Abstract
Seroconversion and cross-reactivity in cattle infected with Anaplasma marginale or a recently described Ehrlichia species (BOV2010 from British Columbia, Canada) were investigated. The study used 76 samples from 20 animals, a commercially available competitive enzyme-linked immunosorbent assay (cELISA) for bovine anaplasmosis, and an indirect fluorescent antibody test (IFAT). Blood smear examination and/or polymerase chain reaction assay were performed to confirm or rule out the presence of Anaplasma or Ehrlichia. Samples comprised 3 groups. Group 1 consisted of 24 samples from 9 cattle naturally infected with Ehrlichia sp. BOV2010. Group 2 had 13 samples from 3 A. marginale–infected cattle from Manitoba, Canada. Group 3 had 39 samples, consisting of 26 from 5 calves experimentally infected with Ehrlichia sp. BOV2010, 10 from 2 calves experimentally infected with A. marginale from cattle (Manitoba) or bison (Saskatchewan), and 3 from an uninfected calf. All samples from cattle naturally or experimentally infected with Ehrlichia sp. BOV2010 or A. marginale were seropositive for A. marginale by both cELISA and IFAT, except 3 calves euthanized at 28 and 33 days post-inoculation (DPI) that did not seroconvert. Antibodies were detected in 2 experimental animals inoculated with Ehrlichia sp. BOV2010, as early as 28 and 33 DPI by the cELISA and IFAT, respectively, and by 42 DPI for both tests. The current study demonstrates that the specificity of the recombinant major surface protein 5 (MSP5) antigen is not restricted to Anaplasma spp., which reduces the utility of the test for serological diagnosis of bovine anaplasmosis in regions where Ehrlichia sp. BOV2010–infected cattle might exist.
Introduction
Tick-borne pathogens are an increasing problem worldwide, including species of the blood-borne bacterial family Anaplasmataceae, which cause disease in animals and human beings. 5 Species of Ehrlichia and Anaplasma are structurally, genetically, and immunologically similar.1,5,15 Most Anaplasma spp. infect erythrocytes of domestic and wild ruminants. Three Anaplasma species infect other blood cells: Anaplasma phagocytophilum infects neutrophils, Anaplasma bovis infects monocytes, and Anaplasma platys infects platelets. 15 Ehrlichia spp. infect leukocytes of a wide range of host species including human beings, domestic canines, ruminants, equids, and felids, except Ehrlichia ruminantium, which infects endothelial cells of domestic and wild ruminants. Ehrlichia ruminantium is distributed in sub-Saharan Africa and several islands in the Caribbean. 1 Anaplasma marginale causes bovine anaplasmosis in cattle. Although A. marginale is globally distributed, it is absent in Canada except for very few controlled incursions. 11 Both E. ruminantium and A. marginale are important globally to the cattle industry.
The diagnosis of Anaplasma and Ehrlichia infections may include identification of the organism in stained blood smears, polymerase chain reaction (PCR) analysis, and detection of antibodies using various serological tests. Examination of stained thin blood smears for evidence of rickettsemia is a common method used for diagnosing early or acute infections with Anaplasma. 31 However, the evaluation of blood smears can be unreliable, particularly at low levels of rickettsemia. Molecular analyses using PCR and DNA hybridization assay have been used for the detection or confirmation of Anaplasma infection in seropositive cattle.7,9 A sensitive and specific nested PCR targeting the major surface protein (msp5) gene has been developed to identify A. marginale in cattle. 26 Despite the availability of DNA-based assays for the detection of Anaplasma, serologic tests using major surface protein antigens are considered the most practical means of screening large numbers of cattle for evidence of infection. 15 Several serological tests have been developed to detect infections of Anaplasma and Ehrlichia, including the competitive enzyme-linked immunosorbent assay (cELISA), indirect ELISA, card agglutination test, and indirect fluorescent antibody test (IFAT).1,31 These tests may be hindered by lack of specificity within or between the several species of Anaplasma and Ehrlichia, which bear homologous families of immunodominant outer membrane proteins. 1
Currently, the cELISA is the primary test used for diagnosing infection with A. marginale in cattle. The IFAT also can be used as a diagnostic test for anaplasmosis. 31 Results of the IFAT have been demonstrated to have 90.8% agreement with DNA hybridization results for A. marginale infection in carrier cattle. 10 Another study reported 96% agreement between an indirect ELISA and IFAT in detecting antibodies against A. marginale in Brazilian cattle. 18 However, the ELISA is generally preferred to the IFAT because the ELISA can be semiautomated making it possible to evaluate larger numbers of serum samples, and the reading of results is less subjective. 18 The cELISA, based on the use of a monoclonal antibody (mAb) that recognizes major surface protein 5 (MSP5) in A. marginale, Anaplasma centrale, and Anaplasma ovis, was validated using a nested PCR procedure, with estimates of sensitivity and specificity of 96–100% and 95–100%, respectively.14,26 The cELISA test kit a is commercially available for the diagnosis of bovine anaplasmosis in cattle. In a previous study, cattle experimentally infected with A. phagocytophilum tested positive using this cELISA test kit. 6 In another study, cross-reactivity was not demonstrated between human or dog antibodies to A. phagocytophilum and the recombinant MSP5 antigen of A. marginale when tested by cELISA. 25 However, cross-reactivity was evident when recombinant MSP5 antigens of A. phagocytophilum or A. marginale were incubated with heterologous sera using an indirect ELISA format. 25
A newly described Ehrlichia genotype (BOV2010) was recently detected in naturally infected cattle from British Columbia, Canada. 8 Based on the results of cELISA, a the cattle were originally suspected of being infected with A. marginale. 11 The present study was conducted to investigate the specificity of serological diagnosis of A. marginale and Ehrlichia sp. BOV2010 in cattle and the potential for confounding interpretation of cELISA and IFAT results used to detect bovine anaplasmosis.
Materials and methods
Samples
Samples of fresh or frozen serum, as well as ethylenediamine tetra-acetic acid whole blood and/or DNA samples extracted from blood were assigned to 1 of 3 groups, based on origin of the samples (Table 1). All animal use was conducted under the approval of the University of Saskatchewan Committee on Animal Care and Supply in accordance with guidelines of the Canadian Council on Animal Care. 21
Source and grouping of serum samples tested by competitive enzyme-linked immunosorbent assay (cELISA) and indirect fluorescent antibody test (IFAT) for Anaplasma marginale.*

For group 1, infection was confirmed by polymerase chain reaction (PCR) in a previous study (7 animals) 8 or current study (2 animals). For group 2, infection was confirmed by PCR in an earlier study. 11 For group 3, molecular analysis results for 7 animals (A, C–G, I) were reported in a previous study. 8 Molecular analysis of samples from animal H was conducted during the current study.
Group 1
Twenty-four serum samples (1–5 per animal) were collected at different times from 9 adult field cattle from British Columbia naturally infected with Ehrlichia sp. BOV2010. Whole blood and/or extracted DNA obtained from each animal were analyzed by PCR to confirm infection with Ehrlichia sp. BOV2010. Diagnosis of infection in 7 animals from this group was described in a previous study, 8 whereas infection of the other 2 animals was confirmed in the present study following the PCR protocol described previously. 8
Group 2
Thirteen serum samples (1–6 per animal) were collected at different periods from 3 field cattle from Manitoba naturally infected with A. marginale (2009) and stored at −20°C. Infection with A. marginale in these animals had been detected by cELISA and confirmed by PCR analysis of whole blood. 11
Group 3
A series of paired whole blood and serum samples were collected from 8 splenectomized calves experimentally infected with A. marginale or Ehrlichia sp. BOV2010. Five calves were inoculated with single or pooled aliquots of citrated whole blood collected from 7 of the naturally infected British Columbia cattle included in group 1 as described previously. 8 Two positive control calves were inoculated with stabilates of A. marginale from cattle (Manitoba) or bison (Saskatchewan). One calf remained uninfected as a negative control. Samples were collected prior to inoculation and at 14, 28, 33, 42, 56, 70 and 84 days post-inoculation (DPI) or until euthanasia, whichever occurred first.
Serological tests
Serum samples from all 3 groups were tested for antibodies to A. marginale using the commercial cELISA Anaplasma antibody test kit. a Kit controls and test samples were tested in triplicate following the manufacturer’s instructions. The results of the test were considered valid when the negative control had a mean OD650nm range of 0.4–2.1 and the percent inhibition of the positive control was ≥30%. The percent inhibition was calculated as percent inhibition = 100 − [(ABS S/ABS N) ×100], where ABS is absorbance at 650 nm, S = sample, and N = negative control. Samples with percent inhibitions <30% and ≥30% were considered negative and positive, respectively.
The IFAT antigen blood smear slides were prepared from whole blood obtained by serially infecting 2 splenectomized calves with A. marginale as described previously 22 with some modifications. Blood from the positive control splenectomized calf (D) inoculated with a Manitoba isolate of A. marginale as described for the group 3 samples above was monitored daily by examination of blood films stained with Romanowsky stain variant. b At 42 DPI, once parasitemia reached approximately 38%, washed red blood cells from this animal were suspended in sterile phosphate buffered saline (PBS; 1:2 v/v), and a total volume of 87 ml of this suspension was inoculated intrajugularly into a second recipient splenectomized calf (I). Calf I had remained as a negative uninfected control for 42 days but was subsequently used as the recipient to produce the Anaplasma antigen slides for IFAT. When parasitemia in calf I reached 51% at 13 DPI, blood was collected for preparation of antigen blood smears, and the animal was euthanized. This procedure was followed to reduce the incubation period required to maximize parasitemia while minimizing concomitant antibody response and nonspecific background staining in the IFAT. Antigen blood smears were prepared as described previously. 22 Briefly, blood was centrifuged at 1,250 × g for 10 min to remove plasma and buffy coat. Erythrocytes were washed 3 times in PBS (pH 7.4); after washing, cells were suspended in PBS containing 1% bovine serum albumin (1:3 v/v). Smears were prepared, wrapped in aluminum foil, and stored at −70°C until used. In addition to the smears prepared with the Manitoba isolate, antigen blood smears prepared similarly with an A. marginale strain from Brazil were also used. To perform the IFAT, slides were thawed at room temperature and fixed in absolute methanol for 3 min. Sera were titrated at serial 2-fold dilutions from 1:80 to 1:640. Twenty-five microliters of diluted sera from positive or negative controls or test samples were added to squares delineated on the blood smear slides by a hydrophobic pen. c Slides were incubated at 37°C for 30 min. After incubation, slides were washed 3 times in PBS and then incubated for 30 min at 37°C with a fluorescein isothiocyanate (FITC)-labeled rabbit anti-bovine immunoglobulin G (IgG)-heavy and -light chains conjugate (FITC-anti-IgG-H+L) d diluted 1:600 in PBS containing 0.01% Evans blue stain. Slides were washed 3 times for 10 min each in PBS, drained, and then mounted with fluorescence Slow Fade reagent. e Slides were examined for FITC fluorescence by fluorescence microscopy using 40× or 100× objectives and incident light wavelength of 450–490 nm. f Slides were graded as negative (–) or positive (+) using control sera from uninfected and A. marginale–infected calves, respectively, as described below. A negative reaction gave a greenish or red counter staining, while a specific positive reaction was detected as green fluorescent bodies within the infected erythrocytes with fluorescence equal to or higher than the weak positive control.
Prior to testing of the samples in the current study, the IFAT was evaluated using bovine serum samples from an earlier A. marginale transmission study. 16 The sera were collected from splenectomized calves before (0 DPI) and after (12, 20, 26, 31, 35, 41, 51, 61, 71, 82, 121, 191, and 281 DPI) experimental infection with the St. Maries strain of A. marginale. Serum samples were diluted at 1:80, 1:160, and 1:320 dilutions in PBS, applied to squares delineated on slides at 25 µl/square, and then incubated with the FITC conjugate. Samples were graded as negative (–) or positive (+) after being examined by fluorescence microscopy. Negative (0 DPI) with no fluorescence, and positive (20 and 191 DPI) sera representing weak and strong positive fluorescence, respectively, were chosen as negative and positive controls on each slide of test samples in the current study. Also, these same samples were used as reference sera to assess the fitness of the antigen blood smears prepared using the Manitoba isolate and Brazil strain of A. marginale. Based on this, a 1:160 antibody titer was established as the cut-off value using antigen smears from either source.
Blood analysis
Fresh blood films stained with a Romanowsky stain variant b were prepared routinely and examined under oil immersion at 100× magnification for evidence of parasitemia in animals sampled in groups 1 and 3. DNA extraction, purification, and molecular analysis have been described previously. 8 Briefly, to confirm A. marginale infection and/or infection with other species of the Anaplasmataceae family in the animals sampled in the current study, nested PCR assays were performed on available blood or extracted DNA samples for animals in groups 1 and 3, and PCR-positive products were cloned and sequenced to verify the infection status of the animals (data not shown). For animals in group 2, confirmation of A. marginale infection by PCR analysis was reported previously. 11
Statistical analysis
Agreement between cELISA and IFAT was estimated by calculating the kappa value. Kappa values of ≥0.81, 0.61–0.80, 0.40–60, and ≤0.40 represent excellent, substantial, moderate to good, and slight to poor agreement, respectively. 29
Results
Serum samples collected from the 9 British Columbia cattle naturally infected with Ehrlichia sp. BOV2010 (group 1) were seropositive by cELISA and IFAT. However, microscopic examination of blood smears from these animals did not reveal any intra-erythrocytic bacteria. Serological testing of all samples from Manitoba cattle infected with A. marginale (group 2) also gave positive results by both cELISA and IFAT (Table 1). Serology results for all tested samples were in agreement with the results of molecular analysis.
When banked serum samples from calves experimentally infected with the St. Maries strain of A. marginale were tested using antigen blood smear slides from both Manitoba and Brazil sources, positive results were obtained as early as 20 DPI. Both antigen sources gave consistent IFAT results when used to test serum samples from all groups included in the present study.
The cELISA and IFAT results for serially collected samples from the inoculated calves in group 3 are shown in Table 2. In 2 Ehrlichia sp. BOV2010–inoculated calves from this group, antibodies were first detected at 28 and 33 DPI by cELISA, and reached maximum inhibition values at 33 DPI and 84 DPI for animal A and C, respectively (Fig. 1). The IFAT results supported those of the cELISA with seropositivity first detected at 33 and 42 DPI for animal A and C, respectively (Table 2). The other 3 Ehrlichia sp. BOV2010–inoculated calves (E–G) remained seronegative with both tests until their euthanasia at 28, 28, and 33 DPI, respectively. Animals E–G were euthanized early due to clinical disease of unknown etiology. Seroconversion in both A. marginale–positive control calves (D, H) was first detected at 42 DPI by both cELISA and IFAT. Blood smears from these animals revealed a few erythrocytes with A. marginale starting at 30 DPI for animal D and 28 DPI for animal H. Serum and blood samples from the negative control calf I were consistently negative for A. marginale and Ehrlichia sp. by cELISA, IFAT, and blood smear examination. Serum samples from calf C, inoculated with Ehrlichia sp. BOV2010 and calf D, inoculated with a Manitoba isolate of A. marginale showed positive staining patterns when tested by IFAT. Serum from the control calf I was IFAT negative (Fig. 2). A comparison of all results obtained using the 2 serologic assays is presented in Figure 3. The overall agreement between cELISA and IFAT was excellent with a kappa value of 0.941.
Results of competitive enzyme-linked immunosorbent assay (cELISA) and indirect fluorescent antibody test (IFAT) for Anaplasma marginale performed on serum samples serially collected from cattle experimentally infected with Ehrlichia sp. BOV2010 or A. marginale or from uninfected control.*

Molecular analysis results for 7 animals (A, C–G, I) were reported in a previous study. 8 Molecular analysis of samples from animal H was conducted during the current study. Animals A, C, E–G were infected with Ehrlichia sp. BOV2010. Animals D and H were infected with A. marginale. Animal I served as a control (uninfected) for 42 days. Subsequently this animal was used as a recipient of a Manitoba isolate of A. marginale from donor animal D to produce antigen blood slides for the IFAT and then euthanized 13 days after inoculation to prepare antigen blood slides. + = positive test; – = negative test; DPI = days post-inoculation; ND = not determined.
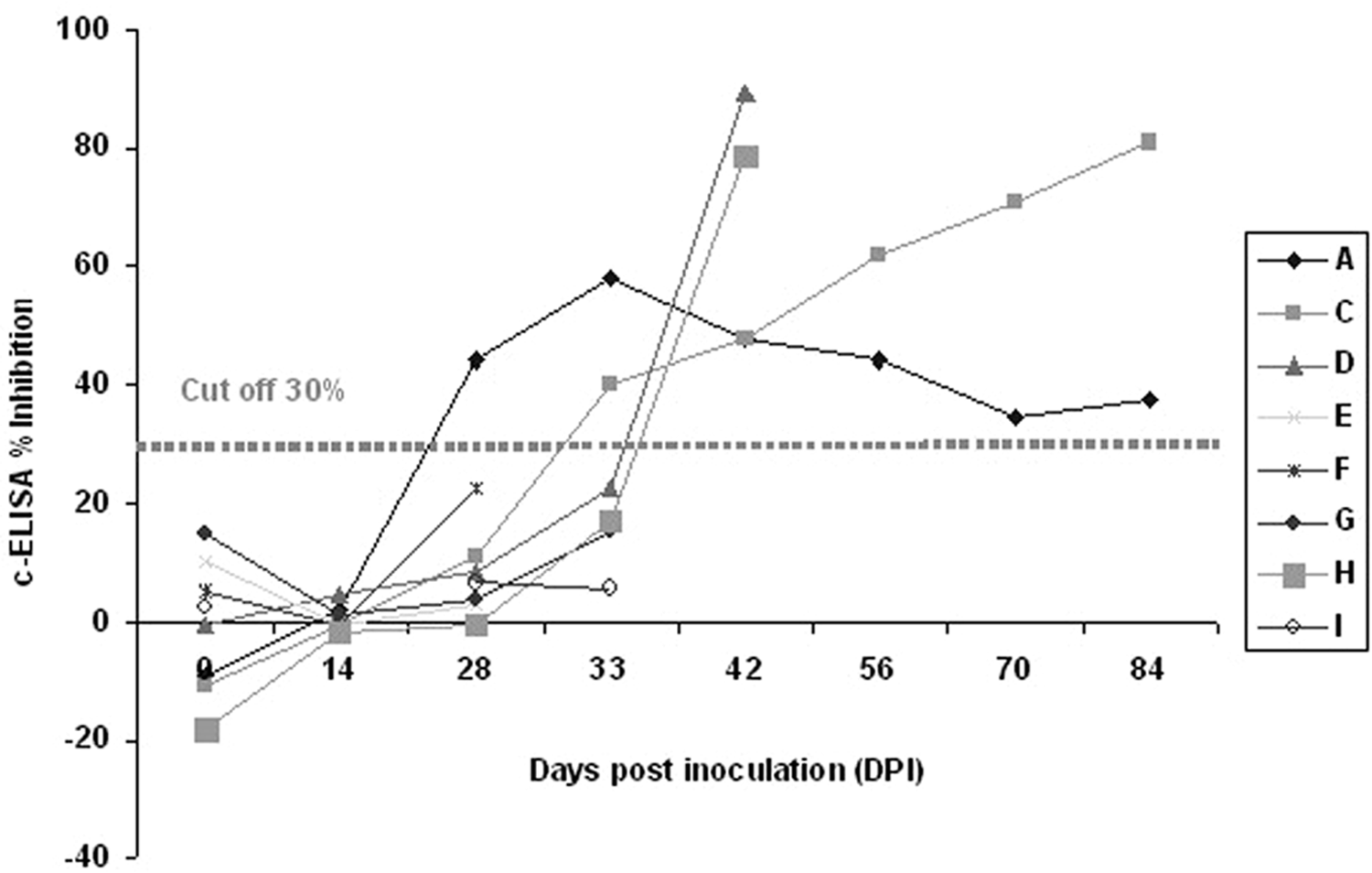
Competitive enzyme-linked immunosorbent assay (cELISA) percent inhibition mean values obtained for serum samples from splenectomized calves inoculated with blood from cattle naturally infected with Ehrlichia sp. BOV2010 (calves A, C, E–G) or with stabilates of Anaplasma marginale (calves D, H). Calf I was a noninfected negative control. Calves were euthanized at various times starting at 28 DPI.
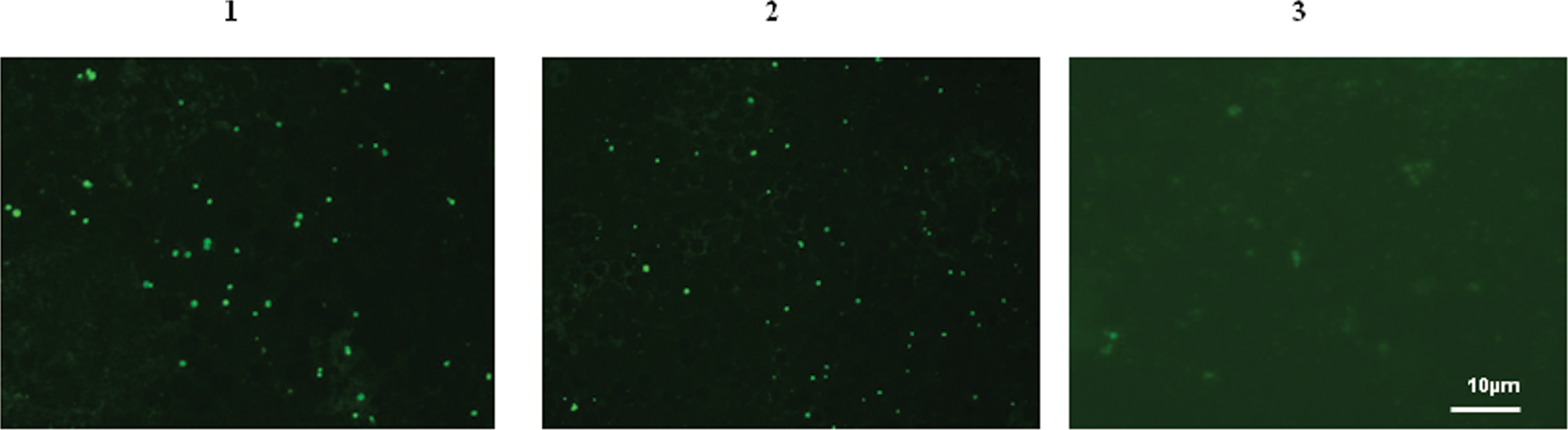
Photomicrograph of indirect fluorescent antibody test (IFAT) results for serum samples tested using antigen from a Manitoba isolate of Anaplasma marginale:

Comparison of competitive enzyme-linked immunosorbent assay (cELISA) and indirect fluorescent antibody test (IFAT) results for the detection of antibodies to Anaplasma and Ehrlichia sp. BOV2010. The dotted line represents the cut-off value for the cELISA. The cELISA was considered positive at percent inhibition ≥30%. Total number of serum samples tested = 76. IFAT+ and IFAT– indicate positive and negative results, respectively.
Discussion
A recent finding of Ehrlichia sp. BOV2010 in North American cattle has been confirmed by molecular and bioassay studies and is the first report of ehrlichiosis in cattle in North America. 8 The only other Ehrlichia species known to naturally infect cattle is E. ruminantium (formerly Cowdria ruminantium). 1 The present study provides evidence of serological cross-reactivity between Ehrlichia sp. BOV2010 and A. marginale using the commercially available cELISA test kit for Anaplasma and an IFAT using A. marginale antigen–coated slides.
The cELISA used in the current study is based on a mAb (ANAF16C1) that recognizes MSP5 in A. marginale, A. ovis, and A. centrale. 30 The present study demonstrates that cattle naturally or experimentally infected with Ehrlichia sp. BOV2010 8 were seropositive for A. marginale by both cELISA and IFAT (Tables 1, 2). It appears that the epitope recognized by this mAb may also be expressed in Ehrlichia sp. BOV2010. However, it is possible that the cross-reactivity is not restricted to MSP5 but may include epitopes of other proteins of Ehrlichia sp. BOV2010 and A. marginale as suggested by the IFAT results, due to the presence of homologous families of immunodominant outer membrane proteins that are expressed in several Ehrlichia and Anaplasma species. 1
Cattle in regions where A. marginale is endemic could be exposed to infections with other rickettsial pathogens that may induce antibodies cross-reactive with A. marginale proteins, leading to false-positive serological test results. This has been demonstrated for the MAP1 diagnostic antigen of E. ruminantium whereby serological cross-reactions between E. ruminantium and Ehrlichia canis, Ehrlichia chaffeensis, A. bovis, and A. phagocytophilum have been reported using IFAT and indirect ELISA.12,13 17,19,28 Similarly, serological cross-reactions between E. chaffeensis and Ehrlichia ewingii infections in human beings have been reported using IFAT.3,23 However, not all E. ewingii–infected dogs or human beings develop antibodies cross-reactive to E. chaffeensis.20,23 Ehrlichia canis, which appears to be most closely related to Ehrlichia sp. BOV2010 in cattle, 8 also serologically cross-reacts with E. chaffeensis.2,24 Thus, Ehrlichia sp. BOV2010 may also confound serological testing for E. ruminantium as well as A. marginale in cattle.
The commercial cELISA test kit is widely accepted as the most reliable test for screening cattle for A. marginale infection. The IFAT using bovine blood films with A. marginale as a source of antigen, although less preferred due to lower throughput of samples and nonspecific fluorescence, is also recognized as an acceptable test for diagnosing anaplasmosis in cattle. 31 The cross-reactivity reported in the present study has important implications for the use of these tests in domestic and wild animals that are susceptible to infection with both Anaplasma and Ehrlichia, such as cattle, and in particular in areas where both infections may coexist. In such cases, results obtained by cELISA and IFAT should be interpreted carefully. Since a seropositive animal could be infected with A. marginale, Ehrlichia spp., A. centrale, or A. ovis, differentiation should be based on further testing by PCR analysis using the appropriate specific primers for each pathogen followed by sequencing of amplified products.
Only 2 out of 76 serum samples tested produced negative IFAT and positive cELISA results (Fig. 3). These disparate results were obtained for sera from calves A and C, which were experimentally infected with Ehrlichia sp. BOV2010. The IFAT first detected animals A and C as positive at 33 and 42 DPI, respectively, whereas the cELISA detected them as positive at 28 and 33 DPI, respectively. Differences in detectable seroconversion between the 2 test methods and between animals could be attributed to variations in the sensitivity of the assays or in the immune response of individual animals. However, the overall agreement between the 2 tests was excellent as indicated by kappa value = 0.941. Experimental animals E and G, similarly infected with Ehrlichia sp. BOV2010, remained seronegative by both tests, presumably because animals E and G were euthanized at 28 and 33 DPI, respectively and had not yet generated a detectable immune response. Molecular analysis of blood from experimentally infected cattle confirmed infection with Ehrlichia in all animals except 1 animal (F) 8 euthanized at 28 DPI that also remained seronegative by both cELISA and IFAT.
Using the commercial cELISA test kit, previous studies have used cut-off inhibition values of both 30% and 42% for cELISA in serological surveys of A. marginale in Canadian cattle.4,27 In the present study, a cut-off value of 30% inhibition was used as recommended by the manufacturer. a Seven serum samples from Ehrlichia-infected animals, with cELISA values, which would be interpreted as negative using a 42% instead of 30% cut-off inhibition, were positive by both IFAT and PCR. Thus, when test results of cELISA interpreted using a 42% inhibition cut-off were compared with results interpreted with the recommended 30% inhibition cut-off, the relative sensitivity of the test decreased to 85.7% instead of 96% and the kappa value from 0.94 to 0.80. Although the sample size in the current study was small (76 tested sera), results are consistent with performance of the cELISA in previous anaplasmosis serological studies reported with large populations.4,27 The cross-reactivity between Ehrlichia sp. BOV2010 and A. marginale demonstrated in the current study indicates that increasing the inhibition cut-off value of cELISA would significantly decrease the sensitivity of this test for both infections.
The present study indicates that both cELISA and IFAT can detect antibodies to Ehrlichia sp. BOV2010 and A. marginale infections in cattle, but neither assay can differentiate between these infections. Diagnostic specificity of serological tests for Ehrlichia might be improved by using cell culture–derived Ehrlichia sp. BOV2010 antigens for serological testing. Such a step would entail preparation of mAbs against specific surface proteins from Ehrlichia sp. BOV2010 to test antigen blood film slides using IFAT or ELISA plates coated with highly specific proteins. A serological assay specific for Ehrlichia could then be used to differentiate between Anaplasma and Ehrlichia.
The serologic cross-reactivity between A. marginale and Ehrlichia sp. BOV2010 reported in the current study limits the diagnostic accuracy of the cELISA and IFAT for A. marginale in cattle. Therefore, positive results derived from these assays should be interpreted with caution, and whenever possible confirmed by molecular analysis.
Footnotes
Acknowledgements
The authors thank all the CFIA employees who provided assistance directly or indirectly in support of this study. Special thanks to Dr. Betty Golsteyn-Thomas and staff at the Lethbridge Laboratory for providing blood and serum samples from field animals. The authors are greatly indebted to the staff of the Centre for Food-borne and Animal Parasitology for the excellent technical support, especially Jane Benjamin, Robyn Ostrander, and Yunxiu Dai. Dr. Murray Lankester provided helpful laboratory and organizational support. Professor Odilone Vidotto, Department of Preventive Veterinary Medicine, State University of Londrina-Brazil, provided A. marginale antigen blood smear slides (Brazil strain) for IFAT in addition to advice on preparing these slides in our laboratory. The animal care and blood collection were provided by staff at the Animal Care Unit, Western College of Veterinary Medicine, University of Saskatchewan. Special thanks to the various CFIA employees who reviewed the manuscript and provided helpful comments.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Financial and in-kind support for this research was provided by the Canadian Food Inspection Agency.
