Abstract
Testing for specific antibodies against Bovine viral diarrhea virus (BVDV) in pooled serum may present an opportunity to decrease the cost of screening for herds of high seroprevalence and increased likelihood of active infection. Experimental serum pools (n = 280) were created by combining equal aliquots of serum from between 5 and 25 individuals. A further 188 serum pools were generated from field serum samples. All pools and individual sera were tested for BVDV-specific antibodies by enzyme-linked immunosorbent assay (ELISA), according to manufacturer’s instructions. Pools returned repeatable results, with coefficients of variation generally below 10%. The presence of serum from a persistently infected (PI) individual in the pool had no significant effect on the ELISA sample-to-positive (S/P) ratio. The results revealed that a single strong antibody-positive individual could maintain a positive result (at the manufacturer’s threshold) in pools of up to 128, while even a single weak-positive animal would generate a positive result in pools of up to 8. The S/P ratio of the pool was positively related to the within-pool prevalence of antibody-positive individuals. However, as the strength of the individual positive animals contributing to the pool had a large effect on the pool S/P ratio, the S/P ratio could not be used to accurately predict the within-pool prevalence of field serum pools. An alternative method of S/P ratio interpretation was pursued, and a two-graph receiver operating characteristic analysis allowed segregation of pools into low, medium, and high risk with good results when applied to field serum pools.
Keywords
Introduction
Bovine viral diarrhea (BVD) is one of the world’s most prevalent infectious diseases of cattle, and has significant economic impacts on cattle production systems. 10 Consequently, time and cost efficient diagnosis of BVD at both the individual and herd level is vital to enable reduction in the prevalence of infection and, in turn, the financial impact of disease.
At present, various diagnostic options are available and have been reviewed. 9 Such options include methods for the detection of BVD virus (BVDV) or viral antigen and of BVDV-specific antibody. Virus or antigen detection methods, such as antigen enzyme-linked immunosorbent assay (ELISA) or reverse transcription polymerase chain reaction (RT-PCR) can be used to identify persistently infected (PI), immunotolerant animals that are the result of fetal infection prior to the development of immunocompetence. 2 These PI individuals are at the center of the disease’s epidemiology; their identification and subsequent removal from cattle populations reduces the incidence of BVDV infection. 11 However, PI animals tend to represent only approximately 0.5–2% of animals in an endemically infected population. 7 As such, it is valuable to perform preliminary screening of herds to identify those most likely to be actively infected (e.g., containing a PI individual). For this purpose, detection of previously BVDV-exposed, seropositive cattle by antibody testing (by serum neutralization or ELISA) can be applied. 10 High within-herd seroprevalence is associated with an increased likelihood of active infection. 4
Antibody ELISA can return a result on serum or milk from individual animals; however, if conducted individually for a whole herd, testing can quickly become prohibitively expensive. In dairy herds, bulk milk antibody testing is a convenient, cost-efficient method of assessing the within-herd prevalence of BVDV-exposed cattle—a higher bulk milk result is indicative of high within-herd antibody prevalence and, in turn, higher likelihood of active infection. 4 However, bulk milk testing is limited to lactating cattle. Therefore, an equivalent bulk testing method for use in nonmilking stock, such as beef cattle, young stock, or dry cows, would be a valuable tool for BVDV diagnosis. The present study aimed to evaluate the use of pooled serum as a diagnostic sample for the identification of herds with high within-herd seroprevalence and, therefore, high likelihood of active BVDV infection.
Materials and methods
Serum pool creation and testing
Experimental serum pools
A total of 187 individual serum samples (collected for previous research or diagnosis) from 94 unvaccinated animals (some serial testing) were tested for the presence of BVDV-specific antibodies by ELISA, a performed according to manufacturer’s instructions. A sample-to-positive (S/P) ratio was calculated for each sample and an S/P ratio >0.3 was considered positive. Seropositive samples originated from cattle blood sampled during investigation of a herd endemically infected with BVDV. The cattle, aged 1–6 years, were naturally exposed to BVDV infection prior to sample collection. In total, 85 positive and 102 negative samples were used for pooling.
A total of 280 experimental serum pools were created by combining equal amounts of several individual serum samples in a microtube and mixing by pipetting. The within-pool prevalence of antibody-positive individuals, the number of individuals contributing to the pool (pool size), and the presence or absence of serum from a PI individual in the pool were varied. Eight unique pools were created for each of 35 combinations of pool size, within-pool prevalence, and PI contribution (Table 1), resulting in a total of 280 pools. Seropositive and seronegative individual samples were nonselectively allocated to a pool. Each (of 8) unique pools at any given combination of pool size, within-pool prevalence, and PI contribution was created using different seropositive and seronegative samples than the remaining 7 unique pools. Variation in the strength of seropositivity of samples was intentionally not stratified, but was recorded and considered retrospectively for distributional bias. All pools were tested in triplicate by ELISA, a with results expressed as mean S/P ratio.
Specifications of the 35 combinations of within-pool prevalence of antibody-positive animals, presence or absence of serum from a persistently Bovine viral diarrhea virus (BVDV) infected (PI) animal in the pool, and the number of individuals contributing to the pool (pool size) that were used to generate 280 experimental serum pools (8 pools per combination) to evaluate testing of serum pools by commercially available enzyme-linked immunosorbent assay as a method of identifying herds with high within-herd seroprevalence of BVDV-specific antibodies.
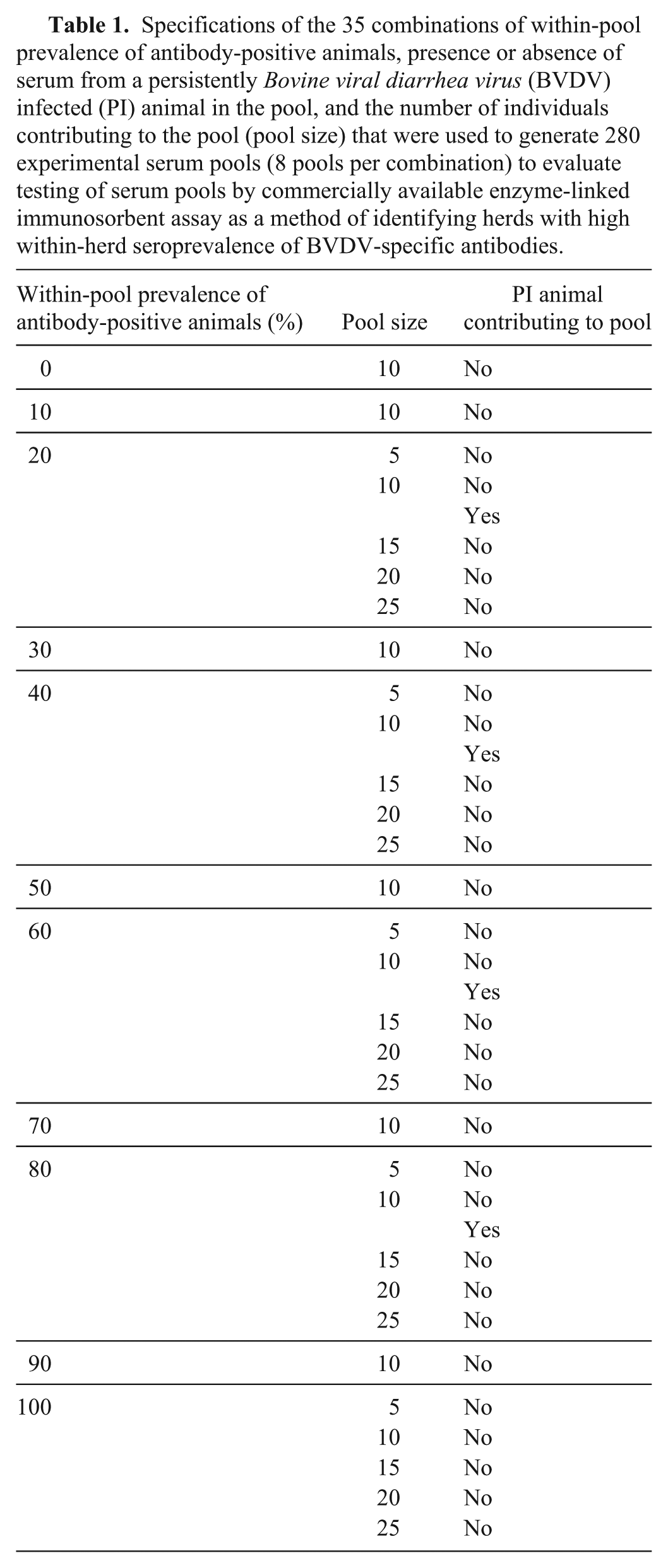
Field serum pools
Serum from approximately 8 heifers (<2 years) and 8 mature age cows was collected from each of 69 cattle herds that had not been vaccinated against BVD. Actual sample size varied from 5 to 9 animals per cohort, and 9 to 17 animals per herd. Herds varied in size from <20 to >100. Three serum pools were created for each herd—a heifer pool, a cow pool, and a total herd pool. In 1 herd, cow samples were unable to be obtained, resulting in only 1 pool being created for that herd. In another herd, error in sample labeling resulted in heifer and cow sera being indistinguishable, resulting in only 1 (total herd) pool being created. All individual sera (n = 962) and serum pools (n = 188) were tested for BVDV-specific antibodies by ELISA. a Individual serum samples were considered positive if they returned an S/P ratio >0.3, as per manufacturer’s recommendations. The within-pool prevalence was calculated for each pool (number of antibody-positive individuals/total number of individuals in the pool), and pooled results were expressed as an S/P ratio. Individual sera were also tested by BVDV antigen ELISA b with a positive result taken to indicate a PI animal.
Dilution series
Eight antibody-positive serum samples (mean S/P ratio, when undiluted, ranging from 0.62 to 1.59) were serially diluted in antibody-negative serum. Dilutions were then tested (in triplicate) by ELISA a following the manufacturer’s procedure for neat serum samples. Results were expressed as the mean S/P ratio of 3 replicates.
Repeatability
Twenty-two experimental pools (within-pool prevalence of 0–100%) were selected from the above-mentioned pools (n = 280). Seven replicates of each pool were tested by ELISA a according to manufacturer’s protocol for serum samples.
Statistical analyses
Repeatability, and effect of a PI individual contributing to the pool
Coefficients of variation (CVs) were calculated for each of 22 experimental pools for which 7 replicates were tested to assess repeatability. Two-tailed Student t-tests were performed using a spreadsheet program c to determine any difference between experimental pools containing serum from a PI animal (n = 8) and pools not containing serum from a PI animal (n = 8), at each of 4 within-pool prevalences (20%, 40%, 60%, and 80%), and overall (n = 32 with PI, 32 without PI). Experimental pools used in this analysis all contained 10 individuals. A P value <0.05 was considered to be significant.
Dilution effect
The mean and 95% confidence interval (CI) of the mean S/P ratios from each of the 8 serially diluted positive serum samples were calculated at each dilution. The diagnostic sensitivity (and 95% CI) of the ELISA was also calculated at each dilution, with a mean S/P ratio >0.3 for a diluted sample considered positive.
Effect of pool size on mean S/P ratio of a pool
Multiple regression was performed on the data from the experimental pools using the statistical package R (http://www.r-project.org/) using function lm(), with log(mean S/P ratio +1) fitted as the independent variable and log(within-pool prevalence + 1) and pool size fitted as covariates. A p-value <0.05 was considered to be significant.
Development of a model for the prediction of within-pool prevalence from the S/P ratio of a pool
A power regression line (and 95% prediction interval) was fitted to the data from the experimental pools using the statistical package R using function lm(), with log(within-pool prevalence + 1) fitted as the independent variable and log(mean S/P ratio + 1) fitted as a covariate. Data was back-transformed prior to graphing.
Prediction of within-pool prevalence using pooled S/P ratio for field serum pools
The above power relationship between within-pool prevalence and pooled S/P ratio generated from experimental pools (regardless of pool size) was applied for the purpose of predicting within-pool prevalence, based on S/P ratio of each field serum pool (n = 188). The absolute discrepancy between actual and predicted within-pool prevalence was calculated for each pool. Mean and standard deviation (SD) of the absolute discrepancies for all field pools were calculated.
TG-ROC analysis
All experimental pools with within-pool prevalence >65% were considered to simulate herds that are likely to be experiencing current BVDV infection. 4 Such herds were designated “current.” Parametric two-graph receiver operating characteristics (TG-ROC) analysis was conducted using computer software d to generate a 95% intermediate range for identification of current herds. The upper and lower limits of the intermediate range (representing the thresholds at which 95% diagnostic sensitivity and 95% diagnostic specificity, respectively, are achieved) were used to designate pools as “low,” “medium,” and “high” risk.
Predicting herd risk for field serum pools
The limits generated by TG-ROC analysis of experimental pool data were applied to field serum pools. Each field serum pool was designated as “low,” “medium,” or “high” risk based on the S/P ratio of the pool. The proportion (and 95% CI) of field pools with within-pool prevalence >65% was calculated for low-, medium-, and high-risk pools. Mean (with 95% CI) within-pool prevalence was also calculated for low-, medium-, and high-risk pools.
Results
Repeatability, and effect of a PI individual contributing to the pool on S/P ratio
The CV of S/P ratios remained below 10.5%, and 90% (18/20) remained below 10%, for all experimental serum pools with within-pool prevalence >0 that were assessed for repeatability by testing of 7 replicates. The 2 (of 20) pools that did not meet the CV <10% criteria returned CVs of 10.3% and 10.4%, and had within-pool prevalences of 100% and 20%, respectively. The 2 experimental serum pools with within-pool prevalence of zero that were assessed for repeatability returned CVs of 62.9% and 131.7%, respectively. However, the high CVs observed were a function of low mean S/P ratios of 0.027 and 0.011, respectively. The SDs of these 2 samples were 0.017 and 0.014, respectively, as compared to SDs varying from 0.028 to 0.135 for the pools with within-pool prevalence greater than zero. Two-sided Student t-tests revealed no significant differences (P > 0.05) in mean S/P ratio between experimental pools of 10 individuals containing serum from a PI individual and those not containing serum from a PI animal, at any within-pool prevalence (20%, 40%, 60%, and 80%) or overall (Fig. 1).
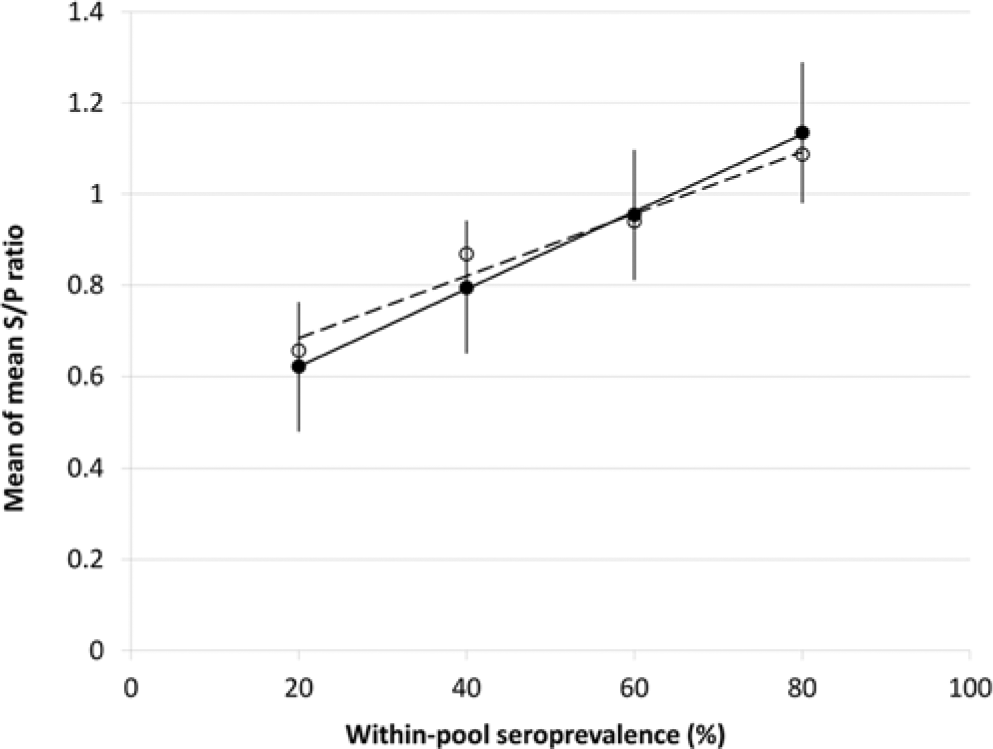
The mean of the mean sample-to-positive (S/P) ratios of 3 replicates each of serum pools containing (n = 32; solid points, solid line; r2 = 0.99) and not containing (n = 32; hollow points, dashed line; r2 = 0.97) serum from a persistently infected individual at varying within-pool prevalence of antibody-positive animals, when tested by commercially available Bovine viral diarrhea virus (BVDV) antibody enzyme-linked immunosorbent assay for specific antibodies against BVDV.
Dilution effect, and effect of pool size on the mean S/P ratio of a pool
The mean S/P ratio of each of the 8 individuals tested decreased as the dilution increased (Fig. 2). The individuals that had the highest S/P ratios when not diluted tended to maintain higher S/P ratios at subsequent dilutions (Fig. 2). The mean of the mean S/P ratios from each of the 8 individuals decreased exponentially as dilution increased, dropping below the manufacturer’s positivity threshold of an S/P ratio >0.3, when diluted 1:64.
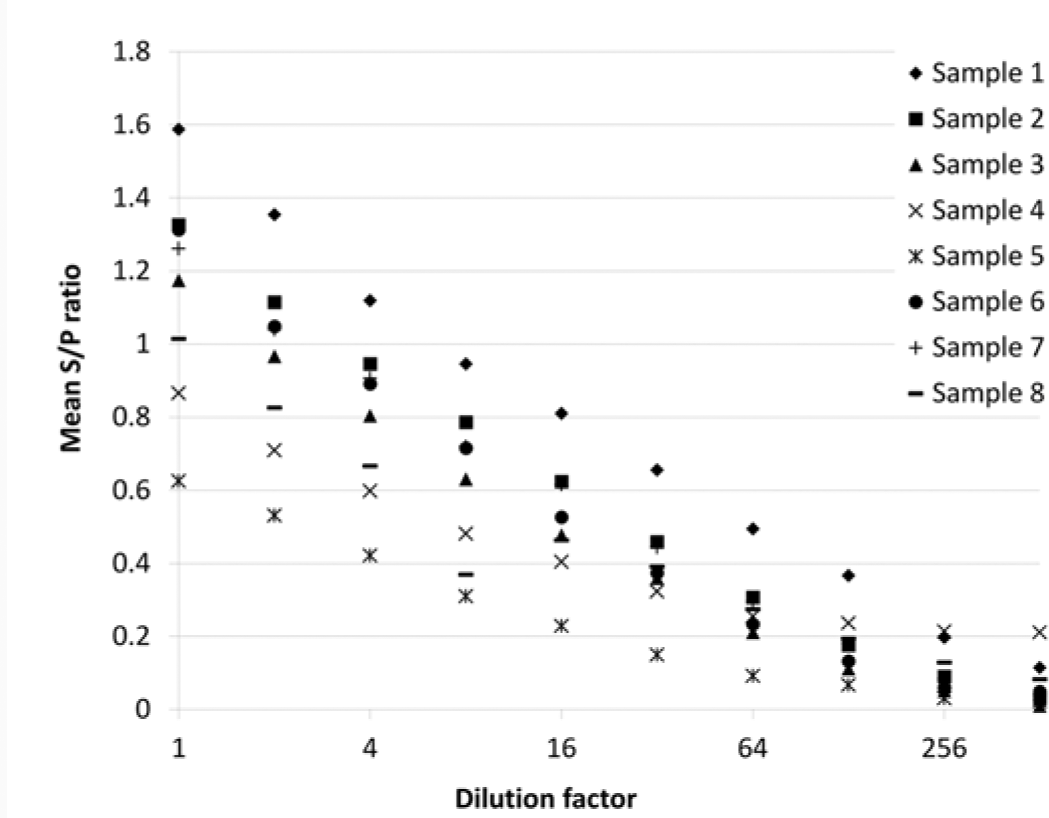
The effect of serial dilution of Bovine viral diarrhea virus (BVDV) antibody–positive serum samples (n = 8) in antibody-negative serum on the mean sample-to-positive (S/P) ratio of 3 replicates of the individual sample when tested for BVDV antibodies by commercially available enzyme-linked immunosorbent assay.
By considering an individual positive when its mean S/P ratio was >0.3, the diagnostic sensitivity of detection of a positive serum sample when diluted in negative serum (i.e., a single positive animal in a serum pool) was observed to be 100% at dilutions of 1:2, 1:4, and 1:8 (corresponding to pool sizes 2, 4, and 8, respectively). The diagnostic sensitivity dropped to 87.5% (95% CI: 64.6–100.0%; 7/8 positive individuals detected) at dilutions of 1:16 and 1:32, before dropping to 25% (95% CI: 0–55.0%) and 12.5% (95% CI: 0–35.4%; 2/8 and 1/8 individuals detected) at dilutions of 1:64 and 1:128, respectively. No samples tested positive at dilutions of 1:256 and 1:512, producing a diagnostic sensitivity of zero. Regression analysis showed that pool size had no significant effect on the mean S/P ratio of a pool (p > 0.05).
Prediction of within-pool prevalence of field serum pools using pooled S/P ratio
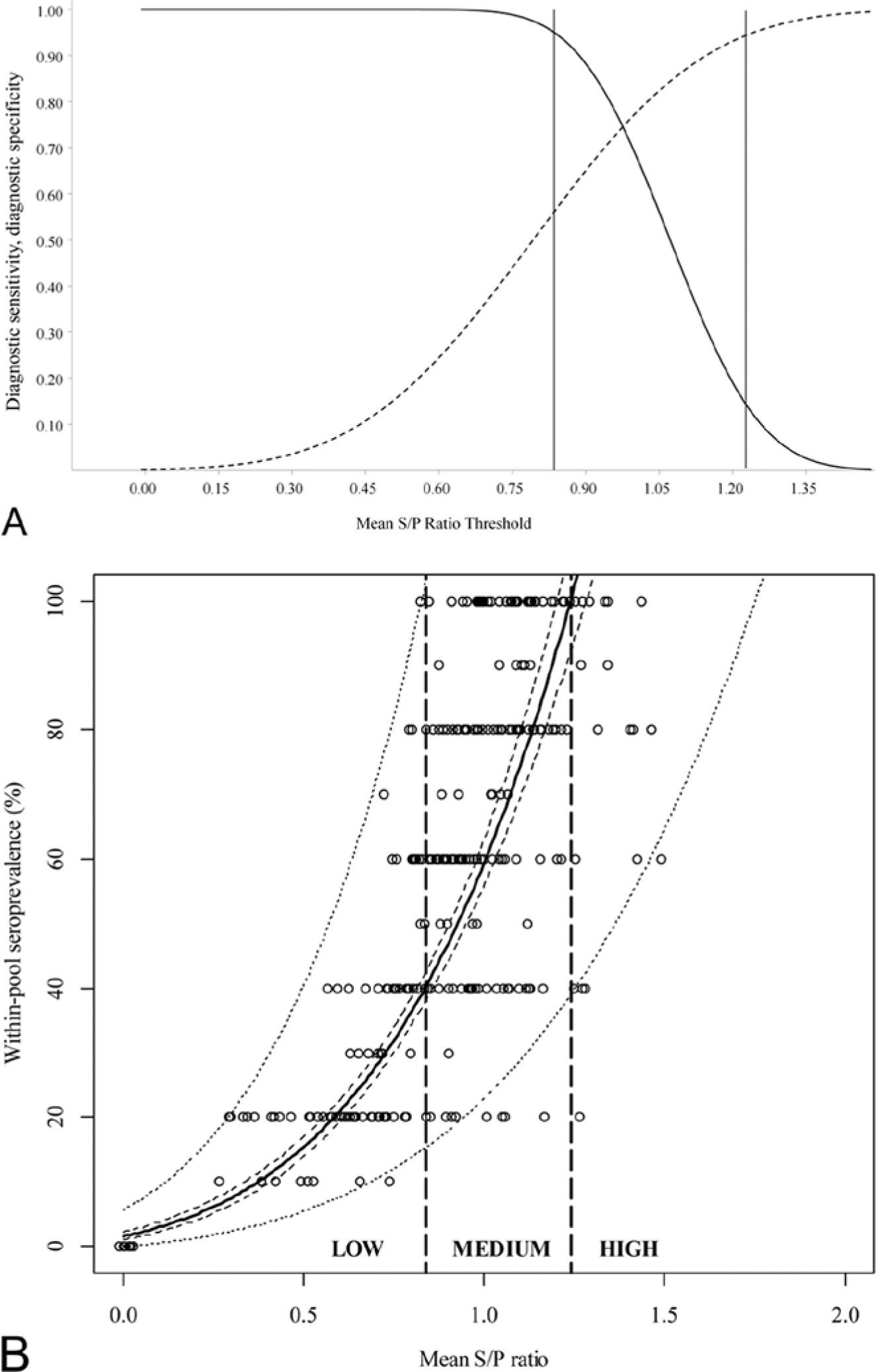
When the power regression model generated from experimental pool data (Fig. 3B) was applied for prediction of within-pool prevalence from the S/P ratio of field serum pools, the mean absolute discrepancy between the actual and predicted within-pool prevalence was 22.1% (SD = 22.7%).
TG-ROC analysis
The TG-ROC analysis of experimental pool data (Fig. 3A) returned lower and upper 95% intermediate range limits of 0.84 and 1.24 S/P ratio, respectively. At these limits, 95% diagnostic sensitivity and 95% diagnostic specificity, respectively, are achieved for detection of pools with within-pool prevalence >65% (likely to be currently BVDV infected 4 ). Therefore, pools returning an S/P ratio <0.84 were designated as low risk, those returning an S/P ratio 0.84–1.24 were designated medium risk, while those returning an S/P ratio >1.24 were considered high risk (Fig. 3B).
Predicting herd risk for field serum pools
When the thresholds generated by TG-ROC analysis were utilized to classify field serum pools as low risk, medium risk, or high risk, a total of 70 pools was classified as low risk, 41 were medium risk, and a further 77 were high risk. Both the proportion of pools with within-pool prevalence >65% and the mean within-pool prevalence increased significantly from low-risk pools to medium-risk pools, and again, increased significantly from medium-risk pools to high-risk pools (Table 2). The proportions of pools of size 8 and 16 did not vary between low-risk, medium-risk, and high-risk pools. Antigen ELISA on individual field serum samples revealed 2 apparent PI heifers from different herds. All 3 pools from each of these 2 herds were classified as high risk. As herds were sampled, and not thoroughly profiled, the true infection status of remaining herds is unknown.
The proportion of serum pools that had a within-pool prevalence of antibody-positive individuals >65%, and the mean within-pool prevalence of antibody-positive animals for pools of field serum designated as “low,” “medium,” or “high” risk based on the result (expressed as a sample-to-positive [S/P] ratio) of an antibody enzyme-linked immunosorbent assay performed on the serum pool.

Numbers in parentheses indicate the S/P ratio.
Numbers in parentheses indicate the 95% confidence interval.
Discussion
The results presented herein show that serum pools tested by commercially available ELISA for the detection of BVDV-specific antibodies will return reliable, repeatable results (expressed as an S/P ratio). Furthermore, unlike commercial BVDV antigen ELISAs that may experience interference in the presence of specific antibodies, 6 the presence of antigen (in the form of serum from a persistently BVDV-infected animal) did not significantly affect the antibody result of the pool in the current study. This is of particular importance, as interference from antigen would greatly diminish the utility of pooled serum as a screening test for determining the likelihood of a herd to be actively infected and, consequently, have a high seroprevalence of BVDV-specific antibodies. As shown in a previous study, 1 some variation in viral titer of PI animals exists, although the PI serum utilized in the previous study was from an adult animal that returned consistently high results by both antigen ELISA and quantitative RT-PCR on several consecutive occasions, and was, therefore, likely representative of a majority of high viral titer individuals. A more rigorous study of the effects of antigen on antibody detection is warranted to ensure that the findings under the specific conditions of the current study also apply to broader application.
The results showed that pool size had no significant effect on the mean S/P ratio of a pool, and hence, no substantial predictive power was to be gained by utilizing separate predictive models for each pool size. Consequently, a single predictive model that did not account for pool size was applied to field serum pools.
A single model that relates within-pool prevalence to the S/P ratio of the pool allows a simple, direct, and flexible application. In particular, this allows for pools of any size from 5 to 25 to be tested with the same interpretation. The appropriate sample size (and hence, pool size) may vary depending on herd size, estimated prevalence, acceptable error, and desired level of confidence. Indeed, sample size calculations performed in WinEpiscope 2.0 e for the detection of BVDV in South Australian cattle herds revealed an appropriate sample size of 8 individuals from a given cohort, based on mean herd size of 100 and 200 for beef and dairy, respectively, with an estimated within-herd prevalence of 25% and 90% confidence, respectively. Field serum pools of size 8 and 16 were generated accordingly.
The serial dilution data shown herein illustrates that in pools of up to 8 individuals, a single antibody-positive animal (even a weak one) is sufficient to produce a positive result (with 100% diagnostic sensitivity) at the manufacturer’s threshold of S/P ratio >0.3, while a strong positive can still induce a positive result in pools of up to 128 individuals. It is clear from the results of the present study that the S/P ratio ELISA result of a stronger positive individual will maintain a positive result at higher dilutions than a weaker positive (as intuition would suggest), and as such, these strong positives contribute a larger S/P ratio increase when pooled than a weak-positive individual. Given this, it is not surprising that there is substantial variation in the S/P ratio ELISA results of pools of a given within-pool prevalence—the strength of the positive individuals contributing to the pool seems to have a strong impact on the pooled result. As a result, a pool containing few strong positives may return the same result as a pool containing several weak positives. As a strong individual ELISA result suggests recent acute infection (with weaker results more likely to represent historic acute infection), 8 both situations have relevance for BVDV diagnosis. Nonetheless, the wide range of within-pool prevalences corresponding to a particular pooled S/P ratio limits the accuracy of prediction of within-herd prevalence (and, in turn, the likelihood of the herd being infected). This problem was clear when applied to field serum pools in the current study. The mean discrepancy between actual and predicted within-pool prevalence was 22.1%, with a SD of 22.7%. By extension, calculation of a 95% prediction interval generates a potential range of values with a discrepancy of up to 66.6%. Because of this inaccuracy of the prediction of within-pool (and therefore, within-herd) prevalence from a pool S/P ratio, alternative options for interpreting a pool’s S/P ratio were investigated.
It has been acknowledged that herds with seroprevalence of BVDV antibodies >65% are more likely to be actively infected than herds with <65% antibody seroprevalence.3,12 One study revealed that approximately 80% of herds subsequently identified as actively infected had a seroprevalence >65%, while of all herds with >65% seroprevalence 33.4% were genuinely infected. 4 While diagnostic specificity may be limited as recently (but not currently) infected herds are often serologically indistinguishable from actively infected herds, identification of herds with seroprevalence >65% allows selective further investigation. As such, the ability to separate herds with >65% seroprevalence from those with <65% seroprevalence has relevance to BVDV diagnosis and control. On this basis, a within-pool prevalence >65% was used as positivity criteria to perform a TG-ROC analysis. The TG-ROC analysis generated 2 thresholds at which pools with >65% within-pool prevalence can be identified with 95% diagnostic specificity (at the higher threshold), and 95% diagnostic sensitivity (at the lower threshold). These intermediate thresholds of 0.84 and 1.24 were used to split pools into low, medium, and high risk. Herds designated high risk are identified with 95% specificity as having a within-pool (and therefore, within-herd) prevalence >65%. Similarly, herds designated low risk are identified as having <65% within-pool prevalence (95% sensitive for detection of >65% within-pool prevalence at S/P ratio >0.84, corresponds to 95% specific for detection of <65% within-pool prevalence at S/P ratio <0.84). The application of these thresholds to field serum pools revealed a distinct and significant increase in both mean within-pool prevalence and in the proportion of pools with within-pool prevalence >65% from low to medium risk, and again from medium to high risk. Thus, practical application of these thresholds allows for classification of herds according to their risk of >65% seroprevalence and, correspondingly, risk of active infection. Herds classified high risk are most likely to profit from BVDV control measures, while medium-risk herds may benefit from further investigation. Furthermore, identification of herds with low risk of active infection may be indicative of the need for careful protection from BVDV incursion; it is these low seroprevalence herds that are most at risk of severe, acute BVDV outbreaks with potentially devastating financial consequences.
The current study utilized sera from unvaccinated animals. Control and mitigation programs undertaken in areas where vaccination against BVDV is uncommon often utilize serological antibody detection.5,13 However, antibody detection methods are less useful in vaccinated cattle and cattle of unknown vaccination status. In such animals, it may be necessary to apply a p80 or anti–NS2-3 ELISA for the detection of naturally induced antibodies in serum and differentiation from vaccinated animals. 14 The possibility of pooling of serum may also be applicable to these ELISAs but would require validation. Veterinarians and diagnosticians may assess the applicability of the methods reported herein to their individual situations.
Therefore, testing of serum pools has the potential to reduce the cost of serological testing for BVDV antibodies. While collection of serum samples is still more cost and labor intensive than collection of milk samples, pooled serum poses similar laboratory benefits to those recognized for bulk milk testing. This has potential as a valuable screening tool for BVDV infection, with particular application in nonmilking stock, including beef cattle or young stock, where bulk milk antibody testing cannot be used.
Footnotes
Acknowledgements
The authors thank IDEXX Laboratories Inc. for providing the ELISA kits for this study.
a.
IDEXX BVD Total Ab Test, IDEXX Laboratories, Rydalmere, New South Wales, Australia.
b.
IDEXX BVD Serum/Ag Plus, IDEXX Laboratories, Rydalmere, New South Wales, Australia.
c.
Microsoft Excel 2007, Microsoft Inc., Redmond, WA.
d.
CDMT, Freie Universität Berlin, Germany.
e.
CLIVE (Computer-Aided Learning in Veterinary Education), United Kingdom.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
All ELISA kits for this project were supplied by IDEXX Laboratories Inc.
