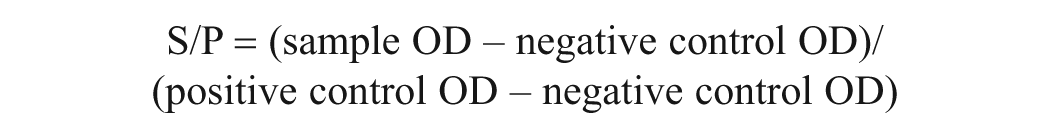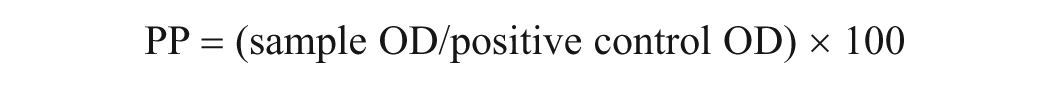Abstract
Bovine viral diarrhea virus is a costly disease of cattle that can be controlled by vaccination, biosecurity, and removal of persistently infected cattle. Development and proficiency testing of assays to identify persistently infected cattle requires substantial quantities of known positive- and negative-sample material. The objective of this study was to determine what sections of bovine skin contained Bovine viral diarrhea virus antigen. Two commercially available antigen-capture enzyme-linked immunoassays were used to test subsamples representing the entire skin of 3 persistently infected calves. Both assays detected Bovine viral diarrhea virus antigen in the samples indicated for use by assay protocol. However, one assay identified all subsamples as positive, while the second assay identified 64.4% of subsamples as positive. These results show that use of samples other than those specified by the assay protocol must be validated for each individual assay. In this study, alternative sample sites and use of the entire hide for proficiency testing would be acceptable for only one of the assays tested.
Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) is a costly disease of cattle in the United States and around the world.2,3 The main reservoirs of BVDV in cattle populations are cattle persistently infected (PI) with BVDV due to early gestational exposure. 5 Persistently infected calves continuously shed large amounts of BVDV. Testing and removing PI calves from herds is useful for controlling spread of BVDV and is economically beneficial. Currently, several diagnostic tests are available for detecting BVDV PI calves, including antigen-capture enzyme-linked immunosorbent assays (ACEs), which detect antigen in skin and serum samples. Ear notch samples are frequently used for BVDV detection and have been used in the licensing of several tests. However, development and validation of ACEs requires substantial quantities of known positive- and negative-sample material, and the amount of ear skin samples that can be harvested from 1 animal is limited. Further, it is desirable to develop panels of known samples to be used in laboratory proficiency testing. While it is commonly held that all haired skin from PI cattle contains BVDV antigen, very little work has been done on skin samples from locations other than ears. The purpose of the current study was to use 2 different ACEs to characterize the availability of BVDV antigen present in the total hide of cattle and to determine if skin from various regions of the body is suitable for validation and proficiency testing.
Three 200- to 300-kg crossbreed beef calves, determined to be persistently infected by ACE,a,8 were used in the present study. Each calf was humanely euthanized by ranch personnel according to ranch protocols. The entire hide was removed from each calf, including the hide on the head and legs down to the coronary bands. At the time of harvest, the hides were divided into nearly equal quarters by folding them in half, front to back, and cutting on the crease and then folding them in half, side to side, and cutting on the crease again. Each quarter was labeled with the calf’s ID and a description of the quarter (FL, FR, RL, and RR, indicating front left, front right, rear left, and rear right, respectively) by placing a labeled ear tag in the quarter. Each quarter was then put into an individual bag and placed on ice for transport to the lab. Quarters were stored at –20°C.
Each quarter was sectioned into squares, 10 cm × 10 cm, starting at the corner of the quarter nearest the middle of the hide. Sectioning was done using a template and common utility knives. Each section was given a unique identifying code to facilitate reconstruction of data maps based on location for analysis. The code consisted of a 2-letter code to identify the quarter (previously mentioned), a number to identify the medial to lateral position, and a letter to identify the cranial/caudal location. Each section was subsampled using a 4-mm leather punch that removed a full-thickness circular sample of skin. The punch was cleaned with alcohol and then rinsed with water between each sample. Each section of hide was placed in a resealable bag and stored at –20°C. Two samples were collected from each section and placed in 1.5-ml microcentrifuge tubes. The tubes were also stored at –20°C prior to ELISA analysis.
Samples from each skin section were analyzed for BVDV antigen using 2 different commercial ACE kits (A a and B b ). One subsample from each section was used for each kit. Each kit was used according to the manufacturer’s protocol. Samples for assay A were soaked in phosphate buffered saline, and an aliquot of the phosphate buffered saline was assayed for the presence of Erns, a structural protein of BVDV. 8 Samples for assay B were soaked in the extraction buffer provided by the manufacturer, and an aliquot of this buffer was assayed for the presence of NS2/3, a nonstructural protein of BVDV. Since multiple plates from each kit were used to analyze all samples, great care was taken to make sure that each plate was treated exactly the same way. Incubation times, wash volumes, and procedure order were identical for each plate. Sample optical density (OD) was measured using a plate spectrophotometer. The sample-to-positive (S/P) ratio was calculated using the following equation.
The OD values of assay B were standardized using percentage positivity (PP) according to protocol instructions, and PP was calculated using the following equation.
The S/P ratio and PP data were used to construct sample signal intensity maps based on hide location.
Data from each kit type were analyzed with a statistical analysis software package. c Frequency tables were constructed to show test results by quarter. Prior to analysis, S/P ratio and PP data were examined for normal distribution. If data were not distributed normally, log transformation was employed to achieve normal distribution. Because of variability seen in assay B results, an examination of the relationship between skin thickness and logPP was conducted.
Assay A returned positive S/P ratios for samples from all sections from each calf. Assay B identified 360 (64.4%) of 618 sections as positive and 220 (35.6%) as negative. Table 1 shows positive and negative results by quarter for assay B.
Number of Bovine viral diarrhea virus sections (positive and negative) for each cattle hide quarter, along with the corresponding percentage for that quarter using the assay B kit.
Numbers in parentheses are percentages. Assay B (PrioCHECK® BVD Ag PI) was manufactured by Prionics AG, Zurich, Switzerland.
The PP data from assay B were right skewed and were log transformed. The results of a linear regression of skin thickness with logPP demonstrated that no significant linear relationship was present.
The data in the current study show that BVDV antigens are present throughout the skin of animals persistently infected with BVDV. In the case of assay A, all skin sections returned a positive result, and overall variation in S/P ratio did not result in erroneous test conclusions for any particular section. Results from assay B demonstrated substantial variability to the point of misclassifying 35.6% of the skin sections. It is important to note that all skin sections that included the ear were positive using both assays. Visual examination of maps of assay B results did not demonstrate a definitive pattern of positive and negative results.
Reasons for the discrepancies between assays remain unknown. One major difference between the 2 assays is their detection target. Assay A targets the Erns protein of BVDV. This protein is associated with the viral envelope and host cell membranes and appears to be readily released into extracellular fluid.6,7 Assay B targets an epitope of the NS2/3 protein, which has been reported as a successful ELISA target for use in identifying animals persistently infected with BVDV. 1 The antibodies found in assay B have also been incorporated into a blocking ELISA as a means of capturing p80 to serve as a target antigen to measure antibodies resulting from BVDV infection. 4 The NS2/3 protein is localized within host cells and is not a structural component of the BVDV virion. 6 Because of its location, NS2/3 requires more intensive extraction methods to make it available for detection. This difference in antigen location and availability may explain some of the differences in response seen between the 2 assays.
The differences in results between the 2 commercial ACE kits examined in the current study reveal the need to validate sample site for any particular BVDV antigen test. In the case of assay A, skin samples from any piece of haired skin were found to be adequate samples for PI status determination as well as proficiency testing. However, skin samples other than the ears returned highly variable results when using the assay B kit. This variability would make using any alternative samples for PI determination or proficiency testing unreliable.
Overall, both tests identified the calves as BVDV positive when using the sample (ear notch) indicated for use by kit instructions. In the present study, assay A was a more robust test for identifying BVDV antigen in skin samples other than ear notches. The use of parts of hides or entire hides for proficiency sampling will still require validation of desired sample sources on an individual assay basis.
Footnotes
Acknowledgements
The authors gratefully acknowledge Gallery Ranch for their cooperation with this project as well as Patricia Federico and Kathryn Fulk for their technical assistance. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
a.
Herdchek Bovine viral diarrhea virus Antigen Detection Kit, IDEXX Laboratories Inc., Westbrook, ME.
b.
PrioCHECK® BVD Ag PI, Prionics AG, Zurich, Switzerland.
c.
SAS Institute Inc., Cary, NC.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Sweiger is a shareholder in Cattle Stats, LLC.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research presented in this article was funded by Cattle Stats, LLC.