Introduction
The most prevalent contagious mastitis pathogen in the United States is Staphylococcus aureus. In 2007, S. aureus was detected in 43% of bulk tanks examined (U.S. Department of Agriculture, National Agricultural Statistics Service: 2008, Milk production, disposition and income: 2007 summary. Available at: http://usda.mannlib.cornell.edu/usda/nass/MilkProdDi//2000s/2008/MilkProdDi-04-25-2008.pdf. Accessed on May 25, 2010). Control programs targeting S. aureus are varied and have been inconsistently applied throughout the dairy industry with equally variable success.
7,11,17
Although there is no single explanation for the inconsistencies, the cost and perceived inaccuracy of diagnosis are acknowledged challenges in the broad application of effective control programs. The success of any mastitis control program lies in the ability to detect subclinical intramammary infections (IMIs). Often present as a subclinical infection, the control of S. aureus is contingent on accurate diagnosis of IMI, yet no agreement exists on what constitutes a definitive microbiologic diagnosis of a S. aureus IMI. As evidenced in a meta-analysis published in 2002, there has been little consistency in defining either the inoculum volume, the number of samples, or colony-forming units (CFU) required to define any type of IMI.
5
However, a recently published report
15
proposed standard microbiologic definitions of IMI caused by major and minor pathogens based on single quarter milk samples. In addition to the current lack of standardized definitions of IMI, much of the information used to describe S. aureus IMI has been collected from experimental infections.
1,2,10,13
Recent investigations examining the molecular relatedness of experimental and naturally occurring strains of S. aureus
14
have challenged the conclusions from previous studies based on experimental infections. Using multilocus sequence typing and pulsed-field gel electrophoresis, it was reported that the reference strain S. aureus Newbould 305, used in the majority of experimental infections since 1958, was significantly associated with teat skin rather than actual IMIs.
14
In light of this new information, previously published works examining the shedding patterns and diagnostic criteria of S. aureus IMIs based on experimentally induced infections
1,3,13
must be re-evaluated and the questions revisited using naturally occurring S. aureus infections as the model. To that end, the goal of the current study was to examine the effect of inoculum volume of milk samples on the detection of naturally occurring S. aureus IMIs.
Microbiologic results from 2 separate field studies examining the epidemiology of naturally occurring S. aureus IMI using 2 different sampling schemes were analyzed: a daily sampling scheme and a weekly sampling scheme. The daily sampling scheme included 21 consecutive daily samples collected during the afternoon milking of 3 sampling periods during the cow's lactation (each period lasting 21 days). The second study used milk samples collected weekly, in the afternoon milking, during the first 10 weeks of each cow's lactation. Milk samples collected daily were from 9 subclinical, S. aureus–positive quarters (7 S. aureus–positive cows). One quarter that was negative for S. aureus from each S. aureus–positive cow (7 total) was also sampled in addition to 7 S. aureus–negative quarters from 4 S. aureus–negative cows (14 S. aureus–negative quarters total). Milk samples collected weekly were from 36 subclinical, S. aureus–positive quarters (27 S. aureus–positive cows) and from 25 quarters negative for S. aureus in S. aureus–positive cows, in addition to 5 S. aureus–negative quarters from 3 S. aureus–negative cows.
Approximately 12 ml of foremilk was collected from each quarter for culture according to National Mastitis Council (NMC) guidelines.
12
Milk samples were kept frozen, were thawed at room temperature for microbiologic culture, and immediately frozen again and kept at −80°C until cultured a second time. The initial culture procedure used standard NMC (2004) guidelines, applying 0.01 ml of milk on trypticase soy blood agar plates (5% sheep blood)
a
using disposable, calibrated loops. Plates were incubated at 37°C and screened at 24 and 48 hr according to NMC (2004) guidelines.
12
A diagnosis of S. aureus was made based on colony morphology, positive catalase test, presence of hemolysis, and a positive coagulase test. A S. aureus IMI was defined as a quarter having at least 2 of the first 3 consecutive samples (daily or weekly) culture positive with ≥1 CFU of S. aureus/0.1 ml. Individual quarter samples collected from S. aureus–positive quarters with ≥1 CFU of S. aureus were considered positive. Samples considered contaminated (3 or more apparent bacterial species) when using 0.01 ml were excluded from the analysis (n = 25). Milk samples that were culture negative for S. aureus using a 0.0-ml inoculum from S. aureus IMI quarters and all samples from non–S. aureus quarters were thawed a second time and cultured using 0.1 ml of milk. Milk samples from S. aureus–positive quarters that were culture positive using 0.01 ml were unavailable for reculture using 0.1 ml and were considered positive at the larger inoculum for the analysis. Sensitivities and specificities of a single microbiologic culture in identifying S. aureus IMI and the comparison of the ability to detect a S. aureus IMI between the 2 inoculum volumes were calculated considering each quarter sample independently
13
(roctab, roccomp, STATA v. 11)
b
applying the definition of a S. aureus–positive quarter (at least 2 of the first 3 consecutive samples culture positive with ≥1 CFU of S. aureus/0.1 ml) and assuming a disease-free status in quarters defined as S. aureus negative. Quarters with no history of S. aureus, but repeatedly testing positive for S. aureus at some point after the third sample, were considered new infections and not included in the analysis (n = 3). Quarters culture positive for S. aureus in cows that were treated with intramammary or systemic antibiotics at anytime during the current lactation were not included in the analysis (n = 2). Culture results from quarters that ceased lactating prematurely and cows that were culled due to illness (other than mastitis) during the study were included provided there was no evidence of antibiotic treatment during the sample period (n = 10).
A total of 1,583 milk samples were included in the analysis. Of the 25 samples considered contaminated only 2 were from S. aureus–positive quarters. A single additional sample of those cultured a second time was considered contaminated using the larger volume. The total number of S. aureus–positive quarters included in the analysis for the daily and weekly sampling was 9 and 34, respectively. The total number of S. aureus–negative quarters included in the analysis of daily samples was 14, and the total number of negative quarters included from weekly samples was 27.
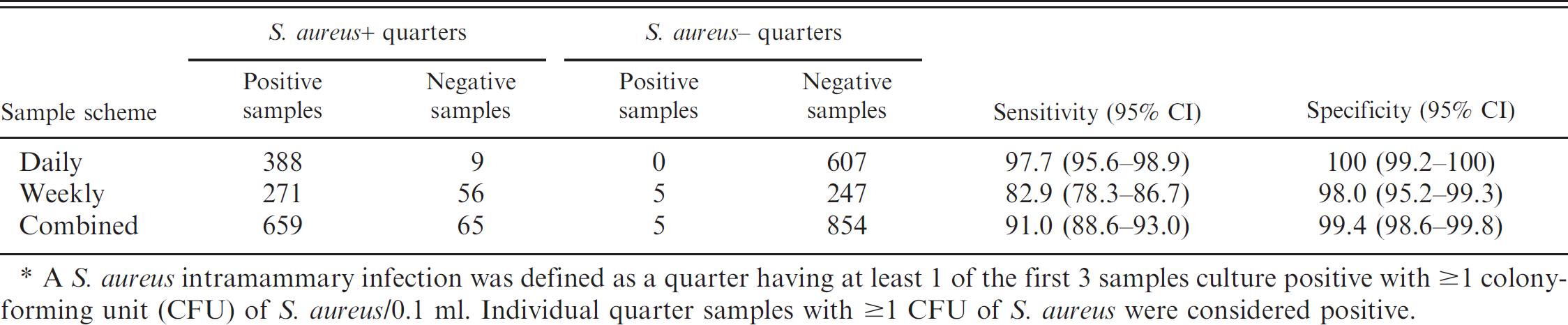
The culture results using a 0.01-ml inoculum are presented in Table 1. Considering a quarter sample culture positive at ≥1 CFU, using a 0.01-ml inoculum, 2% of samples collected daily from known S. aureus–positive quarters were negative, and there were no culture-positive samples from known negative quarters (no false positives). The sensitivity and specificity of microbiologic culture examining daily samples was 97.7% and 100%, respectively. In contrast, examining the weekly samples, 17% of the samples from known S. aureus–positive quarters were negative, and 2% of the samples from known S. aureus–negative quarters were positive resulting in a sensitivity of only 82.9% and a specificity of 98.0%. The CFU count on false-positive quarters using 0.01 ml ranged from 1 to 8 CFU. Of the 56 false-negative samples, 40 (71%) were from 8 of the 34 S. aureus–positive quarters. Combining the results of both studies, the 0.01-ml inoculum resulted in a sensitivity of 91% (95% confidence interval [CI]: 88.6–93.0%) and a specificity of 99.4% (95% CI: 98.6–99.8%).
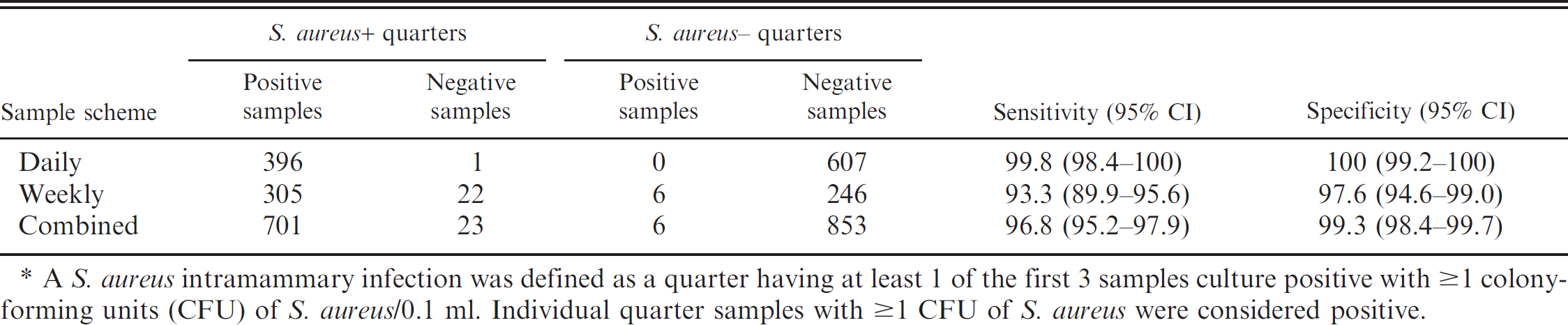
The results of using a 0.1-ml inoculum are presented in Table 2. Considering a quarter sample culture positive at ≥1 CFU, using a 0.1-ml inoculum from samples collected daily, only 0.2% of the samples (n = 1) from known S. aureus–positive quarters were negative, and there were no culture-positive samples from known negative quarters (no false positives). The sensitivity and specificity of microbiologic culture for daily samples were 99.8% and 100%, respectively. Comparing the area of the 2 receiver operating characteristic (ROC) curves for both inoculum volumes, the larger volume proved to be significantly better than the smaller volume (p = 0.004) for detecting S. aureus in the daily samples. Examining the weekly samples, 6% of the samples (n = 22) from known S. aureus–positive quarters were culture negative and 2% (n = 6) were culture positive from known S. aureus–negative quarters (false positives), resulting in a sensitivity of only 93.8% and a specificity of 97.6%. Of the 22 false-negative samples collected weekly, 17 (77%) were represented by 6 of the 34 S. aureus–positive quarters. Again, comparing the 2 inoculum volumes, the larger volume proved to be significantly better than the smaller volume (P < 0.001) in detecting S. aureus. Combining the results of both studies, comparing the area under the 2 ROC curves for both inoculum volumes, using a 0.1-ml inoculum resulted in a sensitivity of 96.8% (95% CI: 95.2–97.9%) and a specificity of 99.3% (95% CI: 98.4–99.7%), which proved to be significantly better than when 0.01 ml was used (P < 0.001). The number of CFU/0.1 ml in false-positive samples ranged from 2 to 95. All of the samples (n = 6) that were S. aureus culture–positive from S. aureus–negative quarters (n = 5) were from S. aureus–positive cows. The single S. aureus–negative quarter that cultured positive twice (using both the smaller and larger inoculum) were samples from a cow with 2 S. aureus–positive quarters. There were no false-positive cultures from S. aureus–negative cows (n = 7).
Great variation in diagnostic schemes prevail throughout mastitis research as there is little agreement on which methods and interpretation of microbiologic culture should be followed in defining a gland as being infected. The widely accepted standard inoculum volume for mastitis diagnostics is 0.01 ml; however, there remains great inconsistency in what has been used and reported in research. Although some investigators have used inoculums of 0.01 ml, others have increased inoculum volumes to 0.05–0.1 ml, thereby increasing the sensitivity of microbiologic screening.
4,8,9
Other investigators have reported variable inoculums within a single study using 0.1 and 0.05,
13
or 0.01–0.03 ml.
10
Considering the daily samples, although statistically significant, there was only a 2% difference in the sensitivity of culture using the larger inoculum. It is important to consider that although the entire study consisted of more than 1,500 samples, the daily study consisted of only 7 cows on a single dairy. In addition, considering each sample independently ignores the clustered nature of the data, potentially resulting in either larger or smaller standard errors. The small number of cows may have also limited the representation of bacterial strains
10
to those that consistently shed higher numbers of bacteria or strains exhibiting cycles of shedding that are longer than 21 days. The study using weekly samples examined milk from 25 cows on 2 different dairies and therefore may reflect a greater diversity in strain types. Because more than 77% of the false-negative weekly samples were represented by only 6 of the 34 S. aureus–positive quarters (using 0.1 ml), the necessity of examining the effects of strain variation and cow-to-cow variation on diagnostics is clear. In addition, although the samples culture positive for S. aureus from S. aureus–positive quarters were not subject to reculture using the larger inoculum, they were used for another study and cultured a second time using the same 0.01-ml volume, all culturing positive again. Last, the possibility of finding more contaminated samples using a larger inoculum was considered. However, given that there were only 2 contaminated samples from S. aureus–positive quarters on initial culture and only a single additional sample was considered contaminated when the larger volume was used, it appears that potential impact of greater contamination on the results when using a larger inoculum volume was negligible.
Using the smaller inoculum, weekly samples detected S. aureus in only 83% of the samples from positive S. aureus quarters. The use of the larger inoculum significantly increased the sensitivity of culture in the weekly samples by 10.9%. Combining the findings of both studies resulted in an overall sensitivity of 96.8, nearly 10% higher than that predicted by other modeling experimental infections.
2
In the present study, every false-positive sample was from a cow with a known S. aureus–positive quarter, and there were no false-positive samples obtained from completely negative cows. In addition, use of the larger inoculum resulted in only 1 additional false positive. With the number of false-negative samples decreasing from 65 (9.0%) to 23 (3.2%), the importance of applying a larger inoculum volume to detect S. aureus IMI in cows shedding comparatively low numbers of S. aureus is clear.
Further implications of the present study include insight into the development of standards for the number of samples required to diagnose a S. aureus IMI. The U.S. Food and Drug Administration's guidelines, published for the collection of efficacy data of mastitis treatments, require recovery of the same organism from at least 2 milk samples collected separately, 5 days apart, after treatment.
16
There are, however, no requirements for the diagnosis of mastitis before treatment. The necessity of repeated (consecutive or duplicate) samples is unclear and at times presents a significant economical hurdle in the diagnosis of S. aureus IMI in management strategies. It has been reported that the percentage agreement between duplicate samples was highest for contagious pathogens, such as S. aureus (94.2%), suggesting that a single quarter sample might be adequate in determining infection status.
6
Recently, the utility of colony count in assessing IMI status, comparing duplicate samples collected at dry-off, was examined, and investigators reported that an IMI could be diagnosed accurately with a single sample when CFU was considered, and that the required number of CFU for a positive diagnosis was dependent on the type of pathogen identified.
15
In that study, the investigators established the utility of a single sample positive with at least 1 CFU of S. aureus/0.01 ml in identifying S. aureus IMI. The current study, examining naturally occurring infections repeatedly over time, supports the investigators' conclusion that a single colony is sufficient to diagnose a S. aureus IMI using single quarter sample.
Based on the findings in the current study in naturally infected cows, using a single quarter sample, S. aureus IMI can be most accurately diagnosed using a standard 0.1-ml inoculum in research and diagnostic settings. A single quarter sample that is culture negative is sufficient to rule out the presence of S. aureus IMI. Meanwhile, a single quarter sample culture positive with ≥1 CFU/0.1 ml should be considered positive. Applying this standard provides an economical approach to S. aureus IMI diagnostics for research as well as practice settings. Quarters like the 6 noted in the present study, infected with S. aureus isolates, at times shedding very low numbers of S. aureus, or shedding at undetectable levels, are of great interest in further investigations. Future studies examining naturally infected quarters during longer periods in addition to examining the genetic relatedness of strains and shedding patterns may further aid in developing a more coherent definition for S. aureus infections