Abstract
Real-time reverse transcription polymerase chain reaction (real-time RT-PCR), histopathology, and immunohistochemical labeling (IHC) were performed on liver specimens from 380 naturally infected cattle and sheep necropsied during the 2010 Rift Valley fever (RVF) epidemic in South Africa. Sensitivity (Se) and specificity (Sp) of real-time RT-PCR, histopathology, and IHC were estimated in a latent-class model using a Bayesian framework. The Se and Sp of real-time RT-PCR were estimated as 97.4% (95% confidence interval [CI] = 95.2–98.8%) and 71.7% (95% CI = 65–77.9%) respectively. The Se and Sp of histopathology were estimated as 94.6% (95% CI = 91–97.2%) and 92.3% (95% CI = 87.6–95.8%), respectively. The Se and Sp of IHC were estimated as 97.6% (95% CI = 93.9–99.8%) and 99.4% (95% CI = 96.9–100%), respectively. Decreased Sp of real-time RT-PCR was ascribed to cross-contamination of samples. Stratified analysis of the data suggested variations in test accuracy with fetuses and severely autolyzed specimens. The Sp of histopathology in fetuses (83%) was 9.3% lower than the sample population (92.3%). The Se of IHC decreased from 97.6% to 81.5% in the presence of severe autolysis. The diagnostic Se and Sp of histopathology was higher than expected, confirming the value of routine postmortem examinations and histopathology of liver specimens. Aborted fetuses, however, should be screened using a variety of tests in areas endemic for RVF, and results from severely autolyzed specimens should be interpreted with caution. The most feasible testing option for countries lacking suitably equipped laboratories seems to be routine histology in combination with IHC.
Keywords
Introduction
Rift Valley fever (RVF) is a mosquito-borne zoonotic disease caused by a virus of the family Bunyaviridae, genus Phlebovirus. It is responsible for extensive outbreaks of disease in livestock in Africa with significant mortality and economic impact. 20 The ecology of the disease involves an endemic and an epidemic transmission cycle. 28 Evidence of low levels of virus transmission has been confirmed in domestic ruminants, wild ruminants, and humans in endemic areas. 1 Sporadically, epidemics of RVF occur over large areas following exceptionally heavy rains that cause substantial increases in vector-competent mosquito populations. 27 In livestock, especially sheep and cattle, epidemics of RVF are characterized by high numbers of abortions (90–100% of pregnant ewes in all stages of gestation), sudden death in young animals and severe liver pathology.4,5 Disease susceptibility varies with age and species. Peracute hepatic disease occurs in lambs and calves less than one month old, with estimated mortality proportions as high as 90–100% in lambs and 10–70% in calves. 1 Mortality in adult ruminants is approximately 10–30% in sheep and 5–10% in cattle. 1
The majority of pathological lesions occur in the liver and are similar in all ruminants, but the extent of liver involvement varies with age.4–7,10,11,14 Gross changes in the liver include enlargement with sharply defined pale or dark red foci ranging in size from pinpoint to several millimeters. The principal histological lesion of RVF is random foci of apoptosis, 26 accompanied by hemorrhage and a mild infiltrate of neutrophils and mononuclear inflammatory cells. Hepatotrophic viruses, acute poisoning with a variety of hepatotoxic plants and bacterial septicemias can present with hepatic lesions that may resemble those associated with RVF. 28 The World Organization for Animal Health (OIE) recommends the use of virus isolation, agar gel immunodiffusion, nucleic acid amplification techniques and immunolabeling to confirm the presence of RVFV. 32 Specific antibodies to RVFV are demonstrable by virus neutralization, enzyme-linked immunosorbent assay (ELISA), and hemagglutination inhibition. 32 Virus neutralization is considered the gold standard for confirming RVFV infection, 20 but the procedure is time consuming and expensive making it unsuitable for use in an epidemic when the disease is spreading rapidly. Real-time reverse transcription polymerase chain reaction (real-time RT-PCR), histopathology, and immunohistochemical labeling (IHC) are the diagnostic methods most often used in South Africa to confirm or exclude a diagnosis of RVF in necropsied animals. Numerous nucleic acid amplification assays have been developed and evaluated for detection of RVFV,8,13,20 and IHC has been used to detect the distribution of viral antigen in the tissues of lambs and calves.8,24,30 Validated estimates of diagnostic accuracy in naturally infected livestock, however, have not been published. Therefore, the objective of the present study was to estimate the diagnostic sensitivity and specificity of real-time RT-PCR, histopathology, and IHC using Bayesian latent class methods. A secondary objective was to estimate stratum-specific values based on species, age, degree of specimen autolysis, and the presence or absence of tissue pigments.
Materials and methods
Outbreak description
An extensive outbreak of RVF occurred in South Africa in 2010. The first report was made at the end of January and documented the sudden death of 230 young lambs in the Bultfontein and Brandfort areas of the Free State Province (World Animal Health Information Database [WAHID]: 2010, South Africa, immediate notification, Rift Valley fever. Report date February 19, 2010. Available at: http://www.oie.int/wahis_2/public/wahid.php/Reviewreport/Review?page_refer=MapEventSummary&reportid=8967; accessed May 21, 2013). 21 Additional cases (n = 80) were reported in the beginning of February, and a diagnosis of RVF was confirmed on February18, 2010 by real-time RT-PCR and IHC. The epidemic rapidly spread throughout the Free State Province and subsequently extended into the Eastern Cape, Northern Cape, Western Cape, and North West Provinces. 21 Isolated outbreaks were also reported in Gauteng, Limpopo, and Mpumalanga Provinces with none reported in KwaZulu-Natal. The outbreak affected mainly sheep but also cattle, goats, African buffalo, camelids, and other wild animals. 21 The last cases were reported in the Northern Cape Province at the end of August 2010 (WAHID: 2010, South Africa, Rift Valley fever. Follow-up report no. 17. Report date November 29, 2010. Available at: http://www.oie.int/wahis_2/public/wahid.php/Reviewreport/Review?page_refer=MapEventSummary&reportid=8967; accessed May 21, 2013)
Samples size justification
The necessary sample size was calculated for an expected sensitivity and specificity of 0.95, and the desire to estimate this value ±0.05 at the 95% confidence level. A conservative frequentist (rather than Bayesian) approach was employed using exact binomial methods as previously described. 15 The calculated sample size was 97 cases each for RVF-infected and uninfected livestock. The true prevalence was unknown and it was expected that there might be a high prevalence in the sampled population (approximately 75%) so the calculated sample size was multiplied by 4 in an effort to obtain enough samples from RVF uninfected livestock. All samples from cattle, neonates, and fetuses from all Provinces were included in the study with the balance being adult sheep from the Free State Province to obtain the required sample size.
Rift Valley fever outbreak specimen selection
This was a retrospective study of cases submitted to the Agricultural Research Council–Onderstepoort Veterinary Institute (ARC-OVI; Onderstepoort, South Africa), at the behest of the South African Department of Agriculture Fisheries and Forestry (DAFF). All specimens originated from the carcasses of naturally infected animals and fetuses that were necropsied during the 2010 RVF outbreak. Fresh and 10% formalin-fixed specimens were submitted to the ARC-OVI and a private veterinary diagnostic laboratory (IDEXX Laboratories, Pretoria), respectively, for the diagnosis of RVF. Archival paraffin-embedded liver specimens with previously performed real-time RT-PCR results were obtained.
Control specimen selection
An additional 40 blinded control cases were obtained from the archives of the Faculty of Veterinary Science, University of Pretoria, for randomization among the 2010 RVF outbreak cases. These included 8 real-time RT-PCR– and IHC-positive cases of RVF that occurred in 2009 and 32 additional cases due to etiologies that cause liver pathology resembling RVF. In the case of IHC, relevant or irrelevant antibodies were applied to sections to create blinded controls for positive and negative reactions.
Non-RVF controls included 7 cases where hepatotoxic plants caused acute death in adult cattle or sheep, 10 cases of Equid herpesvirus 1 (EHV-1) infection in equine fetuses, 4 cases of infectious bovine rhinotracheitis (IBR) in bovine fetuses, 10 cases of Wesselsbron disease (WBD) in neonatal and adult sheep, and 1 case of salmonellosis in an adult bovine. Cases of EHV-1 infection were added because of the limited availability of control cases in ruminant fetuses.
The EHV-1, IBR, and WBD cases were verified by IHC and characteristic histological lesions. The cases where the etiology was acute poisoning by a hepatotoxic plant were selected on the basis of positive identification of the plant and characteristic histological lesions. Two cases of poisoning by Lasiospermum bipinnatum, 1 of Lantana camara, 2 of Xanthium strumarium, 1 of a Cestrum spp., and 1 of a Senecio spp. were included. The case of salmonellosis had characteristic histological lesions verified by bacterial culture.
Diagnostic testing
Real-time reverse transcription polymerase chain reaction
Nucleic acid extractions and real-time RT-PCR were performed by an immunized individual (M. Romito), under biosecure conditions, at the Biotechnology PCR Laboratory of the ARC-OVI. The assay was optimized for use with 200 µl blood or tissue lysate. RNA was extracted from approximately 0.1-cm3 liver macerated in tissue lysis buffer using an automated system a according to the manufacturer’s instructions. As previously described, 9 novel sense and antisense primer pairs and a 5′ hydrolysis probe, targeting conserved sequences on the G2 gene from the M segment of the viral genome were used. Reactions were carried out using a commercial kit, b which incorporates the polymerase enzyme that has both reverse transcription and DNA-polymerase activity. All reactions were carried out on 96-well plates in a real-time thermocycler, c and each plate included RVFV-positive controls (Smithburne and/or Clone 13 vaccine strain d ), an unrelated RNA control, and a nontemplate control reaction. Each 10-µl reaction consisted of 0.6 µl Mn(OAc)2, 3.7 µl of 2x hydrolysis mix, b 0.5 µl of Enhancer, b 5 pmol of each primer, 1 pmol 5′-FAM and 3′-BHQ-3–labeled probe, 2 µl of nucleic acid extract, and as much water as needed. The cycling profile PCR was run on an automated system c and involved reverse transcription at 63°C for 3 min, initial denaturation at 95°C for 30 sec, and 45 cycles of 95°C for 10 sec and 60°C for 30 sec. Fluorescence was read at the combined annealing–extension step and results analyzed using the supplied system software. c Cases were dichotomized as either positive or negative for RVF. Cases where the test result was suspect or inconclusive (cycle count >35.00 or an atypical amplification curve; i.e., not logistic), but still regarded as suspicious for RVF, were classified as positive.
Histopathology
Formalin-fixed, paraffin-embedded liver tissue was cut into 4-µm thick sections and stained with hematoxylin and eosin (HE). Prior to examination of the sections the original laboratory number of each slide was covered with a blank label, thoroughly mixed on a table top and each allocated a new sequential index. Slides were examined by the principal investigator under light microscopy and classified as RVF-positive or -negative based on the presence or absence of characteristic lesions. The researcher was blinded to specimen origin and results of other testing. The severity of autolysis was qualitatively graded as mild, moderate, or severe, and the presence or absence of tissue pigments (i.e., acid hematin, hemosiderin, bilirubin, and lipofuscin) was recorded. Mild autolysis was defined as cases where morphological detail within all the cells and histological structures of the liver was distinct. Moderate autolysis denoted cases where cytoplasmic borders were still distinct, but some cells showed fading of nuclei or stained uniformly eosinophilic; putrefactive bacteria were present. Severe autolysis was defined as cases that showed complete loss of basophilia and where cells had indistinct cytoplasmic borders, nuclei had faded completely, all the red blood cells had lysed, and many putrefactive bacteria were present.
Immunohistochemical labeling
Polyclonal hyperimmune mouse ascitic fluid e to RVFV was used as the primary antibody for the RVFV immunolabeling. Polyclonal hyperimmune mouse ascitic fluid e was used as primary antibody for Wesselsbron virus (WBV) immunolabeling. Rabbit polyclonal antiserum f raised against the Ab4 (neuropathogenic) strain of EHV-1 and monoclonal hyperimmune mouse ascitic fluid to Bovine herpesvirus 1 (BHV-1) g was used as primary antibodies for the EHV-1 and BHV-1 immunolabeling, respectively. Twenty of the control specimens were labeled with antibody against RVFV while the remaining 20 were labeled with antibody against EHV-1, BHV-1, or WBV (Table 1).
Sections from the 380 field cases and 40 samples selected for randomization among the 2010 Rift Valley fever (RVF) outbreak cases were variously labeled with relevant or irrelevant antibodies, as indicated, and randomly dispersed among the field cases.*

RVFV = Rift Valley fever virus; EHV-1 = Equid herpesvirus 1; BHV-1 = Bovine herpesvirus 1; WBV = Wesselsbron virus; RVF = Rift Valley fever; IBR = infectious bovine rhinotracheitis; WBD = Wesselsbron disease.
Mouse polyclonal antiserum to RVFV.
Rabbit polyclonal antiserum to EHV-1.
Mouse monoclonal antiserum to BHV-1.
Mouse polyclonal antiserum to WBV.
The RVFV antibody was an irrelevant antibody for all these cases.
Four micrometer–thick tissue sections were cut from formalin-fixed, paraffin-embedded tissues and mounted on positively charged microscope slides. h Immunohistochemical labeling was performed manually following previously described methods. 23 Briefly, the standard immunoperoxidase method included routine deparaffinizing with 2 changes of xylene, rehydration through graded alcohol baths to distilled water, and incubation with 3% hydrogen peroxide. This was followed by heat-induced epitope retrieval (HIER) or enzymatic digestion (Table 2), followed by blocking of nonspecific epitopes and incubation with the primary antibody. The avidin–biotin complex (ABC) immunoperoxidase detection system was used to detect target antigens in sections treated with anti-RVFV, anti–BHV-1, and anti–EHV-1 serum. Sections were sequentially incubated with the secondary antibody and the peroxidase-conjugated avidin. To visualize WBV antigen, a polymer detection system was used. i Following incubation with the primary antibody, sections were incubated with the Novolink polymer. For the purpose of contrasting the brownish tissue pigments (especially bile, lipofuscin, or hemosiderin in liver sections) all sections were immersed in a NovaRED substrate. j Sections were then counterstained with Mayer hematoxylin, routinely dehydrated through increasing alcohol concentrations and xylol, mounted using entellan, and coverslipped. The same randomizing and labeling procedure described for the HE slides were performed independently for the immunolabeled slides. Slides were examined by the principal investigator for positive labeling using a light microscope and results were dichotomized as either positive or negative for RVF.
Antibody reagents, antigen retrieval, and detection systems used in immunohistochemical labeling.*
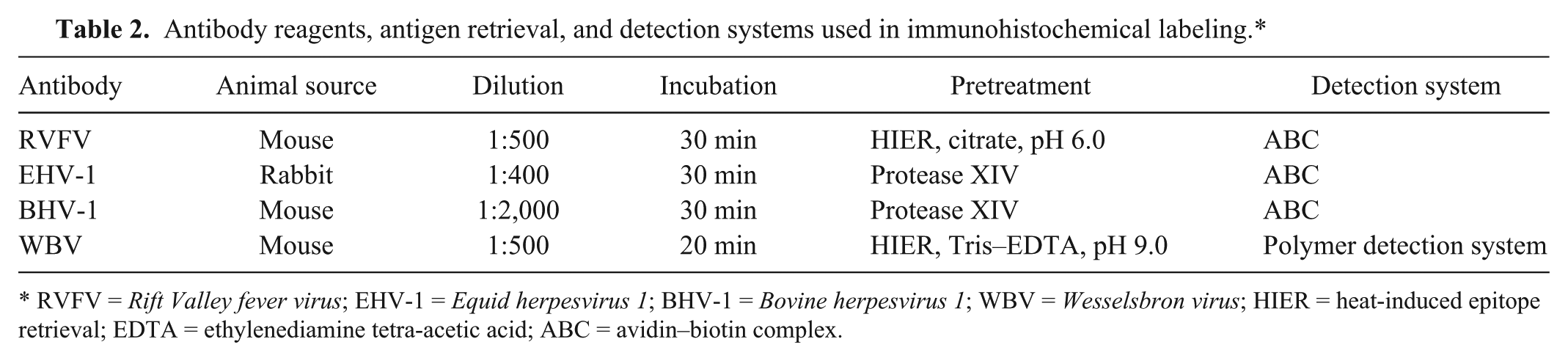
RVFV = Rift Valley fever virus; EHV-1 = Equid herpesvirus 1; BHV-1 = Bovine herpesvirus 1; WBV = Wesselsbron virus; HIER = heat-induced epitope retrieval; EDTA = ethylenediamine tetra-acetic acid; ABC = avidin–biotin complex.
Statistical analysis
Diagnostic sensitivity (Se) and specificity (Sp) of real-time RT-PCR, histopathology, and IHC were estimated in a latent-class model using a Bayesian framework.2,12,18,25,29 The basic statistical model was constructed for 3 conditionally independent tests and a single sampled population. 2 Conditional independence for the 3 tests was assumed as they were each based on a different biological principle. 29 Histopathology relies on the observation of histological lesions using light microscopy. An indirect ELISA based on the recombinant N nucleoprotein 17 showed that the antibody used in the immunoperoxidase techniques detects epitopes on the RVFV nucleoprotein, as opposed to PCR that detects conserved sequences on the M segment of the viral genome.
Beta prior probability distributions were elicited for unknown parameters (Table 3). A non-informative prior probability (i.e., uniform distributions in which all values between 0 and 1 are equally likely) was used to model prevalence since data concerning sample prevalence was not available. Sensitivity and Sp prior values for real-time RT-PCR were modeled according to the previous experience of one of the authors (M. Romito) with expertise using this assay. The most probable value for Se was determined to be 0.98 (assumed to be the median), while the expert was 95% sure that it was at least 0.95. The most probable Sp value of real-time RT-PCR during an epidemic was considered to be 0.78 and it was thought to be at least 0.60 with 95% certainty. Published information concerning the diagnostic accuracy of histopathology and IHC is limited. Therefore, the prior probability distribution for Se and Sp of histopathology was extrapolated from the results obtained from the 40 control cases. The most probable value for Se was determined to be 0.88 (7/8) and alpha and beta of the distribution were adjusted to equal the sample size (n = 8) forming the basis for this estimation. The prior distribution for Sp was based on an estimate of 0.86 (18/22) and the alpha and beta adjusted based on the sample size (n = 22). Immunohistochemical labeling correctly classified all control cases and therefore, noninformative prior probabilities were employed.
Beta prior probability distributions for unknown parameters in the Bayesian model used to estimate the diagnostic test accuracy and prevalence of real-time reverse transcription polymerase chain reaction (real-time RT-PCR), histopathology, and immunohistochemical labeling (IHC) to detect Rift Valley fever virus in liver specimens.
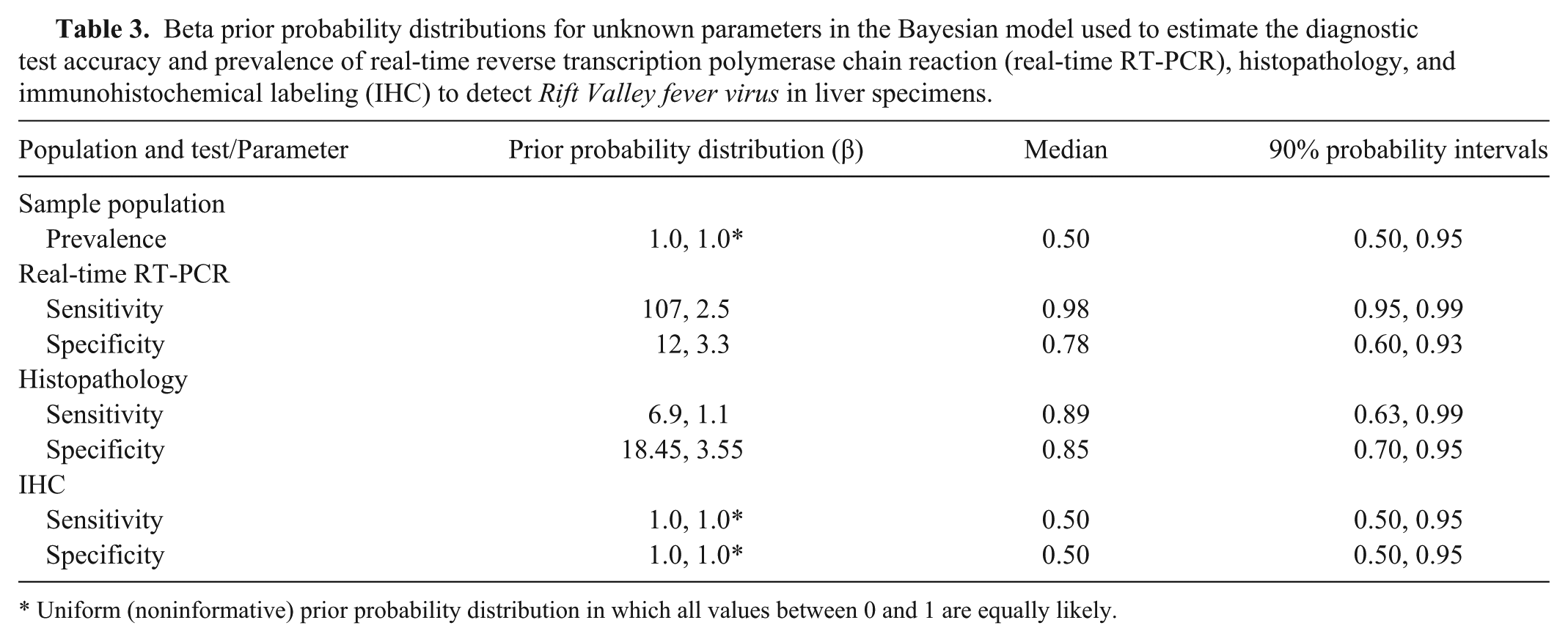
Uniform (noninformative) prior probability distribution in which all values between 0 and 1 are equally likely.
Diagnostic test accuracy was estimated by Markov chain Monte Carlo methods, utilizing available statistical software. k The first 100,000 iterations were discarded as the burn-in, and inferences were made based on the subsequent 20,000 iterations. Unstratified and stratified analyses were performed in respect to species (cattle or sheep), age category (adult, neonate, or fetus), level of autolysis (mild, moderate, or severe), and the presence or absence of tissue pigments (hemosiderin, bilirubin, or lipofuscin).
The convergence between 2 chains was assessed using the Gelman–Rubin statistic available in the software. k Autocorrelation among iterate values was assessed, and only every fifth value was retained to reduce the impact of this correlation. Medians and percentiles (2.5th–97.5th) of the posterior distributions were used as the point estimates and credibility intervals (CIs), respectively. A sensitivity analysis was conducted in which noninformative prior probability distributions were added for real-time RT-PCR and histopathology.
Results
Descriptive results
A total of 400 animals from the 2010 RVF outbreak were selected for study and included 127 cattle (74 adults, 21 neonates, 32 fetuses) and 273 sheep (137 adults, 68 neonates, 68 fetuses). Liver could not be detected in 20 of the HE-stained and/or immunolabeled sections. Complete test results, therefore, were available for a total of 380 animals and included 119 cattle (71 adults, 20 neonates, 28 fetuses) and 261 sheep (130 adults, 65 neonates, 66 fetuses).
Forty-nine percent (188/380) were positive on real-time RT-PCR, histopathology, and IHC whereas 33% (124/380) were negative on all assays (Table 4). Sixty-six percent (79/119) of cattle were positive on real-time RT-PCR, 54% (64/119) on histopathology, and 51% (61/119) on IHC. For the sampled sheep, percentages positive on real-time RT-PCR, histopathology and IHC were 68% (178/261), 56% (147/261) and 55% (144/261) respectively.
Cross-classified test (T) outcomes from real-time reverse transcription polymerase chain reaction (real-time RT-PCR), histopathology, and immunohistochemical labeling (IHC) used to detect Rift Valley fever virus in liver specimens.
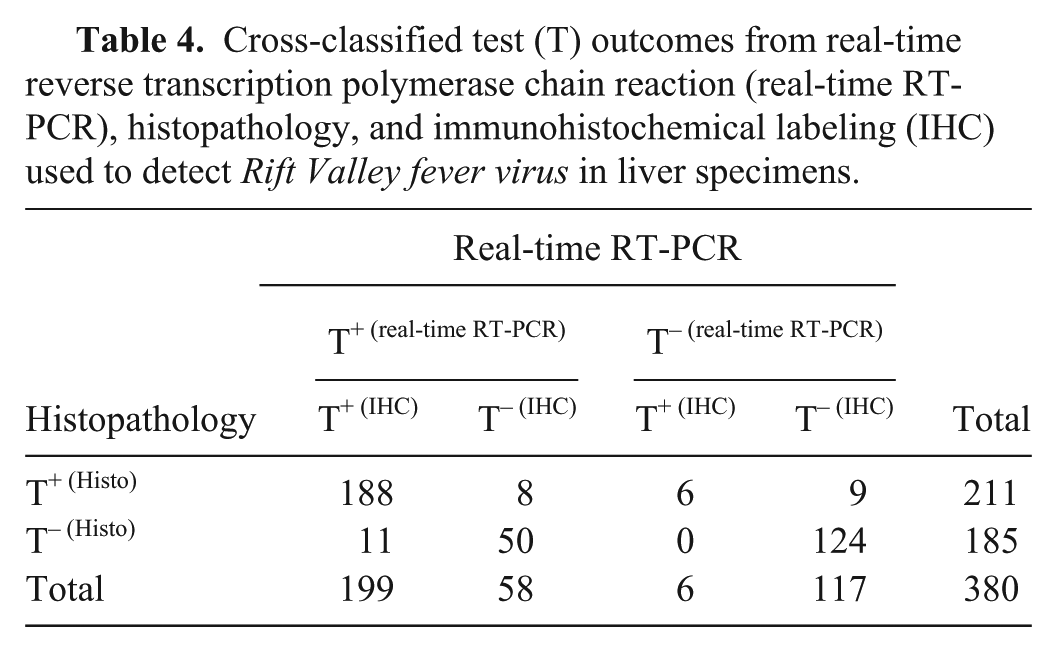
The estimated prevalence in the sample population was 52.7% (95% CI of 47.7–57.8%). Stratified analysis suggested a slightly higher prevalence in sampled sheep (56.8% with a CI of 50.5–62.9%) compared to cattle (51.1% with a CI of 42.1–60.1%). Prevalence was 49.6% (CI of 42.4–56.8%) in adult animals, 56.9% (CI of 46.3–67.3%)in neonates, and 64.5% (CI of 54.2–73.9%) in fetuses (Table 5).
Estimates of Rift Valley fever diagnostic test accuracy and prevalence in cattle and sheep in which the true infection status was unknown, as determined by using a Bayesian approach. The unstratified sample population and stratified populations were independently modeled and analyzed to allow for the estimation of stratum-specific values of diagnostic test accuracy.*
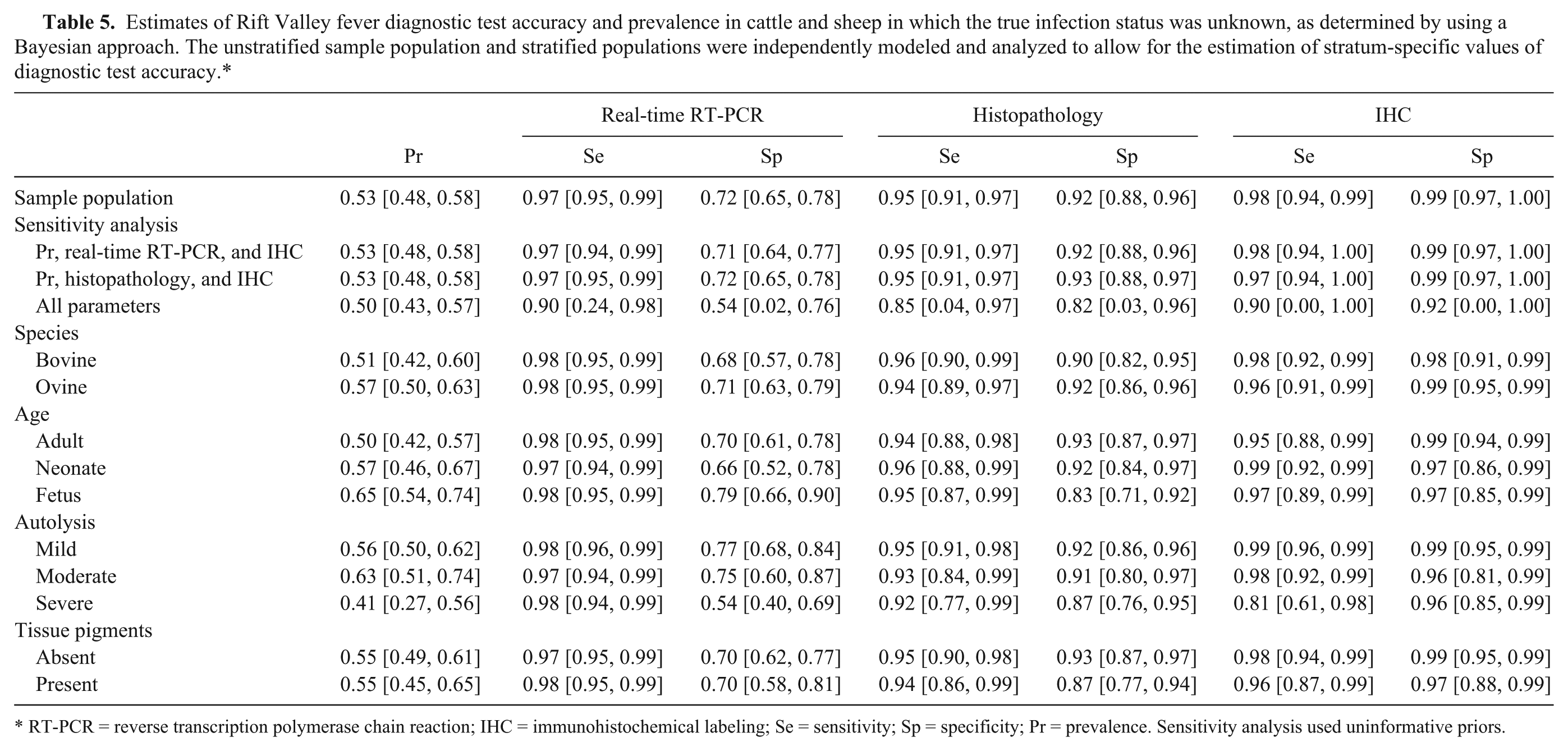
RT-PCR = reverse transcription polymerase chain reaction; IHC = immunohistochemical labeling; Se = sensitivity; Sp = specificity; Pr = prevalence. Sensitivity analysis used uninformative priors.
Eighty percent (35/40) of the control specimens stained with HE were correctly classified. Lesions detected in the cases of plant hepatotoxicosis, EHV-1, and salmonellosis were correctly identified as inconsistent with RVFV infection. Histopathology incorrectly classified 3 of the 4 IBR cases and 1 of the 10 WBD cases as positive for RVFV infection. One of the 2009 RVF cases was incorrectly classified as WBD. Immunohistochemical labeling correctly classified all of the control cases.
Sensitivity analysis
The model with noninformative priors for all unknown parameters would not converge (Table 5). Independent models for the sensitivity analysis with non-informative priors for real-time RT-PCR and IHC and then histopathology and IHC were both able to converge. Estimates from these models for prevalence, Se and Sp varied by 1% or less compared to results obtained when informative priors were used for real-time RT-PCR and histopathology.
Real-time reverse transcription polymerase chain reaction
The Se and Sp of real-time RT-PCR were 97.4% (95% CI = 95.2–98.8%) and 71.7% (95% CI = 65–77.9%) respectively. Stratified analysis suggested that the Se of real-time RT-PCR did not vary by evaluated categories (varied by 1–2% relative to the Se estimate for the sample population). Specificity was slightly more variable with the lowest value in severely autolyzed specimens (54.3% with 95% CI of 40.3–68.5%). Values for Sp in the age and tissue pigment strata varied by only 1 to 2% relative to the values obtained for the sample population.
Histopathology
The Se of histopathology for the sample population was 94.6% (95% CI = 91–97.2%). Sensitivity also remained within a narrow limit in all evaluated strata and only varied by 1–3% relative to the overall Se estimate. Generally, the morphology of the lesions was very distinctive. Lesions were similar in cattle and sheep but the extent of liver involvement varied with age. In adult animals, foci of hepatocellular death were random (Fig. 1A) and could be multifocal, focally extensive, or confluent bridging. Neonates and fetuses tended to present with cell death involving almost all hepatocytes (Fig. 1B). Liver injury was generally accompanied by hemorrhage and a mild to moderate infiltrate of neutrophils, lymphocytes, and Kupffer cells (Fig. 1C). Scattered hyperplastic Kupffer cells were also present within the sinusoids. A mild infiltrate of mononuclear inflammatory cells in the portal tracts (Fig. 1C) and involvement of hepatocytes within the limiting plate (Fig. 1A) was also a common finding. Affected hepatocytes displayed features of apoptosis (Fig. 1C) that included dissociation of cells, with shrinkage and rounding, hypereosinophilic cytoplasm, and karyorrhexis or karyolysis. Apoptotic bodies were identified as small cytoplasmic fragments (Fig. 1C). Destruction of the bile ducts was not observed. Surviving hepatocytes usually had varying degrees of micro- or macrovesicular degeneration and anisokaryosis. A very distinctive feature in many cases (especially in neonates and fetuses) was randomly located foci of well circumscribed cytolysis (Fig. 1B). These foci varied in size and number and contained dense aggregates of hepatocellular debris, degenerate neutrophils, and scarce hyperplastic Kupffer cells. Rod to oval-shaped or round eosinophilic intranuclear inclusions were occasional observed in injured hepatocytes. However, inclusion bodies were often difficult to detect and less common in the livers of adult animals compared to fetuses or neonates.
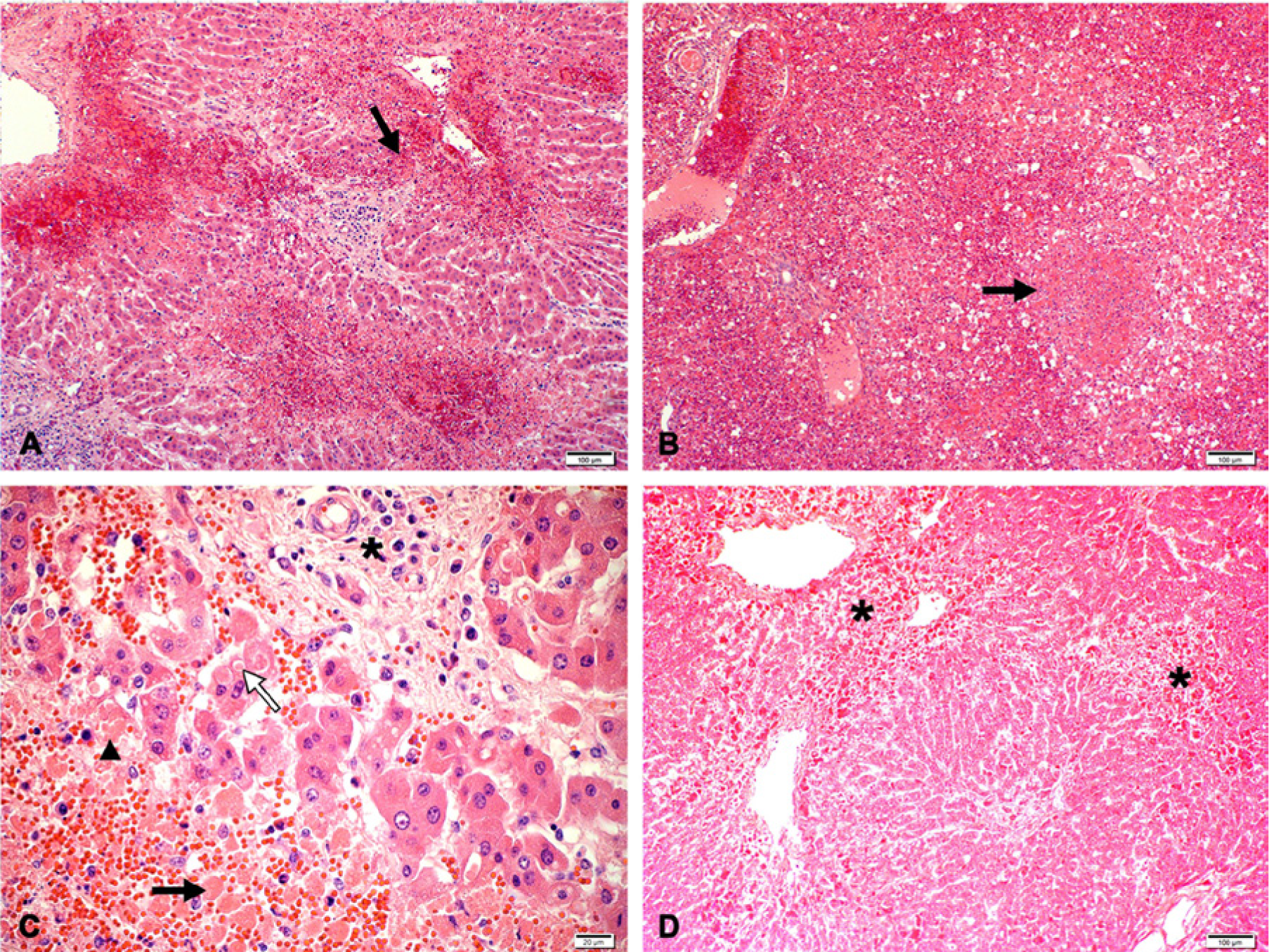
Hematoxylin and eosin–stained sections showing lesions typical of Rift Valley fever virus infection in the liver of cattle and sheep.
The Sp of histopathology was 92.3% (95% CI = 87.6–95.8%) overall and slightly more variable among strata with the lowest value in fetuses (83% with 95% CI of 71.3–91.6%). Lesions in some fetuses were more subtle, and included cellular dropout, scattered small round, disassociated cells, and marginated chromatin in the nuclei. The absence of typical eosinophilic intranuclear inclusions or foci of intense cytolysis in some cases made a diagnosis difficult.
Severe autolysis and the presence of tissue pigments had minor influences on the Sp of histopathology causing a decrease of 4.9% (from 92.3% to 87.4%) and 5.5% (from 92.3% to 86.8%) respectively. The presence of severe autolysis (Fig. 1D) obscured morphological details within the cells and histological structures in the liver so that apoptosis was not always easy to identify.
Immunohistochemical labeling
The overall Se and Sp of IHC was 97.6% (95% CI: 93.9–99.8%) and 99.4% (95% CI: 96.9–100%) respectively. With the exception of specimens with severe autolysis, both Se and Sp of IHC remained within a narrow limit in all strata and only varied by 1 to 4% relative to the Se estimate for the sample population.
Immunolabeling results indicated that hepatocytes are the predominant target of RVFV infection in the liver (Fig. 2A). Fine diffuse to coarse granular labeling was observed in the cytoplasm of apoptotic hepatocytes, and immunolabeled cytoplasmic fragments were frequently detected within the sinusoids and central veins. Areas of hepatocellular death seem to become devoid of immunolabeling with lesion maturity (Fig. 2B). Positively labeled Kupffer cells and degenerate neutrophils were sparse and convincing labeling of endothelial cells could not be detected. Immunolabeling was not detected in nuclei, remnants of nuclei, or in association with intranuclear inclusion bodies (Fig. 2C). Sensitivity decreased to 81.5% (95% CI = 60.1–97.5%) when specimens were severely autolyzed. Sixty-five cases were classified as severely autolyzed on histological examination, and 23 of the cases demonstrated positive immunolabeling that varied from sparse to widespread (Fig. 2D).
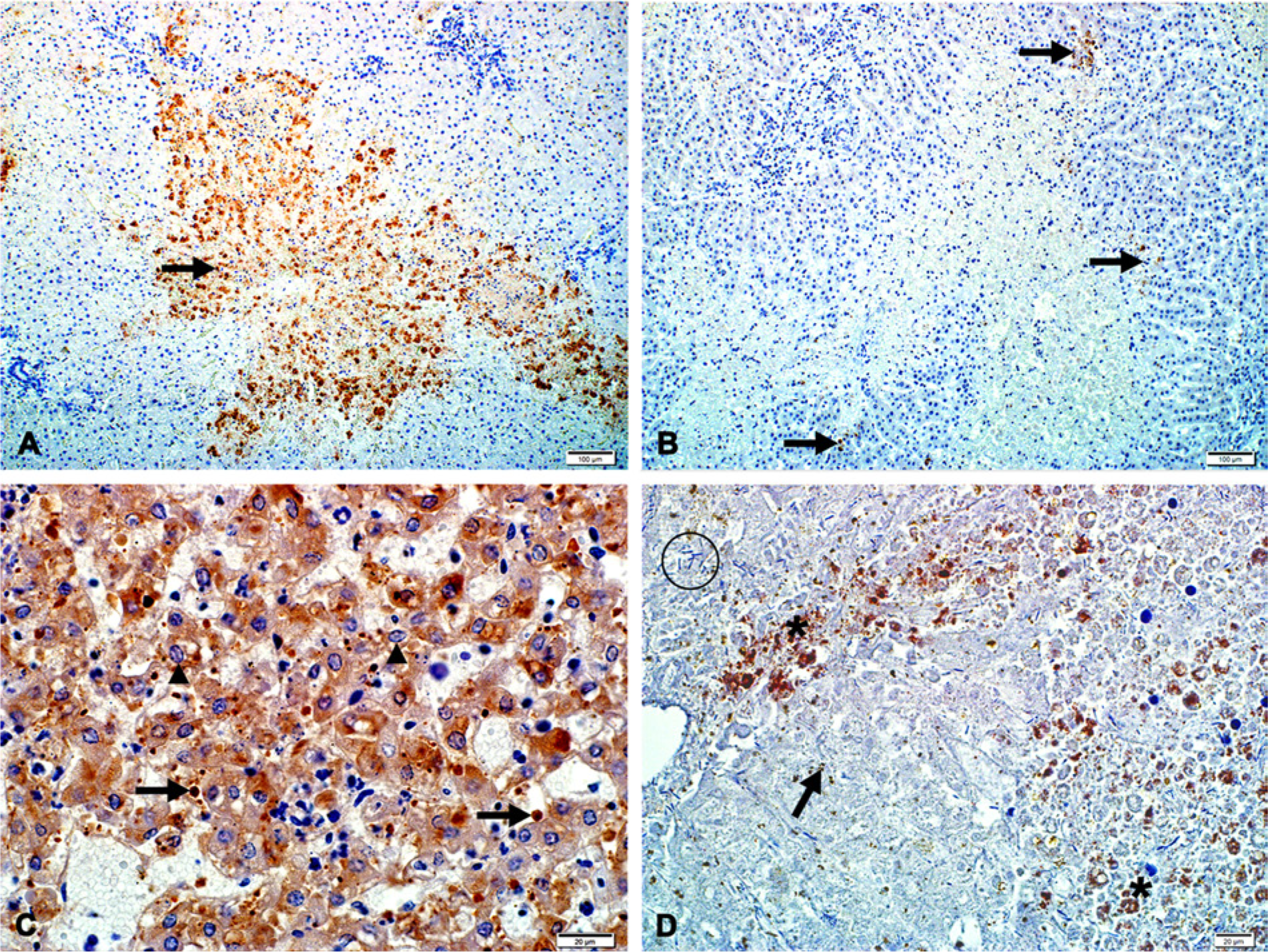
Rift Valley fever virus antigen labeling in the liver of cattle and sheep.
Discussion
A definitive diagnosis of RVF is challenging for a number of reasons. Primarily, the virus poses a health risk to laboratory personnel and routine diagnostic laboratories are precluded from performing diagnostic methods such as virus isolation, hemagglutination inhibition, polymerase chain reaction, or virus neutralization tests prescribed by the OIE.16,32 Specimens that might contain infective virus particles should only be tested in biosecure reference laboratories, and preferably, by immunized personnel. 20 In addition, these tests often require expensive equipment, highly skilled laboratory technicians and continued support from companies that supply the materials and consumables required for the test. 20 A laboratory safe antigen detection ELISA employing a recombinant nucleocapsid protein has been developed 20 and approved by the OIE. Low sensitivity (70%), however, suggests that this test is unsuitable for use during an epidemic. A further difficulty is that most of the recommended diagnostic techniques also require suitable fresh specimens to be collected and transported in a secure and contamination-free manner. 16 Suitable fresh specimens are often not available because the diagnosis was not suspected when the gross examination was conducted or formalin-fixed specimens are more conveniently dispatched over long distances.
In January 2010, an impending RVF outbreak in South Africa was suspected following exceptionally heavy rains, extremely high mortality rates among young lambs and gross and microscopic lesions typical of RVF. The diagnosis was confirmed using real-time RT-PCR and IHC and other diagnostic techniques were rarely used since they were either too lengthy to perform, too expensive or offered no advantage over the use of real-time RT-PCR and IHC. The present study attempted to estimate the diagnostic accuracy of histopathology, real-time RT-PCR and IHC using field samples and a Bayesian latent class approach.
Under the conditions of the current study, real-time RT-PCR was a highly sensitive assay for the detection and quantification of RVFV in liver specimens. The Se of real-time RT-PCR was 97.4% and only 6 cases (1.6%) tested negative for real-time RT-PCR but positive on both histopathology and IHC. However, the extraordinary analytical sensitivity of PCR makes this test very susceptible to false positive reactions 3 thus leading to an apparent reduced specificity. The Sp of real-time RT-PCR was 71.7% and 13% of cases (50/380) tested positive for real-time RT-PCR and negative on both histopathology and IHC. A cause for concern during large-scale disease outbreaks, where abortion storms are characteristic, is the risk for false positive reactions, due to viral contamination at sampling sites and equipment in the field, postmortem halls, and testing laboratories. Veterinary staff and the capacity of postmortem examination facilities may be overwhelmed during an outbreak and many carcasses of animals that originate from different farms may be examined simultaneously in the same room. Another concern is that the procedures of sampling in the field and submission to the laboratory are often inappropriate, with excessive amounts of material being submitted for testing, often in inadequate containers not suitably leakproof. Additionally, the processing of tissue specimens for nucleic acid extraction is tedious and prone to contamination risks and high levels of PCR products (amplicons) generated by large numbers of positive specimens can contaminate other specimens, reagents, or work surfaces at the testing laboratory. This is particularly so because extremely high titers of virus can be present in tissue samples.19,20
The Se (94.6%) and Sp (92.3%) of histopathology was higher than expected. Nine of the field cases (2.4%) were classified as positive on histopathology but tested negative on real-time RT-PCR and IHC. Single cases of RVF may be confused with acute poisoning with plants such as Senecio spp., Crotalaria spp., Lasiospermum bipinnatum, Cestrum spp., Pteronia pallens and Hertia pallens, and Microcystis aeruginosa, as well as bacterial septicemias, including pasteurellosis, salmonellosis, and anthrax. 28 Other abortifacient agents include Brucella spp., Leptospira spp., Chlamydia spp., Campylobacter spp., and Coxiella burnetii infection but these conditions can be excluded with histological examination of the liver, bacterial culture, and toxicological or serological investigation. 28 Two other important differentials that might prove difficult to exclude using histopathology alone are WBD in sheep and IBR in bovine fetuses or neonates. Three of the 4 IBR cases and 1 of the 10 WBD cases were incorrectly classified as positive for RVF and 1 of the 2009 RVF cases was classified as WBD. Microscopically, foci of hepatocellular injury in IBR cases may resemble the foci of cytolysis described for early cases of RVFV infection, and acute severe cases of WBD in sheep can resemble RVF.
Eleven of the field cases (2.9%) were classified as negative on histopathology but tested positive on real-time RT-PCR and IHC. A possible explanation is that early in the course of the disease (18 hr or less postinfection), only isolated hepatocytes show degeneration or death, 10 and typical apoptosis and hemorrhage with a mild mixed inflammatory infiltrate is more readily identified as the disease progresses. The 11 cases were reexamined and hepatocellular injury with or without inflammation was detected in 8 cases but the pattern of cell injury or the type of inflammatory reaction was considered inconsistent with previous descriptions of the histopathology associated with RVFV. Other diseases such as Escherichia coli septicemia, Clostridial spp. enteritis or mastitis, Haemonchus contortus parasitemia, or infection with a variety of arthropod-borne diseases such as bluetongue disease, C. burnetii, or Anaplasma spp. are not uncommon during the rainy season in South Africa; coinfections with these disease agents might explain why histopathology associated with RVFV was not apparent in some of the specimens. Sampling error might also be a contributing factor. Frequently only one section of liver is submitted for testing. However, RVFV does not affect the liver uniformly and it is possible that a specimen might have been taken from a part of the liver where typical histological lesions had not yet developed or where liver injury had advanced to the point where antigen could no longer be detected using IHC. At least 2 cases were detected where 2 or more sections from the liver were examined revealing strong positive immunolabeling in one section with typical histological lesions but no lesions or positive immunolabeling in other sections.
The Se (97.6%) and Sp (99.4%) of IHC were quite high and none of the field cases were classified as positive for IHC but tested negative on real-time RT-PCR and histopathology. Additionally, all control cases were correctly classified. The high Sp of IHC is due to the fact that the characteristic positive immunolabeling of the cytoplasm of hepatocytes can be correlated with the presence of cytopathology typical for RVFV infection. A previous study 30 investigated the distribution of viral antigen in 12 newborn lambs infected with RVFV and detected positive immunolabeling in 11 of the lambs. Only small groups of injured hepatocytes labeled positive for RVFV in lambs euthanized early in the course of the disease whereas viral antigen was dispersed throughout the liver lobules in lambs euthanized later during the course of disease.
In contrast, 2% of the field cases (8/380) were classified positive on histopathology and real-time RT-PCR but tested negative on IHC. False-negative results are sometimes obtained with IHC due to advanced autolysis and the loss of antigenic epitopes. 22 The 8 cases were re-examined and 6 of them were found to be severely autolyzed, which would support the presumption that antigenic epitopes become lost or too widely dispersed within cells because of disintegration of organelles or vesicles where viral proteins normally transit. False negative results are also reported to occur with prolonged fixation of specimens in formalin with a pH of 6 or less because those conditions cause the formation of acid hematin pigment. 22 However, there was no convincing evidence that excessive acid hematin pigment caused false negative IHC results. Twenty-two percent of the cases (84/380) had substantial acid formalin and 52 of these cases were positive, and 24 negative, on all three tests. Only 9 had inconsistent test results, and 7 of these were severely autolyzed. Sampling error or reader error could also explain false negative IHC results. Viral proteins might be absent from completely destroyed sections of liver or scant positive immunolabeling can be missed, which is likely to increase when case loads are high or when inexperienced personnel evaluate slides.
The stratified analysis suggested differences in test accuracy in fetuses and severely autolyzed specimens. The Sp of histopathology in fetuses (83%) was 9.3% lower than the value obtained for the sample population (92.3%). Five of the 9 field cases that were classified as positive on histopathology but tested negative on real-time RT-PCR and IHC, were fetuses. This emphasizes the need to screen aborted fetuses in areas endemic for RVF using a variety of tests because a definitive diagnosis based on histopathology alone can be difficult when lesion are subtle.
In severely autolyzed specimens, the Se of IHC decreased by 16.1% and the Sp of real-time RT-PCR by 17.4%. Twenty of the 65 cases (54%) classified as severely autolyzed tested positive on all 3 tests while 15 tested negative on all 3. Sixty-three percent (19/30) of the incorrectly classified cases were negative on histopathology and IHC but positive on real-time RT-PCR. There is no plausible biological explanation for this decrease in the Sp of real-time RT-PCR. In fact, it would appear that RNA of RVFV is resistant to degradation in autolyzing tissues. To mimic clearance of nucleocapsid protein in decomposing tissues from RVFV-infected animals, sheep and bovine RVFV spiked liver homogenate were incubated at 37°C for a period of 8 days and tested at intervals with a sandwich ELISA developed for the detection of nucleocapsid protein of RVFV. 31 The ELISA detected antigen in spiked bovine and sheep liver homogenates up to at least 8 days. Conversely, the antibody used to detect RVFV using IHC detects epitopes raised against nucleoproteins of the virus and it is possible that viral proteins become too widely dispersed to detect by light microscopy. The assumption is that a marked decrease in Se of histopathology and IHC in severely autolyzed specimens caused an apparent decrease in Sp of real-time RT-PCR. Therefore, IHC results from severely autolyzed specimens should be interpreted with caution.
No attempt was made to estimate an unbiased population prevalence because the study was designed to estimate diagnostic accuracy. Therefore, no inferences can be drawn concerning the true prevalence of RVF during the 2010 outbreak in South Africa. However, the estimated prevalence obtained (52.7%) suggests that veterinarians or animal health technicians readily recognized the macroscopic lesions caused by RVFV and selected cases for testing accordingly. Seemingly, the typical lesions in sheep (56.8%) and neonates (56.9%) were more readily identified than in adult cattle (49.6%), which is in agreement with the findings of previous research.4–7,10,11,14 Selection of cases from fetuses based on the occurrence of abortion storms had the highest prevalence (64.5%).
A limitation in this study was the restricted availability of suitable control specimens in fetuses. Additional cases of IBR infection in bovine fetuses would have been preferable to cases of EHV-1 infection in equine fetuses. However, EHV-1 cases were substituted since this is the only disease causing hepatocellular injury in fetuses, for which an immunohistochemical test was developed at the faculty. It was further reasoned that fetal tissues of horses cannot be easily distinguished microscopically from those of cattle. The control cases that were used, however, served their intended purpose in this study, namely to compel the observer to motivate a decision of positive or negative for RVF in the case of histopathology or IHC circumspectly.
In conclusion, the high estimated Sp (99.4%) of IHC and the low Sp of real-time RT-PCR (71.3%) suggests that the definitive diagnosis or exclusion of RVF should not rely on a single PCR test and that IHC could be an effective confirmatory test for real-time RT-PCR–positive field cases necropsied during an epidemic. The high Se of real-time RT-PCR (97.4%) and IHC (97.6%) suggest that both tests would be appropriate screening tests. It is also likely that the diagnostic Sp of real-time RT-PCR would increase substantially during periods when fewer mortalities due to RVFV infection occur and the risk of cross contamination of specimens is markedly reduced in postmortem facilities or testing laboratories. Another advantage of real-time RT-PCR is that it can detect viremia in blood samples of in contact animals and animals, such as adult cattle and goats, which are less likely to die during the course of infection. However, most countries have a limited number of biosecure reference laboratories that are able to perform real-time RT-PCR, and the capacity of these facilities can be rapidly overwhelmed during an outbreak. The diagnostic Se and Sp of histopathology was higher than expected confirming the value of routine post mortem examinations and histopathology of liver specimens. Based on the availability of appropriate antibodies, the most feasible RVF testing option in countries that do not have suitably equipped laboratories, and where disease is often not diagnosed in livestock until after human cases, 20 would be routine histopathology screening with IHC confirmation. In this respect, an important advantage of IHC is that it reduces the human health risk during shipment because it is based on formalin-fixed specimens that will not contain infectious virus and therefore can be safely transported over long distances or even across international borders.
Footnotes
Acknowledgements
The authors want to thank many colleagues at the National Department of Agriculture, Fisheries and Forestry, Directorate of Veterinary Services for providing access to original diagnostic data. The authors are also grateful for colleagues at IDEXX Laboratories (Pretoria), Vetdiagnostix (Pietermaritzburg), and PathCare (Cape Town) for providing the samples.
a.
MagNA Pure LC Total Nucleic Acid High Performance Kit (catalog no. 05 323 738 001), used with a MagNA Pure LC system; Roche Diagnostics GmbH, Sandhofer, Mannheim, Germany.
b.
LC 480 One-step RNA Master (Hydrolysis Probes) Kit (catalog no. 04 991 885 001), Roche Diagnostics GmbH, Sandhofer, Mannheim, Germany.
c.
LightCycler 480 Real-Time PCR System, Roche Diagnostics GmbH, Sandhofer, Mannheim, Germany.
d.
Onderstepoort Biological Products, Pretoria, South Africa.
e.
National Institute for Communicable Diseases, Johannesburg, South Africa.
f.
Animal Health Trust, Newmarket, Suffolk, United Kingdom.
g.
Veterinary Medical Research and Development, Pullman, WA.
h.
SuperFrost Plus, Menzel-Gläser, Portsmouth, NH.
i.
Catalog no. RE7150-K, NovoLink Min Polymer Detection System; Leica Biosystems, Newcastle upon Tyne, United Kingdom.
j.
Catalog no. SK-4800, Vector Laboratories, Burlingame, CA.
k.
WinBUGS Version 1.4, MRC Biostatistics Unit, Cambridge, United Kingdom.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Departments of Paraclinical Sciences and Veterinary Tropical Diseases of the Faculty of Veterinary Science, University of Pretoria and the Institutional Research Theme (IRT): Biotechnology and the Management of Animal and Zoonotic Diseases of the University of Pretoria.
