Abstract
Many commercial antibody detection enzyme-linked immunosorbent assay (ELISA) kits for Q fever utilize the Nine Mile (Montana tick) strain of Coxiella burnetii as antigen. An ELISA kit manufactured in France employs ovine placenta-sourced antigen and has been used in Europe. Sera from goats experiencing a Q fever abortion storm in the United States were used to compare the sensitivity and specificity of these 2 ELISA formats and the Q fever complement fixation test (CFT). Latent class estimates of sensitivity ranged from 97% to 100% with a specificity of 95–100% for the 2 ELISA kits. Estimates for sensitivity and specificity of the CFT were 89% and 82%, respectively. There was not a significant increase in ELISA sensitivity observed with the ovine-sourced antigen kit in this study. Real-time polymerase chain reactions performed on a portion of the sera found that 15 out of 20 sera were congruent across 4 tests for positive and negative sera.
The intracellular bacteria Coxiella burnetii is the etiologic agent for Q fever (QF) in human beings and livestock. 14 This zoonotic agent is distributed among a world-wide range of animal hosts, including mammals, arthropods, birds, fish, rodents, and reptiles. 10 The bacteria are shed by mammals in urine, feces, milk, or birthing products and are very resistant to degradation. 14 Q fever in sheep and goats can present with reproductive problems, abortion, or stillbirths. Goat outbreaks are generally associated with human exposures, and cattle are considered asymptomatic carriers. 10 Large numbers of bacteria are shed from cattle, sheep, and goats at parturition and remain viable as dried spore-like material in the environment. Aerosolized spores are the predominant route of transmission for C. burnetii both in human beings and animals. 14 Viable agent has been detected on wool after 12–16 months, in dried urine after 49 days, and in dust for 120 days. 3
Numerous reports from Europe described QF antigens and their influence on the results of serological testing. Researchers in 1955 described discrepant results with complement fixation test (CFT) between use of a Nine Mile (NM, tick strain from Montana) and Henzerling (human strain from Italy) strain as the test antigen. 15 An ELISA measuring the serological response to an ovine aborted placenta antigen (Cb01) has been evaluated by numerous researchers in Europe who observed increased sensitivity when compared to a similar ELISA using the NM strain antigen.6,12 The European Food Safety Authority has reported higher diagnostic sensitivity with ELISAs using ruminant C. burnetii isolate antigens when compared to those using NM antigens. 3
Testing for Coxiella by polymerase chain reaction (PCR) is documented in the literature using milk, vaginal swabs, or fecal samples. 12 However, a report from Japan indicated testing of serum by PCR may also be a viable alternative in both human beings and dogs. 9 A Japanese veterinary clinic reported transmission of Coxiella among 2 animal health technicians, 2 dogs, and 3 cats diagnosed by testing serum by PCR. The diagnosis was confirmed by isolation of Coxiella bacteria from the sera of the technicians and the dogs. 9 Retrospective surveillance studies in Japan have found seropositivity rates greater than 20% in healthy individuals with at-risk animal exposures suggesting that Coxiella could be a reemerging disease in Japan. 5
The National Veterinary Services Laboratories (NVSL) received goat sera from an owner reporting 14 abortions from a herd of 50 pregnant does. Ninety-one sera submitted approximately 4 months after the abortion storm were tested with the QF ELISA kit a (German NM antigen, kit A), QF ELISA kit b (French Cb01 antigen, kit B), and QF CFT c (German NM antigen). The CFT closely followed the Manual of Diagnostic Tests and Vaccines for Terrestrial Animals (OIE) protocol, and the ELISAs were conducted following the manufacturers’ instructions. Twenty of the sera were examined for Coxiella DNA by real-time PCR. Assays were performed with published primers for C. burnetii. 8
Briefly, CFT was performed on serum that had been heat inactivated at 56°C for 30 min to destroy the endogenous complement. The serum was added with 2-fold serial dilutions to a 96-well microtiter plate. Controls were included to detected anti-complementary and nonspecific reactions, Coxiella-positive and -negative antisera, and the plates were incubated overnight in a refrigerator with test sera, complement, and Coxiella antigen. Sensitized sheep red blood cells (RBCs) were added as indicators the following day, and the plates were centrifuged to pellet intact RBCs. The highest dilution with a 100% fixation was identified as the end point titer (RBC pellet with clear supernate). The OIE described CF titers at 1:80 and above found within a population of 6 goats, 6 ewes, or 10 cows as evidence of an active infection, and titers in the range of 1:10 through 1:40 as evidence of prior exposure. 14 For the ELISA kits A and B, goat sera were incubated on the plates with inactivated C. burnetii antigen from either NM or Cb01 strain coated on the wells, respectively. Serum QF antibodies were bound to the coated antigen, and the plate was washed to remove unbound antibody. Q fever peroxidase-labeled anti-ruminant IgG was added to kit A (NM) plates, and a Q fever peroxidase-labeled IgG was added to kit B (Cb01) plates; the plates were incubated and washed. Substrate solution was added to the respective plates for color development which was measured as the spectrophotometric optical density (OD) at 450 nm. The cutoff for positivity for both kits were determined with the %OD, which was calculated as %OD = 100 × (OD value of test sample − OD value of negative control)/(OD value of positive control − OD value of the negative control). For the Cb01 antigen kit, samples with a %OD greater than 40% were considered positive, and samples with a %OD of 40 or less were considered negative. For the NM kit samples reporting a %OD greater than 40% were considered positive, and those sera between 30% and 40% were considered suspect for QF antibodies. Those sera less than 30% were considered negative. For the purpose of the current study, the suspect animals identified on the NM kit were treated as negative results.
Sera samples for real-time PCR were mixed with 3-fold excess of lysis solution, d shipped frozen, and stored at −70°C until being processed for PCR testing. Genomic material was extracted from serum samples using the manufacturer’s DNA extraction method d with modifications. Briefly, 1 ml of sample was mixed with 200 µl of chloroform, incubated at room temperature for 10 min, and centrifuged 12,000 × g for 10 min at 4°C. The organic phase was treated with 0.3 ml of 100% ethanol, centrifuged at 2,000 × g for 5 min at 4°C to yield a DNA pellet which was dissolved in 500 µl of water and stored at 4°C. After additional cleaning using commercially available kits, e the purified DNA was stored at −20°C.
The presence of C. burnetii was evaluated by PCR using established primer setsf,8: forward primer isocitrate dehydrogenase (icd)-439F (5′-CGTTATTTTACGGGTGTGCCA-3′), reverse primer icd-514R (5′-CAGAATTTTCGCGGAAAATCA-3′), and fluorescent probe icd-464TM (5′-FAM-CATATTCACCTTTTCAGGCGTTTTGACCGT-TAMRA-3′). Each 20-µl PCR mixture contained 0.5 µM forward primer, 0.5 µM reverse primer, 0.1 µM TaqMan probe, and 5 µl of serum-extracted DNA in 2× universal PCR mix. g Real-time PCR g assays were carried out for each sample in duplicate running a protocol with 1 min at 95°C (initial denaturation) followed by 40 cycles of 15 sec at 95°C and 1 min at 60°C (amplification and detection). PCR results were analyzed using standard manufacturer software. A sample was considered “positive” for C. burnetii genomic material if its associated amplification curves exhibited Ct < 31 with a spread ΔCt ≤ 5% between duplicate sample runs.
As shown in Figure 1, there was good agreement between the NM and Cb01 ELISAs with goat sera sampled from the farm with the abortion storm. The maximum %OD for the NM data was about 200%, and the maximum for the Cb01 data was more than 260%. Calculated percentages were higher across all of the Cb01 data points as demonstrated in Figure 1, by the shift to the upper half of the graph for those positive points above 40%. Table 1 shows 13 animals that met the OIE CFT criteria for an active outbreak, with titers at 1:80 or greater. 14 The CFT titers from Table 1 listed 68 out of 91 animals with titers that range from 1:10 to 1:160. The total counts across all 3 serology tests listed in Table 2 shows that 63 out of 91 animals tested positive by both ELISA kits and CFT. There were 14 sera negative for all 3 tests making 77 out of 91 (85%) of the sera congruent across all serological testing methodologies.
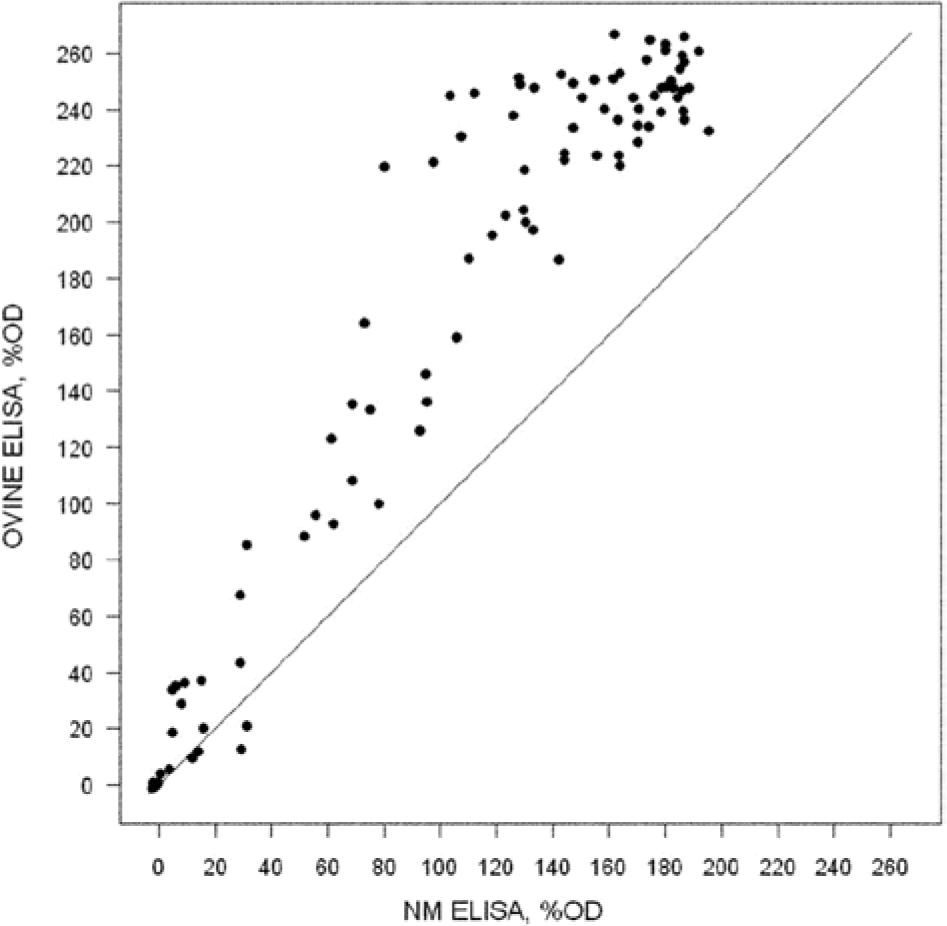
Calculated percentage optical density (%OD) for each goat serum tested with the Nine Mile (NM) and ovine aborted placenta (Cb01) antigen enzyme-linked immunosorbent assay (ELISA) kits. Values for NM ELISA are found on the x-axis, and values for the Cb01 ELISA are found on the ovine y-axis. Each serum is plotted on the graph as (X, Y) product corresponding to (NM %OD, Cb01 %OD). The samples with %OD greater than 40% are considered positive with either ELISA kit.
Goat serum complement fixation test (CFT) titers.*

The count of sera at each CFT titer is listed. There are 23 sera labeled as less than 1:10, which are reported as negative at 1:10. The World Organization for Animal Health (OIE) describes an active case of Coxiella burnetii as CFT titers positive at 1:80 or greater while those at 1:10–1:40 reflect prior exposures.
Number of goat sera by result set.*
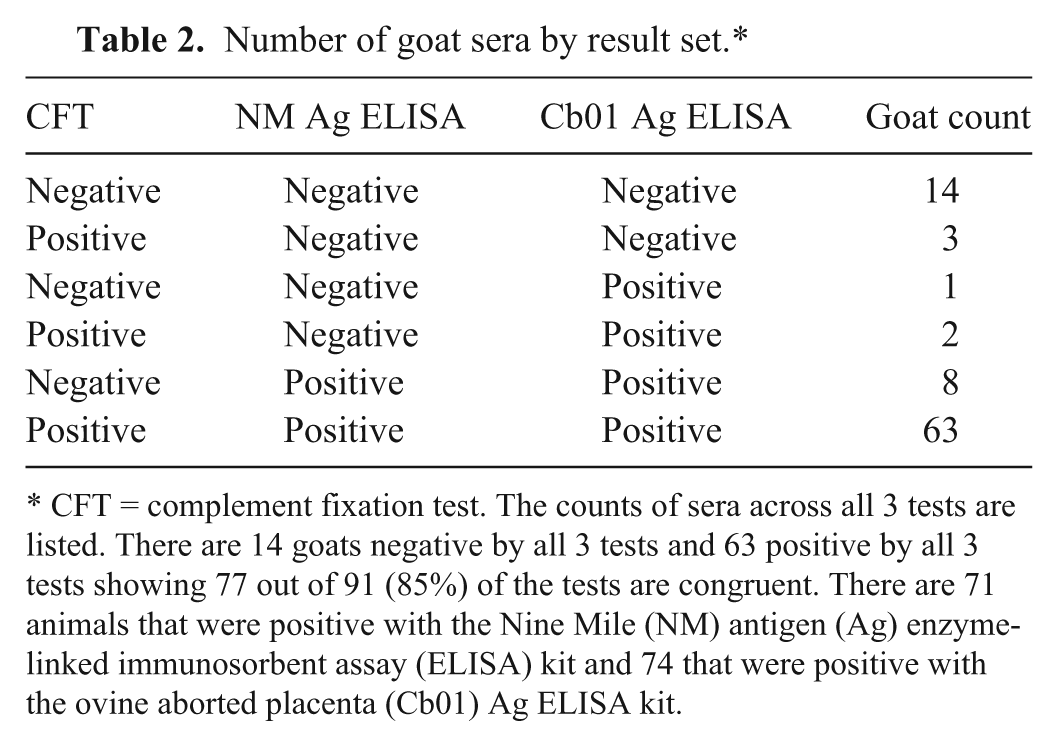
CFT = complement fixation test. The counts of sera across all 3 tests are listed. There are 14 goats negative by all 3 tests and 63 positive by all 3 tests showing 77 out of 91 (85%) of the tests are congruent. There are 71 animals that were positive with the Nine Mile (NM) antigen (Ag) enzyme-linked immunosorbent assay (ELISA) kit and 74 that were positive with the ovine aborted placenta (Cb01) Ag ELISA kit.
Previous studies at NVSL with 154 goats comparing the CFT c (NM antigen) and ELISA a (NM antigen) in diagnostic sera from healthy animals demonstrated kappa values of 0.85 (95% confidence interval [CI]: 0.77, 0.99) and good agreement again between CFT and ELISAs. 2
For the current study, latent class analysis was performed on serology tests and diagnostic estimates for sensitivity (Se) and specificity (Sp) were calculated.7,11 A reference standard was not used in estimating the diagnostic sensitivity Se and diagnostic specificity Sp, rather Se and Sp were estimated from a latent class model. The estimates were obtained using an expectation-maximization (EM) algorithm. This model does not assume that the true disease status is known; rather it is a latent variable. The estimates for Se and Sp have the same interpretation here as they would if the true disease status is known, namely the probability of test positive given disease positive and the probability of test negative given disease negative, respectively. The Sp was 1.00 (95% CI: 0.90, 1.00) for the NM ELISA and 0.95 (95% CI: 0.90, 1.00) for the Cb01 ELISA. The Se was 0.97 (95% CI: 0.92, 0.99) for the NM ELISA kit, and 1.00 (95% CI: 0.97, 1.00) for Cb01 ELISA kit. There was no increase in diagnostic sensitivity observed with the Cb01 ELISA kit with this population of goats. As to be expected the Se and Sp was lower for the CFT: 0.89% (95% CI: 0.80, 0.95) and 0.82 (95% CI: 0.62, 0.95), respectively. Any of the 3 tests would have successfully detected the etiology of the abortion storm in these goats.
A smaller pilot project with these goats was conducted to evaluate if there was any correlation of serology and PCR results on the same serum sample. In Table 3, CFT titers are compared with PCR results. Positive and negative CFT results in this table also reflect congruent positive and negative results on 2 ELISA kits. There were 10 sera positive by all 4 tests (CFT, ELISA NM, ELISA Cb01, and PCR) and 5 sera negative by all four tests which represent 75% of the 20 sera tested by all methods. There were 5 sera with incongruent immunoassay versus PCR results (i.e., positive antibody yet negative PCR or negative antibody with positive PCR). More testing will be required to evaluate these relationships. No attempts were made to determine if the sera were infectious.
Complement fixation test (CFT) and polymerase chain reaction (PCR) results in goat serum collected during a Coxiella burnetii abortion storm.*
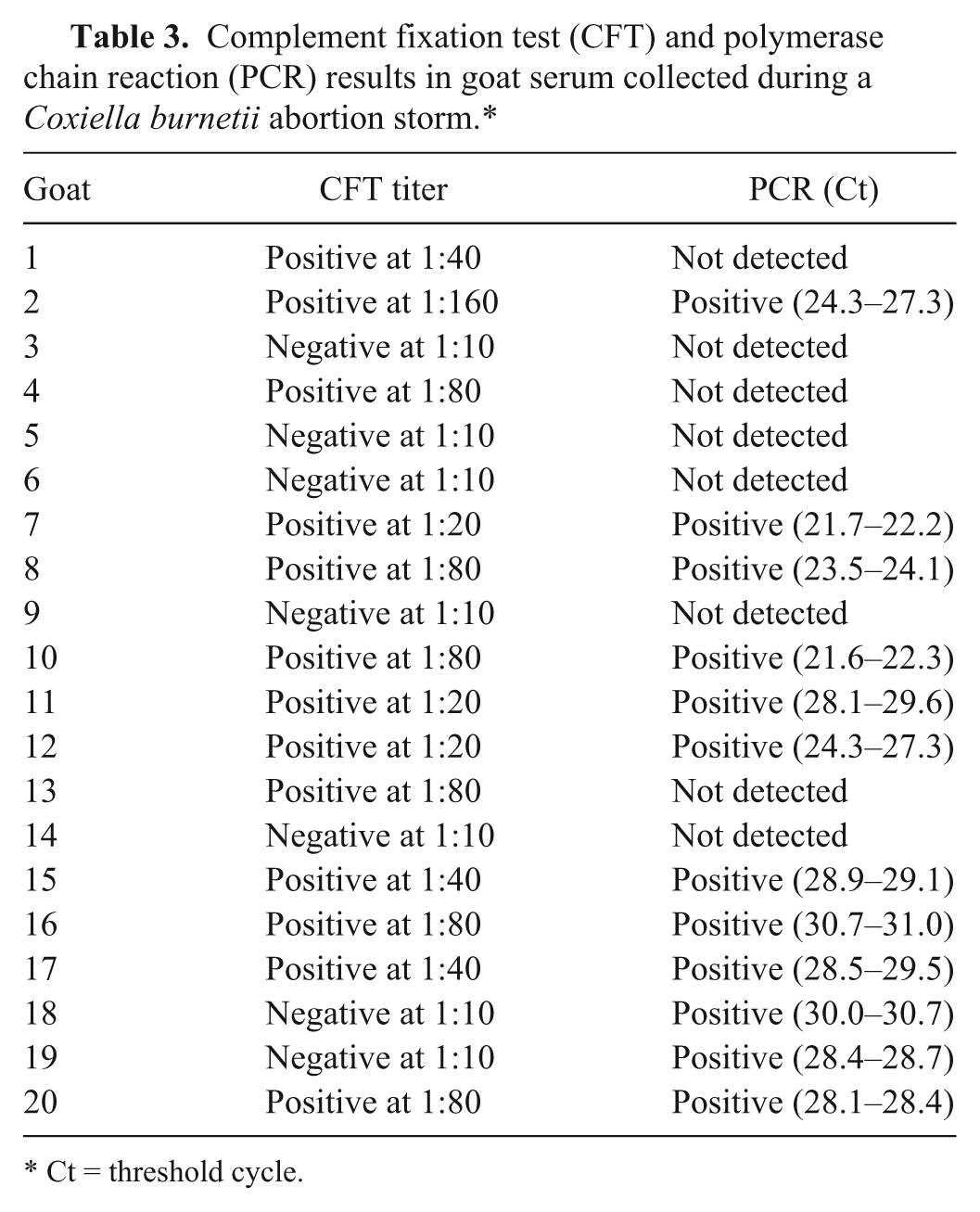
Ct = threshold cycle.
Two of the five incongruent sera were negative on all 3 serology tests but positive on PCR. There are several explanations that could support these results. In herds of French goats with abortions, at least 24% of those goats shed bacteria were detected by PCR (vaginal mucus, feces, and milk), and were also seronegative with NM ELISA. 13 Alternately, the animals could have been infected via the environmental contamination of C. burnetii abortion storm, and have not yet seroconverted. Researchers evaluating C. burnetii–infected cattle have suggested that the serology-negative/PCR-positive animals (placenta) could represent a group of persistently infected animals much like those found with bovine viral diarrhea exposures. 1 And finally, these goats could be immunotolerant to Coxiella with the immune reaction suppressed by unidentified means.
Three of the 5 incongruent sera were positive on all 3 serology tests, but negative on PCR. Coxiella burnetii has 2 antigenic phases. Phase 1 is considered pathogenic with a full length lipopolysaccharide (LPS) while phase 2 is attenuated with a branched LPS. The ELISA kitsa,b used in the current study contained both phase 1 and phase 2 antigens. Phase 1 (chronic exposure) and phase 2 (acute exposure) antibody responses are well defined in human medicine, but are currently developing in veterinary medicine. Researchers evaluating Bavarian cattle compared commercially available NM ELISA kit (phase 1/phase 2) a to single phase 1 and phase 2 ELISAs that were prepared by the manufacturer. 1 PCR assays were performed on placenta from the same herds. Antibodies to phase 2, but not phase 1, were associated with PC-positive results. 1 It could be possible that the 3 incongruent goat sera contain predominately phase 1 antibodies. Another possible explanation of positive serology is from exposure prior to the abortion storm.
The current study showed essentially no difference in diagnostic sensitivity between 2 different C. burnetii antibody ELISA kits when examining goat serum from a herd experiencing an abortion storm. To date, there is little information from Europe on the nature or mechanism of increased sensitivity associated with the ovine antigen in the literature. 4 There was high seroprevalence in the herd examined in the present study, which could explain the lack of diagnostic sensitivity observed. Future studies comparing the 2 ELISA kits will be expanded to examine goats, sheep, and cattle from herds without abortions to observe the impact on sensitivity. The PCR testing merits more work to examine the relationship between Coxiella serology and real-time PCR testing with known positive and negative goat sera. Coxiella DNA was detected by PCR and verified by culture 210 days postinvestigation in Japanese dogs, a length of time well within the average 150-day gestation period for goats. Excreted products, including milk, vaginal swab, and feces may not act as harbingers of Coxiella until parturition. Tandem serum testing for both antibodies and bacteria could provide an opportunity to isolate or cull PCR-positive animals decreasing the potential for infections of other ruminants and the environmental contamination.
Footnotes
Acknowledgements
The authors thank Monica Reising for her assistance with statistical applications.
a.
IDEXX Switzerland AG, Liebefld-Bern, Switzerland.
b.
LSI, Lissieu, France.
c.
Institut Virion, Serion GmbH, Wurburg, Germany.
d.
Trizol LS, Invitrogen Corp., Carlsbad, CA.
e.
QIAamp DNA extraction protocol, Qiagen Inc., Valencia, CA.
f.
PCR primers and fluorescent TaqMan probes; Integrated DNA Technologies Inc., Coralville, IA.
g.
PCR mix, 7900HT Fast Real Time PCR System, system software package SDS 2.4; Applied Biosystems, Life Technologies, Carlsbad, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
