Abstract
The current study tested the hypothesis that removal of maltose binding protein (MBP) from recombinant antigen used for plate coating would improve the specificity of a commercial Anaplasma antibody competitive enzyme-linked immunosorbent assay (cELISA). The number of 358 sera with significant MBP antibody binding (≥30%I) in Anaplasma-negative herds was 139 (38.8%) when tested using the recombinant major surface protein 5 (rMSP5)-MBP cELISA without MBP adsorption. All but 8 of the MBP binders were rendered negative (<30%I) using the commercial rMSP5-MBP cELISA with MBP adsorption, resulting in 97.8% specificity. This specificity was higher than some previous reports, so to improve the specificity of the commercial cELISA, a new recombinant antigen designated rMSP5–glutathione S-transferase (GST) was developed, eliminating MBP from the antigen and obviating the need for MBP adsorption. Using the rMSP5-GST cELISA, only 1 of 358 Anaplasma-negative sera, which included the 139 sera with significant (≥30%I) MBP binding in the rMSP5-MBP cELISA without MBP adsorption, was positive. This resulted in an improved diagnostic specificity of 99.7%. The rMSP5-GST cELISA without MBP adsorption had comparable analytical sensitivity to the rMSP5-MBP cELISA with MBP adsorption and had 100% diagnostic sensitivity when tested with 135 positive sera defined by nested polymerase chain reaction. Further, the rMSP5-GST cELISA resolved 103 false-positive reactions from selected sera with possible false-positive reactions obtained using the rMSP5-MBP cELISA with MBP adsorption and improved the resolution of 29 of 31 other sera. In summary, the rMSP5-GST cELISA was a faster and simpler assay with higher specificity, comparable sensitivity, and improved resolution in comparison with the rMSP5-MBP cELISA with MBP adsorption.
Introduction
Anaplasmosis is a tick-borne disease of ruminant livestock in tropical and subtropical regions of the world caused by rickettsia of the genus Anaplasma, including A. marginale, A. centrale, A. ovis, and A. phagocytophilum. 3 Anaplasma marginale is responsible for almost all outbreaks of clinical bovine anaplasmosis. 34 Anaplasma species are transmitted by ticks and possibly by mechanical transfer of infected blood to susceptible cattle by biting flies and fomites.3,34 As Anaplasma invades and multiplies within mature erythrocytes, acute disease is manifested with anemia, weight loss, abortion, and death in infected cattle. 3 In animals that survive acute disease, Anaplasma causes lifelong persistent infection.11,12,26,27 These persistently infected animals are clinically healthy, but serve as reservoirs for continued transmission of the pathogen to other animals.9,11,12,26,27,31 Regulations, particularly in Anaplasma-free countries, bar the import of animals persistently infected with A. marginale into regions where the disease is not regarded as endemic. 3 The economic loss due to anaplasmosis in the United States alone is estimated to be greater than $300 million per year.6,20 Therefore, control of A. marginale infection is enhanced by identification of carrier cattle using a specific and sensitive serodiagnostic assay.
Several diagnostic assays have been developed and used in the field, but diagnostic performance of different assays is highly variable. Such assays have been used to detect carrier cattle and include complement fixation (CF), 13 card agglutination,13,21 indirect enzyme-linked immunosorbent assay (ELISA), 8 dot ELISA, 22 competitive ELISA (cELISA), 19 and indirect fluorescent antibody test (IFAT).13,29 Most of these assays have diagnostic specificities higher than 96%2,3,10,22–25,29; however, the diagnostic sensitivities vary greatly.10,34 Complement fixation test has a very high specificity, but reports of sensitivity range from 20% 4 to 70%. 13 The CF test is no longer considered reliable for disease certification of individual animals due to low sensitivity.4,13,34 Card agglutination also has a high specificity, but the reports of sensitivity vary between 84% 13 and 98%. 21 The indirect ELISA has a specificity of 98.4–99.6% with a sensitivity of 87.3%.8,23 One indirect ELISA based on the use of a normal red blood cell antigen as the negative antigen and an A. marginale–infected red blood cell antigen as the positive antigen has been found to be reliable, 8 but more cumbersome than tests using only 1 antigen. 8 The dot ELISA has a sensitivity of 93% and a specificity of 96%.22,34 The IFAT has a sensitivity of 98% with a specificity of 90% 13 primarily because of nonspecific fluorescence due to non–Anaplasma-specific antibodies adhering to infected erythrocytes.22,34 Polymerase chain reaction (PCR)-based methods can detect A. marginale parasitemia at 0.0001% infected erythrocytes, but at this level only a proportion of carrier cattle would be detected. 34 A comparative study reported that PCR has a low sensitivity of 67% due to variable parasitemia levels in chronically infected animals. 10 In addition, PCR methods need significant effort to prevent false-positive results, which affect the diagnostic specificity of the assay. 32
A recombinant major surface protein 5 (rMSP5)-based cELISA has proven useful for serological detection of Anaplasma-infected animals in previous studies.3,19,34 The antigen in this cELISA is rMSP5 fused with maltose binding protein (MBP). Major surface protein 5 is present in all recognized Anaplasma species, and the MSP5 epitope defined by monoclonal antibody (mAb) ANAF16C1 is broadly conserved among Anaplasma spp. with the possible exception of A. phagocytophilum, as there are conflicting reports for this species.30,33 Furthermore, the Anaplasma cELISA is reported to detect antibodies in Ehrlichia-infected cattle. 1 The diagnostic specificity of the rMSP5-MBP cELISA is 100% when tested with bovine sera collected in nonendemic areas. 19 The same cELISA has 95% specificity and 96% sensitivity when tested with samples evaluated by nested PCR from anaplasmosis-endemic areas. 32 The reported performance of the commercial cELISA from a study in Australia is 99.5% specificity and 98% sensitivity for A. marginale, 21 whereas a study in Switzerland has a 99.2% sensitivity and a 83.3% specificity, but indicated the specificity may have been underestimated. 7 A study comparing the commercial cELISA with a duplex real-time reverse transcription PCR assay has a cELISA specificity of 97.3% and a sensitivity of 65.2% 28 ; the low cELISA sensitivity could be related to the specificity of the PCR assay used.
Even though the rMSP5-MBP cELISA has reasonable diagnostic specificity, the presence of 3–5% or more false-positive results7,21,32 causes problems when interpreting results from individual animals and in eradication programs. During the initial commercialization of the rMSP5-MBP cELISA, a MBP adsorption step was added to improve assay specificity, and all kits made by VMRD Inc. included this adsorption step. However, there are concerns from kit users about possible false-positive tests with some sera using the commercial rMSP5-MBP cELISA with MBP adsorption. In the current study, the hypothesis was tested that removal of MBP from the recombinant antigen used for plate coating would further decrease the number of false-positive results and improve the specificity of the assay. The rMSP5-MBP fusion protein was replaced with a rMSP5-GST fusion protein along with some changes to the antigen coating procedure. Based on the results in the present study, the rMSP5-GST cELISA significantly improved the diagnostic specificity, the resolution of samples near the cutoff, and the simplicity of the assay.
Materials and methods
Sera
Sera from Anaplasma noninfected cattle (n = 358) were collected from dairy herds maintained in barns free of ticks that transmit Anaplasma and that had no history of clinical anaplasmosis. The sera were used for analysis of the diagnostic specificity of the rMSP5-MBP cELISA with MBP adsorption and the newly modified rMSP5-GST cELISA.Anaplasma-positive sera (n = 135) were obtained from cattle with positive results by both serology and nested PCR assays 32 and were used to evaluate the diagnostic sensitivity of the new rMSP5-GST cELISA. The number of samples (n = 493) used to evaluate diagnostic sensitivity and specificity resulted in a statistical power for a likely diagnostic sensitivity or diagnostic specificity of 95% confidence within 5% margin of error. An additional 163 sera were selected from a large number of diagnostic samples submitted to the Washington Animal Disease Diagnostic Laboratory (Pullman, Washington) for Anaplasma testing. At least some of the 163 sera were possible false-positive samples because they were all negative in a real-time PCR assay 5 but were repeatedly positive in the rMSP5-MBP cELISA.
rMSP5-MBP cELISA
The rMSP5-MBP cELISA that included the MBP adsorption step was performed according to the manufacturer’s recommended procedure using a commercial kit. a An ELISA microplate absorbance reader b was used to measure and record optical densities (ODs) at 620 nm. The percent inhibition (%I) of test serum was calculated as: %I = 100 (1 − sample OD ÷ mean negative control). Samples with ≥30%I were considered positive. Some sera were also tested without the MBP adsorption step as a way to screen for MBP antibody binding.
Cloning and expression of rMSP5-GST
The MSP5 gene of A. marginale was cloned into pGEX-2T vector
c
containing GST fusion protein in the expression product of the inserted gene. Recombinant MSP5-GST in the pGEX-2T vector was transformed to BL21 cells
d
and expressed as follows. Briefly, 200 ml of an overnight culture of pGEX-2T/MSP5-transformed BL21 cells was inoculated into 1.8 liters of 2X YT medium
e
containing 0.2% glucose and 0.01% ampicillin and grown at 37°C for 3 hr. Following the addition of 0.048 g of isopropyl-β-
rMSP5-GST cELISA
The rMSP5-GST cELISA was performed as follows using all buffers and solutions from the commercial rMSP5-MBP Anaplasma antibody cELISA kit. a Fifty microliters of rMSP5-GST diluted in antigen diluting buffer was added to 96-well immunoassay plates h treated with 0.04 µg/well of glutathione–bovine serum albumin, incubated overnight at 4°C, and then incubated 2 hr at 37°C with 200 µl of blocking buffer i per well. The blocking buffer was removed, and the plates were dried overnight. The antigen-coated plates were stored individually in polyester film bags j at 4°C until used.
For the cELISA, 50 µl of test sera was added to duplicate wells of the plate and incubated at room temperature for 1 hr. Wells were then washed twice with 250 µl of wash buffer per well. Each well received 50 µl of mAb ANAF16C1 conjugated to horseradish peroxidase diluted in conjugate diluting buffer, and the plates were incubated at room temperature for 20 min. After wells were washed 4 times with 250 µl of wash buffer per well, 50 µl of tetramethylbenzidine substrate was added to each well, and the plates were incubated at room temperature for 20 min. The reactions were then stopped with 50 µl of 1.5% sodium fluoride solution per well. The result was read and calculated as in the same manner described for the rMSP5-MBP cELISA in the preceding section.
DNA extraction and PCR
DNA was extracted from whole blood with a commercial kit, k according to the manufacturer’s instructions. DNA of each sample was eluted in AE buffer (10 mM Tris–HCl and 0.5 mM EDTA, pH 9.0) and used for PCR. Nested PCR or real-time PCR to detect Anaplasma DNA in extracted samples was performed according to methods previously reported.5,32
Western blot analysis for detection of anti-MSP5 antibody in bovine sera
Western blot analysis to detect Anaplasma-specific antibodies in bovine sera was performed using rMSP5-GST according to the previously described method with some modifications. 19 Briefly, rMSP5-GST was boiled for 3 min in sample buffer l and run in sodium dodecyl sulfate–polyacrylamide gel electrophoresis. m Transfer to nitrocellulose was performed by standard techniques. 19 Membranes were blocked in Tris–Tween-20 buffer containing 5% milk. The rMSP5 band was detected with bovine sera followed by treatment with peroxidase-conjugated donkey anti-bovine IgG.n,33
Data analysis
The prevalence of sera with MBP binding antibodies in the Anaplasma-negative sera was estimated by the proportion with ≥30%I in the rMSP5-MBP cELISA performed without the MBP adsorption step. Sera with ≥5%I difference between with and without MBP adsorption in the rMSP5-MBP cELISA were scored as positive for MBP binding. The diagnostic specificity of the rMSP5-MBP with MBP adsorption and rMSP5-GST cELISAs was estimated from the proportion of the 358 Anaplasma-negative sera with a negative cELISA result. Diagnostic sensitivity of both cELISAs was evaluated as the proportion of the 135 Anaplasma-positive sera defined above with a positive cELISA. Receiver operating characteristic (ROC) plot analysis using spreadsheet software o was carried out to evaluate the cutoff for positive and negative detection by the improved new cELISA through comparison with positive and negative reference panels described above in serum panel section.14–16
Results
Analysis of sera from Anaplasma noninfected cattle using 3 cELISA formats
The percentage of sera with MBP-binding antibodies in the 358 sera from Anaplasma noninfected cattle and their impact on the specificity of the rMSP5-MBP cELISA with MBP adsorption were analyzed. One hundred thirty-nine (38.8%) of the 358 sera were positive (≥30%I) in the rMSP5-MBP cELISA without MBP adsorption, and the other 220 sera were negative (<30%I; Fig. 1A). To determine if the positive results were associated with MBP binding antibodies, the 358 sera were evaluated using the rMSP5-MBP cELISA with MBP adsorption. As expected, the 219 sera negative for MBP binding antibodies were also negative. Furthermore, 131 of the 139 sera that were positive using the rMSP5-MBP cELISA without MBP adsorption were negative in the rMSP5-MBP cELISA after MBP absorption, leaving 8 sera that were considered false positives (Fig. 1B; Table 1). Six of the 8 sera had significant MBP antibody binding (≥5%I difference between 2 cELISAs with and without MBP adsorption), whereas 2 did not (Table 1). For the 358 sera from Anaplasma noninfected cattle, the diagnostic specificity of the rMSP5-MBP cELISA with MBP adsorption was 97.8%.
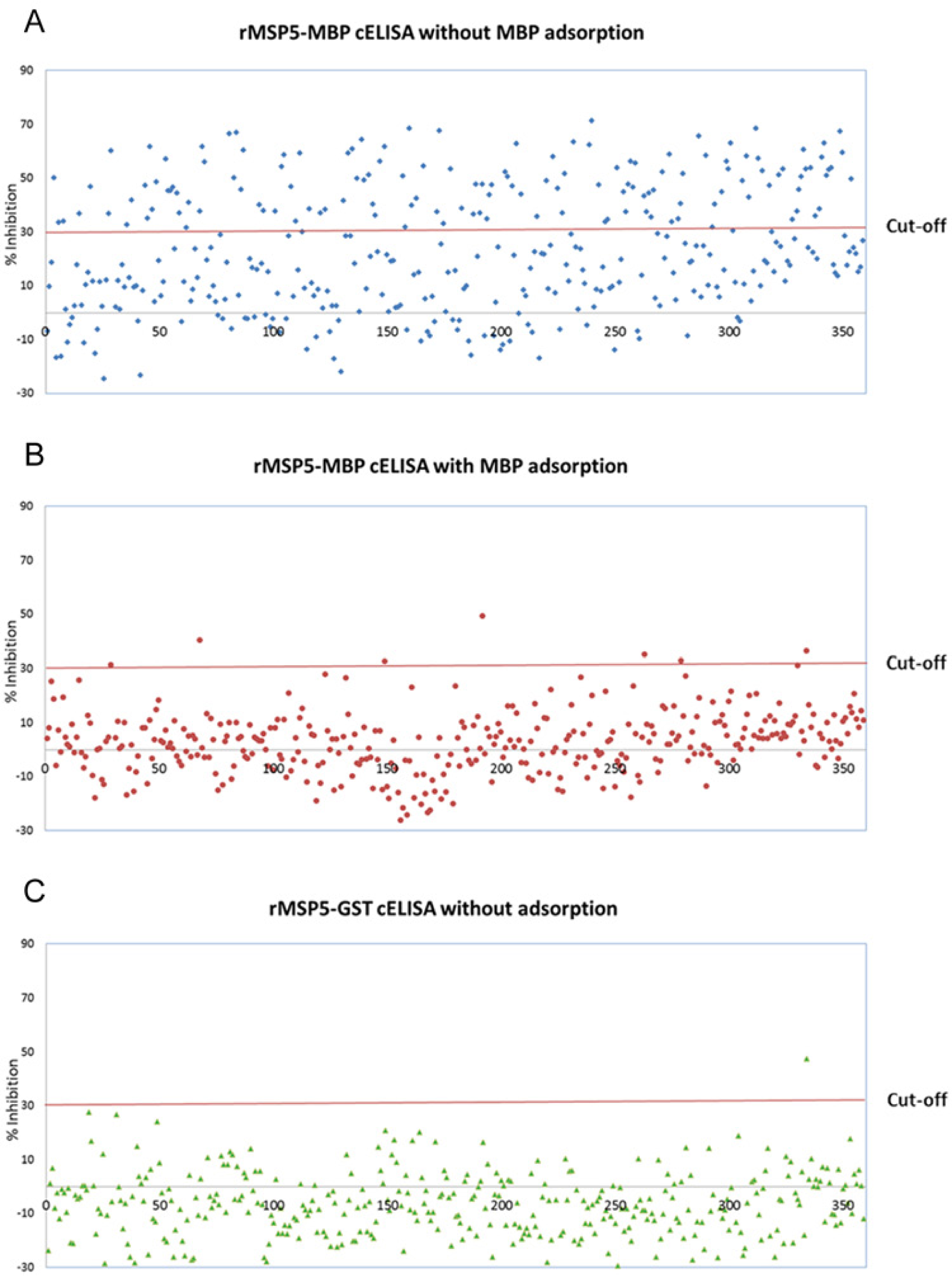
Analysis of sera from Anaplasma-negative herds using 3 different competitive enzyme-linked immunosorbent assay (cELISA) formats. All sera (n = 359) were analyzed using recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) cELISA with (
Sera with discrepant results between recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) with MBP adsorption and rMSP5–glutathione S-transferase (GST) competitive enzyme-linked immunosorbent assays (cELISAs).*
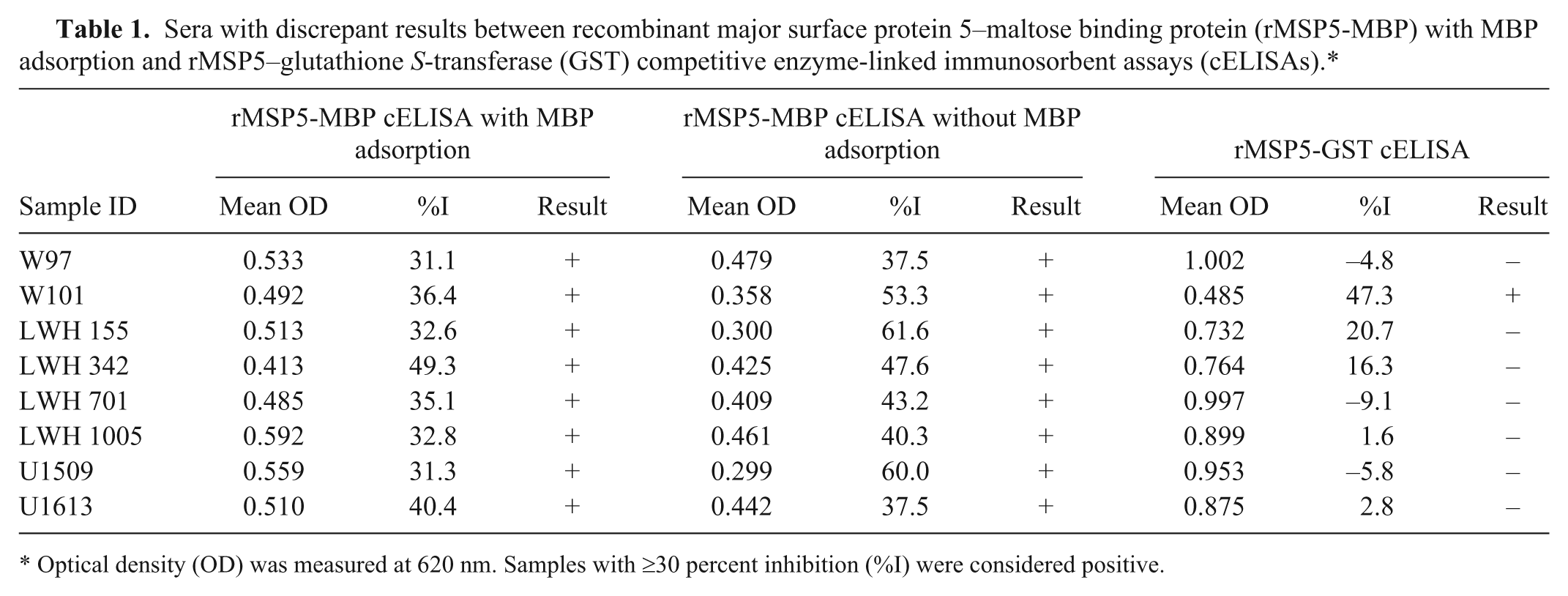
Optical density (OD) was measured at 620 nm. Samples with ≥30 percent inhibition (%I) were considered positive.
When the same set of 358 sera from Anaplasma noninfected cattle was analyzed with the rMSP5-GST cELISA, which does not require MBP adsorption, 357 sera were negative (<30%I) and 1 was positive (Fig. 1C). The positive serum (W101) was 1 of 8 false-positive sera identified in rMSP5-MBP cELISA with MBP adsorption (Table 1). The positive serum had a 47.3%I in the rMSP5-GST cELISA and a 36.4%I in the rMSP5-MBP cELISA after MBP adsorption (Table 1). The 8 sera with false-positive results in the rMSP5-MBP cELISA with MBP adsorption, which included the 1 rMSP5-GST cELISA false-positive serum (W101), were evaluated by Western blotting. None of the 8 sera had significant Anaplasma antibody detected by Western blot (Fig. 2). Moreover, sera with negative results, but with a %I just below the cutoff (25–29.9%I) in the rMSP5-MBP cELISA with MBP adsorption, were also negative in the rMSP5-GST cELISA, but with even lower %I (−19.5%I to 14.4%I; Fig. 1B, 1C; Table 2). For the 358 sera from Anaplasma noninfected cattle, the diagnostic specificity of the new rMSP5-GST cELISA that did not require a MBP adsorption step was 99.7%.

Western blot analysis of sera with discrepant results between recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) with MBP adsorption and rMSP5-glutathione S-transferase (GST) competitive enzyme-linked immunosorbent assays: lane A: molecular weight markers (10, 17, 28, 35, 48, 63, 75, 100, 130, and 180 kDa); lane B: W97; lane C: W101; lane D: LWH155; lane E: LWH342; lane F: LWH701; lane G: LWH1005; lane H: U1509; lane I: U1613; lane J: positive control bovine serum; lane K: negative control bovine serum; lane L: Anaplasma MSP5-specific monoclonal antibody ANAF16C1; lane M: molecular weight markers.
Comparison of assay resolution between recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) and rMSP5–glutathione S-transferase (GST) competitive enzyme-linked immunosorbent assays (cELISAs) using Anaplasma-negative sera with percent inhibition (%I) near the cutoff (25–29.9%I).*
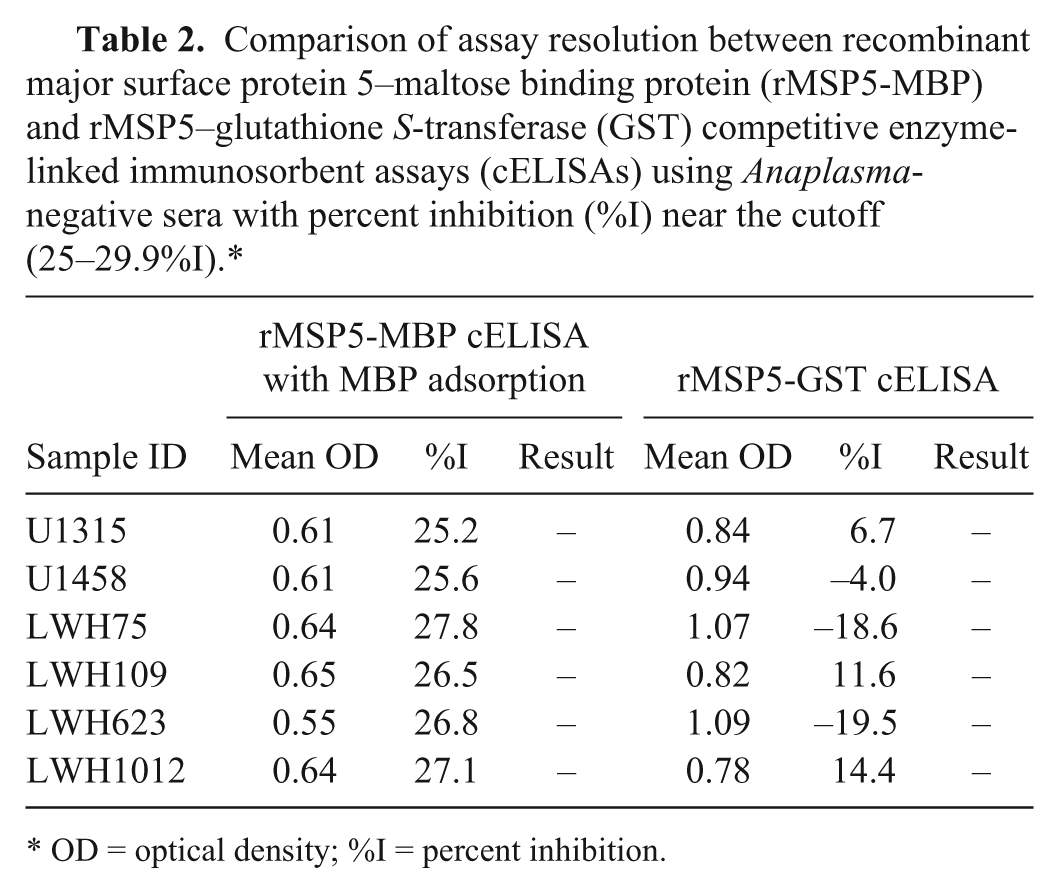
OD = optical density; %I = percent inhibition.
Comparison of 2 cELISAs in analytical and diagnostic sensitivities
The analytical sensitivities of the 2 cELISA formats (rMSP5-MBP cELISA with MBP adsorption and rMSP5-GST cELISA without MBP adsorption) were compared with panels made with 2-fold serial dilutions of 3 Anaplasma-positive sera and with sera collected sequentially from a calf between 0 and 25 days postinfection (dpi). In the comparison using 3 panels made with 2-fold serial dilutions of 3 Anaplasma-positive sera, the 2 assays had the same end points of positive detection, demonstrating the same analytical sensitivity (Table 3). In the comparison of sequential sera, the rMSP5-MBP cELISA with MBP adsorption first detected antibody in sera collected 8 dpi while the rMSP5-GST cELISA first detected antibody in sera collected 9 dpi (Fig. 3).
Comparison of analytical sensitivity between recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) with MBP adsorption and rMSP5–glutathione S-transferase (GST) competitive enzyme-linked immunosorbent assays (cELISAs) using sensitivity panels made with serially diluted Anaplasma seropositive samples.*
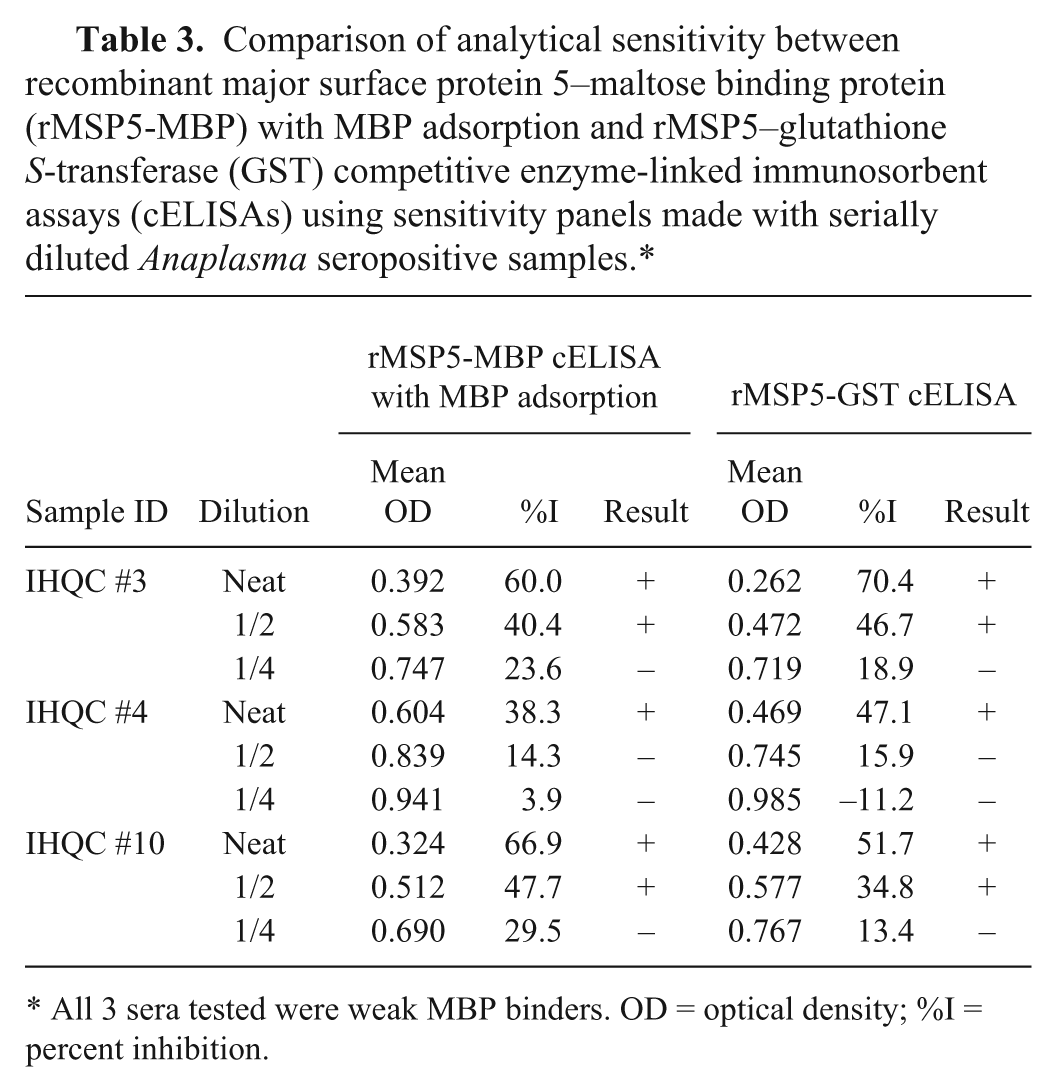
All 3 sera tested were weak MBP binders. OD = optical density; %I = percent inhibition.
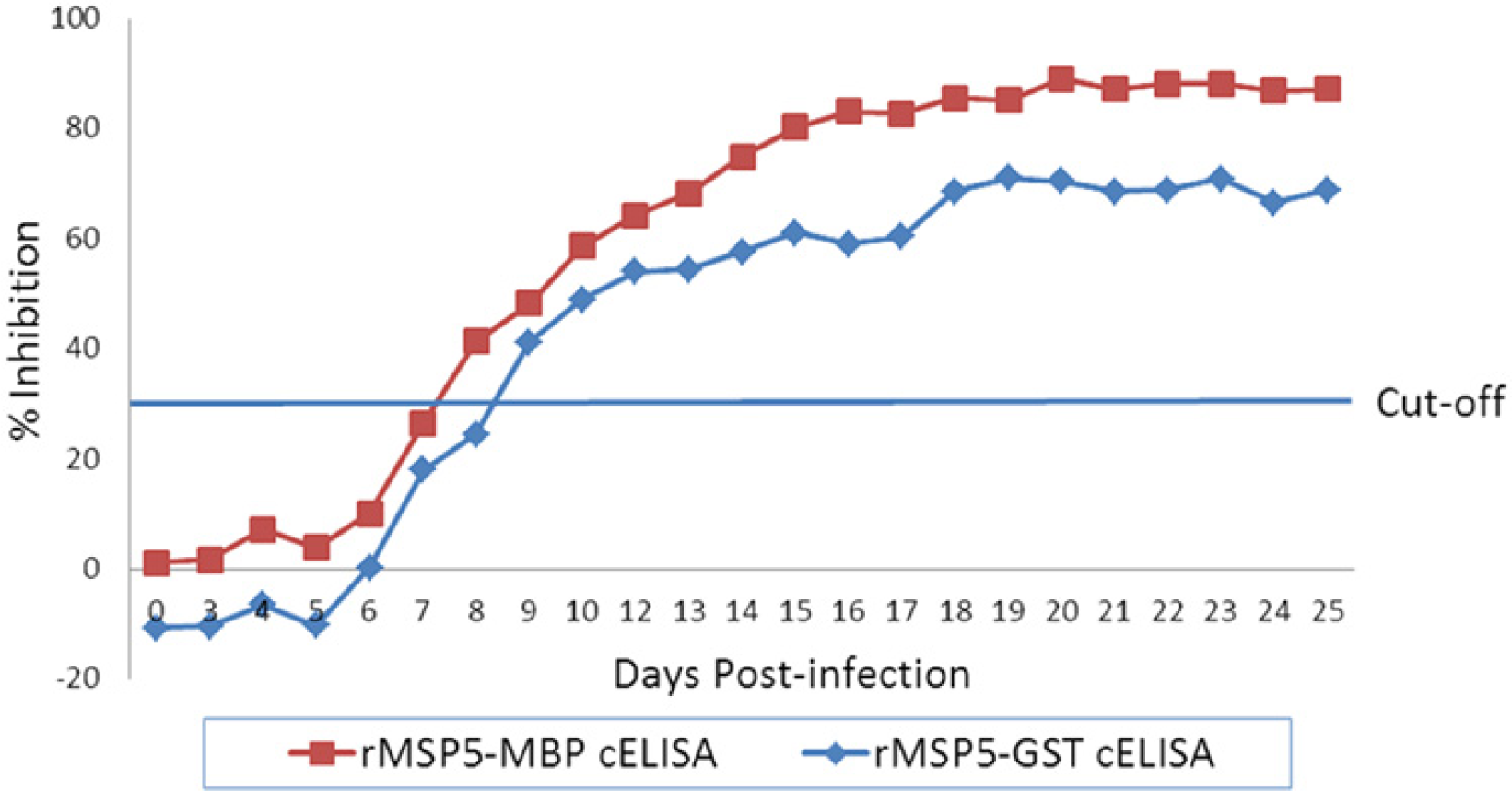
Comparison of analytical sensitivity between recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) with MBP adsorption and rMSP5–glutathione S-transferase (GST) competitive enzyme-linked immunosorbent assays (cELISAs) using sera sequentially collected after experimental infection of a calf with Anaplasma marginale St. Mary strain.
To further evaluate the performance of the rMSP5-GST cELISA, the diagnostic sensitivity was determined with 135 Anaplasma-positive sera that were defined by positive results in nested PCR. All 135 sera were positive using the rMSP5-GST cELISA, resulting in 100% diagnostic sensitivity. The 135 sera were also all positive using the rMSP5-MBP cELISA with MBP adsorption. Furthermore, the resolution of 5 of 6 positive sera near the cutoff (30–40%I) using the rMSP5-MBP cELISA with MBP adsorption were significantly improved with the rMSP5-GST cELISA as the %I were higher by 15.5–44.3%I (Table 4). However, 1 of the 6 sera (serum B-S07-078) had a slightly lower %I using the rMSP5-GST cELISA (Table 4). For the 135 Anaplasma-positive sera, both the rMSP5-GST cELISA and the rMSP5-MBP cELISA with MBP adsorption had 100% diagnostic sensitivity, and the rMSP5-GST cELISA improved the resolution of most weak-positive samples near the cutoff.
Comparison of assay resolution between recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) with MBP adsorption and rMSP5-glutathione S-transferase (GST) competitive enzyme-linked immunosorbent assays (cELISAs) using positive sera with percent inhibition (%I) near the cutoff.*
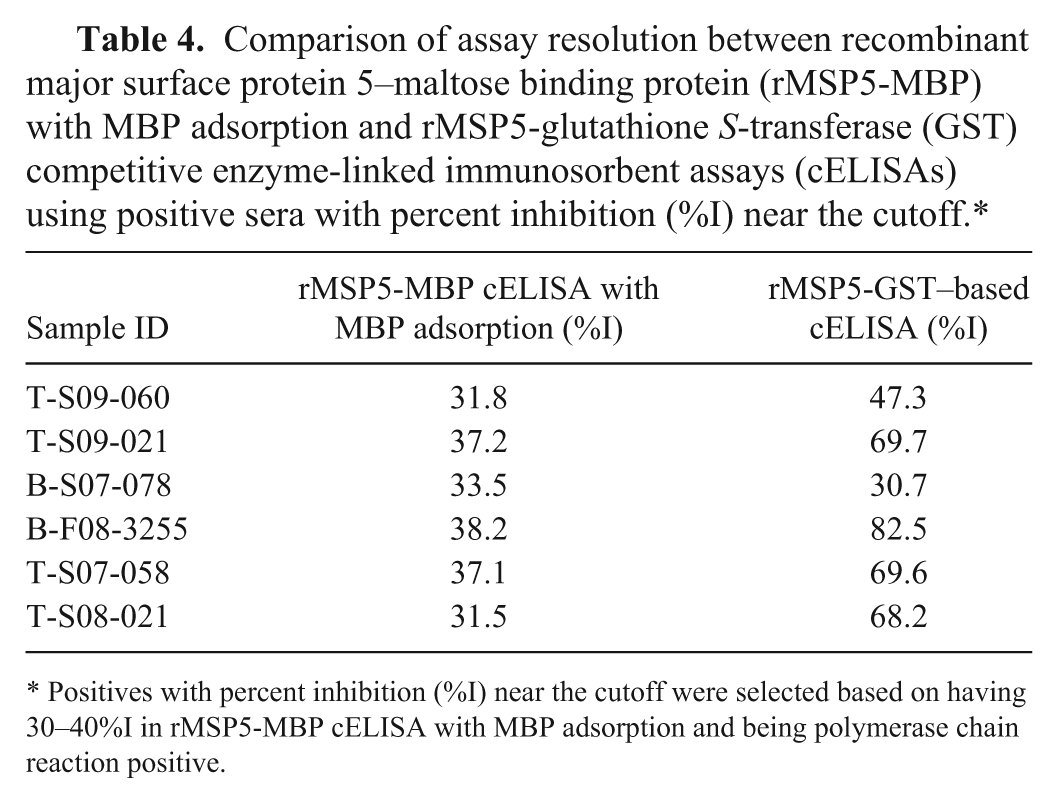
Positives with percent inhibition (%I) near the cutoff were selected based on having 30–40%I in rMSP5-MBP cELISA with MBP adsorption and being polymerase chain reaction positive.
Evaluation of analytical specificity of 2 cELISAs
The analytical specificities of the rMSP5-MBP with MBP adsorption and rMSP5-GST cELISAs were examined with sera positive to Babesia bigemina and Babesia bovis, which are other intra-erythrocyte parasites in cattle. All these sera were negative in both cELISA formats, indicating high analytical specificity (Table 5).
Evaluation of analytical specificity of competitive enzyme-linked immunosorbent assay (cELISA) using sera with antibodies to other intra-erythrocyte bovine parasites, Babesia bovis and Babesia bigemina.*
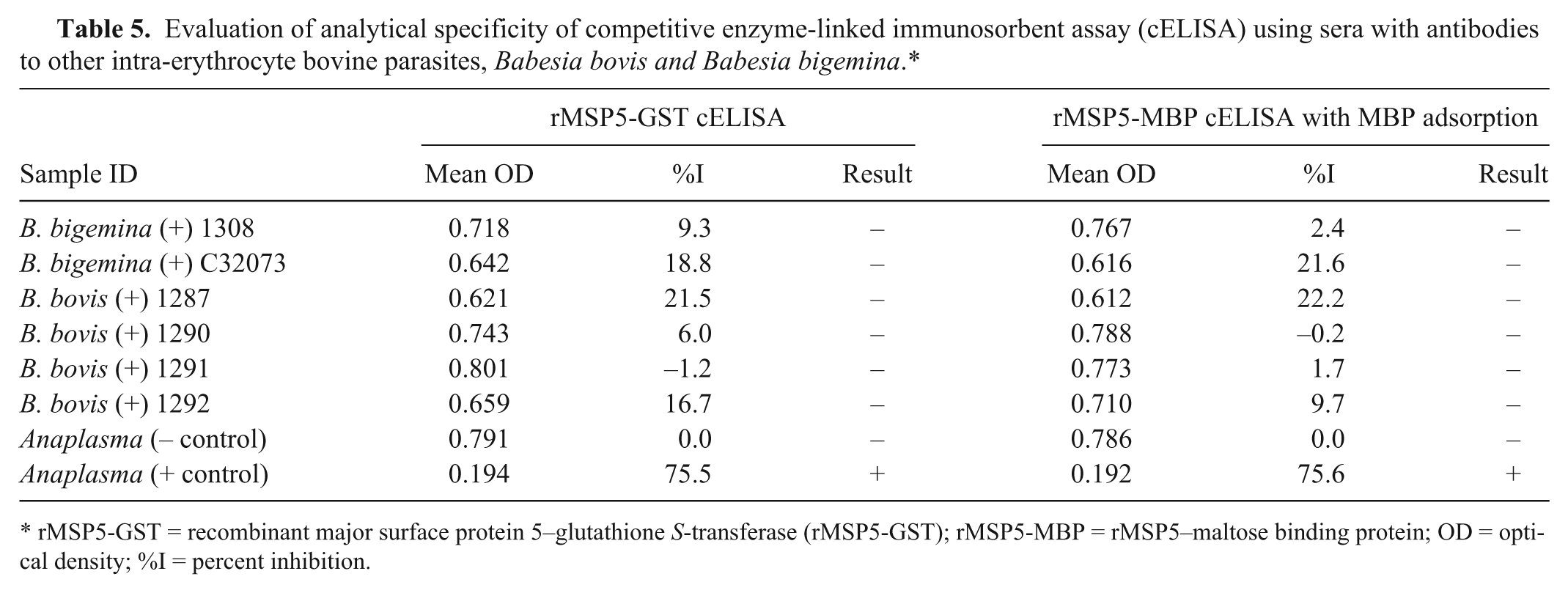
rMSP5-GST = recombinant major surface protein 5–glutathione S-transferase (rMSP5-GST); rMSP5-MBP = rMSP5–maltose binding protein; OD = optical density; %I = percent inhibition.
Evaluation of a serum panel containing possible false positives using rMSP5-GST cELISA
After validation using Anaplasma-negative and -positive serum panels, the rMSP5-GST cELISA was used to evaluate 163 sera containing some possible false positives. The sera were selected based on positive results using the rMSP5-MBP cELISA with MBP adsorption and negative results using real-time PCR. The 163 sera were retested using the rMSP5-MBP cELISA with MBP adsorption, and tested using the rMSP5-MBP cELISA without MBP adsorption and the rMSP5-GST cELISA. Based on the results from the rMSP5-GST cELISA, 29 of 163 sera were positive, and the remaining 134 were negative for Anaplasma antibody (Table 6). The presence of Anaplasma antibody detected by the rMSP5-GST cELISA in the 29 sera was confirmed by IFAT as positive. Furthermore, the 29 sera were positive in both the initial and retest using the rMSP5-MBP cELISA with MBP adsorption (Table 6). The 134 sera that were negative using the rMSP5-GST cELISA, but had false-positive results in the initial rMSP5-MBP cELISA with MBP adsorption, were classified into 3 groups. Group 1 contained 76 of the 134 false positives, and these sera had high levels of MBP antibody suggesting that incomplete MBP adsorption accounted for the false-positive reactions in both the original and retest using the rMSP5-MBP cELISA with MBP adsorption (Tables 6, 7). Group 2 contained 27 of the 134 false positives that did not have MBP antibody, and there was no obvious explanation for these 27 false-positive reactions in both the original and retest using the rMSP5-MBP cELISA with MBP adsorption (Tables 6, 7). Group 3 contained 31 of the 134 false positives. The 31 sera were positive in the original, but negative in the retest using the rMSP5-MBP cELISA with MBP adsorption (Table 6). However, the 31 sera had a %I near the cutoff (25–29.9%I) in the retest with the rMSP5-MBP cELISA with MBP adsorption, suggesting repeatability problems near the cutoff with this assay (Table 7). The rMSP5-GST cELISA improved the resolution of 29 of the 31 sera in group 3, as all 31 were negative and only 2 of the sera had a %I near the cutoff (25–29.9%I) and 29 had <25%I. Thus, the rMSP5-GST cELISA resolved 103 false-positive reactions obtained using the rMSP5-MBP cELISA with MBP adsorption in groups 1 and 2 and improved the resolution of most of the samples in group 3.
One hundred sixty-three suspected false positives were selected based on positive recombinant major surface protein 5–maltose binding protein (rMSP5-MBP) competitive enzyme-linked immunosorbent assay (cELISA) with MBP adsorption in initial assay and negative real-time polymerase chain reaction results. GST = glutathione S-transferase.
False positive in rMSP5-MBP cELISAs with MBP adsorption likely from residual MBP antibody binding.
Cause of false positive in rMSP5-MBP cELISAs with MBP adsorption unknown.
Percent inhibition (%I) near cutoff (25–29.9%I) possibly resulting in false positives in the initial assay with rMSP5-MBP cELISA with MBP adsorption.
All assays positive.
Representative data from evaluation of a serum panel containing possible false positives using 3 competitive enzyme-linked immunosorbent assay (cELISA) formats.*
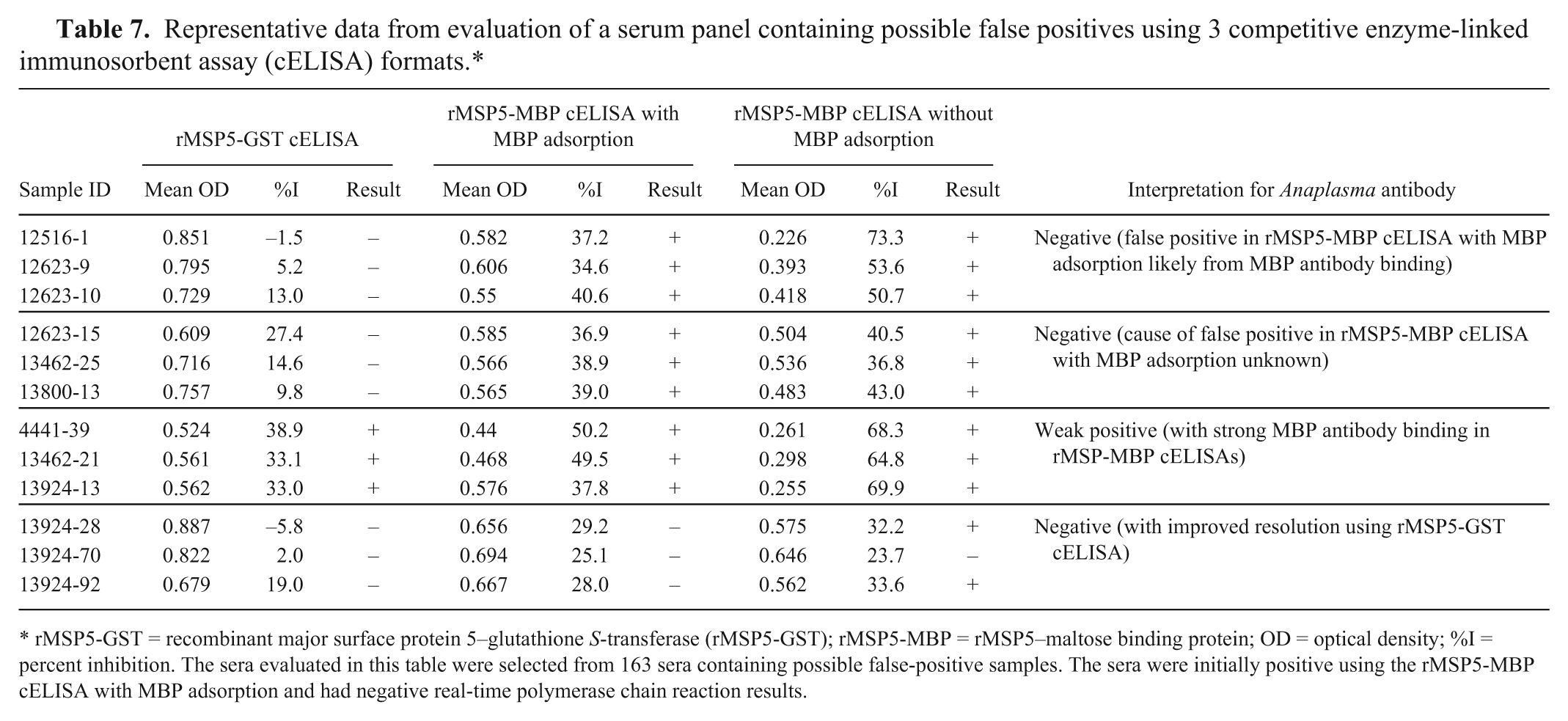
rMSP5-GST = recombinant major surface protein 5–glutathione S-transferase (rMSP5-GST); rMSP5-MBP = rMSP5–maltose binding protein; OD = optical density; %I = percent inhibition. The sera evaluated in this table were selected from 163 sera containing possible false-positive samples. The sera were initially positive using the rMSP5-MBP cELISA with MBP adsorption and had negative real-time polymerase chain reaction results.
Evaluation of rMSP5-GST cELISA cutoff resulting in the best combination of diagnostic sensitivity and specificity
To confirm that the ≥30%I cutoff for defining seropositive samples and <30%I for defining seronegative samples determined in previous studies using a commercial rMSP5-MBP cELISA was still optimal for the improved rMSP5-GST cELISA, data from rMSP5-GST cELISA reference positive and negative samples was analyzed by ROC. In this analysis, the best correlation between rMSP5-GST cELISA and reference panel data was observed using a cutoff of ≥30%I for positive samples and <30%I for negative samples (Fig. 4).
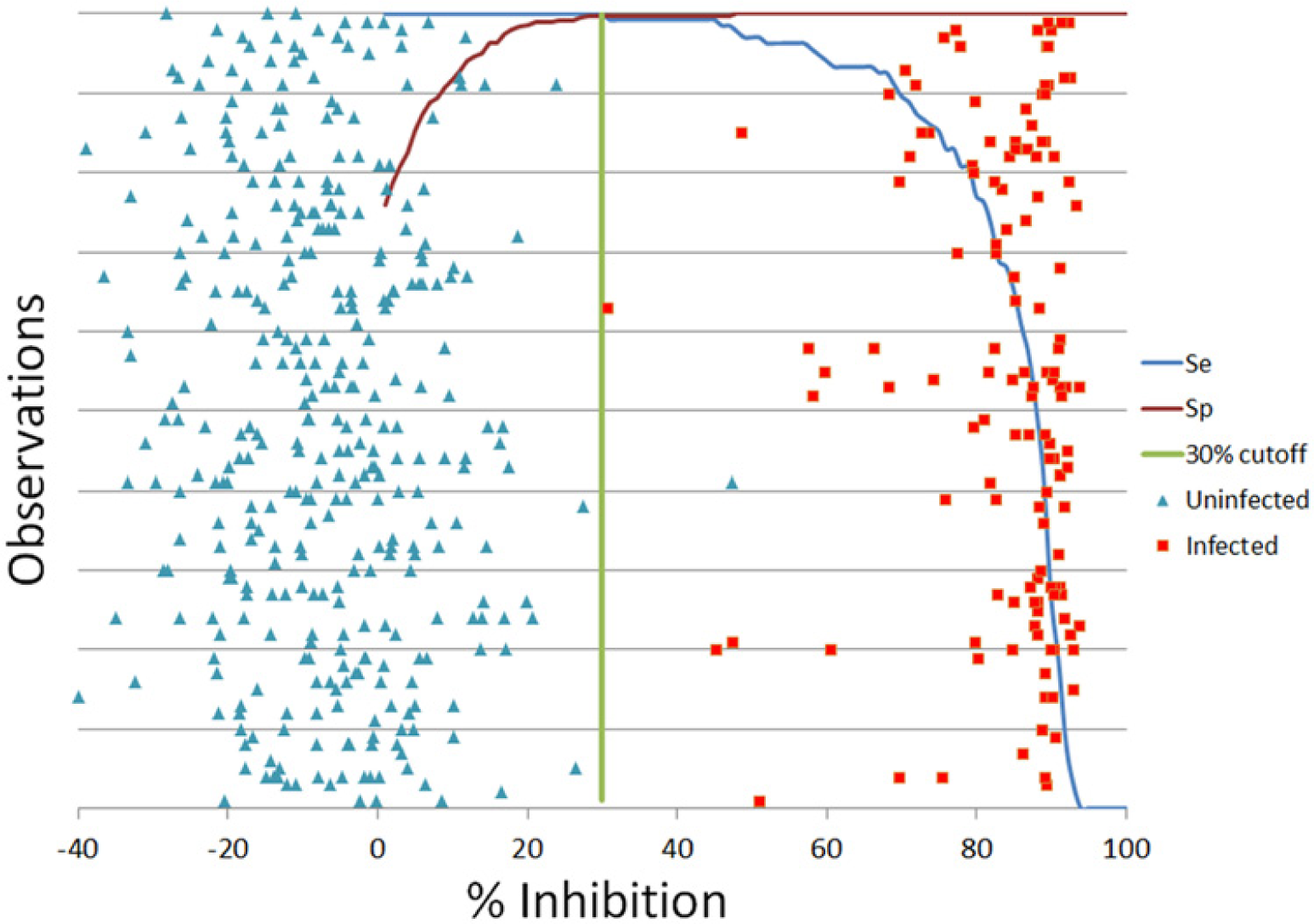
Receiver operating characteristic plot analysis of improved competitive enzyme-linked immunosorbent assay data. Green line is at 30 percent inhibition, which resulted in the best combination of diagnostic sensitivity (Se) and specificity (Sp).
Discussion
The current commercially available rMSP5-MBP cELISA with MBP adsorption has a better diagnostic performance and simplicity of use than other available assays. However, the World Organization for Animal Health Terrestrial Manual states “Caution is advised on its use for disease or disease-free certification,” due to minor deficiencies in specificity.” 34 Further improving the diagnostic sensitivity and specificity of this assay would be an aid to producers, diagnosticians, regulatory organizations, and governments endeavoring to control and/or prevent the import of bovine anaplasmosis.
In the current study, the rMSP5-MBP cELISA with MBP adsorption for Anaplasma antibody detection was significantly improved by changing the coating antigen to rMSP5-GST, resulting in a more specific, simpler, and faster assay. Replacement of the MBP fusion protein with GST in the coating antigen construct particularly improved diagnostic specificity by eliminating almost all false positives due to MBP antibody binding, achieving a diagnostic specificity of 99.7% with a panel of 358 sera from Anaplasma noninfected cattle. This was done while retaining diagnostic sensitivity of 100% with a panel of 135 Anaplasma-positive sera, comparable to the rMSP5-MBP cELISA with MBP adsorption. An additional benefit of the rMSP5-GST cELISA included significant improvement in resolution with sera having %I near the cutoff, giving clearer differentiation between positive or negative results with fewer samples with a %I in the gray zone (25–35%I). In addition to improved diagnostic performance, the rMSP5-GST cELISA procedure was simpler and quicker to run because it eliminated the need for the MBP-reactive antibody adsorption step.
The percentage of Anaplasma-negative bovine sera with significant MBP antibody binding (≥30%I) was surprisingly high at 38.7% when tested in the rMSP5-MBP cELISA without MBP adsorption. Maltose binding protein is reportedly part of the important maltose/maltodextrin system of Escherichia coli, which is responsible for the uptake and efficient catabolism of maltodextrins. 18 In some cases, fusion to MBP can promote the proper folding of the attached protein into its biologically active conformation. Maltose binding protein seems to be capable of functioning as a general molecular chaperone in the context of a fusion protein. 17 However, anti-MBP responses in cattle may be common due to the ubiquity of E. coli in the environment and its frequency as the etiological agent of enteric infections in bovine species. Although most of the sera with MBP antibody were resolved by the MBP adsorption step in the current commercial rMSP5-MBP cELISA, 6 of the sera with MBP antibody were still false positives possibly because of incomplete MBP adsorption, and 2 other sera without MBP antibody were false positives, causing up to 1.8% of false-positive results (98.3% specificity) in the 358 sera from Anaplasma noninfected cattle evaluated. The 98.3 % specificity in the present study is higher than the 95% previously reported, but the previous study used sera from an area with endemic A. marginale infections. 32 The misinterpretation caused by false positives in the rMSP5-MBP cELISA with MBP adsorption could be a critical issue in tests for disease-free certification, which is commonly needed for export of economically important breeding cattle.
Inclusion of GST as a fusion protein in the cELISA antigen did not hamper the manufacture of kits or the performance of the assay. The GST fusion protein is reported to be less efficient as a fusion partner of recombinant protein expression when compared to MBP due to lower efficiency as a molecular chaperone. 17 Maltose binding protein is also known to be a far more effective solubilizing agent than GST as a fusion partner. In spite of the less favorable features as a fusion protein, the rMSP5-GST cELISA had extraordinarily good diagnostic performance and better resolution of samples with %I near the cutoff.
On top of major improvements in diagnostic specificity and resolution of samples with a %I near cutoff, the rMSP5-GST cELISA has additional merits. First, in spite of a slightly downward movement of the negative sera %I distribution due to the elimination of MBP-reacting antibodies (Figs. 1C, 3), analytical sensitivity was not negatively affected, showing a comparable level to the rMSP5-MBP cELISA with MBP adsorption. Interestingly, the first detection of Anaplasma antibody response using both cELISA formats was 1–2 days earlier than detection of parasitemia by Giemsa staining (data not shown), suggesting reliable analytical sensitivity. Furthermore, the rMSP5-GST cELISA resolved false positives due to both MBP binding and unknown mechanisms as demonstrated by comparison of the 2 cELISA formats with selected field sera containing possible false-positive samples (Table 6).
In summary, the rMSP5-GST cELISA resolved 3 types of problems observed in the rMSP5-MBP cELISA, including MBP binders, nonspecific binders of unknown mechanism, and sera with %I near the cutoff (25–35%I). The improved cELISA maintained reliable analytical sensitivity and analytical specificity in addition to producing 100% diagnostic sensitivity and 99.7% diagnostic specificity using the cutoff of 30%I determined by ROC analysis. The diagnostic sensitivity and specificity results need to be further evaluated in other laboratories with additional Anaplasma-positive and -negative sera. In conclusion, based on the high diagnostic performance demonstrated in the current study, the rMSP5-GST cELISA appears to be a simpler and more reliable serodiagnostic tool for bovine anaplasmosis with various applications including epidemiological monitoring and disease/disease-free certification. The assay will be commercially available when field trials in other laboratories and additional regulatory licensure requirements are successfully completed.
Footnotes
Acknowledgements
The authors appreciate Ethan Adams and Amanda Grimm (VMRD Inc.), and Ralph Horn and James Allison (ADRU, USDA) for their technical assistance or review of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the general research funding of VMRD (Veterinary Medical Research and Development) Inc., Pullman, WA.
a.
Anaplasma antibody cELISA, VMRD Inc., Pullman, WA.
b.
ELISA microplate absorbance reader, Labsystems Multiscan MS, Thermo Scientific, Waltham, MA.
c.
pGEX-2T vector, GE Healthcare Technologies, Piscataway, NJ.
d.
BL21 competent cells, GE Healthcare Technologies, Piscataway, NJ.
e.
YT medium, Difco Laboratories Inc., Detroit, MI.
f.
Isopropyl-β-
g.
Triton X-100, Sigma-Aldrich, St. Louis, MO.
h.
Stripwell 96-well immunoassay plates, Corning Inc., Acton, MA.
i.
Blocking buffer, VMRD Inc., Pullman, WA.
j.
Mylar bags, IMPAK Co., Los Angeles, CA.
k.
QIAamp DNA blood mini kit, Qiagen Inc., Valencia, CA.
l.
Sample buffer, Bio-Rad Laboratories, Hercules, CA.
m.
Sodium dodecyl sulfate-polyacrylamide gel, Bio-Rad Laboratories, Hercules, CA.
n.
Peroxidase-conjugated donkey anti-bovine IgG, Jackson Immunoresearch Laboratories, West Grove, PA.
o.
Excel 2010, Microsoft Corp., Redmond, WA.

