Abstract
Autosomal-dominant polycystic kidney disease (ADPKD) is the most prevalent inherited genetic disease of cats, predominantly affecting Persian and Persian-related cats. A point mutation (C→A transversion) in exon 29 of the PKD1 gene causes ADPKD, and is the specific molecular target for genetic diagnosis in cats. The current study describes a newly developed touchdown polymerase chain reaction (PCR) to detect this single point mutation, using 2 primers specific for the mutant allele, adapted from an existing multiplex amplification refractory mutation system (ARMS PCR). Furthermore, correlations between the clinical outcomes of tested animals and the results of the genetic test were investigated. A total of 334 cats were tested, 188 from the Veterinary Hospital of Small Animals at the University of Brasilia, and 146 from an anti-rabies vaccine campaign of the Federal District. A total prevalence of 9% was evident among the samples, with 33% of the Persian cats testing positive, and 7% of the Brazilian long- and shorthaired cats testing positive. Prevalence was not correlated with gender or hemogram. Positive animals exhibited hyperglobulinemia (P = 0.02). This research demonstrated that the mutation does not only occur in Persian and Persian-related cats, and that a touchdown PCR can be used to diagnose ADPKD.
Feline polycystic kidney disease (PKD), also known as autosomal-dominant polycystic kidney disease (ADPKD), is one of the most important causes of kidney failure in cats, and the most prevalent inherited feline disease. 12 It is characterized by cysts of less than 1 mm to more than 1 cm in size. These cysts can appear in renal parenchyma, and occasionally in the liver, pancreas, and spleen. 5
Two forms of expression of the gene encoding polycystin-1 (the PKD1 gene) induce ADPKD. Homozygous dominant carriers do not survive to term, as this genotype results in a serious and lethal form of the disease, causing early intrauterine death or renal failure. 15 ADPKD carriers are heterozygous. Thus, the offspring of 2 homozygous negative (noncarrier) cats are all healthy. Half of the offspring resulting from a mating between a heterozygous carrier cat and a homozygous negative cat are predicted to be healthy (homozygous negative for the deleterious gene) and half are predicted to be carriers. The offspring of a mating between 2 carrier cats is predicted to be 25% healthy, 50% carrier, and 25% stillborn. 6
Autosomal-dominant polycystic kidney disease is linked to a C→A transversion in exon 29 of the feline PKD1 gene. This mutation results in a stop codon at position 3284, which corresponds to a loss of 25% of the C-terminus of the polycystin-1 protein, and this form of the protein is a target for molecular detection in ADPKD carriers. 12 Polycystin-1 is responsible for regulating cell proliferation, cell differentiation, and apoptosis, and when its activity is reduced, lesions and cellular changes in the renal parenchyma occur more often. 2
Prevalence studies of this illness suggest high incidences in the United States, Australia, England, Germany, France, and Italy. Collectively, the global ADPKD prevalence is approximately 30–38%. 11 While it affects 37–49% of Persian and Persian-related breeds worldwide, it is also present in many other breeds due to cross-breeding, including British Shorthairs, American Shorthairs, and Scottish Folds; thus, there are no specific phenotypic traits that are reliably indicative of heterozygosity. Notably, the percentage of cats carrying the mutation that actually have cysts cannot be estimated because cyst development depends on age and the severity of the disease. 11
Autosomal-dominant polycystic kidney disease is an inherited disease affecting mainly Persians and Persian-related cats. 10 However, given the high frequency of mating between Persians and other breeds, including Siamese, British Shorthair, American Shorthair, and mixed-breed cats, veterinarians should test all cats for PKD.11,15 The objectives of the current study were to develop a touchdown polymerase chain reaction (PCR) technique for ADPKD diagnosis, determine the occurrence of ADPKD in a cat population in Brazil, and analyze associations between laboratory findings and PCR results.
Blood samples were collected from a total of 334 domestic cats of the Federal District, Brazil (188 from the Veterinary Hospital of Small Animals at the University of Brasilia [Hvet-UnB] and 146 from the anti-rabies vaccination campaign), by venipuncture of cephalic or jugular veins into ethylenediamine tetra-acetic acid–coated tubes for complete blood cell count, a and into tubes containing a clotting activator for biochemical analysis. b Samples from 3 Persian cats confirmed as ADPKD positive by ultrasound screening were used as positive controls for all PCR tests. The PCR negative control was ultrapure water and cats without clinical signs and cysts, confirmed by ultrasound and negative for ADPKD PCR. Male cats comprised 51.7% of the samples, and 48.3% were from female cats. Mean age was 5.8 years, and ranged from 3 months to 20 years. The Ethics Committee on Animal Use (UnB, Brasilia, Brazil) approved the research (UnBDOC #43930/2012).
DNA for the PCR was extracted using a commercial kit, c in accordance with the manufacturer’s instructions, and stored at –20°C prior to use. All amplifications were performed on the same thermal cycler. d Prior to testing for the PKD1 gene of interest, all samples were tested in a PCR that detects the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) enzyme, to verify DNA quality, integrity, and the presence of PCR inhibitors as previously described using GAPDH-F (CCTTCATTGACCTCAACTACAT) and GAPDH-R (CCAAAGTTGTCATGGATGACC) primers. 3
A touchdown PCR was developed to detect the PKD1 mutant gene (GenBank accession no. AY612847) using PKD-F3 (AGAGGCAGACGAGGAGCACT) and PKD-R2 (GCCTCGTGGAGAAGGAGGT) primers. 10 The final mix volume was 25 µl, containing 1× PCR buffer, 1 µl of primers (10 pmol), 2 mM MgCl2, 0.25 mM of each deoxyribonucleotide triphosphate, e 1 U of Taq DNA polymerase, e and 5 ng of DNA sample. The initial denaturation step was 5 min in duration at 94°C, followed by 10 cycles of 1 min at 94°C, 1 min at 67°C (decreasing 1°C per cycle), and 1 min at 72°C, followed by 30 cycles of 1 min at 94°C, 1 min at 59°C, and 1 min at 72°C, with a final extension step of 10 min at 72°C, producing a 277-bp fragment in samples positive for the PKD1 mutant gene.
As a confirmatory PCR test for the mutation detected via the touchdown PCR protocol, a restriction fragment length polymorphism (RFLP) PCR was performed as previously described, 12 using PKD1F (CAGGTAGACGGGATAGACGA) and PKD1R (TTCTTCCTGGTCAACGACTG) primers, in all of the samples that had tested positive for the PKD1 mutant gene. The final amplification product for exon 29 was 559 bp in all samples. PKD1 mutation causes a restriction enzyme site alteration for MLY1, f resulting in 2 fragments of 316 bp and 243 bp in samples containing the mutant allele, whereas for negative cats only 1 fragment (559 bp) remains after digestion.
All amplification fragments were separated on 1.5% agarose gels at 80 V. Gels were visualized by ultraviolet exposure g after ethidium bromide h staining. Descriptive statistics and comparisons thereof were based on means and standard deviations. Kolmogorov–Smirnov and Shapiro–Wilk tests were used to determine normality in both groups (G1-positive × G2-negative), for each category. The Mann–Whitney U test was used to analyze nonnormally distributed data, and Student’s t-test was used to compare normally distributed data. Differences in proportions were assessed via the chi-square test, equal proportions test, and odds ratios with an appropriate confidence interval (CI).i,j Results were considered statistically significant if P < 0.05. Throughout the course of the research, positive controls for all PCR tests always amplified products of the expected size, while negative controls showed no signs of amplification (Fig. 1).
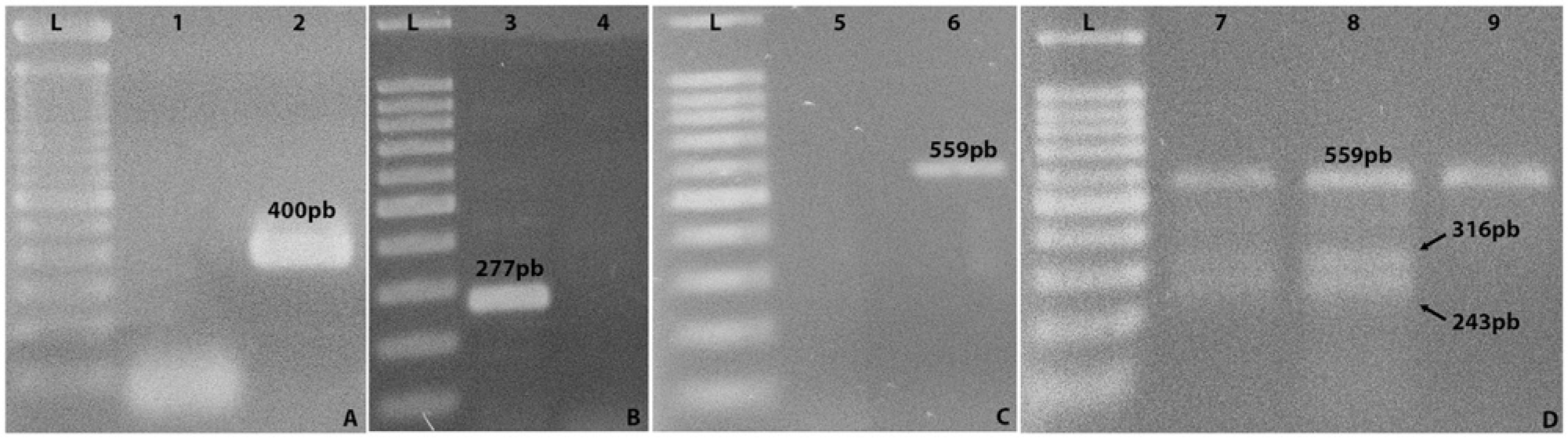
Agarose gel electrophoresis stained by ethidium bromide (0.01%) from positive control polymerase chain reaction (PCR) assays.
The current study reports the occurrence of ADPKD as detected by PCR in domestic cats in Brazil. Table 1 shows the total number of animals and the prevalence of PKD1 mutant gene–positive cats as identified by touchdown PCR, according to location, sex, and breed. In the present study, 9% of the cats tested were positive for the PKD1 mutant gene. Collective prevalence in cats from the Hvet-UnB was 15%, including animals of all breeds, as compared with a collective prevalence of 4% in samples acquired via the anti-rabies vaccination campaign. Typically, patients from clinics and hospitals displayed behavioral alterations that were first noticed by their owners; such clinical signs and conditions characteristically allowed for detection of a disease, which could explain the difference in prevalence (P < 0.001).
Total number of animals and autosomal-dominant polycystic kidney disease prevalence according to location, sex, and breed.*
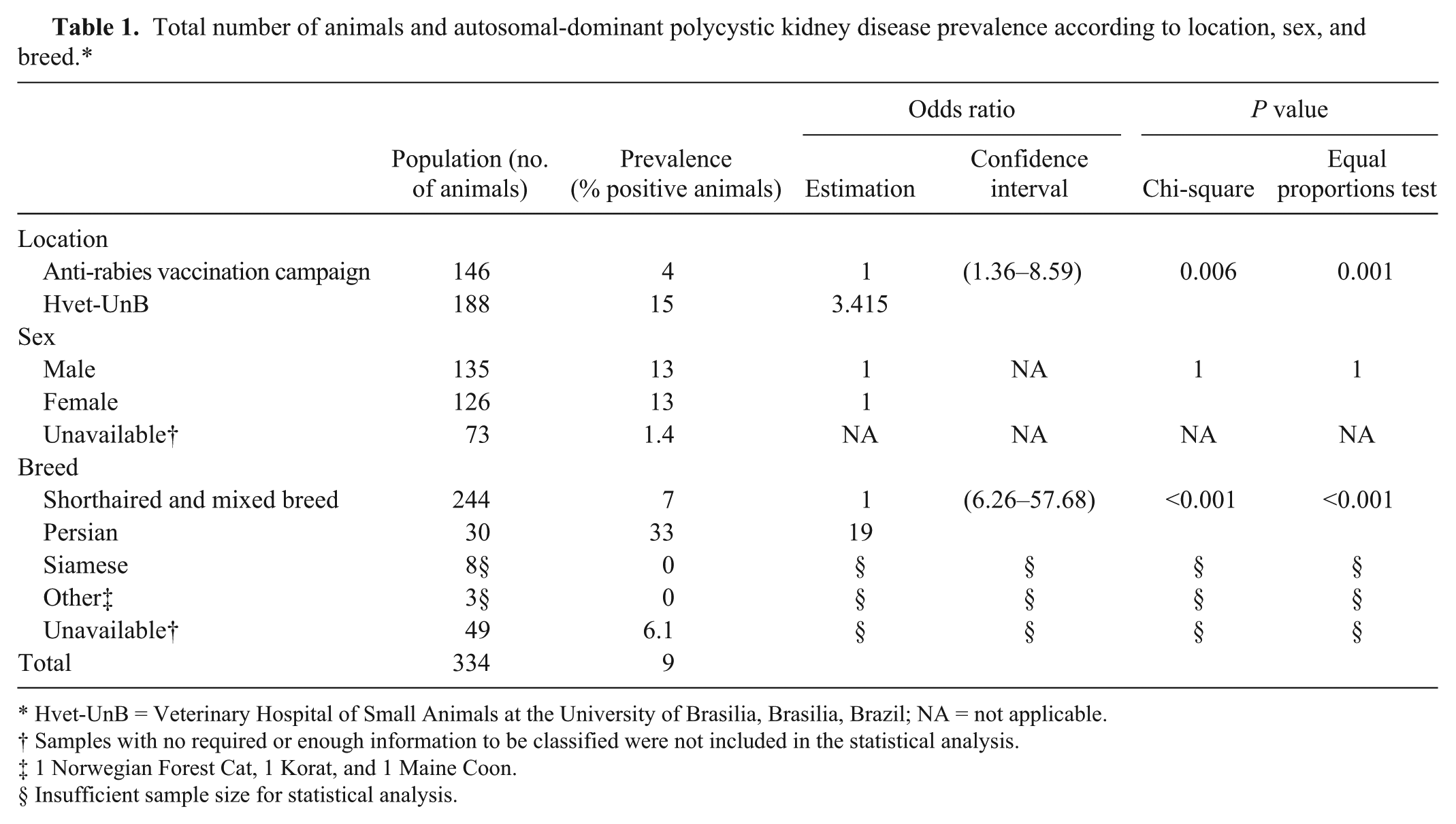
Hvet-UnB = Veterinary Hospital of Small Animals at the University of Brasilia, Brasilia, Brazil; NA = not applicable.
Samples with no required or enough information to be classified were not included in the statistical analysis.
1 Norwegian Forest Cat, 1 Korat, and 1 Maine Coon.
Insufficient sample size for statistical analysis.
In the current study, the prevalence of the PKD1 mutant allele in Persian cats was 33%, which is consistent with the 30–38% prevalence reported for Persian cats worldwide. 11 Most research uses groups of purebred cats, and approximately 80% of breeds have some genetic influence from the Persian lineage, 12 including mixed breeds. There is a concern in testing all cats because ADPKD prevalence differs in each breed, and there is, to date, no data published on mixed-breed cats. 1 In the present study, 7% of mixed-breed cats were positive for the PKD1 mutant gene by PCR, which suggests that the mutation is present in other lineages of the domestic cat population.11,13 Other breeds of cat evaluated in the study were negative for the PKD1 mutant gene as assessed by PCR (Table 1), but the numbers assessed were too small to warrant their inclusion in statistical analysis; such low numbers cannot be deemed representative of their respective breeds in any wider sense. However, in conjunction with the existing literature, the results suggest that all breeds should be tested for ADPKD mutation. Genetic tests should help identify ADPKD carriers, and these cats should in turn not be included in breeding programs, in order to prevent the ongoing transmission of this disease. 11
The presence of the PKD1 mutant gene analyzed in the current study is the main cause of ADPKD in cats, but notably other mutations in different exons of the PKD1 gene have been reported, as have environmental factors that could influence PKD progression. 12 According to studies of human ADPKD, 85% of cases reportedly result from a mutation in the PKD1 gene on chromosome 16p13.3, and the remaining 15% are due to a mutation on chromosome 4q21-23, within the PKD2 gene; thus, a genetically similar occurrence may be seen in cats. 12
The touchdown PCR developed in the present study was adapted from a multiplex PCR 10 that is simpler and faster than the RFLP PCR method previously described.12,15 Comparison of these techniques confirms that a touchdown PCR approach can yield the same results as the complex and expensive RFLP PCR technique.
Persian and Persian-related cats should be tested for ADPKD before breeding. Even when cat breeders know that they should never interbreed carriers of the PKD1 mutant gene, this continues to happen for several reasons: 1) carriers do not express clinical signs of ADPKD early in life; 2) many breeders are still unaware of the disease and its impact; 3) ultrasound is either unavailable or cost-prohibitive; 4) breeding decisions are made before adequate accuracy of diagnosis; and 5) the disease is highly prevalent; thus, many catteries could lose approximately 40% of their breeding population. Genetic testing for ADPKD provides an accurate and efficient method of selecting breeding cats, and thus could function to remove ADPKD from breeding programs. 12 Unfortunately, many breeders avoid seeking diagnoses, fearing losing breeding animals. There was no statistically significant association between the PKD1 mutant gene and sex (P = 1), suggesting that the gene mutation has no sex predisposition. ADPKD is an autosomal-dominant disease, and this result is consistent with previous studies. 5
Hemogram analysis showed no differences between the means of any parameters (data not shown). Erythrogram, leukogram, and platelet values remained within reference ranges. 8 With regard to biochemical parameters, the only statistically significant difference was between the mean analysis of globulins (P = 0.02), where positive animals (5.83 ± 1.21 g/dl) had a value above reference, 9 greater than negative animals (5.05 ± 1.16 g/dl). Increased globulin concentration may be an indicator of inflammatory processes, usually nonspecific acute phase responses, which may also require evaluation of other markers such as albumin, C-reactive protein, transferrin, and haptoglobins.4,14 It is noteworthy that most samples were from veterinary hospital animals, which possibly introduced bias.
Veterinarians should be able to provide owners and breeders with a sound knowledge base with regard to the diagnosis and treatment of genetic disorders. This includes fostering an ability to explain the results of genetic tests, and being aware of the emotional ramifications that this news may bring in cases of a positive diagnosis. In ADPKD-positive animals, the genetic mutation may be a sign of premature death or shortened lifespan in unhealthy cats. A conscientious breeder and/or owner can help and encourage other owners to undertake genetic diagnoses to reduce the prevalence of ADPKD in domestic cat populations and also promote an understanding that carrier cats need veterinary care and more frequent clinical evaluations. 7 Quantifying the prevalence of ADPKD is critical for patient control because the simplest way to prevent gene transmission is sterilizing carriers, and not introducing them into breeding programs. For this, genetic testing is the most valuable and effective diagnostic method.
In conclusion, the current study showed that a touchdown PCR method was successful for detection of the PKD1 mutation gene in domestic cats, and should be a diagnostic method in clinical routine examinations, especially in breeding programs. The overall prevalence in the sampled population was 9%, including 7% of mixed-breed cats, suggesting that mutation is not limited to purebred animals. Persian cats exhibited a prevalence of 33%, which is high and consistent with other studies in different countries.
Footnotes
a.
Vet abc, HORIBA Instruments Brasil Ltda, São Paulo, Brazil.
b.
Bio2000, Bioplus, Barueri, São Paulo, Brazil.
c.
Illustra Blood genomicPrep Mini Spin kit, GE Healthcare Technologies, Piscataway, NJ.
d.
C1000 thermal cycler, Bio-Rad Laboratories, Hercules, CA.
e.
Invitrogen Brasil Ltda, Vila Guarani, São Paulo, Brazil.
f.
New England Biolabs Inc., Ipswich, MA.
g.
UV transilluminator, UVP LLC, Upland, CA.
h.
Vetec, Sigma-Aldrich, St. Louis, MO.
i.
Excel, Microsoft Corp., Cambridge, MA.
j.
SPSS Statistic 19, IBM SPSS, Armonk, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with regard to the research, authorship, or publication of this article.
Funding
The authors wish to thank CNPq (National Council of Scientific and Technological Development) and CAPES (Brazilian Federal Agency for the Support and Evaluation of Graduate Education) for their support with regard to scholarship funding.
