Abstract
The purpose of this study was to determine the effect of blood contamination of cerebrospinal fluid (CSF) on the results of indirect fluorescent antibody tests (IFATs) for Sarcocystis neurona and Neospora hughesi. The in vitro study used antibody-negative CSF collected from non-neurologic horses immediately after euthanasia and blood samples from 40 healthy horses that had a range of IFAT antibody titers against S. neurona and N. hughesi. Serial dilutions of whole blood were made in seronegative CSF to generate blood-contaminated CSF with red blood cell (RBC) concentrations ranging from 10 to 100,000 RBCs/μl. The blood-contaminated CSF samples were then tested for antibodies against both pathogens using IFAT. Blood contamination of CSF had no detectable effect on IFAT results for S. neurona or N. hughesi at any serologic titer when the RBC concentration in CSF was <10,000 RBCs/μl. At concentrations of 10,000-100,000 RBCs/μl of CSF, positive CSF results (IFAT titer ≥5) for S. neurona and N. hughesi were detected only when the corresponding serum titers were ≥160 and ≥80, respectively. The IFAT performed on CSF is reliable for testing horses for equine protozoal myeloencephalitis caused by S. neurona or N. hughesi, even when blood contamination causes the RBC concentration in CSF to be up to 10,000 RBCs/μl.
Equine protozoal myeloencephalitis (EPM) is caused by infection of the central nervous system of horses and ponies with the protozoan parasites Sarcocystis neurona, 7 and, less commonly, Neospora spp., 3 , 11 , 13 particularly Neospora hughesi. 2 , 8 , 14 Equine protozoal myeloencephalitis is often a progressively debilitating disease that induces clinical signs that may include weakness, ataxia, loss of proprioception, incoordination, muscle wasting, behavioral changes, and death. 9 , 12 Antemortem diagnosis of EPM is based upon a thorough neurologic examination, evaluation of serum and cerebrospinal fluid (CSF) for the presence of antibodies to S. neurona and N. hughesi, and the elimination of other neurologic disorders. It is important to note that the presence of antibodies in serum indicates only exposure to, and not necessarily clinical infection with, the respective parasites. At this time, definitive diagnosis of EPM relies on immunohistopathological examination of neural tissue at postmortem. 10
Until recently, the Western blot (WB) test was the standard diagnostic test used for serologic surveys and the clinical diagnosis of EPM caused by S. neurona. When performed on serum, the overall sensitivity and specificity of the WB for the detection of clinical EPM were estimated at 80% and 38%, respectively, for horses with signs of neurologic disease. 4 Specificity of the WB test when used on
CSF was estimated at 44%. 4 A serum indirect fluorescent antibody test (IFAT) was recently evaluated for the diagnosis of EPM caused by S. neurona by using an arbitrarily chosen cut-off serum titer of 80 for a positive test. When compared to the WB test on serum where a weak positive WB was considered a positive result, the IFAT had the same sensitivity (88.9%) but increased specificity (IFAT 100%, WB 87.2%). 5 Sensitivity and specificity of the IFAT on CSF of horses naturally infected with S. neurona were recently estimated to be 100% and 99%, respectively, at a cut-off titer of 5. 6 An IFAT test for N. hughesi antibodies has been validated by testing serum and CSF collected before and after experimental infection with N. hughesi. 16
Blood contamination of CSF has previously been shown to yield false positive results in WB tests for S. neurona antibodies. 15 The influence of blood contamination of CSF on IFAT results has not yet been evaluated. The purpose of this study was to determine the effect of contamination of antibody-negative CSF with blood collected from seropositive horses on the results of IFA tests for S. neurona or N. hughesi performed on CSF.
Blood samples were collected into evacuated tubes (serum and ethylenediaminetetraacetic acid [EDTA]) from 40 healthy nonneurologic horses that were previously shown to have a wide range of serum IFAT titers to both S. neurona (range <40-2,560) and N. hughesi (range <40-320). After clot formation at room temperature, serum tubes were centrifuged (4,190 × g for 5 minutes), and the serum was then removed and frozen at −80°C until tested. Serum samples were tested for antibodies against S. neurona and N. hughesi using the IFAT as previously described. 5 , 16 Sera were initially diluted at 1: 40, and the reciprocal end-point titer was determined for each positive sample as the last dilution with evidence of distinct, whole-parasite fluorescence. The concentration of red blood cells (RBCs) in the blood samples collected in EDTA was measured within 40 minutes of collection using a hemacytometer.
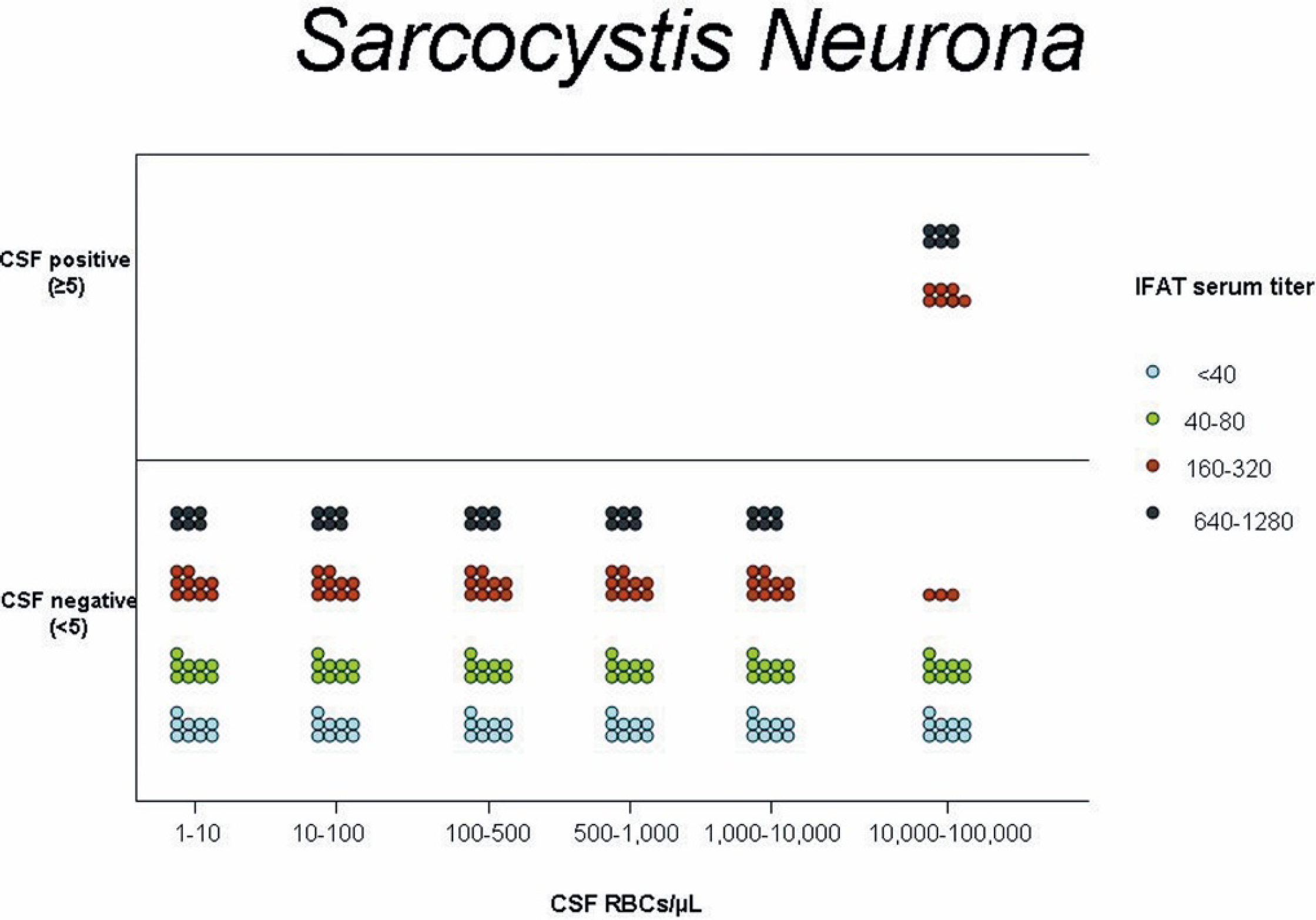
Graphic representation of the serum titer and number of cerebrospinal fluid (CSF) positive and negative horses based on indirect fluorescent antibody testing for Sarcocystis neurona after iatrogenic blood contamination of CSF at varying amounts of red blood cell concentrations.
Serum was collected from 5 neurologically normal horses that were scheduled for euthanasia at the Veterinary Medical Teaching Hospital for reasons other than neurologic disease. Serum collected was processed as described above. Cerebrospinal fluid (approximately 60–70 ml) was then collected from the cisterna magna of each horse by introducing an 18-gauge spinal needle via the atlanto-occipital space immediately after euthanasia with 100 ml of Beuthanasia-D administered rapidly through a catheter placed in the jugular vein. One aliquot of each CSF sample was submitted for cytology and IFA testing; approximately 60 ml of CSF from each horse was centrifuged (8,200 × g for 15 minutes) and the supernatant was removed and stored in 10-ml aliquots at −80°C. From the 5 horses, only CSF samples without blood contamination (<1 RBC/μl) that tested negative (<5) for both pathogens by IFAT were saved and used as surrogate CSF for the blood contamination study.
For each of the 40 healthy horses, the serum titer to both S. neurona and N. hughesi was determined by IFAT. The concentration of RBCs in the blood of each horse (range 6.29–9.89 × 106/μl; median 7.71 × 106/μl) was used as the basis for making serial dilutions of whole blood with banked antibody-negative CSF to create blood-contaminated CSF samples with RBC concentrations in the following 6 ranges: 10,000-100,000 RBCs/μl CSF; 1,000-9,999 RBCs/ml CSF; 500–999 RBCs/μl CSF; 100–499 RBCs/μl CSF; 10–99 RBCs/μl CSF; and <10 RBCs/μl CSF. Initially, a 1: 10 dilution was made between the EDTA blood sample and the surrogate CSF to obtain RBCs in the 100,000-1,000,000 range, followed by 5 serial 1:10 dilutions of EDTA blood to surrogate CSF. To obtain results for the 500–999 RBCs/μl range, a 1:1 dilution was made from the 1,000-9,999 RBCs/μl category. Each range of blood-contaminated CSF was tested using IFAT for antibodies against both pathogens as previously reported. 4 , 6 Results of the IFA tests on CSF were reported as negative (<5) or positive (≥5), and positive samples were diluted to an end-point titer. Negative and positive controls, consisting of negative CSF from the same banked uncontaminated sample and positive CSF from previous positive cases with no blood contamination, were run with each sample tested. The percentage of positive samples was evaluated for each combination of serum IFAT titer in the contaminating blood and the number of RBCs in the blood-contaminated CSF sample. Because only CSF samples with RBC concentrations in the 10,000-100,000 RBCs/μl range tested positive for antibodies, Spearman's rank correlation coefficient was used to evaluate the correlation between titer and the percentage of positive samples only for the subset of samples with RBC concentrations within this range. Significance was set at P < 0.05.
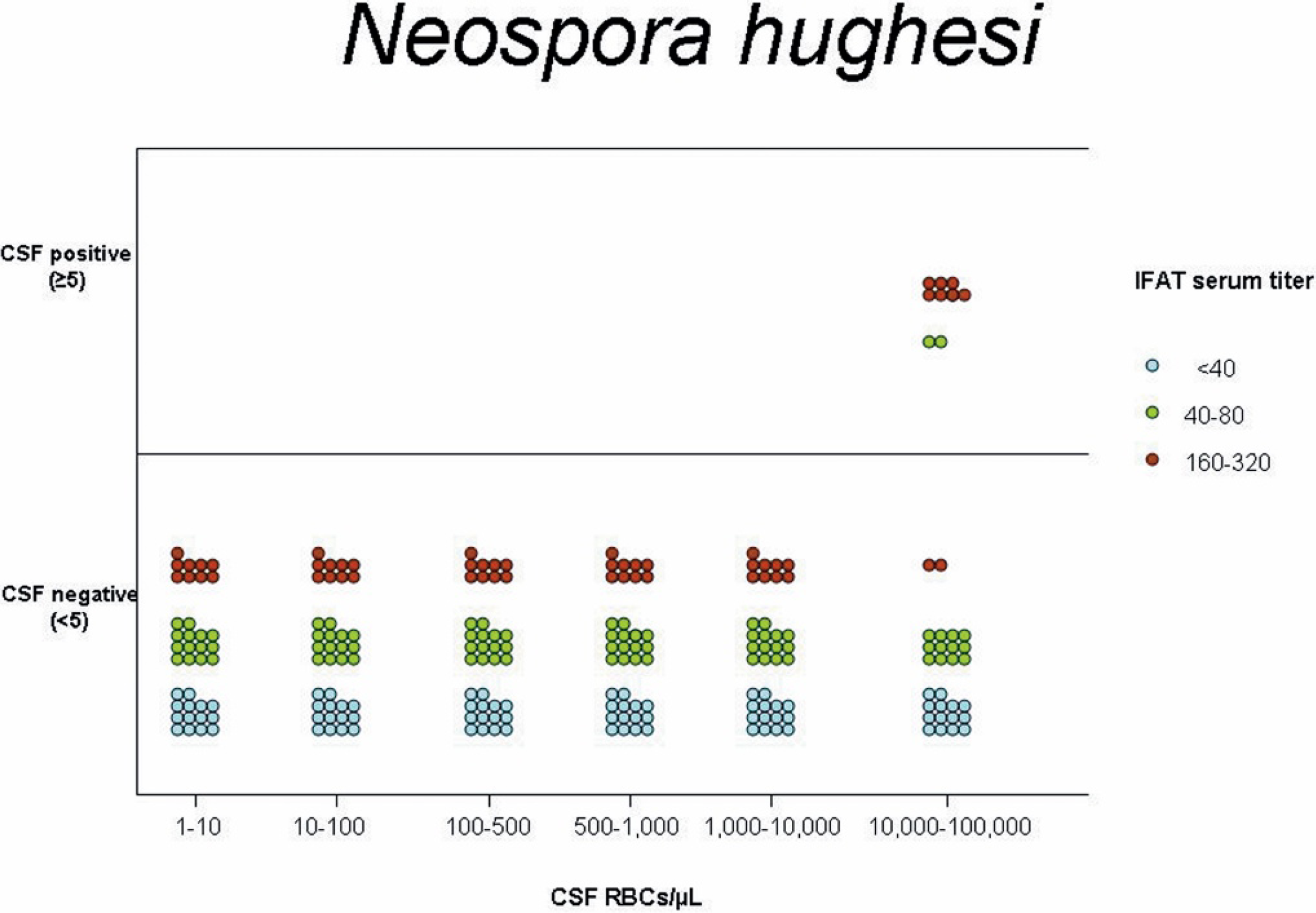
Graphic representation of the serum titer and number of cerebrospinal fluid (CSF) positive and negative horses based on indirect fluorescent antibody testing for Neospora hughesi after iatrogenic blood contamination of CSF at varying amounts of red blood cell concentrations.
No effect of blood contamination on IFAT results for S. neurona or N. hughesi was found at any serologic titer when the CSF was contaminated with <10,000 RBCs/μl CSF. At concentrations of 10,000-100,000 RBCs/μl of CSF, positive IFAT results at a titer of ≥5 were detected only when serum titer in the contaminating blood was ≥160 for S. neurona (Fig. 1). Correlation that approached significance was found between the 7 serum titer values (<40, 40, 80, 160, 320, 640, ≥1,280) and the percentage of positive samples (Spearman's ρ = 0.14, P = 0.05). At concentrations of 10,000-100,000 RBCs/μl of CSF, positive IFAT results at a titer of ≥5 were detected only when the corresponding serum titers was ≥80 for N. hughesi (Fig. 2). Although there was a strong trend for higher titer values in contaminating blood to generate a higher percentage of IFAT-positive CSF samples within the range of N. hughesi IFAT titers of <40-320, this trend did not reach significance (Spearman's ρ = 0.95, P = 0.06).
The finding that blood cell contamination of CSF may cause false-positive IFAT results for both S. neurona and N. hughesi is in agreement with results obtained using a WB test for S. neurona in another in vitro blood contamination study. 15 As in that study, the present results showed that the titer of antibody in the contaminating blood and the amount of blood both influence the outcome of blood contamination on the results of antibody tests for EPM performed on CSF. It is not possible, however, to directly compare the results of the 2 studies because the IFAT is a quantitative test, whereas the WB is qualitative. Western blot test results were characterized as strongly immunoreactive, moderately immunoreactive, weakly immunoreactive, and negative when compared to controls. 15 To the authors' knowledge, the degree of WB immunoreactivity has not been correlated with IFAT titer. Regardless, the extent of blood contamination necessary to create false-positive IFAT results for both S. neurona and N. hughesi appears to be much higher than that needed to generate false-positive results on the WB test for S. neurona. Contamination with blood that has an IFAT titer of ≥160 and ≥80 for S. neurona and N. hughesi, respectively, in amounts sufficient to cause RBC concentrations in CSF to exceed 10,000 RBCs/μl is necessary to cause false positive results (≥5) on IFA tests performed on the CSF. In contrast, false-positive WB test results on CSF were found with low levels of blood contamination that yielded RBC concentrations in CSF as low as 8 RBCs/μl. 15 In the present study, as in the WB study, the higher the titer in contaminating blood, the higher likelihood of false-positive results. 15
Albumin quotient (AQ) and the immunoglobulin G (IgG) index have been used as indicators of increased permeability of the blood-CSF barrier and to estimate the degree of iatrogenic blood contamination of CSF. 1 Albumin quotient and IgG index were not measured in the current study because contamination of the CSF samples with small volumes of blood that were strongly or moderately immunoreactive on a WB test for S. neurona was previously shown to have no effect on AQ. 15 The authors of that study concluded that the number of RBC in a CSF sample may be a more sensitive indicator of whole blood contamination than AQ. 15 Contamination of CSF with blood that was strongly immunoreactive to S. neurona resulted in an increase in IgG index in CSF, 15 a result that would be expected with intrathecal production of IgG, thereby making the IgG index difficult to interpret. Because of these discrepancies, a recent consensus statement by the American College of Veterinary Internal Medicine does not recommend the use of AQ or IgG index for the diagnosis of EPM. 10
Potential limitations of the current study include the relatively small number of horses in the higher serum IFAT groups (S. neurona ≥ 640, N. hughesi ≥ 160). Several horses with high serum IFAT titers (S. neurona ≥ 640, n = 2 horses; N. hughesi ≥ 320; n = 7 horses) had repeated blood samples taken and used to contaminate CSF; however, when these samples were excluded from analysis, statistical findings were unchanged. Because this study was performed in vitro, future in vivo studies may be necessary to confirm the effect of iatrogenic blood contamination during collection of CSF on the results of IFA tests for S. neurona and N. hughesi on CSF.
In conclusion, iatrogenic blood contamination of CSF appear to falsely affect IFAT results for EPM caused by S. neurona and N. hughesi only when the degree of blood contamination is high and results in a large number of RBCs (>10,000/μl) in the CSF sample to be tested. The clinical relevance of a positive (≥5) CSF IFAT titer to S. neurona and N. hughesi should be interpreted with caution if blood contamination causes the RBC concentration in CSF to exceed 10,000/μl and the corresponding serum titer is ≥160 for S. neurona and ≥80 for N. hughesi, because false-positive results may occur.
Acknowledgements. Funding for this study was provided by the Center for Equine Health at the University of California at Davis. The authors thank Catherine Glines for assistance in collecting cerebrospinal fluid samples.
