Abstract
The aims of the present study were to determine the prevalence of hemoplasmas in cats and dogs from the Barcelona area of Spain with the use of species-specific quantitative polymerase chain reaction (qPCR) assays and to evaluate any associations between hemoplasma infection, clinical presentation, and vector-borne infections. Blood samples from cats (191) and dogs (182) were included and were classified as healthy (149) or unhealthy (224). Ethylenediamine tetra-acetic acid blood samples underwent DNA extraction and qPCR analysis. Mycoplasma haemofelis, ‘Candidatus Mycoplasma haemominutum’, and ‘Candidatus Mycoplasma turicensis’ were detected in cats, whereas Mycoplasma haemocanis and ‘Candidatus Mycoplasma haematoparvum’ were detected in dogs, with prevalences of 3.7%, 9.9%, 0.5%, 14.3%, and 0.6%, respectively. In cats, no association between hemoplasma infection and health status, age, breed, presence of anemia, Feline leukemia virus status, and other vector-borne infections was found, but outdoor access (P = 0.009), male sex (P = 0.01), and Feline immunodeficiency virus status (P < 0.001) were significantly associated with hemoplasma infection. In dogs, sex, age, health status, presence of anemia, and breed were not significantly associated with hemoplasma infection, but a significant association was found between hemoplasma infection and vector-borne infections (P < 0.001). The present report documents the occurrence of feline ‘Candidatus M. turicensis’ and canine ‘Candidatus M. haematoparvum’ infections in Spain.
Three feline hemoplasma species exist: Mycoplasma haemofelis, ‘Candidatus Mycoplasma haemominutum’, and ‘Candidatus Mycoplasma turicensis’, 8,10,15,20 whereas dogs can be infected by 2 hemoplasma species: Mycoplasma haemocanis and ‘Candidatus Mycoplasma haematoparvum’. 1,7,14,18 The pathogenicity of hemoplasmas species varies from being asymptomatic to inducing severe hemolysis. The most pathogenic feline species is regarded as M. haemofelis. 19 Concurrent diseases and infections appear to influence the pathogenicity of the other feline hemoplasma species. Clinical cases of hemoplasmosis in dogs have occasionally been reported, but cofactors such as splenectomy, immunosuppression, or concurrent infections seem to play a role in pathogenesis of disease. 6,14
Worldwide geographical variation in the prevalence of the different feline and canine hemoplasma species has been reported. 1,15,22 In Europe, prevalence studies with the use of polymerase chain reaction (PCR) for the diagnosis of hemoplasmas in cats have only been performed on domestic cats in Switzerland, Germany, northern Italy, and the United Kingdom. 2,5,9,19 Moreover, prevalence studies that use PCR for the diagnosis of M. haemocanis and ‘Candidatus M. haematoparvum’ have only been performed on domestic dogs in France, Switzerland, Japan, northern Tanzania, and Trinidad. 1,7,12,18 Additional prevalence data for other countries are needed, and no information exists regarding hemoplasma prevalence in Spain. The aims of the present study were to determine the prevalence of hemoplasmas in cats and dogs presenting to the Veterinary Teaching Hospital of the Universitat Autònoma de Barcelona in Spain with the use of real-time quantitative PCR (real-time qPCR) assays and to evaluate any possible associations between hemoplasma infection, clinical presentation, and other vector-borne infections.
Blood samples from cats (n = 191) and dogs (n = 182) admitted to the Veterinary Teaching Hospital of the Universitat Autònoma de Barcelona between May 2005 and December 2007 were entered into the study. All cats and dogs were from Barcelona or the immediate surrounding area. The clinical records of all cases were reviewed to obtain data on signalment (sex, age, and breed). Additionally access to outdoors and Feline immunodeficiency virus (FIV) and Feline leukemia virus (FeLV) status a were recorded for the feline cases. On the basis of information in the clinical records (reason for presentation, clinical signs, physical examination findings, hematology, and serum biochemistry results), the health status of the cats and dogs was grouped as healthy or unhealthy. The animals were also classified according to the presence or absence of anemia (anemia defined as a packed cell volume of <29% for cats and <35% for dogs). Polymerase chain reaction amplification and quantification of Leishmania infantum, 4 and amplification and sequencing of Ehrlichia spp., Anaplasma spp., Rickettsia spp., Bartonella spp., Hepatozoon spp., and Babesia spp. had already been performed on all samples as previously described, 16,17 and results of these PCR assays were collated for analysis.
Characteristics of the 191 feline samples: breed, gender, lifestyle, age, Feline leukemia virus (FeLV) and Feline immunodeficiency virus (FIV) status, health status, and presence of anemia or other vector-borne coinfection of all cats testing positive or negative for hemoplasma polymerase chain reaction (PCR). *
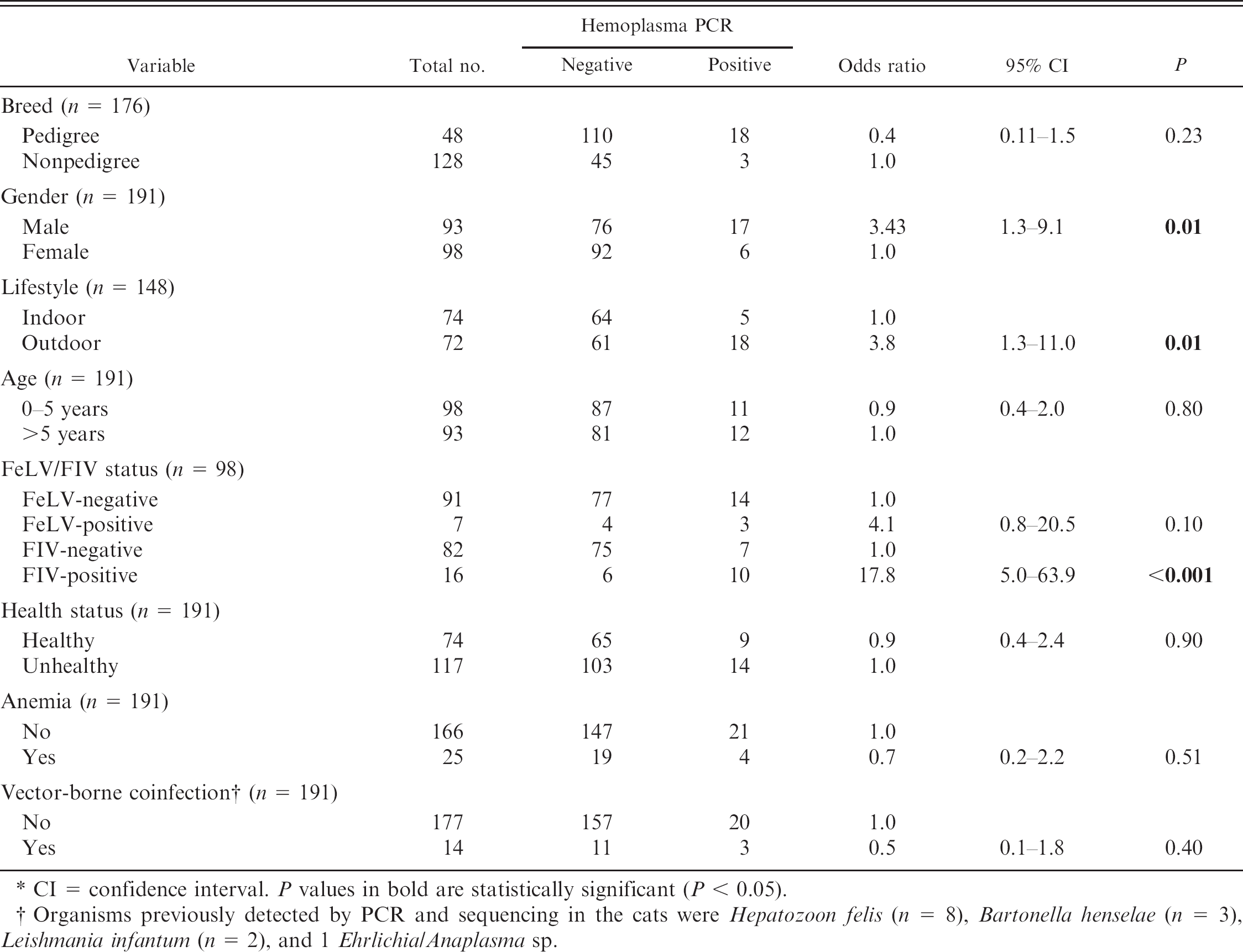
CI = confidence interval. P values in bold are statistically significant (P < 0.05).
Organisms previously detected by PCR and sequencing in the cats were Hepatozoon felis (n = 8), Bartonella henselae (n = 3), Leishmania infantum (n = 2), and 1 Ehrlichia/Anaplasma sp.
DNA was extracted from 0.5 ml of ethylenediamine tetra-acetic acid anticoagulated blood as described previously. 4 Polymerase chain reaction amplification of 5 μl of DNA (equivalent to 5 μl of initial blood volume assuming 100% extraction and PCR efficiency) was performed with the use of distinct real-time qPCR assays 9 to identify M. haemofelis, ‘Candidatus M. haemominutum’, and ‘Candidatus M. turicensis’ in the feline DNA samples, and to detect M. haemocanis and ‘Candidatus M. haematopar-vum’ 1 in the canine samples. All qPCR assays incorporated an internal amplification control (feline 28S ribosomal [r]DNA or canine glyceraldehyde-3-phosphate dehydrogenase [GAPDH]) to confirm the presence of amplifiable DNA and an absence of PCR inhibitors in the qPCR. 1,9 Positive control DNA samples obtained from cats and dogs infected with each of the hemoplasma species, and a negative control sample (water), were included in each PCR run. The PCR was performed in a real-time PCR detection system b as described previously. 1,9
Statistical analyses were performed with the use of a statistical package. c Chi-square analysis or Fisher's exact test was used. Statistical significance was set at P < 0.05.
Table 1 summarizes the characteristics of the feline samples, whereas Table 2 summarizes the hemoplasma PCR results. Twenty-three of the 191 cats (12%) were hemoplasma qPCR–positive, with ‘Candidatus M. haemominutum’ being most prevalent, followed by M. haemofelis and ‘Candidatus M. turicensis’. Dual, but not triple, infections were detected. The feline 28S rDNA threshold cycle (Ct) values ranged from 25.6 to 29.0 (‘Candidatus M. haemominutum’ positive), from 25.3 to 28.4 (M. haemofelis positive), and 28.7 for the single ‘Candidatus M. turicensis’–positive sample. The feline 28S rDNA Ct values for the hemoplasma-negative samples ranged from 25.3 to 27.1. Outdoor access (P = 0.01), FIV-positive status (P < 0.001), and male sex (P < 0.01) were significantly associated with feline hemoplasma infection (Table 1).
Hemoplasma detection by quantitative polymerase chain reaction in 191 feline samples. *

CI = confidence interval.
Table 3 summarizes the characteristics of the canine samples, whereas Table 4 summarizes the hemoplasma PCR results. Twenty-six of the 182 dogs (14.3%) were hemoplasma qPCR–positive; all 26 dogs were positive for M. haemocanis, including 1 dog dual infected with M. haemocanis and ‘Candidatus M. haematoparvum’. The canine GAPDH Ct value ranged from 24.9 to 27.3 for the M. haemocanis–positive samples and was 22.8 for the ‘Candidatus M haematoparvum’–positive sample. The canine GAPDH Ct values for the hemoplasma-negative samples ranged from 24.5 to 29.4. Only coinfection with other vector-borne infections was significantly associated with positive hemoplasma infection (P < 0.001; Table 3), although unhealthy dogs tended to be more likely to be hemoplasma qPCR positive (P = 0.05).
The current study documents the occurrence of M. haemofelis, ‘Candidatus M. haemominutum’, ‘Candidatus M. turicensis’, M. haemocanis, and ‘Candidatus M. haematoparvum’ in the Barcelona area of Spain. To the authors’ knowledge, this is also the first report of ‘Candidatus M. turicensis’ and ‘Candidatus M. haematoparvum’ DNA in 1 cat and 1 dog, respectively, in Spain. The study does have certain limitations because of the relatively small number of cases analyzed, which precluded performance of multivariable analysis, and statistical analysis could only be performed for hemoplasma-positive status rather than individual hemoplasma species. Nevertheless, the findings expand the knowledge of canine and feline hemoplasmas.
Characteristics of the 182 canine samples: breed, gender, age, health status, and presence of anemia or vector-borne coinfection of all dogs testing positive or negative for hemoplasma polymerase chain reaction (PCR). *
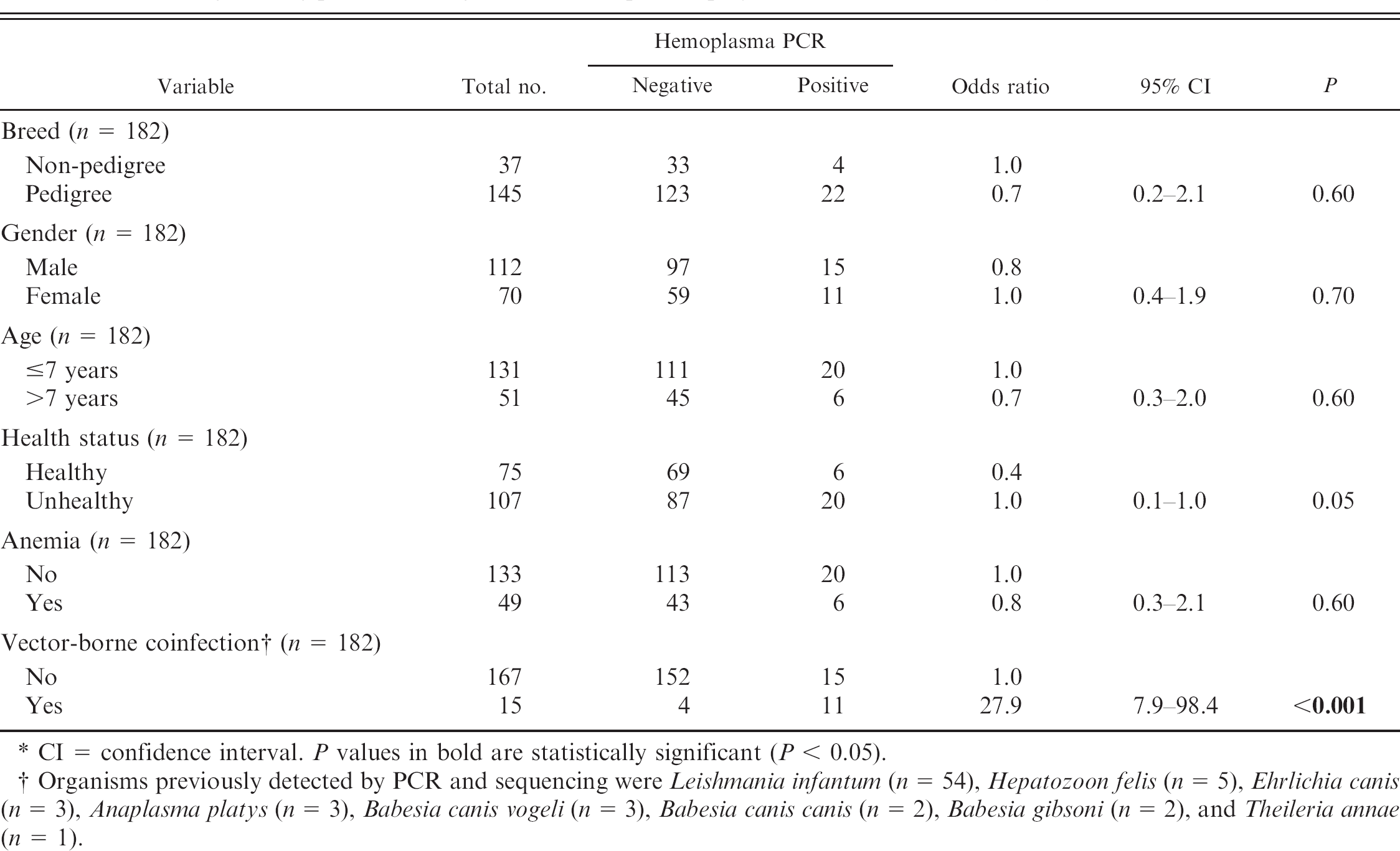
CI = confidence interval. P values in bold are statistically significant (P < 0.05).
Organisms previously detected by PCR and sequencing were Leishmania infantum (n = 54), Hepatozoon felis (n = 5), Ehrlichia canis (n = 3), Anaplasma platys (n = 3), Babesia canis vogeli (n = 3), Babesia canis canis (n = 2), Babesia gibsoni (n = 2), and Theileria annae (n = 1).
Hemoplasma detection by quantitative polymerase chain reaction in 182 canine samples. *

CI = confidence interval.
The prevalence of the feline hemoplasma species detected in the present study is very similar to that found in recent studies from Switzerland 19 and the United Kingdom, 9 but significantly lower than that reported in German 2 and Italian studies. 5 The canine hemoplasma prevalence rates obtained in the present study differ greatly from studies previously reported in France and Switzerland, 7,18 in which infection with ‘Candidatus M. haematoparvum’ was more prevalent or similar to that with M. haemocanis, but results are similar to those for dogs in Trinidad and Tanzania, where M. haemocanis was the most commonly detected canine hemoplasma species. 1 Difficulties exist in comparing results from different studies because small sample sizes might not truly represent whole populations and different types of animals are sampled in different studies (e.g., variable percentages of healthy and sick animals, demographics). Vector distribution could also influence results. Rhipicephalus sanguineus is thought to be a vector for canine hemoplasmas, 13 and different Rhipicephalus sanguineus tick distributions might influence results. 1,7,18
No association between anemia and hemoplasma infection was found, in contrast to associations previously reported between anemia and feline hemoplasma species. 5,22 Clinical signs attributable to the hemoplasmas in cats and dogs are variable, and severity of illness depends on the infecting hemoplasma species and presence of concurrent diseases or infections. 5,6,14,19 It is possible that acute hemoplasma infections primarily result in anemia, in contrast to chronic infections, as has been previously suggested. 19 The sampling conditions of naturally infected animals in the current study were not able to distinguish between acute and chronic infections, but sampling of chronically infected animals could explain the failure to find an association between anemia and hemoplasma infection.
The finding that outdoor FIV-infected male cats are more likely to be hemoplasma infected supports the suggestion that hemoplasma transmission can occur via fighting. 3,5,19 However, the mode of transmission of hemoplasma infection in field cases has not been confirmed, although transmission by fleas and ticks has been investigated. 21,23 In the present study, presence of coinfection with other vector-borne pathogens was significantly associated with a positive hemoplasma qPCR result. The current study is the first report in Europe to investigate and document such an association and might reflect similar vectors being involved in the transmission of these infections or a more complex host immune response associated to coinfection. 11
The results of the present study confirm the presence of M. haemofelis, ‘Candidatus M. haemominutum’, ‘Candidatus M. turicensis’, M. haemocanis, and ‘Candidatus M. haematoparvum’ in cats and dogs from the Barcelona area of Spain, with prevalences of 3.7%, 9.9%, 0.5%, 14.3%, and 0.6%, respectively. To the authors’ knowledge, this is the first report of ‘Candidatus M. turicensis’ in cats and ‘Candidatus M. haematoparvum’ in dogs in Spain.
Acknowledgements. This study was funded in part by the Wellcome Trust (grant 077718).
Footnotes
a.
SNAP® FIV/FeLV Combo Test, IDEXX Laboratories, West-brook, ME.
b.
Bio-Rad Laboratories Ltd., Hemel Hempstead, UK.
c.
Statistical Package for the Social Sciences version 14.0, SPSS Inc., Chicago, IL.
