Abstract
The Silkie is a typical Chinese breed of chicken. In 2012, batches of Silkies were found to have diffuse tumor-like nodules on their skin after feather removal, when they were slaughtered at about 60 days old. Gross examination showed no visible neoplastic lesions on the visceral organs and peripheral nerves, except slight splenomegaly in individual chickens. The disease was prevalent, with high condemnation rates for skin lesions, which caused great economic losses to the company. Tissues, including skin, visceral organs, and peripheral nerves, were collected for histologic examination. Heparinized blood samples were collected for virus isolation and identification. Marek’s disease virus (MDV), Reticuloendotheliosis virus (REV), and Avian leukosis virus (ALV) were analyzed, using polymerase chain reaction (PCR) tests. Histologic examination showed that all of the tumor-like nodules on the skin were lymphomas. Lymphoproliferative lesions occurred mostly on the skin and only a few on the viscera, including the liver and proventriculus. Infected chick embryo fibroblasts showed clear cytopathic effects; indirect fluorescent antibody test for envelope glycoprotein B was positive. In addition, PCR indicated the presence of MDV serotype 1 infection without REV and ALV. A phylogenetic tree of the Meq gene showed that the isolate (SD121201) and Chinese reference strains, which are very virulent MDVs, are in the same clade. It was concluded that the Silkies tested were infected with MDV serotype 1. The Marek’s disease epidemic has been controlled using CVI988/Rispens vaccines.
Keywords
Marek’s disease (MD) is a lymphoproliferative disease of domestic chickens caused by the highly infectious cell-associated alphaherpesvirus MD virus serotype 1 (MDV-1). Marek’s disease virus strains have been classified into 3 serotypes, namely serotype 1 (Gallid herpesvirus 2), serotype 2 (MDV-2; Gallid herpesvirus 3), and serotype 3 (Meleagrid herpesvirus 1), also known as herpesvirus of turkeys, 4 which have major differences not only in the genome but also in their biological features. Marek’s disease virus serotype 1 infection results in a rapid onset of T-cell lymphomas within several weeks of infection. Tumor infiltration can be found in various organs and tissues, including the viscera, peripheral nerves, skin, gonads, and musculature. 7 Serotype 1 MDV strains, which can be further classified into 4 pathotypes, namely mild (m), virulent (v), very virulent (vv), and very virulent plus (vv+) strains, include all the oncogenic strains and their attenuated forms. 9 Marek’s disease virus infection in the feather follicle epithelium (FFE) is productive and is the sole source of enveloped fully infectious virus that infects susceptible chickens. 3 Chickens are infected with MDV naturally by inhalation, and the source of infection is usually poultry house dust containing feather dander. 7 If house dust such as feathers is not handled properly, the virus will spread from FFE to the external environment. To date, MD is controlled by vaccination and good management practices. 7 However, chickens could be infected simultaneously with vaccine and challenge strains of MDV in the field. 1 Skin-type MD was still the main reason for condemnation of meat-type chickens. 5
In the case reported herein the level of condemnation rates was nearly 15%, and led to great economic loss to the company. The aims of the present study were to make a definite diagnosis by means of comprehensive diagnostic procedures and investigation.
In December 2012, samples of Silkies from a rearing company in Shandong Province were received at the College of Veterinary Medicine, Shandong Agricultural University (Tai’an, Shandong Province, China) that were found to have tumor-like nodules on the skin. Samples were submitted for pathological examination. After initial examination, the chickens were suspected of being infected with MDV-1, and an investigation was conducted. The company concerned has reared chickens for slaughter at 60 days old in a fixed pattern since 2008. The company hatches chicks, which are dispersed to different farms to rear, and then collects the chickens for slaughter. Given the short management cycle, the chickens were not vaccinated against MDV, and did not show obvious lesions. The disease was first detected in June 2012, and spread from October to December 2012. Meanwhile, 48 batches of Silkies slaughtered at approximately 60 days old were surveyed; approximately 20,000 Silkies were tested from each batch. The statistics showed that 46 batches of chickens had tumor-like nodules on their skin, and the incidence of tumors in each batch varied from 6% to 15%; the high condemnation rates of Silkies led to huge economic losses to the company.
Fifty-six live Silkies with cutaneous tumor-like nodules on the neck or thigh were collected from a chicken farm in Shandong province for laboratory examination. Blood was obtained from the wing vein into heparinized tubes. The chickens were then euthanized by carotid artery bleeding after intraperitoneal injection of pentobarbital sodium, and tissue samples were collected, including the skin, liver, spleens, lung, kidneys, heart, proventriculus, bursa of Fabricius, and peripheral nerves. One portion of each tissue was fixed in 10% formalin solution for 24 hr then dehydrated, embedded in paraffin, and cut into 5-μm sections. The sections were stained with hematoxylin and eosin and examined by microscopy. Another portion was stored at −70°C for further analysis.
Chicken embryo fibroblast (CEF) cells were prepared from 11-day-old specific pathogen–free embryos a as described previously. 2 Secondary CEF cells were maintained in Dulbecco modified Eagle medium supplemented with 1% fetal bovine serum at 37°C with 5% CO2 and seeded to 80−90% confluence in cell culture dishes for virus infection. Heparinized blood samples collected from affected chickens were inoculated into primary CEF cells and cultured for 5 days for each passage. After 2 blind passages, cells were harvested when cytopathic effects became clearly visible in approximately 80% of the cells. The cells were replated in 96-well plates and fixed with chilled acetone. 8 Indirect fluorescent antibody test (IFAT) was then performed using specific primary monoclonal antibody to MDV-1 (clone BA4 b ; Cui Z, Lu C, Qin A, et al.: 1996, Monoclonal antibodies against serotype 1-specific and group-common epitopes on the glycoprotein b of Marek’s disease viruses. In: Current research on Marek’s disease: Proceedings of the 5th international symposium on Marek’s disease, East Lansing, Michigan, September 7–11, ed. Silva R, pp. 233–238. American Association of Avian Pathologists, Jacksonville, FL). Fluorescein isothiocyanate–labeled anti-mouse immunoglobulin G c was used to bind the primary antibody.
To test the viruses using polymerase chain reaction (PCR), 2 sets of primers were designed to detect MDV (forward primer, 134625–134604, 5′-TCATCAGGGTCTCCCGTCACCT-3′; reverse primer, 133599–133620, 5′-AGAGATGTCTCAGGAGCCAGAG-3′) and Reticuloendotheliosis virus (REV; forward primer, 257–274, 5′-CATACTGAGCCAATGGTT-3′; reverse primer, 531–550, 5′-AATGTTGTAGCGAAGTACT-3′), respectively. Tissue DNA from 56 samples was extracted from the liver using a commercial kit d and then used as templates for PCR. The PCR amplifications were carried out with rTaq polymerase e using the following cycle conditions for all 3 reactions: 95°C for 5 min, followed by 30 cycles of 94°C for 30 sec, 51°C for 1 min, and 72°C for 1 min. A final extension step was conducted at 72°C for 10 min. For Avian leukosis virus (ALV) detection, the specific PCR used the primer pair H5/H7 (forward primer, 5258–5277, H5: 5′-GGATGAGGTGACTAAGAAAG-3′; reverse primer, 5783–5802, H7: 5′-CGAACCAAAGGTAACACACG-3′) for subgroup J and H5/AD1 (reverse primer, 5327–5345, AD1: 5′-GGGAGGTGGCTGACTGTGT-3′) for subgroups A–D. 6 DNA was isolated from the liver using a high-purity viral nucleic acid kit. f The PCR products were purified using a gel extraction kit. g After purification, the target DNA fragment was cloned into the pMD18-T Easy vector for sequencing. h For all reactions, DNA isolation and PCR amplification were performed in least 2 independent experiments. Multiple nucleotide and amino acid sequences of the Meq gene were aligned using the Jotun Hein method using commercial software. i The reference sequences used for comparison were retrieved from the GenBank database: GA (AF147806), MD-5 (AF243438), 0297 (AF493553), 0304 (AF493554), GX070079 (EU427304), N (AF493557), G2 (AF493556), YL040920 (DQ174459), GXY2 (EF546430), 549a (DQ534530), 548a (DQ534532), 595 (DQ534533), 648a (DQ534534), 686 (DQ534535), CU-2 (DQ534537), 814 (AF493551), CVI988-BP5 (DQ534536), RB1B (DQ534541), RM-1 (DQ534542), CVI988 (Intervet) (DQ534538), JM/102W (DQ534539), and GX0101 (JX844666).
Postmortem diagnosis showed that tumor-like nodules were mostly found on the skin of chickens, especially on the neck, in various sizes (0.1–1.3 cm in diameter; Fig. 1A–1C). The nodules were solid and off-white in color. In contrast, the chickens had no obvious neoplastic lesions on the liver, spleen, lung, kidneys, heart, proventriculus, testes (or ovaries), peripheral nerves, and bursa of Fabricius. Only a few chickens showed slight swelling of the proventriculus and spleen (Fig. 1D).
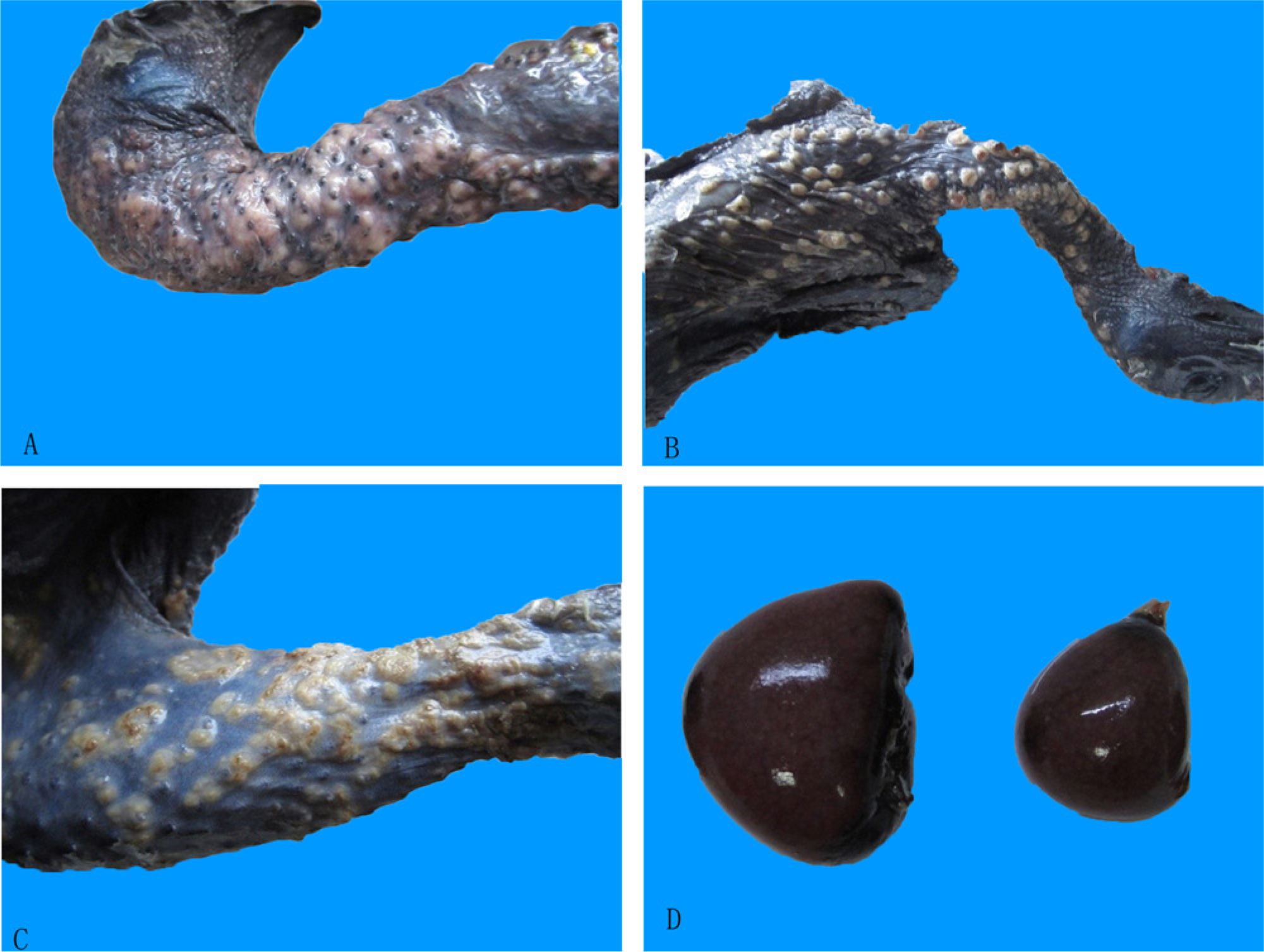
Gross lesions in diseased chickens.
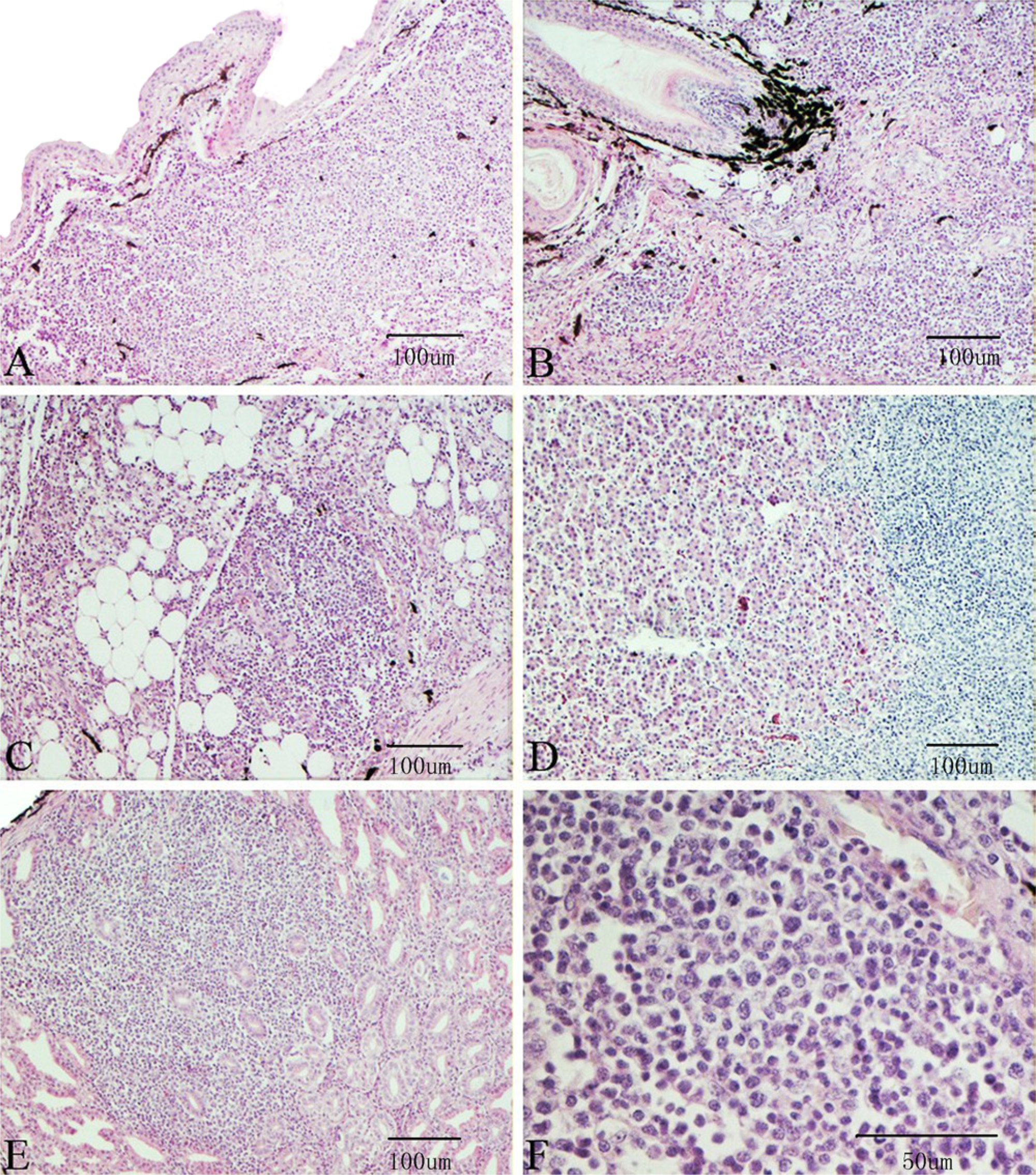
By histologic examination, lymphoproliferation was located mainly in the dermis layer around the feather follicles (Fig. 2A, 2B) and subcutaneous tissue (Fig. 2C). It could also be found in the liver (Fig. 2D), in the submucosa or among the glands of the proventriculus (Fig. 2E), under the tunica mucosa of the bronchi, and in the kidney and spleen (not shown). Proliferating lymphocytes showed varying morphology and size (Fig. 2F).
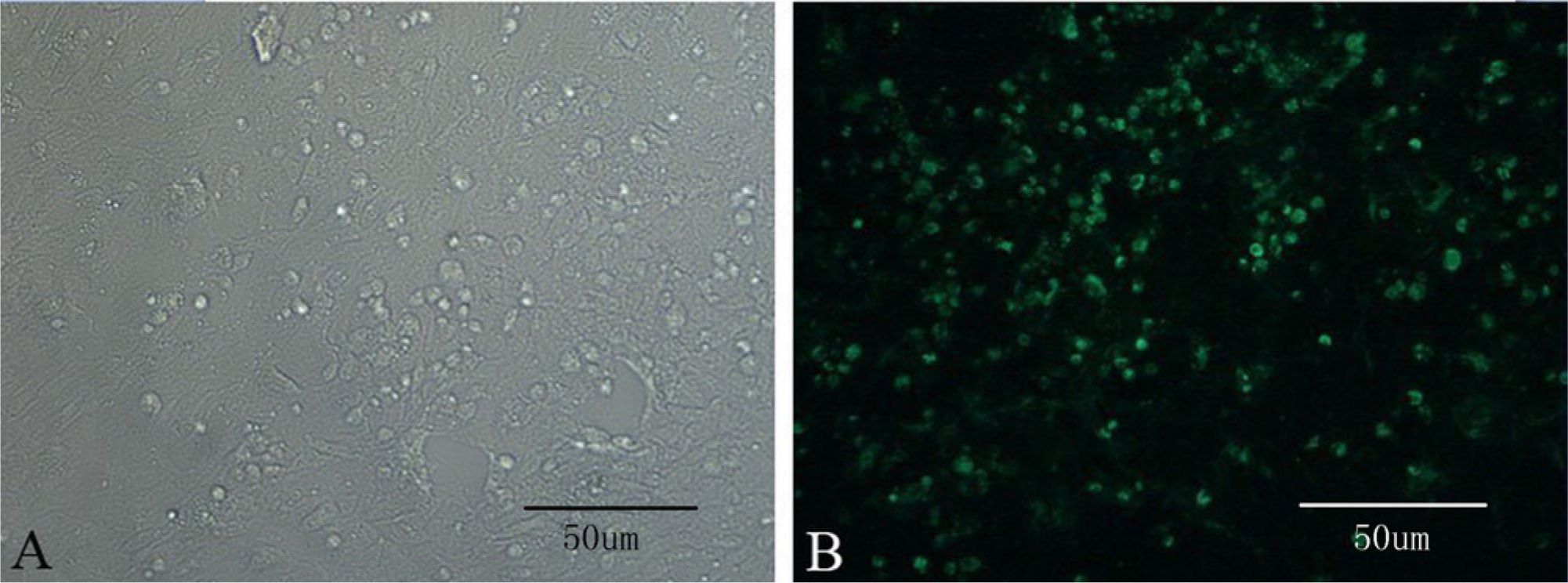
After 2 blind passages, cytopathic effects became clearly visible in infected CEF cells (Fig. 3A). The IFAT was performed against MDV-1 (Fig. 3B).
The MDV PCR analysis showed that 51 out of 56 samples were positive, while all samples were negative for REV and ALV. Sequence analysis of the isolates (SD121201) showed that the length of the fragment was 1.1 kb, which contained the Meq gene open reading frame. The similarity of the nucleotide and deduced amino acid sequences of the Meq gene between the isolated strains and the reference strains was 98.9.0–100% and 97.4–100%, respectively. The isolated strain SD121201 shared 100% nucleotide sequence and 100% amino acid sequence homology with GX0101, GX070097, and GXY2. Using commercial software, i the isolate showed no insertion at 180, which has been associated with attenuation or low virulence in reports from other countries. A phylogenetic tree based on the Meq gene sequences of the isolates and 22 MDV reference strains was produced by the neighbor-joining method using MEGA version 5.0 (http://megasoftware.net/mega.php). The topological accuracy of the tree was estimated by the bootstrap method, with 500 replicates, which is presented in Figure 4. The phylogenetic tree (Fig. 4) showed the isolate was in the same clade as other Chinese isolates and Chinese reference strains.
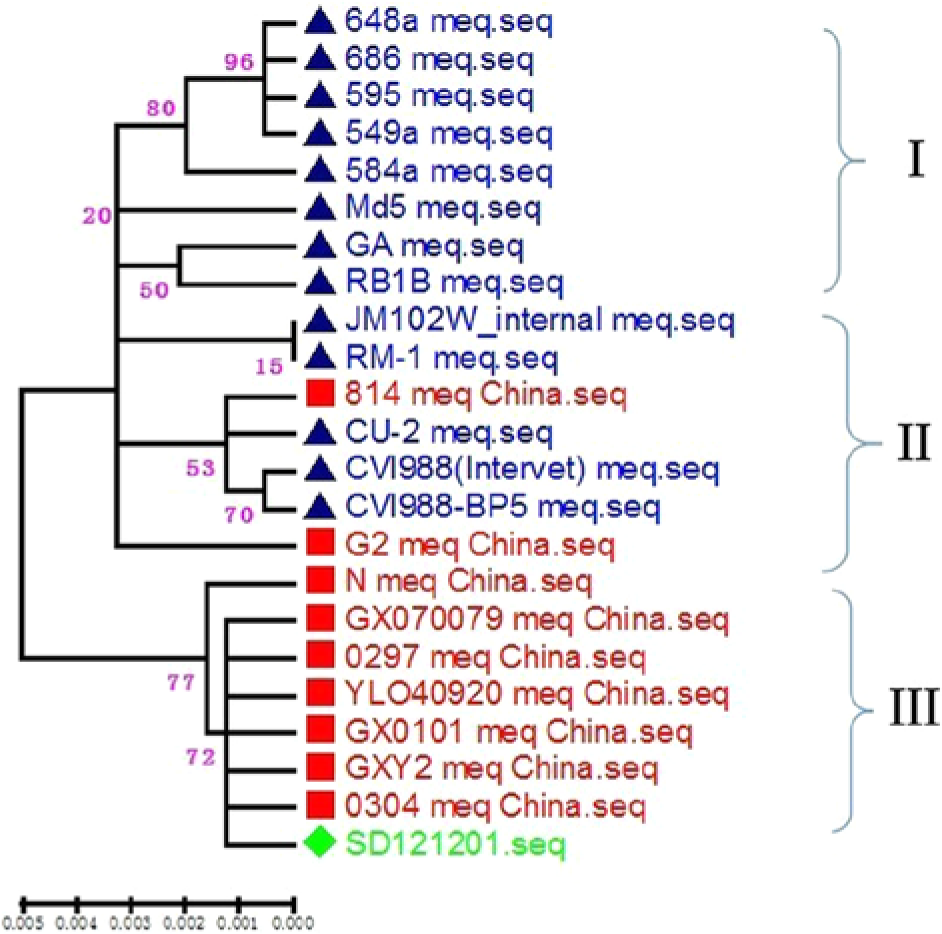
Phylogenetic tree based on the Meq gene sequences of Marek’s disease virus (MDV) isolates and MDV reference strains. An unrooted phylogenetic tree was generated by the distance-based neighbor-joining method using MEGA 5.0 software. The reliability of the tree was assessed by bootstrap analysis with 500 replications. The isolate is marked by a diamond; Chinese reference viruses are marked by filled squares; GA (AF147806), MD-5 (AF243438), 549a (DQ534530), 548a (DQ534532), 595 (DQ534533), 648a (DQ534534), 686 (DQ534535), CU-2 (DQ534537), CVI988-BP5 (DQ534536), RB1B (DQ534541), RM-1 (DQ534542), CVI988 (Intervet) (DQ534538), and JM/102W (DQ534539) are marked by triangles.
The gross lesions and the results of histological examination were consistent with MD. Given that the chickens were not vaccinated with CVI988, the IFAT results indicate that they were infected with MDV-1. After the flocks had been vaccinated with serotype I CVI988/Rispens, there were scarcely any chickens with tumor-like nodules on the skin when slaughtered at 60 days old. Production performance such as average slaughter weight, rearing cycle, feed conversion, and average daily feed from 50 to 60 days, improved significantly (Table 1), which has also improved the accuracy of the diagnosis.
Condemnation rates and production performance comparison before and after vaccination.*
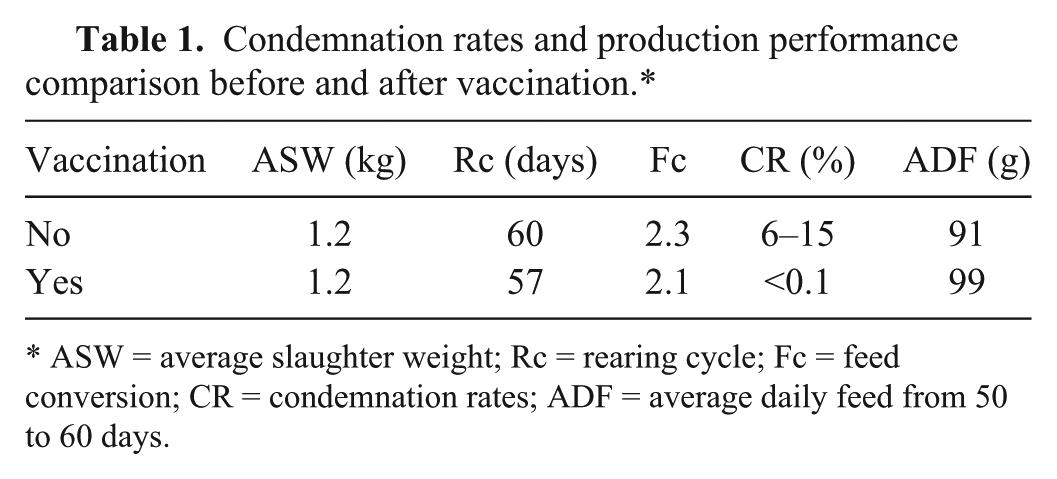
ASW = average slaughter weight; Rc = rearing cycle; Fc = feed conversion; CR = condemnation rates; ADF = average daily feed from 50 to 60 days.
Meq is the only protein in the repeat long region of the MDV genome whose phylogenetic tree reflects the cladogram of the repeat long region DNA sequences. From the phylogenetic tree, 3 main clades were identified. The first clade (I) is composed of 1 virulent (v) strain (GA), 4 very virulent (vv) strains (RB1B, 549a, 595, Md-5), and 3 very virulent plus (vv+) strains (584a, 648a, 686). The second clade (II) consists of attenuated and mildly (m) virulent strains (CVI988-BP5, CVI988 [Intervet], CU-2, RM-1), 1 Chinese vaccine strain (814), and 1 virulent strain, which contained a 177-bp insertion (JM/102 W). The third clade (III) is composed of Chinese isolates and Chinese reference strains. 9 The new strain SD121201 isolated from Shandong Province shares 100% nucleotide sequence and 100% amino acid sequence homology with GX0101, GX070097, and GXY2. The phylogenetic tree showed that the current isolate and the Chinese reference strains are in the third clade, clearly distant from the other reference strains.
It is accepted that MDV infection in the FFE is the only source of infectious virus. The main cause of the outbreak of MD was surmised to be feather drift from chickens infected with MDV. After investigation, some defects were identified in the company’s rearing methodology. Slaughterhouses, hatcheries, and farms were close enough that the virus could spread from one to another. The shed feathers were not handled properly after slaughter. Also, no strict biosecurity measures were found at the hatcheries. Moreover, the chickens were generally not vaccinated against MDV-1 because the rearing cycle occurred within 60 days and the cutaneous lesions were inconspicuous. However, the many infected chickens were potential sources of infection. Furthermore, chickens are reared at high density in Shandong Province, which also contributes to outbreaks and spread of MDV. Although there may be no visible cutaneous lesions, poor performance caused by MDV infection can lead to great economic loss. Therefore, chicken flocks should be vaccinated against MDV on the first day after hatching.
Chickens reared to more than 60 days old are generally vaccinated against MDV, and although they may still become infected with MD occasionally, the incidence rate is no higher than 1%. The neoplastic lesions of MD reported since the 1990s were mainly in visceral organs and nerves, rarely on the skin. Reports of MD in China were reviewed, and 102 reports were published since 1900. All the reports refer to lesions in the visceral organs, 49 in the nerves, and 6 in cutaneous lesions; 12 reports were associated with Silkies, and included 3 cases with skin-type MDV infection, accounting for 12% of reports. Silkies infected with MDV-1 show obvious lesions mainly at 50–80 days; 30 days is the earliest reported start time, significantly earlier than in other breeds of chicken. Therefore, the reasons why skin was predominately involved in the current outbreak were varied, perhaps due to the genetics of the chicken and/or the virus or the receptors in the skin for the virus. Vaccination status, age, and biosecurity may have also been contributing factors.
Footnotes
a.
Jian Sais Poultry Co., Jinan, Shandong Province, China.
b.
Kind gift from Dr. Zhizhong Cui, College of Veterinary Medicine, Shandong Agricultural University, Tai’an, Shandong Province, China.
c.
Invitrogen China Ltd, Shanghai, China.
d.
Axygen Biotechnology Ltd, Hangzhou, Zhejiang Province, China.
e.
Takara Biotechnology (Dalian) Co. Ltd, Dalian, Liaoning Province, China.
f.
Roche (China) Ltd, Shanghai, China.
g.
Axygen, Corning Inc., Tewksbury, MA.
h.
BGI Sequencing, Beijing, China.
i.
MegAlign, DNASTAR Inc., Madison, WI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by the National Natural Science Foundation of China (grant 31172296).
