Abstract
Repeated polymerase chain reaction (PCR) testing of 3 asymptomatic domestic cats were positive for Cytauxzoon felis DNA, suggesting persistent infection. Two cats initially presented with clinical signs consistent with acute cytauxzoonosis and, in both cases, signs of illness resolved after treatment. Parasitemia was detected in peripheral blood smears from these cats upon presentation with illness and, at subsequent follow-up appointments, in the absence of clinical illness. Polymerase chain reaction analysis was positive for C. felis from blood sampled at each time point. A third cat, a housemate of a cat fatally infected with C. felis, was preventatively treated for infection at the time of the housemate cat's death. This contact cat, having never shown signs of clinical illness consistent with cytauxzoonosis infection, had no detectable parasitemia but was positive for C. felis on repeated PCR testing. Detection of asymptomatically infected cats allows for the possibility of a yet unrecognized population of infected domestic cats that may have the capacity to serve as an additional reservoir host for C. felis, altering the currently accepted paradigm of C. felis transmission to domestic cats through bobcats as the reservoir host. In cases of very low parasitemia, more sensitive means of parasite detection, such as PCR testing, may be necessary to detect infected cats. Increased detection of asymptomatically infected cats will aid in understanding the epidemiology of C. felis infection and enhance the ability to prevent this highly fatal infectious disease of domestic cats.
Cytauxzoonosis is an emerging, tick-borne, hemoprotozoal disease of domestic and exotic cats that has been characterized with an extremely high fatality rate. The causative organism, Cytauxzoon felis, is a relatively new pathogen in the United States that was first identified in Missouri in 1973. 16 Subsequently, this parasite has been reported to infect cats in rural and urban regions throughout the south-central, southeastern, and mid-Atlantic United States. 1,2,9,11,12,16,18 The life cycle of C. felis involves a tissue (or leukocytic) phase and an erythrocytic phase. Infected ticks transmit the parasite, which enters host mononuclear phagocytes and undergoes schizogony and binary fission to produce numerous developing merozoites. These infected mononuclear cells line the lumens of veins throughout the body and eventually rupture, releasing the merozoites into circulation. The free merozoites subsequently infect host erythrocytes, resulting in a parasitemia with intraerythrocytic piroplasms that are visible in routine Romanowsky-stained peripheral blood smears. The bobcat (Lynx rufus) has been identified as the natural reservoir host of the parasite. 3,5 In affected cats, the course of disease usually is rapid, and most cats die within 1 week of initial clinical illness. 4,7,10,18 The sporadic occurrence, short course of disease, and high fatality rate of cytauxzoonosis in domestic cats has suggested that they likely serve as incidental, dead-end hosts. 7,13
Although C. felis infection has historically been viewed as uniformly fatal in domestic cats, recent studies 6,14,15,17 have documented cats that have survived parasitic infection. Survival of infected cats allows for the possibility of persistent infections. One recent study identified 3 of 961 free-roaming, feral cats tested from trap–neuter–return programs that were positive for C. felis when tested by polymerase chain reaction (PCR). 8 Clinical signs of cytauxzoonosis were not noted at the time of their capture, although there is limited follow-up clinical information and laboratory testing on these cats to document persistent, subclinical C. felis infection. These findings suggest that, despite a low prevalence rate, apparently healthy, but C. felis–infected, cats exist and may have the capacity to serve as a new reservoir for parasitic infection. The present communication describes the clinical history and laboratory testing of 3 asymptomatic, persistently C. felis–infected domestic cats.
A 13-month-old, 3.9-kg, neutered male, indoor/outdoor, Domestic Shorthair cat from Hot Springs, AR, was referred to a private veterinary practice for lethargy, inappetence, and fever that had first been detected 6 weeks previously. On presentation, the cat was febrile (39.4°C), but the physical examination was otherwise unremarkable. The cat had a history of tick exposure and was not on tick preventative. Ethylenediamine tetra-acetic acid (EDTA)–anticoagulated whole blood was submitted to the Veterinary Diagnostic Laboratory of the Arkansas Livestock and Poultry Commission (ALPC; Little Rock, AR) where Wright-Giemsa–stained blood smears revealed intraerythrocytic piroplasms, morphologically consistent with C. felis. The cat was treated with imidocarb dipropionate a (5 mg/kg IM), following premedication with atropine b (0.04 mg/kg SQ) 30 min prior. Both treatments were repeated 1 week later. Oral enrofloxacin c (3 mg/kg PO q12h) was administered for 14 days. Clinical signs had completely resolved within 2 weeks of presentation, although persistent parasitemia remained. Blood smear review confirmed persistent parasitemia at 5, 10, and 15 months after initial illness, when the cat was clinically healthy (Fig. 1).
A littermate of the first cat, a 6.3-kg, neutered male, indoor/outdoor, Domestic Shorthair cat, living in the same home in Hot Springs, AR, presented to the same practice 3 months after the first cat was referred. The cat had a 24–48-hr history of decreased appetite, lethargy, and fever. The cat had no known history of tick exposure, although application of tick preventative by the owner was inconsistent. On presentation, the cat was febrile (40.6°C) and lethargic. EDTA–anticoagulated whole blood was submitted to ALPC, and Wright-Giemsa–stained blood smears revealed intraerythrocytic piroplasms morphologically consistent with C. felis. An IV catheter was placed, and a constant-rate infusion of Lactated Ringer's solution d was administered at 15 ml/hr for 48 hr. The cat was treated with imidocarb dipropionate a (5 mg/kg IM), following premedication with atropine b (0.04 mg/kg SQ) 30 min prior. Enrofloxacin c (3 mg/kg SQ q24h for 3 days, then 3 mg/kg PO for 11 days) was also administered. Ten days later, at which point clinical signs had resolved, the imidocarb dipropionate a treatment with atropine b premedication was repeated. Blood smear review performed at 2 and 7 months after the initial diagnosis of cytauxzoonosis revealed persistent parasitemia in an otherwise clinically healthy cat.
EDTA–anticoagulated whole blood samples drawn from the infected cats at each appointment were submitted for further study. Wright-Leishman–stained blood smear reviews of each sample confirmed hemoparasites that were morphologically consistent with C. felis. Parasitemia levels at initial diagnosis of cytauxzoonosis and then during follow-up varied between 0.1–0.6% for the first cat and between 0.1–1.0% for the second cat, as quantitated using a Miller ocular and examining 10 counting fields at 100X.
The third case was a 9-year-old, 4.5-kg, spayed female, Domestic Shorthair cat from Columbus, GA, that was prophylactically treated with a single dose of imidocarb dipropionate a (5 mg/kg IM) after another housemate cat died of cytauxzoonosis. Although this cat lived predominantly indoors, she did have close contact with several other indoor/outdoor cats, and she did have a history of known tick exposure. However, both the owners and attending veterinarian report that this cat had never demonstrated clinical signs consistent with cytauxzoonosis. Wright-Leishman–stained blood smears, prepared 20 and 29 months after the prophylactic treatment, did not reveal a detectable parasitemia. Blood from each time point was also submitted for PCR analysis.

Peripheral blood smears from 2 clinically healthy, but persistently parasitemic, cats prepared
Polymerase chain reaction analysis for C. felis was performed on each EDTA–anticoagulated whole blood sample from all 3 cats at each follow-up appointment. DNA was isolated using a commercially available kit, e and the C. felis internal transcribed spacer region 2 (ITS2) plus 5.8S and 28S partial flanking regions were amplified by PCR. The sequences of the forward and reverse oligonucleotide primers used to amplify C. felis ITS2 were 59′-TGAACGTATTAGACACACCACCT-3′ and 5′-TCCTCCCGCTTCACTCGCCG-3′, respectively. These primers were expected to produce an amplicon of 431 base pairs (bp) from genomic C. felis DNA that incorporates the 265-bp ITS2 region. Polymerase chain reaction components consisted of a hot-start Taq polymerase master mix f (containing hot-start DNA polymerase, PCR buffer with 3 mM MgCl2, and 400 μM of each deoxyribonucleotide triphosphate), additional MgCl2 (bringing the final MgCl2 concentration to 2.0 mM), 37.5 pmol of each primer, 4-μl DNA template, and water, for a final volume of 50 μl. For a positive control, DNA was extracted from the blood of a fatally infected cat that had confirmed cytauxzoonosis on necropsy. Two negative controls included both DNA extracted from a clinically healthy cat with no known history of tick exposure (thus believed to be not at risk for C. felis transmission) and water. After initial denaturation at 95°C for 5 min, 38 amplification cycles were performed, each cycle comprised 30 sec at 94°C, 30 sec at 60°C, and 1 min at 72°C, with final extension at 72°C for 10 min. The PCR products were resolved in agarose gels and visualized by staining with ethidium bromide (Fig. 2). Positive DNA bands of the appropriate length were detected from all blood samples. However, repeated PCR testing of the first blood sample from the cat with the undetectable parasitemia was inconsistent after long-term storage at −20°C. These findings may be attributable to the very low level of parasitemia and target DNA available.
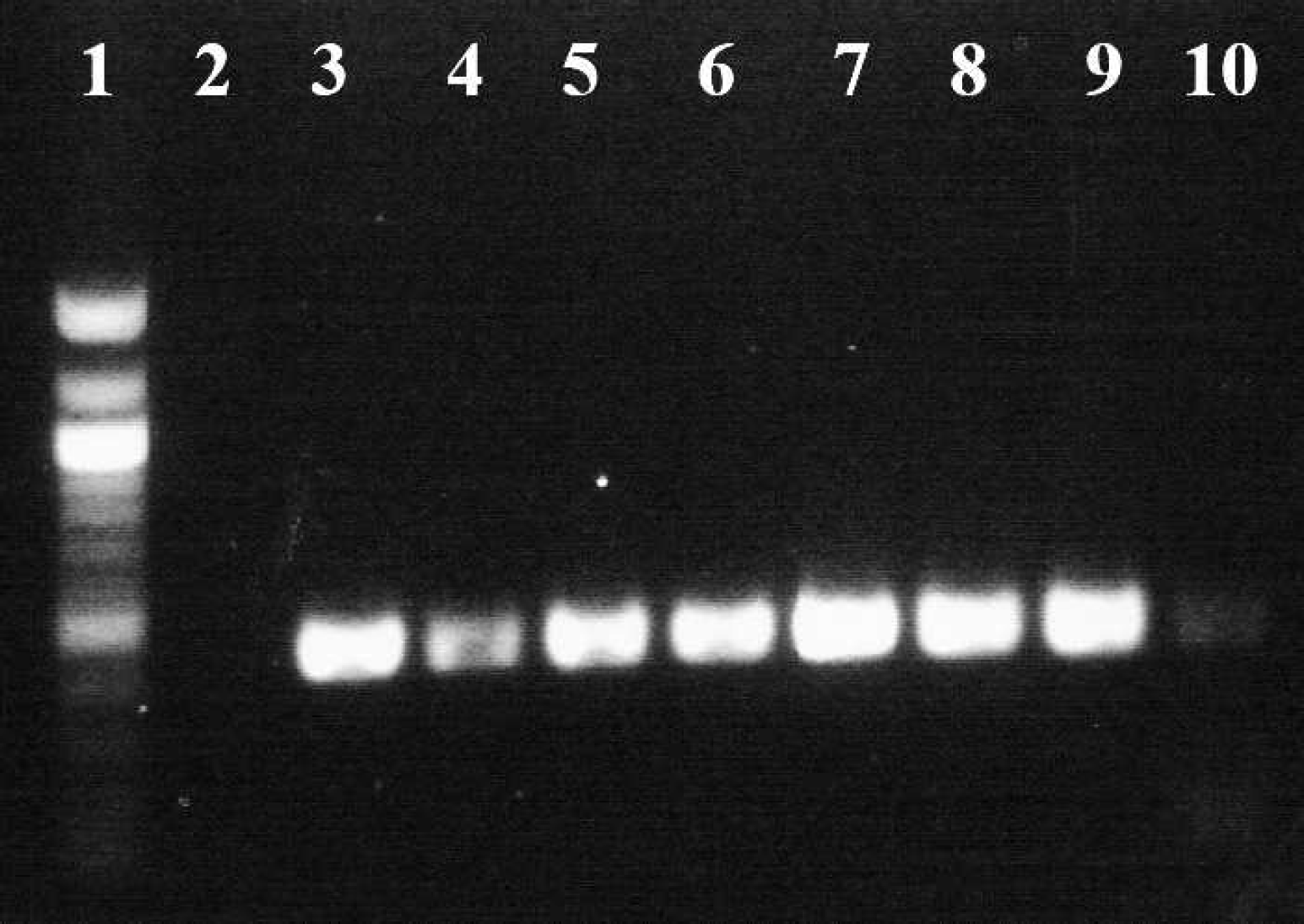
Amplification of the Cytauxzoon felis internal transcribed spacer region 2 (ITS2) from 3 asymptomatic, persistently infected cats. Lane 1, DNA 100-bp ladder; lane 2, negative control (no DNA); lane 3, the first cat on presentation, when clinically ill; lanes 4–6, the first cat 5, 10, and 15 months, respectively, after acute cytauxzoonosis, when clinically healthy; lane 7, the second cat on presentation, when clinically ill; lanes 8 and 9, the second cat 2 and 7 months, respectively, after acute cytauxzoonosis, when clinically healthy; lane 10, the third cat, asymptomatic, 9 months after initial detection of C. felis DNA by polymerase chain reaction.
Positive PCR products from each blood sample were sequenced by a commercial laboratory g and analyzed with Vector NTI software. h The ITS2 sequences were aligned with Vector NTI software for sequence comparison. The ITS2 sequences from each isolate in the present study (all 3 cases at multiple time points) were identical (GenBank EU450801). In comparison to the other complete 265-bp ITS2 sequences deposited in GenBank, the consensus sequence was identical to 2 submissions (GenBank AY699809 and DQ458797) and differed by either 1 or 2 nucleotides from the other 4 entries (GenBank AY695934, AY695935, AY695936, and AY699808).
The present communication reports repeated detection of C. felis in the peripheral blood of 3 asymptomatic, domestic cats as confirmed by PCR analysis with DNA sequencing. Although 2 cats initially presented with clinical cytauxzoonosis, the third cat had never demonstrated clinical signs consistent with acute C. felis infection. This finding allows for a population of yet unidentified domestic cats that may silently harbor the parasite. Because of the inherent insensitivity of blood smear review for the detection of very low levels of parasitemia, more sensitive means of parasite detection, such as PCR analysis, may be necessary to identify asymptomatic carriers of C. felis. However, very low levels of parasite DNA may result in inconsistent PCR results. Therefore, repeated PCR testing or more sensitive analyses, like nested or real-time PCR, may be necessary to identify C. felis–positive cats.
Asymptomatic, persistently infected domestic cats may have the capacity to serve as an additional reservoir host for C. felis, altering the currently accepted paradigm of C. felis transmission to domestic cats through bobcats as the reservoir host. Domestic cat reservoirs would greatly increase the risk of exposure to C. felis infection for other cats, although the ability of those cats to serve as sources of infection for fatal disease is unknown. Increased understanding of the epidemiology of C. felis infection will enhance the ability to prevent this fatal infectious disease of domestic cats.
Acknowledgements. The authors wish to thank Mike Mahoney, medical technologist, and Mariann Clark at the Veterinary Diagnostic Laboratory of the Arkansas Livestock and Poultry Commission for their technical assistance. Funding for this project was provided by The University of Georgia Clinical Research Program.
Footnotes
a.
Imizol, Schering-Plough Animal Health, Union, NJ.
b.
IVX Animal Health Inc., St. Joseph, MO.
c.
Baytril, Bayer HealthCare LLC, Animal Health Division, Shawnee Mission, KS.
d.
Hospira Inc., Lake Forest, IL.
e.
Illustra blood genomicPrep Mini Spin Kit, GE Healthcare UK Limited, Little Chalfont, Buckinghamshire, UK.
f.
HotStarTaq Plus Master Mix, Qiagen Inc., Valencia, CA.
g.
MWG Biotech Inc., High Point, NC.
h.
Invitrogen Corp., Carlsbad, CA.
