Abstract
Sixteen years of adult cattle submissions to the California Animal Health and Food Safety Laboratory System were examined and data captured from cases with anaerobic cultures of intestinal content. Analysis was performed to determine if there were statistical differences between case submission types (nonbloody intestinal content [129 cases], bloody intestinal content [134 cases], and jejunal hematoma [JH; 51 cases]) for the presence of Clostridium perfringens (314 cases), C. perfringens toxinotypes (35 cases), and C. perfringens toxins (51 cases) in the content. Across submission types, significant differences were found in the isolation of C. perfringens between different specimen types (live cow, dead cow, or tissue from a field necropsy) with field samples being the most likely to have C. perfringens detected and live animals the least likely (P = 0.001). In cases of JH, detection of C. perfringens by enzyme-linked immunosorbent assay was more likely when a live or dead animal was submitted (P = 0.023) or when a live animal was submitted (P = 0.019) compared with submission of field necropsy tissues. These differences were not observed when cultures were performed to detect C. perfringens in cases of JH. There were no statistical differences between submission types with regard to any other variables evaluated. Detailed histologic examination of 21 cases of JH suggested disturbance of normal vascular or lymphatic function as the underlying problem in this entity.
Introduction
Jejunal hematoma (JH), also known as hemorrhagic bowel syndrome (HBS) or intestinal hematoma, is an entity that occurs primarily in adult cattle with most clinical cases reported in dairy breeds.3,8 The clinical syndrome is one of sudden death often with clotted blood found in the feces. A small percentage of affected animals may present with abdominal pain prior to death.3,8 On postmortem examination, it is clear that the animals have bled into the intestinal lumen with most cases having 1 or more short segments of small intestine with intramural hematomas. 1 The underlying cause for this entity is not known but the most commonly discussed potential cause is infection with Clostridium perfringens, type A isolates that contain the beta2 toxin gene (cpb2). 7
The objective of the current study was to determine if there were statistical differences between adult cattle diagnosed with JH, cattle with blood in the intestine for other reasons, and cattle without blood in the intestinal lumen in regard to the organisms isolated on anaerobic cultures of the intestine, C. perfringens toxins in intestinal content detected by enzyme-linked immunosorbent assay (ELISA), or genotype of the C. perfringens isolates. Analysis also included infection status for Bovine viral diarrhea virus (BVDV) and Salmonella sp. and liver copper levels. A second goal was to perform a detailed histologic examination of a large number of cases of JH in an effort to determine if the pattern of histologic lesions was consistent with a clostridial infection or if some other pathogenesis was suggested.
Materials and methods
Retrospective data collection
The laboratory information management system for the California Animal Health and Food Safety Laboratory System (CAHFS) was queried for cases between January 1, 1996 and December 31, 2011 in which adult bovine animals (42 live, 97 dead on submission) or tissue from field necropsies (175 cases) were submitted, and anaerobic cultures were performed on some portion of intestine. Data, including history, breed, age, and diagnostic test results, for the 314 individual cases were collated in a spreadsheet. a The breakdown of case submission type (JH, non-JH with bloody content, or nonbloody content) compared to specimen type (live animal, whole carcass, tissue from a field necropsy) is shown in Table 1.
Cross-tabulation of the type of specimen received and the case submission type (gross postmortem findings) in 314 adult bovine cases received by the California Animal Health and Food Safety Laboratory System between January 1996 and December 2011 with anaerobic cultures performed on intestinal samples.*
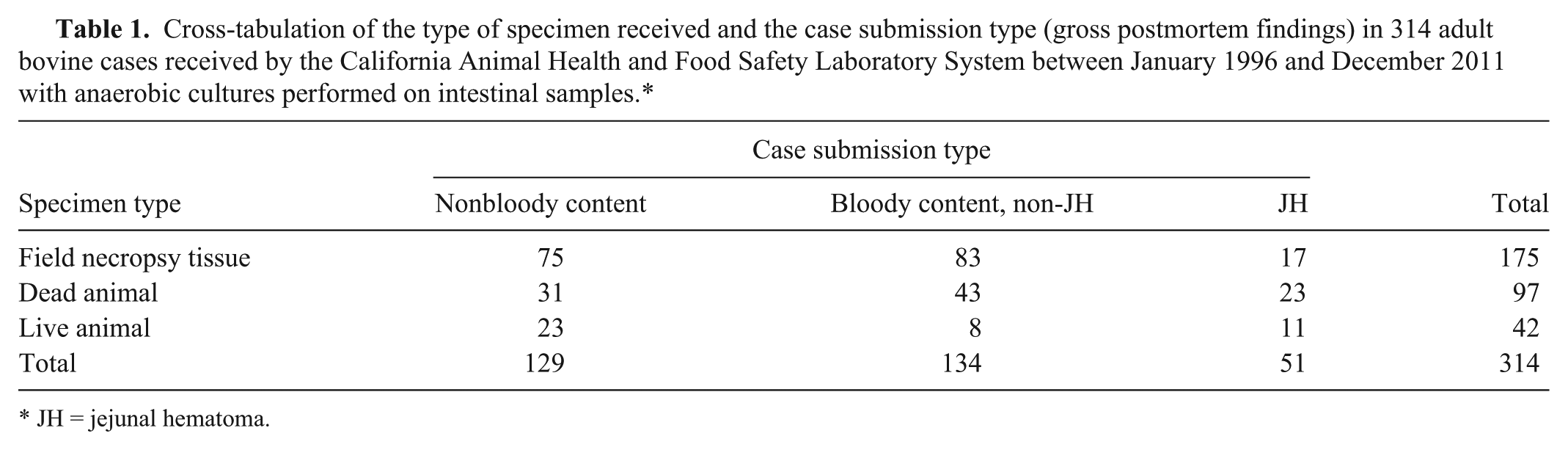
JH = jejunal hematoma.
Anaerobic cultures
Intestinal content was plated to prereduced anaerobically sterilized (PRAS) Brucella agar, phelylethyl alcohol (PEA) agar, and an egg yolk agar (EYA) plate and incubated at 35–37°C under anaerobic conditions. Plates were examined for growth at 1–3 days for the presence of colonies with a double zone of hemolysis on Brucella agar or lecithinase positive on EYA. One to 3 compatible colonies were subcultured to Brucella agar for purity and incubated anaerobically for 18–48 hr and then examined for purity. Once a pure culture was obtained, each colony type was subcultured to a chocolate agar plate and incubated at 35–37°C in 5–10% CO2 and examined at 18–24 and 42–48 hr to check for aerotolerance. If no growth occurred on the chocolate plate, the isolate was worked up as an anaerobic organism. The double zone hemolysis isolates were Gram stained, and those that were large Gram-positive rods with or without spores were tested further. Clostridium perfringens colonies had a double zone of hemolysis on the PRAS Brucella plate and were lecithinase positive and lipase negative on EYA with a reverse Christie, Atkins, Munch-Peterson (CAMP) reaction when tested against Streptococcus agalactiae.
Genotyping
Clostridium perfringens genotyping was performed on 35 isolates by the laboratory of J. Glenn Songer at University of Arizona. Isolates were typed by a multiplex polymerase chain reaction (mPCR) to amplify segments specific for the genes encoding alpha toxin (cpa), beta toxin (cpb), epsilon toxin (etx), iota toxin (ibp), enterotoxin (cpe), and beta2 toxin (cpb2), as previously described. Briefly, bacteria were grown on brain–heart infusion agar plates, b incubated anaerobically overnight at 37°C, and then processed for mPCR analysis using colony lysates as templates. The mPCR products were then separated in 2% agarose gels, stained with ethidium bromide, and examined by ultraviolet transillumination. 6
Clostridium perfringens toxin ELISA
Samples of small and/or large intestinal contents were tested for C. perfringens alpha, beta, and epsilon toxins (CPA, CPB, and ETX, respectively) and presence of C. perfringens organism via a qualitative commercial capture ELISA kit, c following the manufacturer’s instructions, in 51 of the cases. Briefly, the test used 96-well plates sensitized by specific monoclonal antibodies for C. perfringens, CPA, CPB, or ETX. Samples were added to wells, and plates were incubated for 60 min at room temperature, followed by washing and incubation for 60 min with peroxidase-labeled anti–C. perfringens, CPA, CPB, or ETX polyclonal antibodies. Plates were washed again, and a mixture of chromogen substrate (hydrogen peroxide and tetramethyl benzidine) was added. The enzymatic reaction was stopped by acidification with phosphoric acid. Optical densities were read using an ELISA reader with a 450-nm filter. Purified CPA, CPB, ETX, or C. perfringens were used in positive control wells, while the agent and toxins were replaced by buffer in negative control wells. Results were calculated according to the manufacturer’s instructions.
Bovine viral diarrhea virus testing
Testing for the presence of BVDV was performed on 115 of the cases. A variety of methods were used, including immunoperoxidase staining of formalin fixed tissue, fluorescent antibody staining of frozen tissue, and polymerase chain reaction performed on unfixed tissue.2,4,11,13
Salmonella cultures
Cultures of intestinal content for Salmonella sp. were performed on 288 of the cases. Approximately 1 g of intestinal content was added to 10 ml of selenite broth or tetrathionate broth and incubated aerobically at 35–39°C for 18–24 hr. Using a sterile cotton tipped swab or inoculating loop the broth was inoculated onto 1 minimally selective media (Hektoen agar, MacConkey agar, xylose–lysine–deoxycholate or Brilliant Green) and 1 highly selective media (Brilliant Green agar plate with novobiocin (20 µg/ml), xylose–lysine–deoxycholate [XLD] with Tergitol 4 [XLT-4] agar plate, or XLD with novobiocin]. The plates were incubated aerobically at 35–39°C and examined at 18–24 and 42–48 hr. Colonies with morphologic character compatible with Salmonella were subcultured and screened initially using triple sugar iron agar (TSI) and lysine iron agar (LIA) or excluded if urea positive. Compatible colonies were tested using O-antigen poly and group specific antisera. If agglutination occurred and/or the TSI and LIA reactions were compatible, isolates were identified as Salmonella suspects. Confirmatory biochemical testing was performed to assess urease activity, indole, motility, ornithine decarboxylase, citrate utilization, and ONPG. Serotyping was performed on any confirmed Salmonella isolates.
Liver copper levels
Testing for liver heavy metals, including copper levels, was performed on 166 of the cases. The livers were digested with nitric acid at 180°C and subsequently analyzed by inductively coupled argon plasma emission spectrometer.d,12 Accuracy of ICP results for these elements was measured by analyzing standard reference materials (SRMs) and fortified bovine liver tissue. The SRMs used were dogfish liver e and lobster hepatopancreas. f Data were accepted if analyzed standard reference material values were within 2 standard deviations of the certified reference values and spike recoveries were within an 80–120% recovery range. The limit of quantitation for copper on a wet weight basis was 0.1 ppm.
Statistical analysis
Fisher’s exact test was used to compare the proportion of each case submission type (nonbloody intestinal content, JH, and non-JH bloody intestinal content) that tested positive for C. perfringens by culture or ELISA, the proportion of C. perfringens isolates that tested positive for each of the toxin genes, and, similarly, the proportion of intestinal content samples that tested positive for CPA, CPB, ETX by ELISA. The proportion of positive test results for Salmonella spp. and BVDV was also compared between submission types. In addition, copper status was compared (below normal <25 ppm; normal 25–100 ppm; above normal >100 ppm) between submission types to test the hypothesis that copper deficiency and secondary collagen abnormalities might contribute to a bleeding tendency. In addition, the comparison of the proportion of each case submission type that tested positive for C. perfringens by culture or ELISA was stratified by specimen type (live animal, whole carcass, tissue from a field necropsy). All comparisons were performed using commercial software, g and a P value of <0.05 was considered significant.
Histology
Paraffin-embedded blocks from 21 cases of JH were retrieved from the CAHFS archives. Sections that included the interface between the hematoma and nonhematoma portions of the intestine were cut at 5 µm and stained using routine hematoxylin and eosin (HE) methods as well as MacCallum–Goodpasture Gram stain. Archived slides from 30 of the non-JH, adult cattle cases submitted to the Tulare CAHFS laboratory were used for comparison. All of the cases examined for the detailed histologic description were from carcasses or live animals submitted to the laboratories with the exception of 1 case of JH, which contained tissues submitted from a field necropsy.
Results
Retrospective case analysis
Clostridium perfringens was isolated from 209 cases, 79 cases were negative for C. perfringens by culture and 26 cases had the cultures overgrown with Proteus and were removed from the data analysis. For the overall dataset there were no statistical differences detected between the 3 types of case submissions (nonbloody intestinal content, JH, and non-JH bloody intestinal content) with regard to isolation or toxinotypes of C. perfringens, detection of C. perfringens or its toxins by ELISA, isolation of Salmonella, detection of BVDV, or copper status (Table 2). However, C. perfringens was statistically more likely to be found in cases of JH than in cases of non-JH bloody intestinal content or nonbloody intestinal content when ELISA testing was performed for detection of C. perfringens on cows presented alive and euthanized (P = 0.019; Table 3) or on intestinal content from whole animal (live or dead; P = 0.023; Table 3) submissions. This was not true when the analysis was limited to cases in which dead animals (P = 0.149) or tissues from field necropsies (P = 0.286) were submitted (Table 3). When the same comparison was performed between submission types based on the results of anaerobic cultures no significant differences were found (Table 4). There was a statistically significant relationship (P = 0.001) between the isolation of C. perfringens and the type of specimen (field necropsy tissue, dead animal, and live animal) submitted to the laboratory (Table 5) with C. perfringens most likely to be isolated from field necropsy tissue submissions and least likely from live animal submissions. There were no statistical differences between the 3 specimen types with regard to toxinotypes of C. perfringens and C. perfringens toxin detection (Table 5). Fresh date was available for 21 of the cases of JH and on average the animals were 120 days in milk (range: 30–539; median: 100).
Summary of diagnostic test results for samples from adult cattle with different case submission types.*
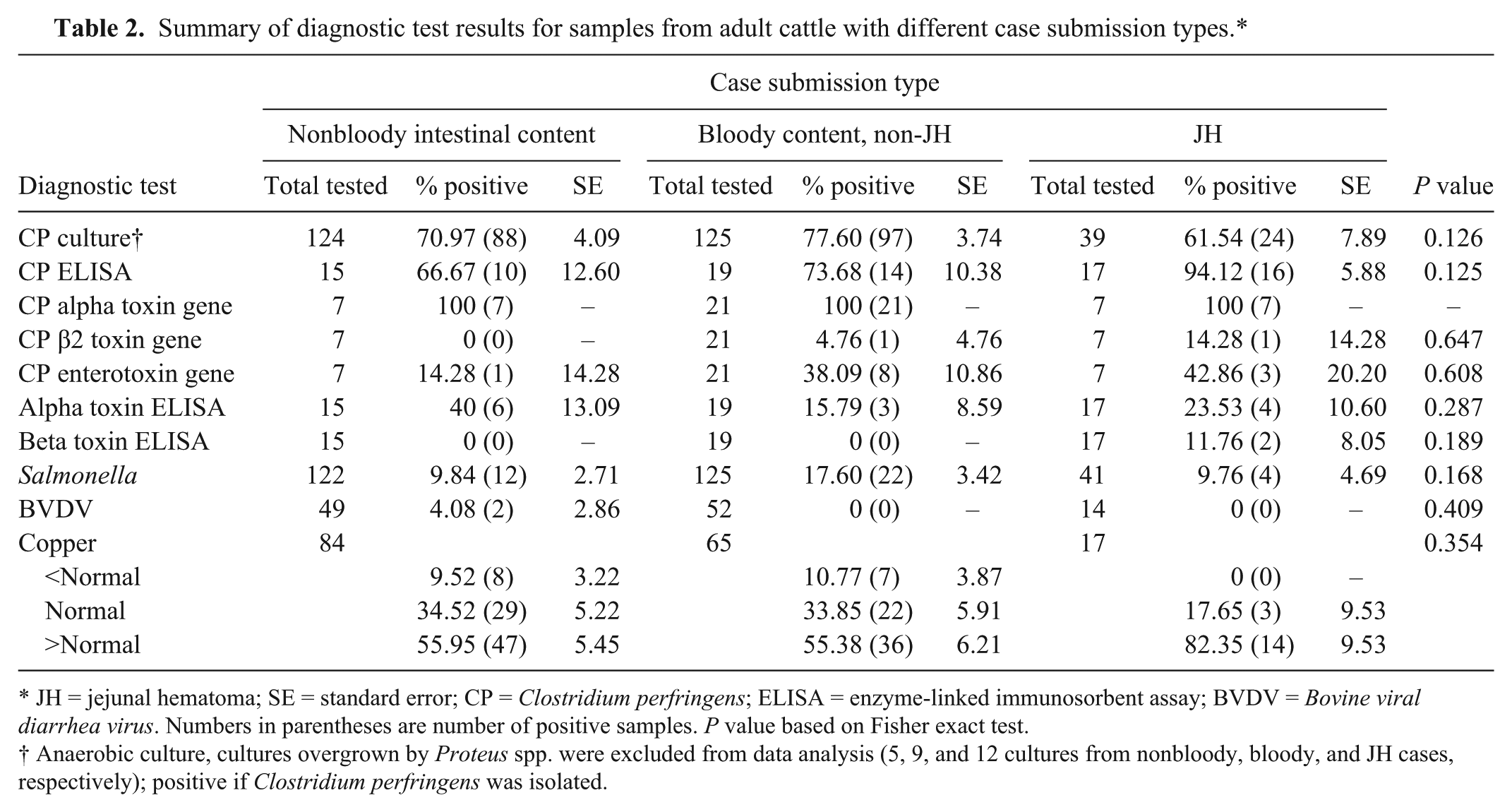
JH = jejunal hematoma; SE = standard error; CP = Clostridium perfringens; ELISA = enzyme-linked immunosorbent assay; BVDV = Bovine viral diarrhea virus. Numbers in parentheses are number of positive samples. P value based on Fisher exact test.
Anaerobic culture, cultures overgrown by Proteus spp. were excluded from data analysis (5, 9, and 12 cultures from nonbloody, bloody, and JH cases, respectively); positive if Clostridium perfringens was isolated.
Comparison of results of Clostridium perfringens enzyme-linked immunosorbent assay testing of intestinal content between case submission types for each of 4 different specimen types.*
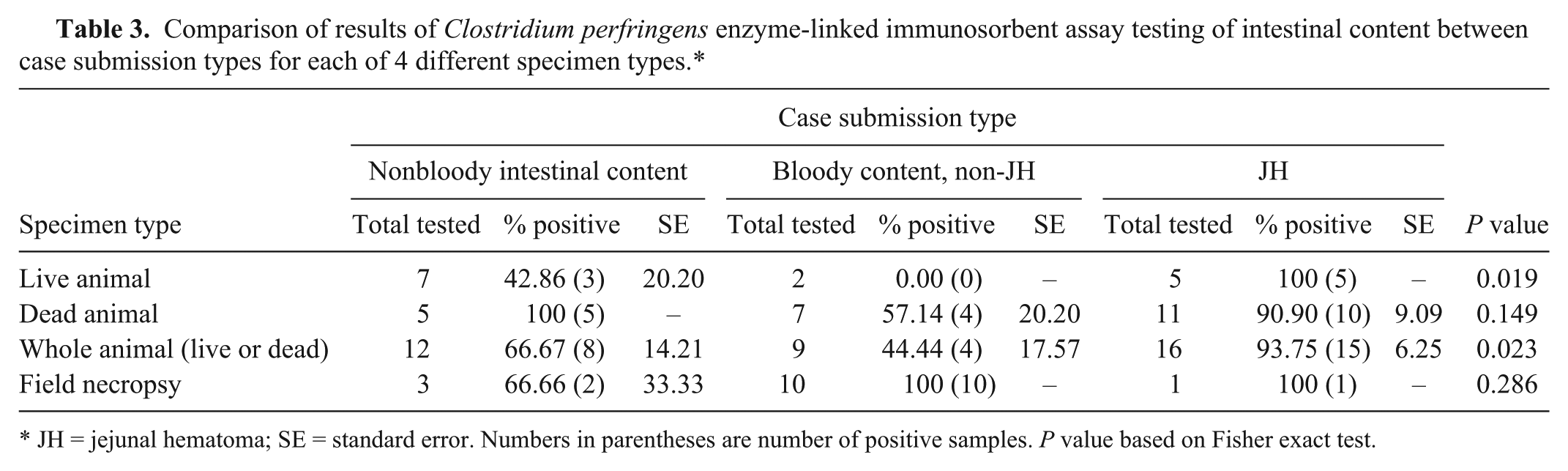
JH = jejunal hematoma; SE = standard error. Numbers in parentheses are number of positive samples. P value based on Fisher exact test.
Comparison of results of anaerobic culture of intestinal content for Clostridium perfringens between case submission types for each of 4 different specimen types.*
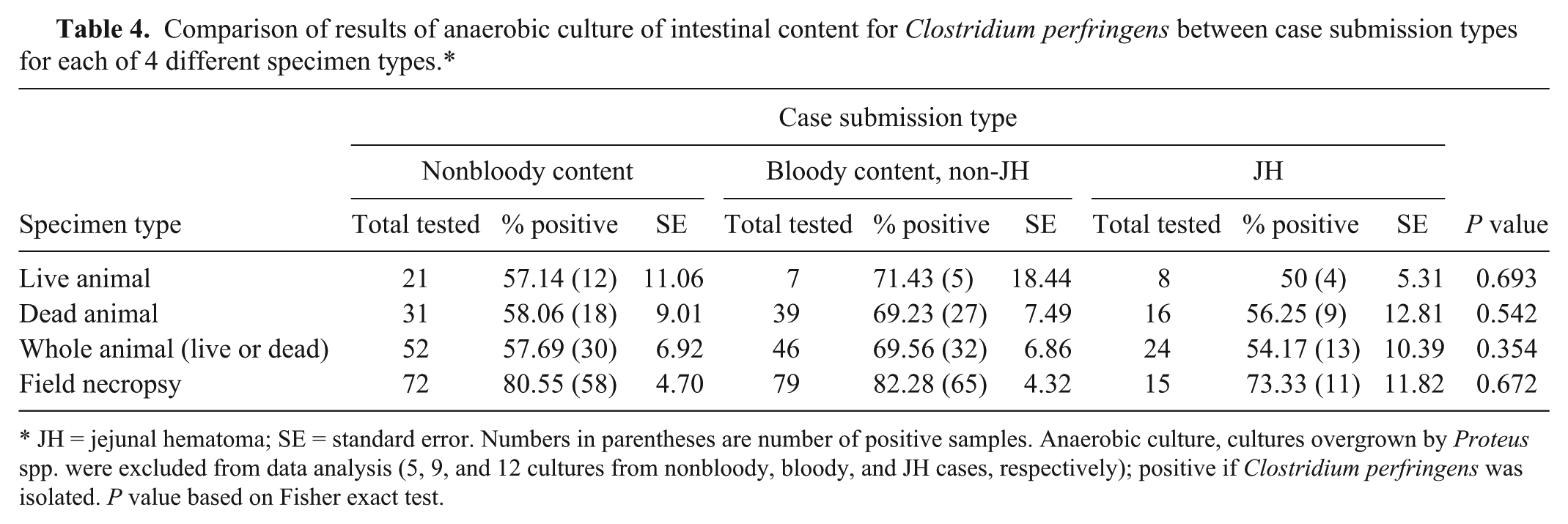
JH = jejunal hematoma; SE = standard error. Numbers in parentheses are number of positive samples. Anaerobic culture, cultures overgrown by Proteus spp. were excluded from data analysis (5, 9, and 12 cultures from nonbloody, bloody, and JH cases, respectively); positive if Clostridium perfringens was isolated. P value based on Fisher exact test.
Summary of diagnostic test results for submissions from adult cattle with different specimen types.
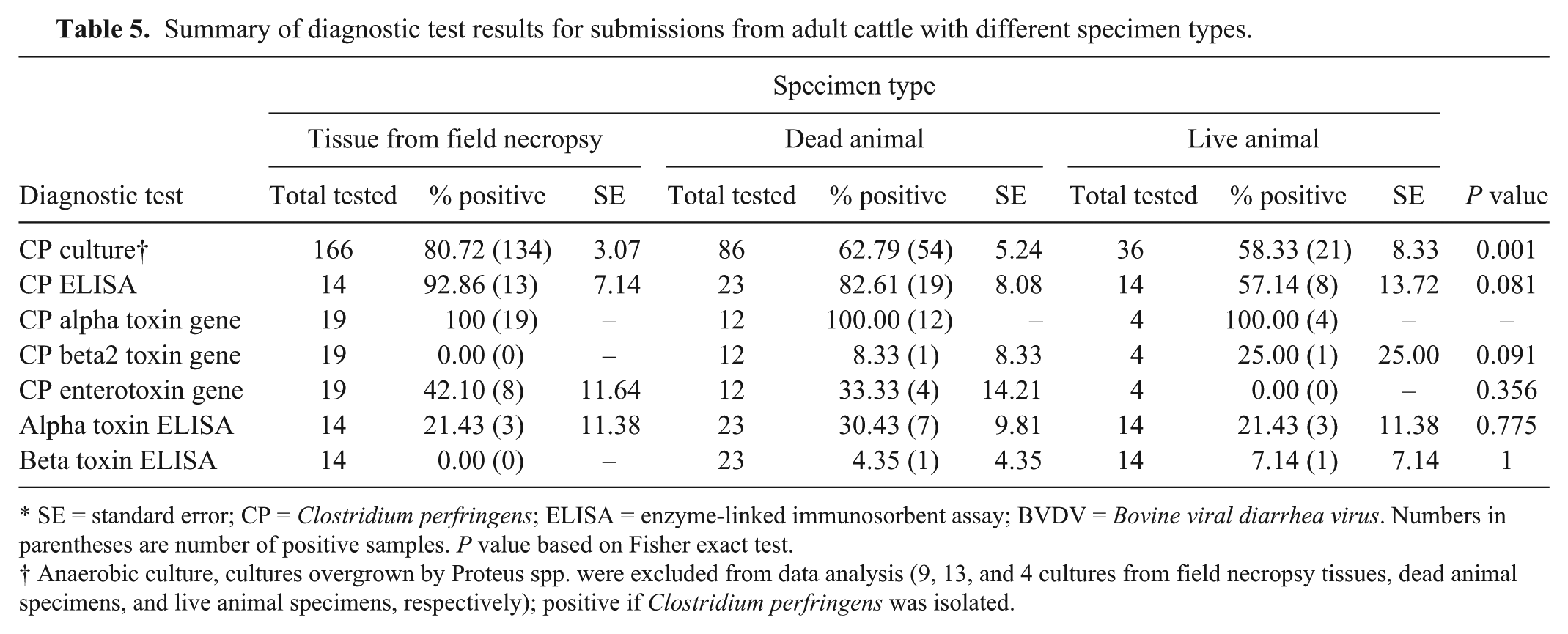
SE = standard error; CP = Clostridium perfringens; ELISA = enzyme-linked immunosorbent assay; BVDV = Bovine viral diarrhea virus. Numbers in parentheses are number of positive samples. P value based on Fisher exact test.
Anaerobic culture, cultures overgrown by Proteus spp. were excluded from data analysis (9, 13, and 4 cultures from field necropsy tissues, dead animal specimens, and live animal specimens, respectively); positive if Clostridium perfringens was isolated.
Histology
On HE-stained sections of both JH and non-JH cases, the nonhematoma portions of the lamina propria in all animals had moderate to large numbers of mixed lymphocytes and plasma cells which is considered within normal limits for the population of animals sampled. Occasionally, there were small numbers of neutrophils in the lamina propria and small amounts of cellular debris on the mucosal surface. Villi were occasionally shortened but were more often of normal length. The mucosal epithelium did not have adherent bacteria on Gram stains but there were occasionally small numbers of mixed bacteria within the superficial debris. While Gram-positive large bacterial rods were often present, they did not predominate in the population of bacteria seen. The interface region between the hematoma and nonhematoma portions of the tissue in JH cases (Fig. 1) typically had an abrupt elevation of the mucosa and the muscularis mucosa was often split with 1 portion staying with the submucosa and the other remaining adherent to the lamina propria, lifting off with the mucosa. The elevated mucosa was variably necrotic or autolytic with some cases only having mild necrosis of the surface epithelium while others had complete necrosis of the mucosa and only a pale eosinophilic ghost of the tissue remained. Often, the elevated mucosa had moderate to large numbers of large, Gram-positive, bacterial rods as part of a mixed bacterial population that was primarily on the luminal surface. The percentage of Gram-positive rods increased with degree of tissue degeneration from either necrosis or autolysis. Both the intact and the elevated mucosa in many of the cases of JH had moderately to markedly dilated villus lacteals, which were either empty or that contained abundant erythrocytes and/or pale eosinophilic somewhat hyalinized material, presumably lymphatic fluid. In contrast, the non-JH cases often had only slightly dilated lacteals and invariably these were empty. In small numbers of the JH cases, there were small numbers of hemosiderophages in the lamina propria (Fig. 1). The hematoma itself consisted of blood with small to moderate numbers of mixed bacteria, including large Gram-positive rods and the number of organisms varied between sections of the affected intestine. The portion of the sections of JH with the elevated mucosa or where the mucosa had sloughed typically had mild to moderate edema of the submucosa with very small to small numbers of loosely arranged neutrophils and occasionally, small foci of submucosal hemorrhage. Submucosal blood vessels in these areas were usually within normal limits but a small percentage of cases had mild to moderate vasculopathy with hyaline change of the vessel wall, mild pleocellular perivascular and subintimal inflammatory infiltrates, and plump lining endothelium (Fig. 2). In cases in which the tissues were more autolyzed, there were small numbers of large Gram-positive bacterial rods scattered within the submucosa, muscular layers, and the subserosa. Three of the cases had mild peritonitis with fibrin and neutrophils on the serosal surface.
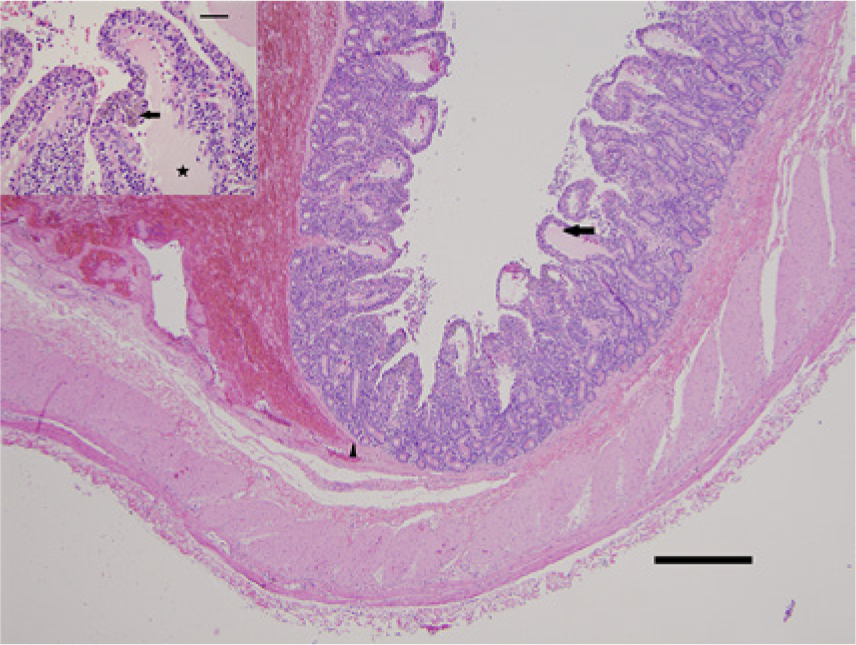
Jejunum; Holstein cow (1) with jejunal hematoma. Low-magnification photomicrograph at margin the hematoma with elevation of the overlying mucosa and an area with intact mucosa. Note dilated villus lacteals (arrow) and splitting of the muscularis mucosa (arrowhead). Hematoxylin and eosin (HE). Bar = 500 µm. Inset: markedly dilated villus lacteals (star) in an area of intact mucosa with an aggregate of hemosiderin-laden macrophages (arrow) within the adjacent interstitium of the villus. HE. Bar = 50 µm.
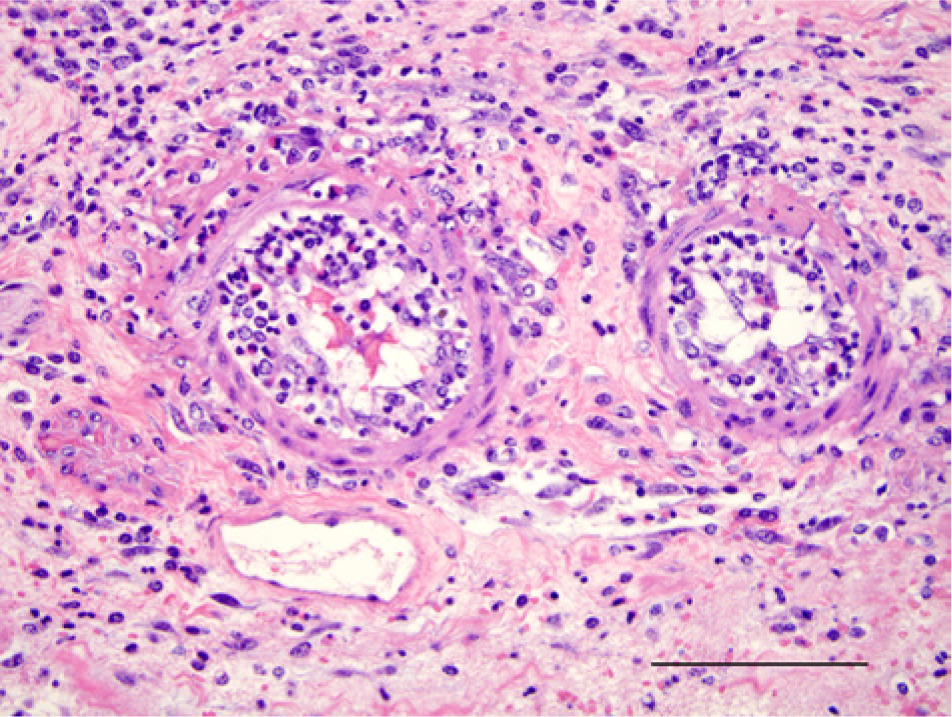
Jejunum; Holstein cow (3) with jejunal hematoma. Submucosal vessels with vasculitis characterized by hyaline change, mixed adventitial and subendothelial inflammation, and plump endothelium in an area of intestinal submucosa with an overlying hematoma (not shown in field). Hematoxylin and eosin. Bar = 100 µm.
Discussion
Hemorrhagic bowel syndrome is a moderately well-described entity,1,7,8 which has significant overlap with the cases described herein. In the current report, the term “jejunal hematoma” is used in preference to HBS because it is believed to more accurately describe the gross and histologic lesions and because it is a more specific term. Previous reports of HBS have suggested an association between the condition and the presence of C. perfringens type A 1 often with isolates containing the gene encoding beta2 toxin (cpb2).7–9 However, attempts to experimentally induce JH by abomasal or jejunal inoculation of healthy cows with cpb2-positive C. perfringens type A were unrewarding. 10 An extensive study of field isolates found that 35.1% of isolates of C. perfringens genotype A in multiple animal species and 12.8% of the isolates from cattle were positive for cpb2. The percentage was higher (47.3%) in cattle with enteritis or abomasitis but only 50% bovine C. perfringens type A isolates positive for cpb2 expressed beta2 toxin in culture. 6 Clostridium perfringens can be found in clinically normal cattle and in a previous study was found in 26 of 30 normal cattle submitted for slaughter (Uzal FA, Blanchard P, Songer G, et al.: 2003, Studies on the so-called “clostridial enteritis” of cattle. In: Proceedings of the 46th Annual Conference of the American Association of Veterinary Laboratory Diagnosticians). In the study reported herein, a statistically significant relationship was not found in the overall dataset between the isolation of C. perfringens, C. perfringens type A, or the cpb2 gene and JH. Also, a statistical relationship was not found between JH and the isolation of C. perfringens in cows submitted alive suggesting that C. perfringens does not play a role in JH. However, the statistically significant relationship between the detection of C. perfringens by ELISA in samples from live cows and JH suggests otherwise. These contrasting findings may be a result of differences in the sensitivity of the 2 methods, ELISA versus culture, in detecting C. perfringens in intestinal content from a live cow. The studies support observations that C. perfringensis a normal inhabitant of the bovine intestine and suggest that this organism proliferates after death regardless of the cause of death. The finding that C. perfringens is likely to undergo postmortem proliferation, something that has not previously reported, suggests that further studies of the role of C. perfringens in JH must rely on cases of live or very recently dead animals in order to minimize the overgrowth of C. perfringens. The cpb2 gene was detected in only 2 of the 35 isolates tested, 1 from an animal with JH and 1 from an animal with bloody content but not JH. There were also 6 other cases of JH in which the cpb2 was not detected in the C. perfringens isolated from the intestine and this suggests this toxin is not necessary to cause JH. The possibility that the CPB2 toxin can be easily destroyed in the intestinal lumen during the postmortem period also suggests that studies of JH should involve live or recently dead animals if the role presence of this toxin is to be investigated. Histologic examination of cases of JH found that the elevated mucosa that overlay the intramural hematoma and, to a lesser extent, the blood within the hematoma itself were the most common locations to find moderate or large numbers of large, Gram-positive bacterial rods consistent with C. perfringens. It was rare to find such organisms or significant inflammation within the more viable submucosal tissue or in the intact lamina propria. These findings suggest that C. perfringens proliferates in the presence of the necrotic or autolyzed tissue and extravascular blood. This was supported when comparisons were made between type of sample submitted (field necropsy tissue, dead animal, live animal) as a proxy measure for degree of autolysis (and presumed availability of free nutrients in the intestinal lumen) and the likelihood of detecting C. perfringens. There was a statistically significant association between a submission of tissues from a field necropsy and the isolation of C. perfringens while it was less likely to isolate the organism in samples from a live cow. In the set of cases reported herein, the animals with JH averaged 120 days in milk (median 100 days in milk) and this is in agreement with a previous study, 2 which found a median of 104 days in milk prior to onset of clinical signs in cases of HBS. In the current study, there was no statistical significance between JH, low liver copper, isolation of Salmonella, or detection of BVDV, indicating that these are unlikely to play a role in the pathogenesis of JH. The histologic description of JH provided herein is in contrast to previous brief descriptions of HBS1,7,8 in which the morphologic diagnosis was typically hemorrhagic enteritis or jejunitis indicating a more significant degree of inflammation in the lesions. In addition, the histologic lesions seen in cases of JH were not typical of those seen in more well-characterized cases of clostridial enteropathies such as hemorrhagic canine gastroenteritis or bovine necrohemorrhagic enteritis produced by C. perfringens type C in calves in which there is acute hemorrhagic enteritis with mucosal necrosis associated with numerous Gram-positive bacilli with morphology compatible with C. perfringens. 5 The mucosal necrosis and bacterial rods in JH occurred primarily in those areas where the mucosa had been physically separated from the submucosa by the intramural hematoma. Histologic examination of the tissues also revealed that, at the margins of the lesion, the hematoma often split the muscularis mucosae or was on the lamina propria side of the muscularis mucosae. The splitting of the muscularis mucosa did not have associated inflammatory infiltrate and is potentially an artifact but it was consistently seen in cases of JH and, in most cases, there was associated hemorrhage indicating the split occurred while the animals were alive. The splitting of the muscularis mucosa by the hematoma, combined with the observation that many of the cases had markedly dilated, blood filled lymphatic lacteals within the villi, the fairly uniform lack of significant inflammation and the presence of hemosiderophages in the lamina propria suggests that the lesion may begin in the lamina propria secondary to some disturbance of blood or lymphatic flow with resulting formation of the hematoma, eventual rupture of the overlaying mucosa and ultimate hemorrhage into the intestinal lumen. There were no consistent postmortem findings (such as organ displacement or torsion) in the JH cases in which whole animals were submitted that might cause increased blood or lymphatic pressure and thereby predispose the cattle to a vascular accident (data not shown). As with any retrospective study, the material available for histologic examination was only that which was collected at the time the original necropsies were performed and, with some of the older cases, only that which was trimmed into cassettes and embedded in paraffin. Because the sites within the intestine and within the hematoma lesions, the postmortem interval and intensity of sampling were not controlled during the original tissue collection, performing and reporting a statistical analysis of the histologic lesions might imply more significance to the results than the lack of a sampling scheme would merit. This, in large part, is why an admittedly subjective evaluation of the histologic sections is included in this article. A better design would be a prospective study in which tissue samples are collected as soon as possible after death from specific locations in the small intestine and within the hematoma lesions, and specific microscopic tissue alterations (such as vascular necrosis, lacteal dilation, etc.) scored for severity and then compared between the submission types and also compared to other parameters tested (i.e., C. perfringens culture, toxins detected, etc.).
This retrospective review of 314 cases in which anaerobic cultures were performed on intestinal samples from adult cattle did not find any significant difference between cattle with nonbloody intestinal content, bloody intestinal content but non-JH, and JH with regard to isolation or detection of C. perfringens organisms, presence of toxin genes within C. perfringens isolates, detection of C. perfringens toxins in intestinal content, detection of Salmonella or BVDV, or liver copper levels. The lack of a statistically significant association between these variables and JH implies these do not play a role in the pathogenesis of JH. When ELISA was performed for the presence of C. perfringens in live animals the organism was statistically more likely to be detected in cases of JH than in the other 2 submission types but this did not hold true when anaerobic culture was performed. Histologic examination of 21 cases of JH found that many of the cases had splitting of the muscularis mucosa at the margin of the hematoma and dilation of villus lacteals suggesting the possibility that some disturbance of blood or lymphatic flow leading to leakage into the lamina propria followed by development of the intramural hematoma underlies this disease.
Footnotes
a.
Excel 2007, Microsoft Corp., Redmond, WA.
b.
Hardy Diagnostics, Santa Maria, CA.
c.
Bio-X Diagnostics SPRL, Jemelle, Belgium.
d.
FISONS, Accuris Model, Thermo Optek Corp., Franklin, MD.
e.
DOLT-4, National Research Council of Canada, Ottawa, Ontario, Canada.
f.
TORT-2, National Research Council of Canada, Ottawa, Ontario, Canada.
g.
Stata MP 11.5, StataCorp LP, College Station, TX.
Declaration of conflicting interests
The author(s) declare that they have no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for the research, authorship, and publication of this article was provided by the California Animal Health and Food Safety Laboratory System, School of Veterinary Medicine, University of California, Davis.
