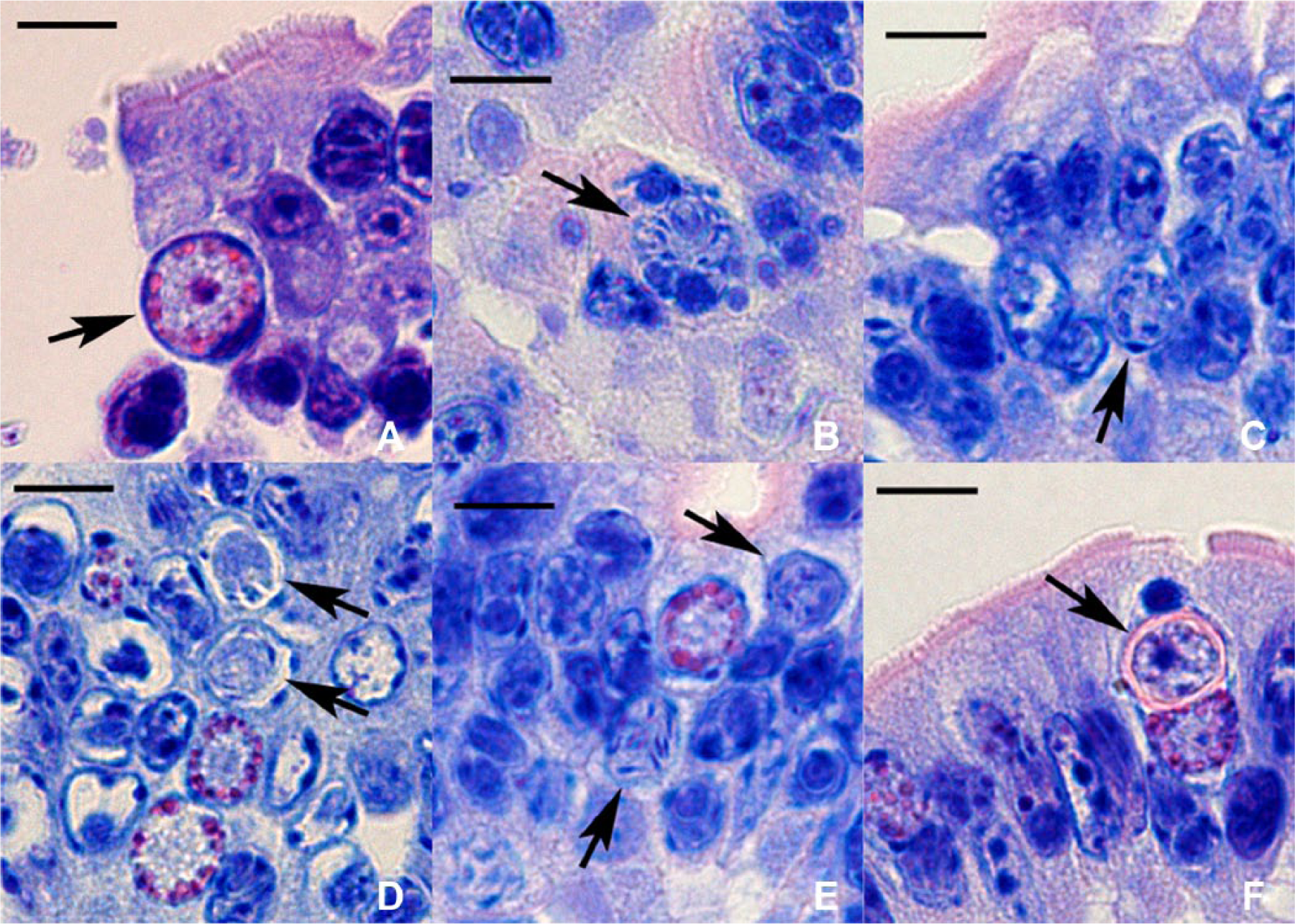
Cyclospora spp.
In case 3, jejunal villi were atrophic, with intranuclear coccidia in many epithelial cells. Meronts and merozoites predominated, but macrogametocytes and macrogametes were occasionally seen. Microgametocytes (Fig. 1C, 1D), microgametes (Fig. 1E), and oocysts (Fig. 1F) were present within the nuclei of a few epithelial cells. There were epithelioid cells just beneath the villus epithelium, and low numbers of intraepithelial lymphocytes. In cases 2 and 3, bacterial organisms were numerous in the intestinal lumen. Except in the ileum of case 2 with sparsely distributed crypt abscesses, no abnormal lesions were detected in the duodenum, ileum, and large intestine in all cases.
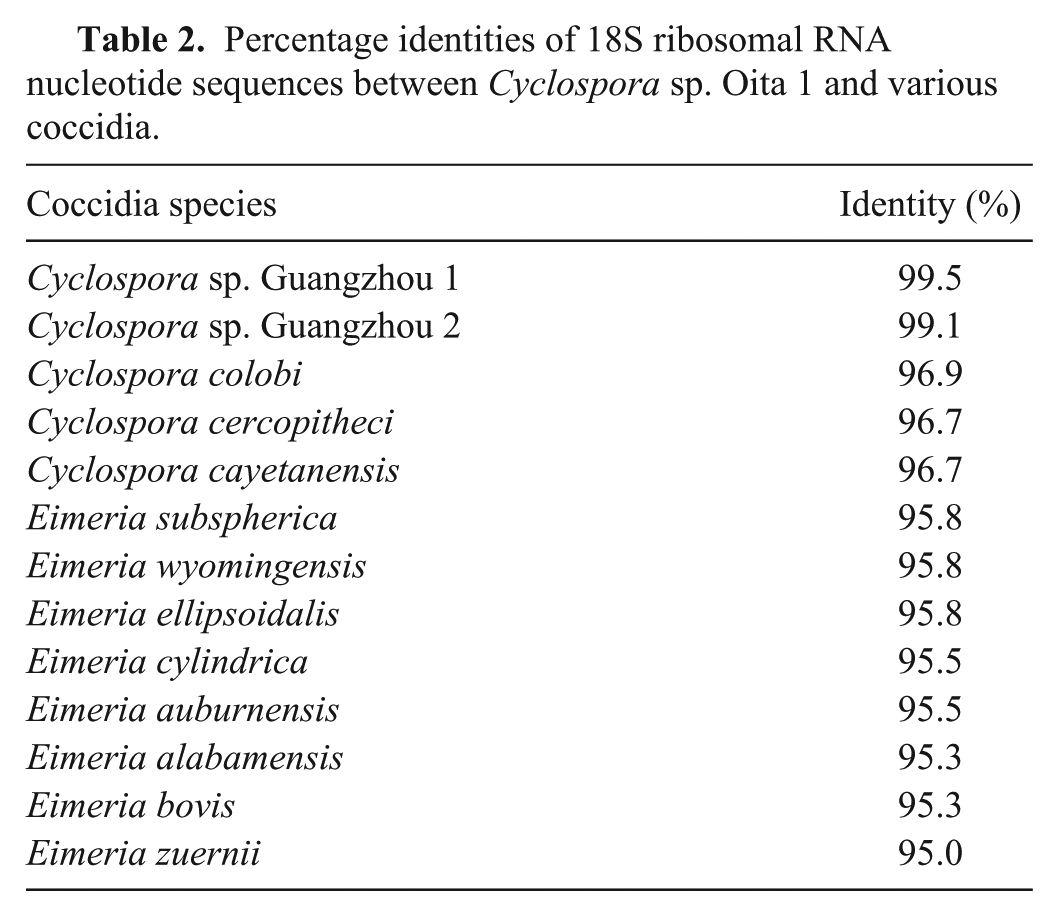
There are a number of factors that support the conclusion that Cyclospora spp. is the etiological agent in the 3 calves described herein. An 18S ribosomal DNA sequence was obtained in case 1, but not in the other cases, perhaps due to the prolonged fixation time or the numerous bacteria in the jejunum. The amplified DNA comprised a sequence of 468 base pairs in length and exhibited 96.2–99.5% and 95.0–95.8% nucleotide sequence identity with the homologous sequence in the genome of various Cyclospora and Eimeria species, respectively (Table 2). The most closely related species was Cyclospora sp. Guangzhou 1, which had been found in fecal samples of cattle in China (Table 2). 11 These results were also supported by phylogenetic analysis of the nucleotide sequences (Fig. 2). The nucleotide sequence reported in the current study was designated Cyclospora sp. Oita 1 and deposited in the DNA Data Bank of Japan (accession no. AB735603).
Percentage identities of 18S ribosomal RNA nucleotide sequences between Cyclospora sp. Oita 1 and various coccidia.
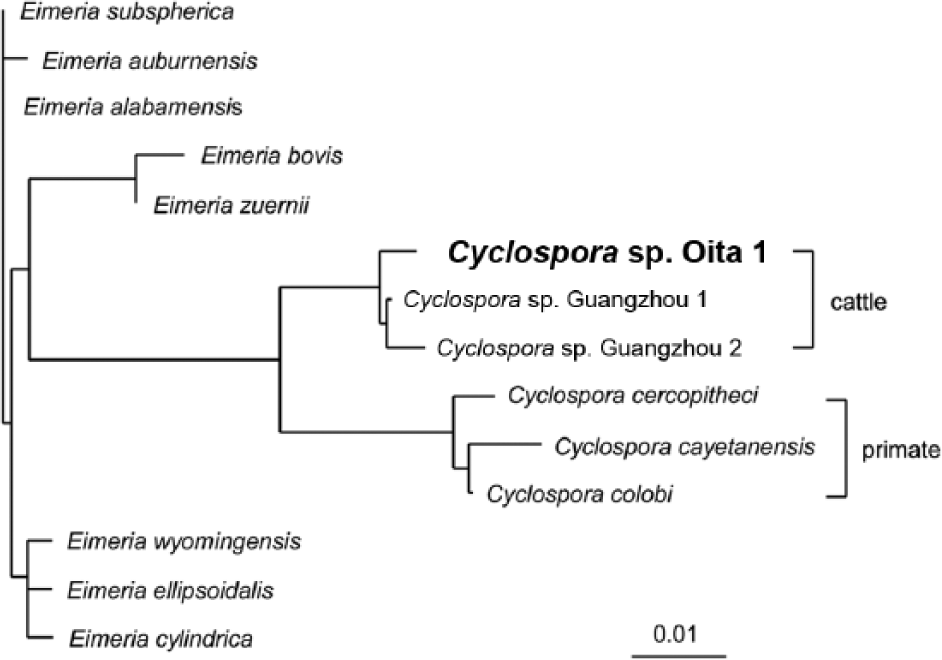
Phylogenetic tree constructed from 18S ribosomal RNA sequences of Cyclospora sp. Oita 1 and other Cyclospora and Eimeria species. DNA Data Bank of Japan accession numbers are as follows: Cyclospora sp. Guangzhou 1, DQ082866; Cyclospora sp. Guangzhou 2, DQ082867; Cyclospora cercopitheci, AF111185; Cyclospora cayetanensis, AF111183; Cyclospora colobi, AF111186; Eimeria subspherica, AY876930; Eimeria auburnensis, AY876927; Eimeria alabamensis, AF291427; Eimeria bovis, EBU77084; Eimeria zuernii, AY876932; Eimeria wyomingensis, AY876931; Eimeria ellipsoidalis, AY876929; Eimeria cylindrica, AY876928.
In the control case involving E. alabamensis, intranuclear merozoites, meronts, and macrogametocytes were readily detected in intestinal epithelial cells. A few macrogametes (Fig. 3A), microgametocytes (Fig. 3B), microgametes (Fig. 3C), and oocysts (Fig. 3D) were present, mainly in epithelial cells located at the crypt base. It could not be ascertained if these latter forms of the organism were located within epithelial cell nuclei, because of their large size or number.
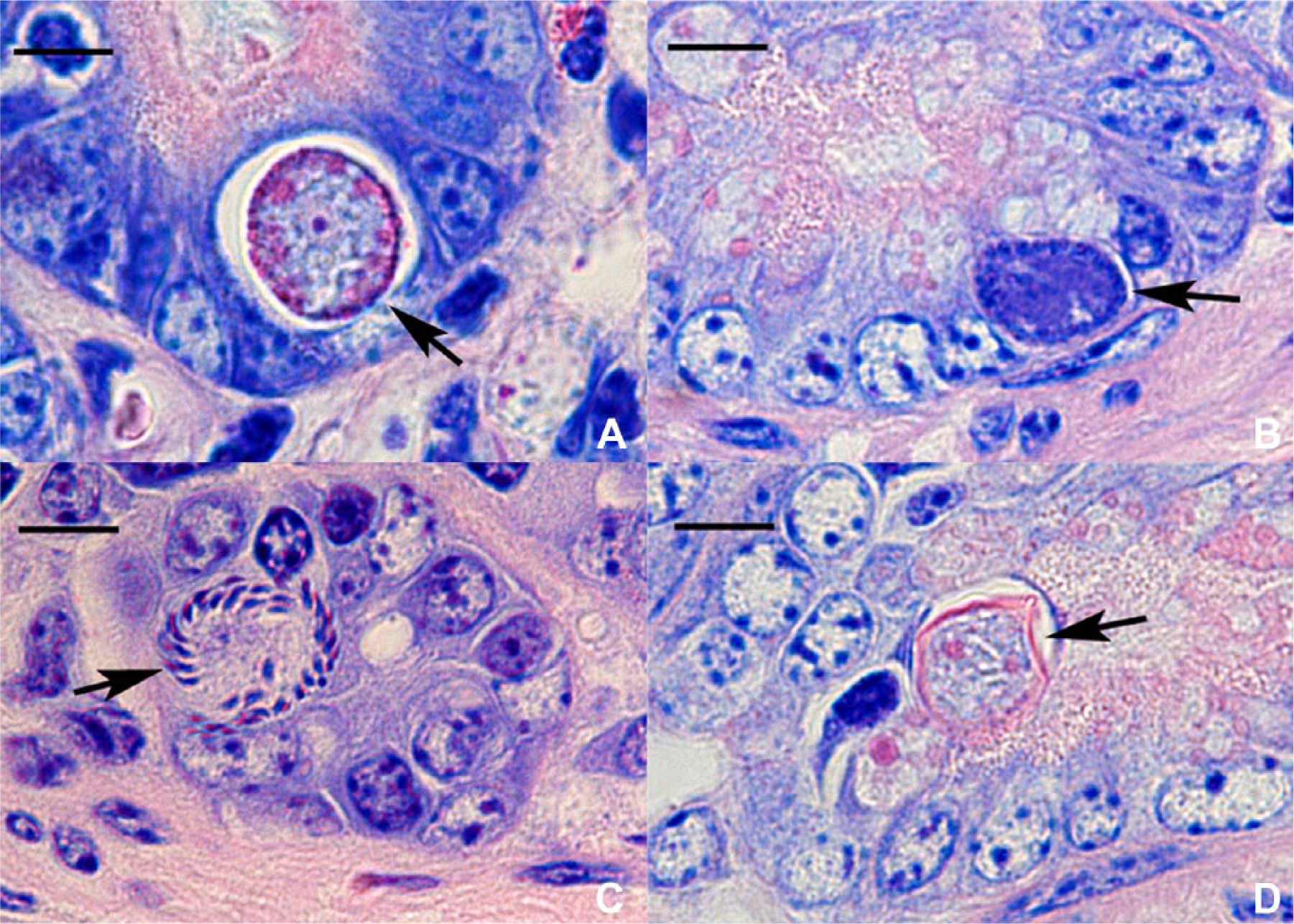
Eimeria alabamensis.
Large microgametocytes of E. alabamensis, capable of developing into many microgametes, have been observed previously in cattle, 3 and similar findings were obtained in the control case of E. alabamensis in the current study. In contrast, far smaller microgametocytes were detected in the current 3 cases, which is consistent with the finding that microgametes from a microgametocyte were few in number. Oocysts of E. alabamensis measure 16–24 μm × 12–16 μm, 10 whereas the size of oocysts in case 3 was similar to Cyclospora spp. in cattle (8 μm in diameter), 11 Cyclospora cayetanensis in human beings (8–10 μm), and C. caryolytica in moles (8–12 μm). 12 On the basis of such results, a diagnosis of intranuclear coccidiosis by Cyclospora spp. was made. Similar Cyclospora-like organisms were detected in 6 out of 168 specimens from dairy farms in China by molecular analysis, 21 and these organisms may be widely distributed in East Asia.
In a case of E. alabamensis infection, nuclear cleavage in epithelial cells was considered to be evidence that merozoites can escape from the host cell nuclei or cytoplasm without inducing their destruction. 14 In contrast, in the present cases, there was widespread desquamation of villus epithelial cells, which contained parasites. Moreover, epithelial cell nuclei with several parasites were disintegrated. Connective tissue was reduced in the lamina propria of the villi in cases 1 and 2, implying that the villi will become atrophic shortly after epithelial desquamation. Case 3, whose lesion was characterized by villous atrophy, was judged to be in a somewhat more advanced stage of enteritis than the other cases. This view was supported by the fact that parasites at the stage of gametogony were more readily observed in case 3.
Eimeria alabamensis induces disease only at extremely large infective doses,2,7 and the presence of many intranuclear parasites in the intestinal villi suggests severe parasitic contamination of the environment.3,18 In case 1, it is not clear why the calf was susceptible to infection. This possible process of pathogenesis resembles E. alabamensis infection. 14 The absence or atrophy of the thymus in cases 2 and 3 suggests that perhaps the calves were already immunocompromised, 19 and therefore susceptible to opportunistic infection. Thus, growth retardation in some congenitally compromised calves may be associated with this type of intranuclear coccidiosis.
Infection caused by Cyclospora spp. is difficult to diagnose. Oocysts are few in number and small, and therefore not easily detected; and the absence of diarrhea (in some cases at least) and gross intestinal lesions may direct the clinician’s attention away from an intestinal coccidial infection. These factors may lead to a poor prognosis for affected calves, as well as making it difficult to define the normal cycling of Cyclospora spp. in the calf population on affected farms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
