Abstract
Infection of pregnant cattle with both species of Bovine viral diarrhea virus (BVDV) can result in reproductive disease that includes fetal reabsorption, mummification, abortion, stillbirths, congenital defects affecting structural, neural, reproductive, and immune systems, and the birth of calves persistently infected with BVDV. Accurate diagnosis of BVDV-associated reproductive disease is important to control BVDV at the production unit level and assessment of the cost of BVDV infections in support of BVDV control programs. The purpose of the current study was to examine the stability of viral nucleic acid in fetal tissues exposed to different conditions, as measured by detection by polymerase chain reaction. Five different types of fetal tissue, including brain, skin and muscle, ear, and 2 different pooled organ samples, were subjected to conditions that mimicked those that might exist for samples collected after abortions in production settings or possible storage conditions after collection and prior to testing. In addition, tissues were archived for 36 months at −20°C and then retested, to mimic conditions that might occur in the case of retrospective surveillance studies. Brain tissue showed the highest stability under the conditions tested. The impact of fecal contamination was increased following archiving in all tissue types suggesting that, for long-term storage, effort should be made to reduce environmental contaminants before archiving.
Infections with Bovine viral diarrhea virus 1 and 2 (BVDV-1 and -2; family Flaviviridae, genus Pestivirus) result in economic loss to producers worldwide.5,6,14 Frequently, infection of immunocompetent adults does not result in clinically apparent disease. However, fetal infection, particularly in the first one-third of gestation, can result in a number of clinically significant manifestations, including fetal reabsorption, mummification, abortion, and congenital defects affecting structural, neural, reproductive, and immune sytems.2,4,7–9 Prominent among congenital defects of the immune system is the establishment of lifelong persistent infections. Accurate diagnosis of BVDV-associated reproductive disease is important to the control of BVDV and the assessment of the cost of BVDV infections. Methods commonly used for diagnosis of BVDV include virus isolation (VI), immunohistochemical staining, antigen-capture enzyme-linked immunosorbent assay (ACE), and polymerase chain reaction–based amplification of viral genome (PCR). 3 The accuracy of these methods, when used to identify BVDV infection of fetuses, may be affected by the breakdown of fetal tissues during collection and storage.
Previously, the stability of BVDV antigen was examined, as measured by detection by ACE in ear punch samples collected from BVDV-infected bovine fetuses, 11 and it was found that exposure to light and/or drying conditions for 7 days did not affect the detection of viral antigen by ACE. The purpose of the current study was to examine the stability of viral nucleic acid in fetal tissues exposed to different conditions, as measured by detection by PCR. Polymerase chain reaction–based testing is offered by a number of diagnostic laboratories and commercial testing entities for screening for BVDV infections. Five different types of fetal tissue, including brain, skin and muscle, ear, and 2 different pooled organ samples, were subjected to different storage conditions. In addition, tissues were archived for 36 months at −20°C and then retested to mimic conditions that might occur in the case of retrospective surveillance studies.
Seven first calf heifers were determined to be negative for virus neutralizing titers in serum against BVDV and BVDV persistent infection using methods described previously. 11 Prior to breeding, 1 heifer was vaccinated with a modified live vaccine a per manufacturer’s label. Heifers were purchased from a commercial producer and before purchase were raised under commercial type cattle feeding conditions. Heifers were bred via artificial insemination as described previously. 11 After purchase, the dams were held in controlled outdoor facilities until inoculation with BVDV. After inoculation, the pregnant dams were individually penned in an indoor facility.
The isolation and characterization of the BVDV-1 strain CA0401186a, used for inoculation, has been described previously. 13 All of the dams were exposed to 5.4 logs TCID of BVDV-1 strain CA0401186a by the nasal route using a glass atomizer b as described previously. 11 The volume of the inoculation dose was 4 ml total, divided into 2-ml aliquots per nostril. The fetuses were between 84 and 86 days gestation at the time of challenge and 115–117 days of gestation at the time that they were harvested by C-section. After harvesting, fetuses were held on ice until processing. Approximately one-half of the thymus, 1 kidney, approximately one-half of the spleen, and the cerebellum were collected for VI. Virus isolation was performed as described previously. 11 The remaining brain, internal organs, ears, and the skin and muscle from one of the back legs were processed as follows. The internal organs were divided into 2 pools. Pool A consisted of the heart, lungs, and thymus; pool B consisted of the spleen, kidney, and intestines. Tissues were weighed and mixed 1:3 (weight:volume [w:v]) with ice cold sterile phosphate buffered saline (PBS) and homogenized with a hand-held homogenizer c in three 30-sec bursts. Ear samples were hand minced prior to homogenization. All other tissues were homogenized directly. Fecal matter, collected from one of the dams, was also mixed 1:3 (w:v) with ice cold sterile PBS and homogenized to produce a slurry. Tissues were homogenized in order to generate uniform samples for comparison of holding and storage conditions.
Duplicate 500-µl aliquots were placed into 24-well tissue culture plates. A 50-µl aliquot of the fecal matter slurry was added to one of the aliquots, and a 50-ml aliquot of PBS was added to the other. Plates were covered with an air-permeable clear membrane d and exposed to various environmental conditions for 7 days. Hereafter these conditions will be referred to as storage conditions. The reasons for choosing the different storage conditions are as follows: −80°C (standard storage temperature used for viral stocks and tissues by many research laboratories), 4°C (typical refrigerator setting), 23.3°C (hereafter referred to as room temperature; to mimic situations in which samples are left out on laboratory benches prior to processing), and 37°C (temperature at which most autolytic enzymes are active). Samples were spiked with fecal material, collected from one of the dams, to mimic environmental contamination that might occur when fetuses are aborted in cow lots. After testing at 7 days, all samples were stored frozen at −20°C for a period of 36 months, thawed, and then retested as described above, to mimic conditions that might be encountered in retrospective studies that use archived samples. Hereafter the holding of the samples for 36 months will be referred to as archiving and the samples held under these conditions will be referred to as archived samples.
A 140-µl aliquot, removed from each sample well, was used to prepare total RNA using a commercial kit e per the manufacturer’s directions. The final volume of RNA template solution was 60 µl. A 10-µl aliquot of this solution was used to amplify a 280 nucleotide region of the 5’– untranslated region by reverse transcription PCR as described previously. 10 Amplicons were visualized by staining with a fluorescent nucleic acid gel stain f following electrophoresis in 3% agarose gels. Amplicons were confirmed to be the virus used in inoculation by sequencing as described previously. 12
The proportion of correct tests results was compared using Fisher exact test for comparing proportions. 1 Proportions were compared between archived and nonarchived samples for each 7-day storage temperature, between different storage temperatures for each combination of archive and fecal status, and between contaminated and noncontaminated with fecal material for each storage temperature and archiving status. Evidence of significance was based on a P value of less than 0.05.
All tissues from the 6 fetuses, carried by the 6 exposed nonvaccinated heifers, were infected with BVDV based on VI. The fetus from the vaccinated exposed heifer was BVDV negative by VI. The results of PCR testing following different storage conditions are summarized in Figure 1. The BVDV status was correctly diagnosed in all 35 samples (30 positive and 5 negative) that were tested immediately after processing and in samples held for 7 days at either −80°C or 4°C regardless of fecal contamination. The quantity of PCR amplicon, based on gel electrophoresis, was similar between tissue types and between fetuses in samples that were tested immediately after processing (data not shown). In the absence of fecal contamination, the proportion of miscalls was not significantly different between archived and nonarchived samples (P < 0.05). However, when samples were contaminated with fecal matter, the proportion of correct calls of archived samples was significantly lower than with nonarchived samples (P < 0.05).
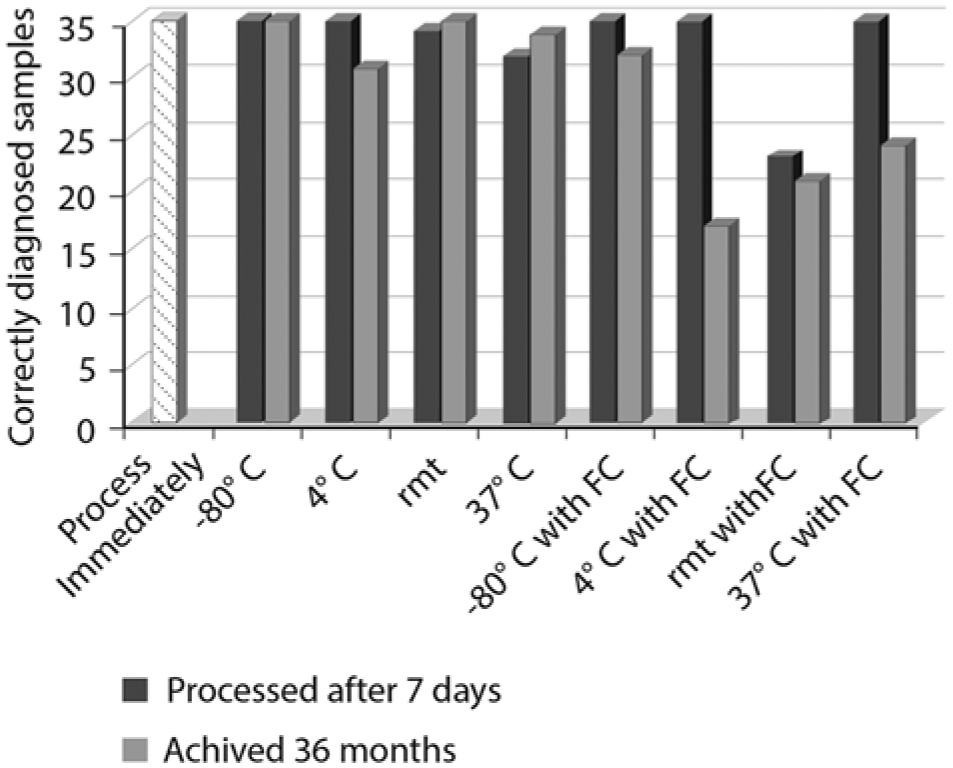
Comparison of rate of correct diagnosis of Bovine viral diarrhea virus in 35 bovine fetal tissues under different storage and archiving conditions. rmt = room temperature (23.3°C); FC = fecal matter contamination.
Overall, there were 96 tests run on each tissue type from fetuses tested positive for BVDV (6 positive fetuses, 4 storage conditions, contaminated and not contaminated with fecal matter, archived and nonarchived). Based on the proportion of samples detected as BVDV positive, the stability of the genomic sequences amplified by the PCR test was highest in brain tissue, followed in order by skin and/or muscle, ear, organ pool A (heart, lungs, and thymus) and finally organ pool B (spleen, kidney, and intestines; Fig. 2). Detection rate in the 5 tissue types ranged from 89 out of 96 (92.7%) for brain tissue to 77 (80.2%) out of 96 for organ pool B.
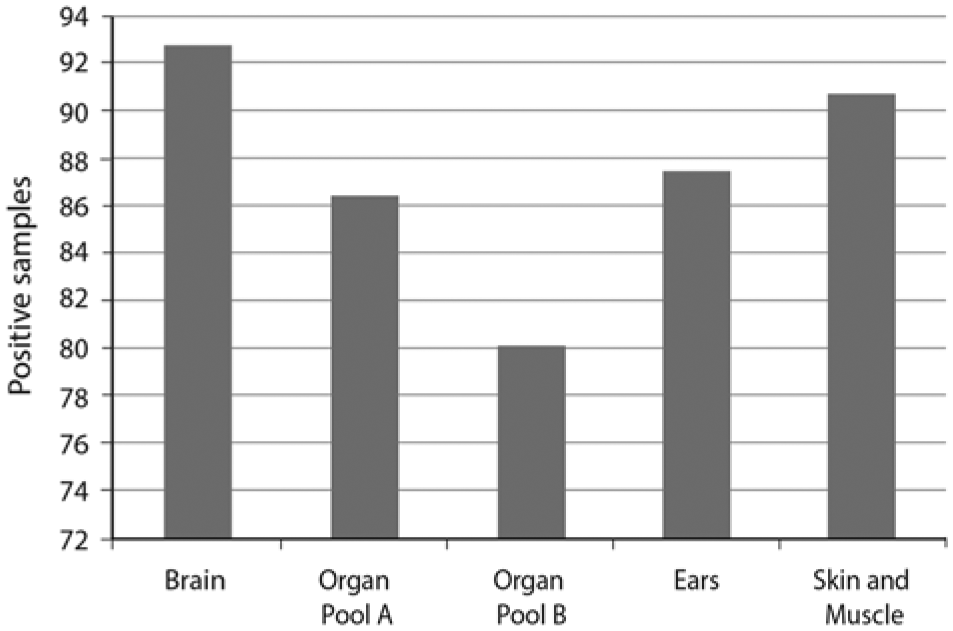
Comparison of detection of Bovine viral diarrhea virus in different bovine fetal tissue types.
While only 1 BVDV-negative fetus was available for testing, it did not appear that any of the storage conditions affected a negative diagnosis or that any of the 5 tissue types used was more prone to false positives or “no calls.” Storage conditions, the presence of fecal contamination, and archiving did, however, affect the rate of false negatives and “no calls” for positive samples. Not unexpectedly, the greatest accuracy was observed in samples that were stored at −80°C after processing, regardless of tissue type, fecal contamination, or archiving. The results demonstrated that archiving of samples at −20°C does not significantly reduce detection if the samples have not been previously contaminated with fecal material. It should be noted that the archived samples were thawed after storage at 20°C for 36 months and thus archived samples went through an extra freeze–thaw cycle compared with nonarchived samples. Many researchers limit freeze–thaw cycles for viral stocks in the belief that every freeze–thaw reduces the amount of viable virus in stocks. However, no decrease in band intensity was observed in any multiple freeze–thaw cycles of nonarchived samples (data not shown) suggesting that the reduced level of detection in these samples was due to archiving and not an extra freeze–thaw cycle. The data suggests that if samples are to be archived, care should be taken to remove contaminants before samples are placed in long-term storage.
Comparison of detection in the 5 tissue types used in the current study revealed that diagnosis of BVDV infection was most reliable using brain tissue. Outside of cases where nervous system congenital defects are suspected, brain samples are seldom submitted to diagnostic laboratories. While recovery of brain tissue may be slightly more difficult than dissecting out other tissues because the skull must be breached, there may be several advantages to using brain tissue for the purpose of diagnosing BVDV infections, in addition to stability of the viral genome. Because of the blood–brain barrier, brain samples are less likely to be contaminated with bacteria. Particularly in early gestation fetuses, brain tissue is in relative abundance and can be easily identified. Furthermore, after expulsion, encasement of the brain in the skull makes it less likely to be in contact with environmental contaminants.
In summary, the results of the present study suggest that fetal tissues are relatively stable under a number of different storage conditions. This suggests that if fetal tissue is available for testing, even if storage conditions have not been ideal, it is worth the effort to test. Furthermore, collection of brain should be considered in the testing strategy for fetal tissues, and effort should be made to reduce environmental contaminants on tissues before archiving.
Footnotes
Acknowledgements
The authors thank Patricia Federico and Kathryn McMullen for their excellent technical support, Harold Ridpath for statistical support, and Michael Marti for preparation of illustrations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
a.
Pyramid 5, Fort Dodge Animal Health, Fort Dodge, IA.
b.
Model 151, Devilviss Healthcare, Somerset, PA.
c.
Tissue-Tearor, BioSpec Products Inc., Bartelsville, OK.
d.
AirPore tape sheets, Qiagen tape sheet; Qiagen Inc., Valencia, CA.
e.
QIAamp viral RNA mini kit, Qiagen Inc., Valencia, CA.
f.
GelRed, Phoenix Research, Candler, NC.
