Abstract
Routine serologic testing for Avian metapneumovirus (AMPV) infection of turkey flocks at slaughter is currently being used to monitor changes in the occurrence of AMPV infection in endemic areas and can also be used to detect the emergence of infection in currently unaffected areas. Because of the costs associated with false-positive results, particularly in areas that are free of AMPV infection, there is a need to obtain improved estimates of flock-level specificity (SP). The objective of this study was to estimate flock-level SP of a program to monitor AMPV infection in turkey flocks at processing using a standard enzyme-linked immunosorbent assay (ELISA). A study was carried out in which 37 AMPV-free flocks from 7 Midwest operations were followed serologically. Six percent, 3%, and 0.2% of total samples tested AMPV positive at 8 weeks, 12 weeks, and at processing, respectively. Overall, flock-level SP increased as the cutoff increased and as age increased. Flock-level SP at processing was 97%, if a cutoff of 1 was used (the flock was classified as positive if at least 1 sample tested positive), and 100%, if any other cutoff was used. Administration of antibiotics (P = 0.02) and vaccination for Bordetella avium (P = 0.08) were positively associated with the probability of (false) positive test results. These findings suggest possible cross-reactions with other infections and highlight the need to consider variable diagnostic performance depending on farm conditions.
Avian metapneumovirus (AMPV; order Mononegavirales; family Paramyxoviridae; subfamily Pneumovirinae; genus Metapneumovirus) 11 is an RNA virus that was first identified in the United States in 1996 from a commercial turkey flock in Colorado following an outbreak of upper respiratory disease. 14 In 1997, the virus was detected in Minnesota, where it became endemic in subsequent years 8 with a flock seroprevalence ranging from 14% to 65%. 9 The virus was later detected in other states, including North Dakota, South Dakota, and Wisconsin. 2,13 The incidence of AMPV outbreaks in domestic turkeys in Minnesota has shown a seasonal trend, with most outbreaks occurring in spring and fall. It has been speculated that this pattern could be explained by migrations of wild birds, which may serve as reservoirs for AMPV. 2 Impact of AMPV infection on flock health includes decreased egg production, morbidity up to 100% in flocks showing clinical disease, and mortality that can be a low as 0.4% or as high as 50%. 7
Routine serologic testing for AMPV infection of turkey flocks at slaughter is currently being used to monitor changes in the occurrence of AMPV infection in endemic areas and can also be used to detect the emergence of infection in currently unaffected areas. The goal of a monitoring program at slaughter is to identify infected flocks through detection of AMPV antibodies. However, serologic assays are not perfect, and false-positive and false-negative results are expected. Consequently, knowledge of the accuracy of the diagnostic test is needed to interpret the observed results appropriately. Avian meta-pneumovirus serologic testing is commonly done using an enzyme-linked immunosorbent assay (ELISA), which has been evaluated to some extent. Accuracy evaluations have been performed using samples from individual birds under experimental infection settings. Reported estimates of diagnostic sensitivity (SE) for ELISA include 98% 12 and 99%, 10 and estimates of 98% 12 and 99.5% 10 for diagnostic specificity (SP). Because the interest of a monitoring program is to identify infected flocks as accurately as possible, and not necessarily individual infected birds, the parameters of diagnostic performance needed are flock-level SE and SP. Flock-level SE is defined as the probability that an infected flock is classified as infected, and flock-level SP is defined as the probability that an uninfected flocks is classified as uninfected. Because of the costs associated with false-positive results, particularly in areas that are free of AMPV infection, there is a need to obtain improved estimates of flock-level SP. The objective of the current study was to estimate flock-level SP of a program to monitor AMPV infection in turkeys at processing using a standard ELISA and to describe possible factors associated with false-positive results.
The objective was accomplished through a prospective study of AMPV-free flocks, which were tested for AMPV antibodies at various times. Turkey producers from a geographic area of Iowa known to be free of AMPV, based on historical evidence of no infection, were invited to participate in the study. A total of 7 farms were enrolled in the study based on their willingness to collaborate by enrolling flocks in the prospective study. Farms can be considered representative of independent, family-owned turkey production facilities in the Midwest. The target sample size for number of flocks was based on an expected value for flock-level SP of 80%, 95% confidence level, margin of error of 20%, and a 50% increase beyond the recommended sample size to allow further statistical analysis. A questionnaire was administered to the farm owner to request information associated with each flock enrolled in the prospective study, including hatch date, placement date, flock size, mortality, vaccinations, treatments, clinical disease, sampling dates, and processing date.
Blood samples were obtained by venipuncture in collaboration with the farm owner at 8 weeks and 12 weeks of age and immediately before processing for slaughter. At each sampling, 20 birds were randomly selected from each flock and bled to obtain 2 ml of blood, which was sent to the Iowa Veterinary Diagnostic Laboratory (Ames, IA) for processing. Sera were stored at −20°C until testing at the Minnesota Poultry Testing Laboratory (Willmar, MN). The current AMPV surveillance protocol in Minnesota involves sampling10 birds per flock; however, based on the known negative correlation between sample size and population-level SP, 5 the number of birds per flock was doubled to obtain a more conservative estimate of flock-level SP.
Samples were tested for AMPV using the same ELISA procedure that is used as part of the routine AMPV monitoring program in Minnesota. 4 Briefly, serum samples were diluted 1:40 in dilution:blocking buffer and added to 2 virus antigen-coated wells (antigen prepared in Vero [African green monkey kidney] cells) and 2 control antigen-coated wells (equally processed uninfected Vero cells) at 50 μl/well. Known positive and negative control sera were included in each plate. The plates were covered, incubated at room temperature for 1 hr, washed 5 times, and blotted dry. The conjugate was diluted in diluting-blocking buffer and added to each well at 50 μl/well. After incubation at room temperature for 1 hr, the conjugate was removed, and the plates were washed 5 times and blotted dry. Substrate solution was added to each well at 100 μl/well. After incubation in the dark for 10 min, the reaction was stopped by adding 25 ml of 2.5 mol sulfuric acid to each well, and the optical density (OD) was read at 490/405 nm. The results were expressed as the average OD difference (ODD) between virus antigen-coated and control wells of each serum. A sample with ODD of ≥0.2 was considered positive. Additionally, samples were tested for Newcastle disease virus (NDV), Avian influenza virus (AIV), and Bordetella avium, as a predetermined respiratory disease panel using commercially available ELISA diagnostic test kits. a
Descriptive statistics (means, proportions, and standard deviations) were used to characterize the management and sanitary conditions of the farms and flocks enrolled in this study. Traditionally, monitoring programs assume that a flock is infected if at least 1 sample tested positive; however, sometimes this flock-level cutoff is arbitrarily increased because of concern with false-positive results. For a given sampling, flocks in the current study were classified based on AMPV infection status using flock-level cutoffs from 1 through 4 positive samples. Flock-level, diagnostic SP for a monitoring program based on testing 20 birds per flock was defined as the proportion of these AMPV-free flocks that were correctly classified as AMPV negative. To account for the correlation of flocks within farms, population-averaged estimates of flock-level SP and 95% confidence intervals (CI) were calculated using a generalized estimating equation model for binomial data and a exchangeable correlation structure. 1 The same model was used to assess the potential effect of flock factors, including flock size, mortality, clinical disease, vaccination, treatments, and serostatus for other infections on the probability of (false) positive AMPV results. The assumption of linearity in the ln(odds) for continuous variables (mortality proportion, proportion positive for NDV, AIV, and B. avium) was assessed by categorizing each variable and visually inspecting a plot of the estimates of ln(odds) against the midpoint of the categories. 6 Because of limited sample size for the statistical analysis, a P-value of, 0.1 was considered statistically significant. All statistical analysis was performed using available computer packages. b,c
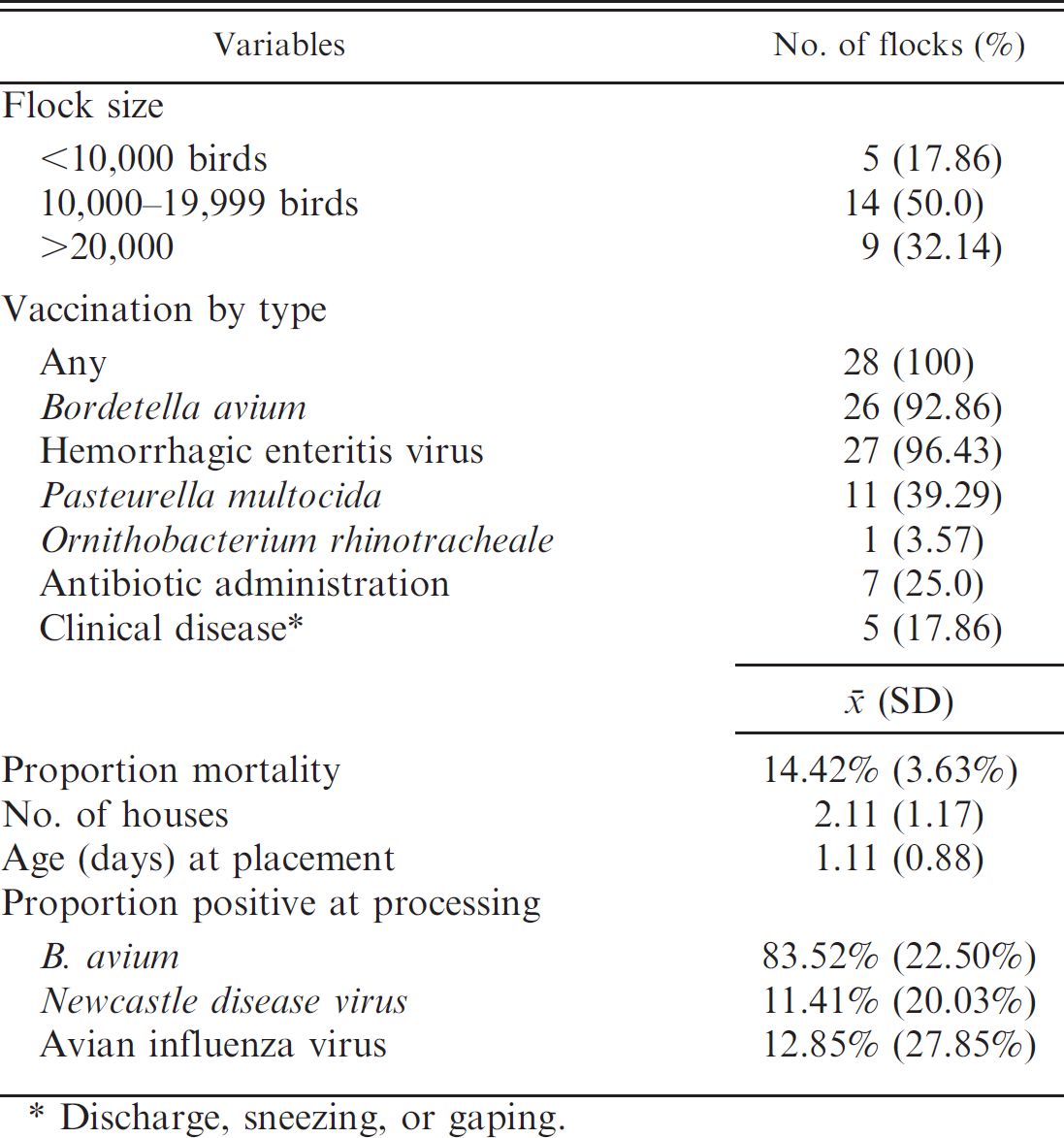
A total of 37 flocks from 7 different farms were followed prospectively from 8 weeks of age until processing at approximately 21 weeks of age. Between 1 and 13 flocks per farm were enrolled. Avian metapneumovirus test results were available for 732 birds at 8 weeks of age, 721 birds at 12 weeks of age, and 465 birds at processing. Questionnaire information was available for 28 flocks, which was used to describe the characteristics of the farms and flocks (Table 1). The majority of the flocks had ≥10,000 birds and were administered routine vaccinations for B. avium and Hemorrhagic enteritis virus. Observation of the distributions of the bird-level crude ODD values showed a shift to the left as age increased (Fig. 1) and, therefore, the number of samples that yielded a positive AMPV ELISA (ODD ≥0.2) decreased with time. A total of 42 of 732 (6%) samples tested AMPV positive at the first sampling, 20 of 721 (3%) at the second sampling, and 1 of 465 (0.2%) at processing.
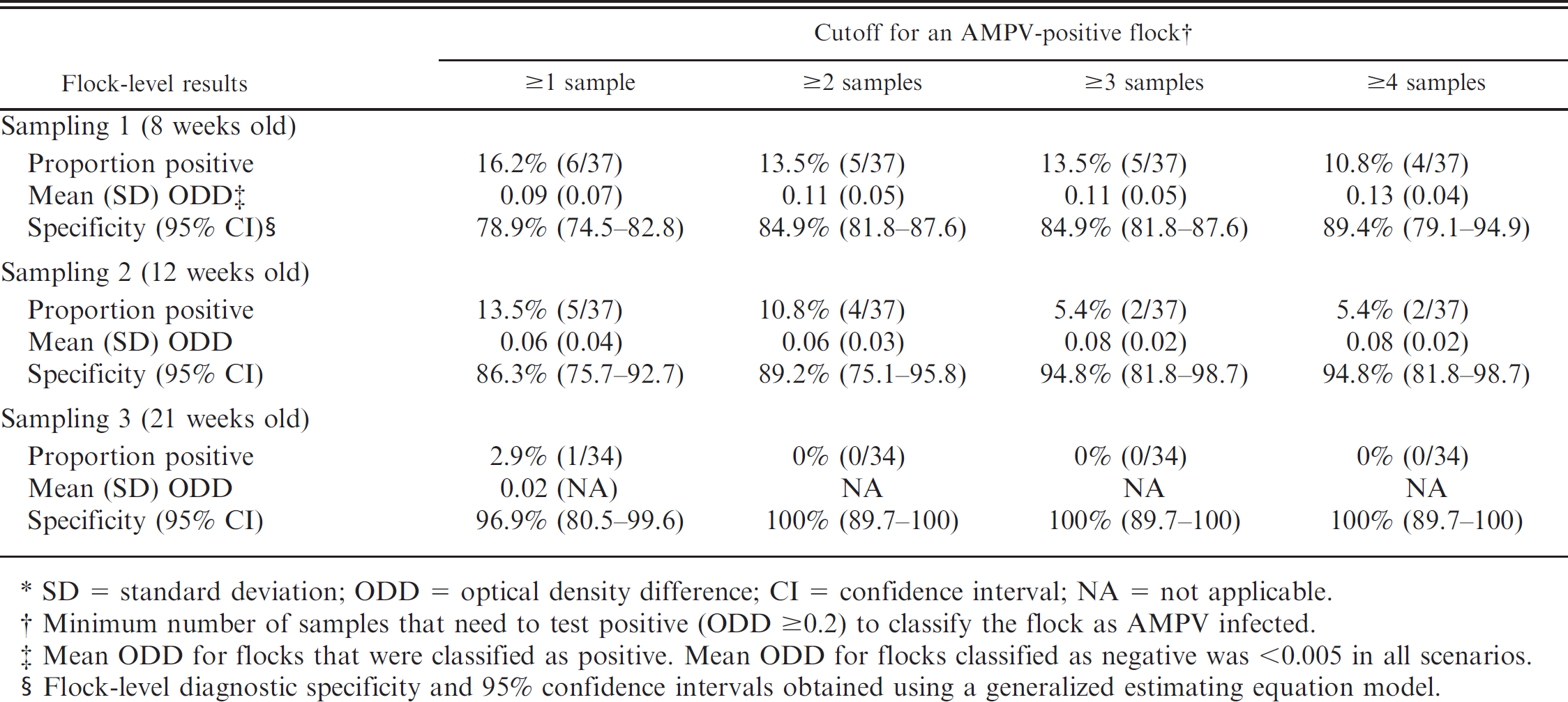
Flock-level AMPV results are shown in Table 2. As expected, increasing the cutoff for classifying a flock as AMPV-infected resulted in a decrease in the number of flocks classified as positive. Using a cutoff of 1 (the flock was classified as positive if at least 1 sample tested positive), resulted in 6, 5, and 1 flock(s) classified as positive at first sampling, second sampling, and at processing, respectively. This decrease in the number of flocks classified as positive as age increased was observed for each classification cutoff. Among flocks that were classified as positive using a cutoff of 1, the proportion of positive samples ranged from 5% to 55% at the first sampling and from 5% to 25% at the second sampling. The single flock that was classified as positive at processing had 6.7% of positive samples. Overall, flock-level SP increased as the cutoff increased and as age increased. For example, flock-level SP with a cutoff of 1 was 79% and increased to 89% when a cutoff of 4 positive samples was used. Because of the decrease in the number of AMPV-positive samples as age increased, by the time of the sampling at processing, flock-level SP was 97%, if a cutoff of 1 was used, and 100%, if any other cutoff was used (Table 2). Consequently, results from the current study indicate that serologic monitoring at processing for the prevalence of AMPV-infected turkey flocks would have high SP, particularly if cutoffs of >1 positive sample are used to classify flocks as positive.
Description of Avian metapneumovirus–free turkey flocks (n = 28) used for the estimation of diagnostic specificity of a monitoring program based on enzyme-linked immunosorbent assay testing at processing.
Discharge, sneezing, or gaping.
It is important to note that increasing the cutoff to >1 positive sample to maximize flock-level SP could potentially decrease the flock-level SE of the program, leading to infected flocks being incorrectly classified as not infected. Investigation of flock-level SE was not the aim of the present study; however, based on reportedly high values of bird-level diagnostic SE 12 and likely high proportion of seropositive birds in infected flocks (within-flock prevalence) by processing time, high values for flock-level SE can be expected. Theoretically, flock-level SE for a monitoring program based on testing 20 birds/flock can be as high as 100%, when assuming a within-flock prevalence of infection of >40%, a cutoff of 1, and a test with a bird-level SE of 98% 12 and a bird-level SP of 99.8% (obtained from the current study). 6 Estimates of the diagnostic accuracy of an AMPV monitoring program (flock-level SE and SP) can be used to calculate the predictive values of test results 5 and to help with their epidemiologic interpretation. For example, assuming a scenario where 15% of the flocks are truly infected, the flock-level SP of 97% obtained here, and a flock-level SE of 100%, would result in a predictive-value positive of 85% (85% probability that a flock classified as infected is truly AMPV infected) and a predictive-value negative of 100% (100% probability that a flock classified as not infected is truly not infected). Knowledge of these diagnostic performance parameters is crucial to design effective AMPV monitoring programs and to interpret results appropriately.
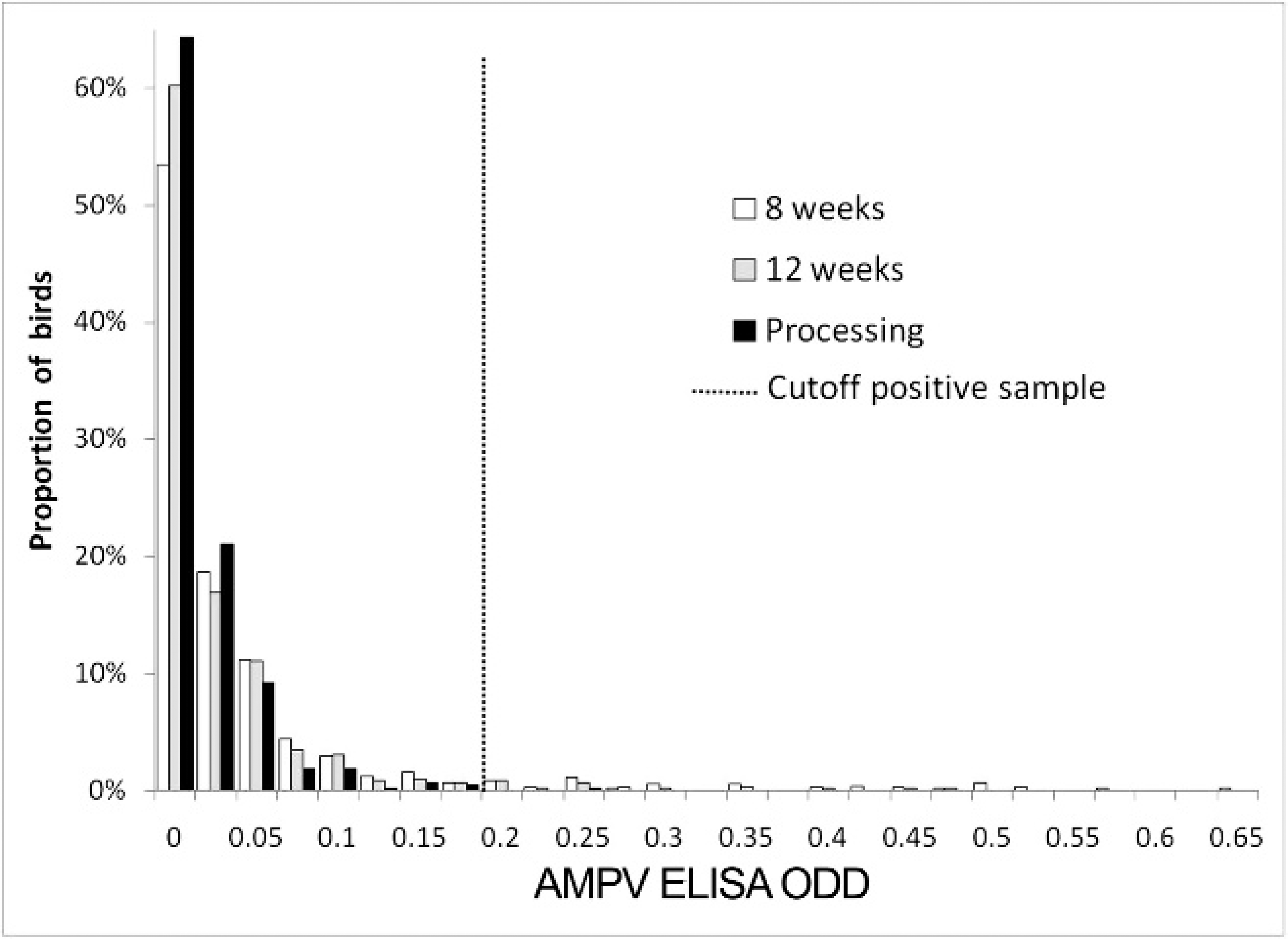
Distribution of enzyme-linked immunosorbent assay (ELISA) titers for Avian metapneumovirus (AMPV) in turkeys from known AMPV-free flocks from Iowa at 8 weeks of age, 12 weeks of age, and at processing. ODD = optical density difference.
An interesting finding of the present study was that the likelihood of (false) positive results decreased as age increased (Table 2). This could be explained by potential cross-reactions with maternal immunity, which wanes gradually over time; however, it commonly disappears by 4 weeks after hatch. 3 Statistical analysis of the factors associated with the probability of a (false) AMPV-positive flock was performed with data from the first sampling and a cutoff of 1. Results suggested that administration of antibiotics any time before the first sampling (P = 0.02) and vaccination for B. avium 14 days before the first sampling (P = 0.08) were positively associated with the probability of (false) positive test results. These associations are indicative of possible cross-reactions of the AMPV test with immune responses to the vaccines and/or other infections that were not examined in this study. The fact that 77% of the vaccinations or treatments occurred before the first sampling may help explain the decrease over time in the number of false positive results that was observed. The potential effect of cross-reactions highlights the need to consider variable performance of diagnostic depending on farm conditions. Overall, findings from this study suggest that monitoring for AMPV at processing is highly specific; however, estimates of SP cannot be extrapolated to other poultry farming systems, such as backyard flocks, or large, vertically integrated commercial farms, or farms from different geographical areas.
Results of a field study to estimate the diagnostic specificity of an enzyme-linked immunosorbent assay for Avian metapneumovirus (AMPV) in turkey flocks at various ages based on individual testing of 20 samples per flock.*
SD= standard deviation; ODD = optical density difference; CI = confidence interval; NA = not applicable.
Minimum number of samples that need to test positive (ODD $0.2) to classify the flock as AMPV infected.
Mean ODD for flocks that were classified as positive. Mean ODD for flocks classified as negative was, 0.005 in all scenarios.
Flock-level diagnostic specificity and 95% confidence intervals obtained using a generalized estimating equation model.
Acknowledgements. The authors thank the turkey producers that participated in the study and the laboratory personnel at the Iowa and Minnesota Veterinary Diagnostic Laboratories and the Minnesota Poultry Testing Laboratory for sample processing and testing. This study was funded by Minnesota Turkey Research and Promotion Council (Project 2005–05) and a U.S. Department of Agriculture-Cooperative State Research, Education, and Extension Service Special Research Grant to the Midwest Poultry Consortium.
Footnotes
a.
Synbiotics Corporation, San Diego, CA.
b.
R: A Language and Environment for Statistical Computing, R Foundation for Statistical Computing, Vienna, Austria.
c.
Excel® 2007, Microsoft Corporation, Seattle, WA.
