Abstract
A competitive enzyme-linked immunosorbent assay (cELISA) was developed by using a whole-cell antigen from a marine Brucella sp. isolated from a harbor seal (Phoca vitulina). The assay was designed to screen sera from multiple marine mammal species for the presence of antibodies against marine-origin Brucella. Based on comparisons with culture-confirmed cases, specificity and sensitivity for cetacean samples tested were 73% and 100%, respectively. For pinniped samples, specificity and sensitivity values were 77% and 67%, respectively. Hawaiian monk seal (Monachus schauinslandi; n = 28) and bottlenose dolphin (Tursiops truncatus; n = 48) serum samples were tested, and the results were compared with several other assays designed to detect Brucella abortus antibodies. The comparison testing revealed the marine-origin cELISA to be more sensitive than the B. abortus tests by the detection of additional positive serum samples. The newly developed cELISA is an effective serologic method for detection of the presence of antibodies against marine-origin Brucella sp. in marine mammals.
Introduction
Brucella sp. isolations in marine mammals were first reported in 1994, and since then there have been numerous reports of isolates from many marine species. 8,10,11,15,21,22,29,31,34 Classically, there are 6 recognized Brucella spp. that infect terrestrial mammals. 23,37 The Brucella biovars isolated from marine mammals have microbiologic growth and genetic characteristics distinct from the classically recognized terrestrial species. 2,8,10,17,20,22,29,32,37 Genetic analyses of the various isolates from marine mammals indicate diversity from the terrestrial species, and there is strong evidence that shows that at least 2, if not more, species or biovars exist in marine mammals. New nomenclature for these marine species has been proposed but not officially accepted. 2,8,12,14,20,38
Serology is an effective and efficient tool for screening animals for the presence of antibodies against Brucella spp., which indicate a current active infection or previous exposure. Multiple studies have detected anti-Brucella antibodies in marine mammals worldwide, from pinniped, cetacean, and mustelid families. 9,18,20,26,27,31–33 Traditionally, a variety of serologic tests specifically designed and validated for Brucella abortus have been used to screen marine mammal sera for the presence of antibodies against the bacterium. Both the specificity and sensitivity of these tests for use with bovine serum are high; however, results of testing in marine mammals are often inconsistent, and comparisons between tests are difficult to make. 21,26 A consensus for determining a positive result often requires that a marine mammal serum sample must test positive on multiple tests. 7
Although B. abortus antigen reacts with serum from marine mammals infected with Brucella spp., it is likely that a serologic test that uses antigen from a marine mammal isolate may be more sensitive. Both primary (lipopolysaccharide [LPS]) and secondary (protein) antigenic determinants may be sufficiently different to affect antigen—antibody affinity among clades of bacteria. 1 Use of a whole-cell antigen versus specific cell wall components may also increase sensitivity by allowing antibody binding to different antigenic epitopes. Recently developed assays for testing marine mammal serum have been briefly described, including both indirect and competitive enzyme-linked immunosorbent assay (ELISA) test methods 16 (Miller WG, Romano TR, Pugh R, et al.: 2001, ELISA for detection of brucellosis in bottlenose dolphins (Tursiops truncatus). In: Proceedings of the 32nd Annual Conference of the International Association of Aquatic Animal Medicine, pp. 142–143. Tampa, FL; Estepp JD, Middlebrooks BL, Patterson RA: 2005, Development of an immunodiagnostic assay for Brucella sp. in the harp seal (Phoca groenlandica) and hooded seal (Cystophora cristata). In: Proceedings of the 36th Annual Conference of the International Association of Aquatic Animal Medicine, p. 97. Seward, AK). The antigens used in these recently developed assays range from cell surface LPS to whole-cell antigen.
The present report describes the development of a cELISA by using whole-cell antigen from a harbor seal (Phoca vitulina) marine Brucella sp. isolate, as well as studies that validate and use the cELISA for testing of marine mammal sera. Because this cELISA measures competitive binding of known positive goat serum and test sera to a common antigen via anti-goat antibody secondary labeling, it alleviates the need for numerous species-specific secondary antibodies, as in indirect ELISA tests, and reduces time and expense for production and testing.
Materials and methods
The cELISA developed in the current study was modified from a previous protocol developed by collaborators at the University of Southern Mississippi (Estepp JD, et al.: 2005, Development of an immunodiagnostic assay for Brucella sp. in the harp seal [Phoca groenlandica] and hooded seal [Cystophora cristata]).
Antigen
A methanol-killed, whole-cell suspension of marine Brucella isolated from a harbor seal (case no. 04-0281, accession no. 311585) was obtained from the National Veterinary Services Laboratory in Ames, Iowa. The suspension was washed at 18,000 rcf for 10 min to remove methanol, and cells were resuspended in sterile 1× phosphate buffered saline solution (PBS). a The final stock of antigen was stored in aliquots at −20°C.
DNA was extracted from aliquots of bacterial suspensions by using spin columns and the Gram-negative bacteria protocol of a commercial DNA purification kit. b The polymerase chain reaction assays performed on this extract were positive for insertions of IS711 elements previously described as specific for marine mammal strains of Brucella. 2,4 Typing of outer membrane protein sequences showed this isolate to be within a subcluster of Brucella pinnipedialis. 6
Control sera
The positive control serum was obtained through polyclonal antibody production in collaboration with Rockland Immunochemicals. c Briefly, after performing a direct ELISA to confirm the absence of antibodies against Brucella antigens, a female Toggenburg goat (OLAW assurance no. A4062-01), that weighed approximately 25 kg, was immunized with the previously described methanol-killed, whole-cell marine Brucella antigen. The immunization protocol included an initial injection of 400 μg, followed by 2 subsequent booster injections of 200 μg each at 1-week intervals, and a final injection of 200 μg at a 2-week interval. Several direct ELISAs were performed c during the project to confirm the development of antibodies against the antigen and to measure serial titer levels. Once an acceptable titer was detected, 120 ml of serum and 1,050 ml of plasma were collected for use in the assay development and as a positive control sample. Goat serum and plasma antibody titers were compared, and no difference was found between the 2 blood components; therefore, plasma was collected, stored, and used because of a larger collection volume. The negative control used for the cELISA was goat plasma obtained from a goat that was never immunized or exposed to Brucella. c
Secondary antibody
Immunoglobulin G (IgG) purification of the previously described goat anti-Brucella plasma was performed by Rockland Immunochemicals. c Briefly, the antibody was purified from monospecific antiserum by a multistep process, which includes delipidation and defibrination of the plasma, followed by salt fractionation and ion exchange chromatography. The purified antibody was then dialyzed extensively against 0.02 M potassium phosphate and 0.15 M sodium chloride, pH 7.2 with 0.01% (w/v) sodium azide. The purified goat IgG was then biotinylated by using a commercial kit. d Excess biotin was removed by dialysis. e The final biotinylated IgG protein solution was stored at −20°C.
Competitive ELISA
Antigen coating was performed by diluting the whole-cell marine Brucella antigen in coating buffer (Na2CO3 0.015 M, NaHCO3 0.0348 M, NaN3 0.003076 M, pH 9.6, 1 liter) to an optical density (OD) at 600 nm of 0.1 (1 × 109 cells/ml). A 96-well microtiter ELISA plate f was coated with 50 μl of antigen solution per well, covered, and allowed to incubate overnight at room temperature (22–25°C) for a minimum of 12 hr and no longer than 18 hr. Two controls were included on each plate: 1) a no-serum control (all steps apply with the exception of the test serum), and 2) a blank control (wells received coating buffer only during each step, with the exception of the color-developing reagent at the final step). Before each step, plates were washed 3 times by using a wash buffer (Na2HPO4 0.04818 M, KH2PO4 0.008818 M, NaCl 0.8213 M, KCl 0.01609 M, NaN3 0.018458 M, Tween 20 3.00 ml, pH 7.4, 6 liters) on an automated plate washer. g A 2% blocking buffer with bovine serum albuminh was prepared by dilution in coating of buffer, and 50 μl was added to each well. The plate then was incubated for 30 min at 37μC followed by a wash step. A 1:10 dilution of test sera and positive and negative controls were prepared in incubation buffer (0.03 M Na2HPO4, 0.02998 M KH2PO4, 0.41065 M NaCl 24.0, 1.50 ml Tween-20, pH 7.0, 3 liters). Fifty microliters of the diluted sera were added to antigen-coated wells. All serum samples and controls were tested in triplicate. The plate was covered and allowed to incubate for 30 min at 37°C followed by a wash step.
A 1:9000 dilution of the secondary biotinylated goat anti-Brucella antibody IgG was prepared in incubation buffer, and 50 μl was added to designated wells. The plate was covered and allowed to incubate for 30 min at 37°C followed by a wash step. A 1:30,000 dilution of the enzyme substrate ExtraAvidin-labeled alkaline phosphatase i was prepared in incubation buffer and 50 μl added to each designated well. The plate was covered and allowed to incubate for 30 min at 37°C followed by a wash step.
The color-developing reagent was prepared by using a 1× concentration solution of diethanolamine substrate buffer 5× j and diluted in NANOpure water. A developing solution was then prepared by dissolving two 5-mg tablets of ρ-nitrophenol phosphate k into each 5 ml of 1× substrate buffer, and 50 μl of the developing solution was added to each well. The plate was covered and allowed to incubate for 30 min at room temperature. Plates were read on an ELISA plate reader, l at a wavelength of 405 nm. In cases in which the plate could not be read immediately, 50 μl of 3 N NaOH was added to each well to stop the reaction. No significant differences were found between plates read with and without NaOH immediately after the incubation period.
Data analysis
The output from the ELISA plate reader was analyzed by using KC4 analysis software. m The following formula was used to calculate percent inhibition: percent inhibition = 100 − [(A/B) × 100], where A indicates the mean OD value of test/positive/negative control serum, and B indicates the OD value of no-serum control.
Determination of cutoff values
The cutoff values for the positive, negative, and suspect ranges were determined by testing 15 serum samples from confirmed culture-positive bottlenose dolphins (Tursiops truncatus; Brucella spp. were isolated during clinical illness or at necropsy). An additional 8 serum samples were tested from confirmed culture-negative bottlenose dolphins. In addition, by using the cutoff values determined from the cetacean samples, serologic results obtained from the following small group of pinniped species that were determined to be culture positive at necropsy: Phoca vitulina (n = 3), Zalophus californianus (n = 1), Callorhinus ursinus (n = 1), and Phoca groenlandica (n = 1).
Test comparisons and cross reactivity
Serum samples from 48 wild common bottlenose dolphins were tested in a blind study with the newly developed cELISA, and results were compared with assays that detect antibodies against B. abortus. The B. abortus tests included Rivanol, Card, and buffered acidified plate antigen test (BAPA) tests and were performed based on protocols from the U.S. Department of Agriculture, Animal and Plant Health Inspection Service, National Veterinary Services Laboratories. n Dolphin serum samples were collected during capture—release operations for health assessments in Sarasota Bay, Florida (n = 10), 36 Indian River Lagoon, Florida (n = 17), and Charleston, South Carolina (n = 21), 30 between June and August 2005. Serum samples were also obtained from 28 wild Hawaiian monk seals (Monachus schauinslandi) as part of an ongoing health assessment study between 1998 and 2001 in the Northwestern Hawaiian Islands. The samples were screened in a blind study with the newly developed cELISA, and results were compared with previously reported results that used multiple B. abortus serologic assays, including BAPA, indirect ELISA, fluorescence polarization assay, and cELISA tests. 26 Brucella abortus (n = 3), Brucella melitensis (n = 2), and Yersinia enterocolitica 0:9 (Y09; n = 2) positive sera were tested to evaluate cross-reactivity of terrestrial Brucella antibodies with the cELISA.
Results
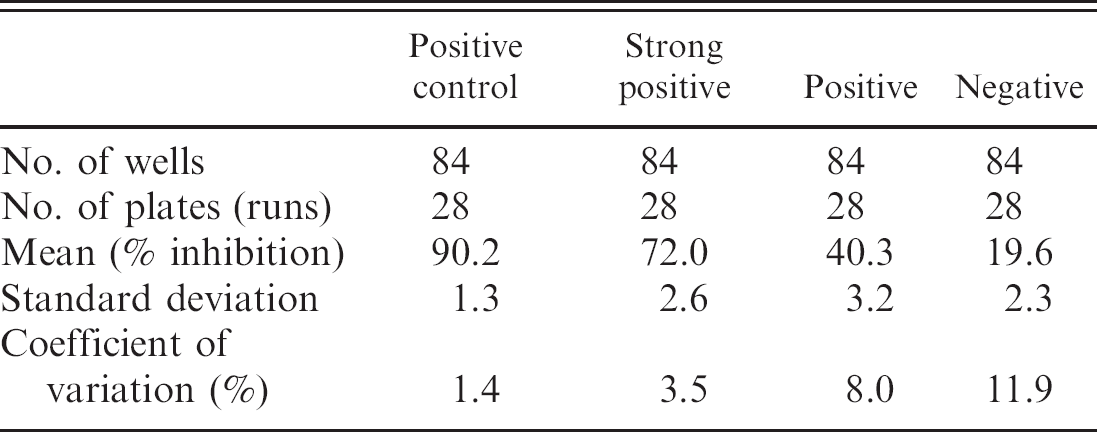
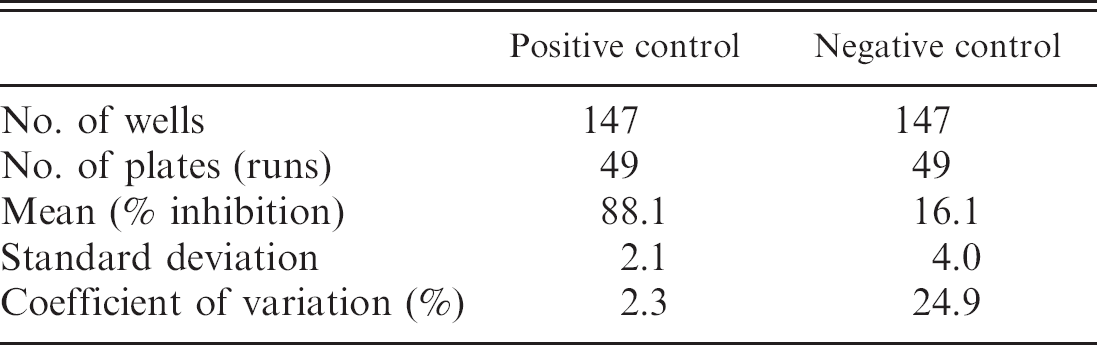
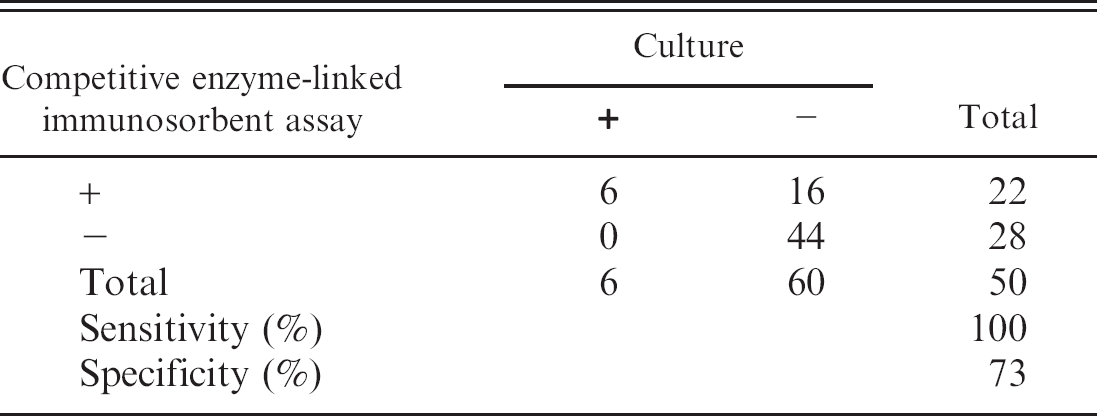
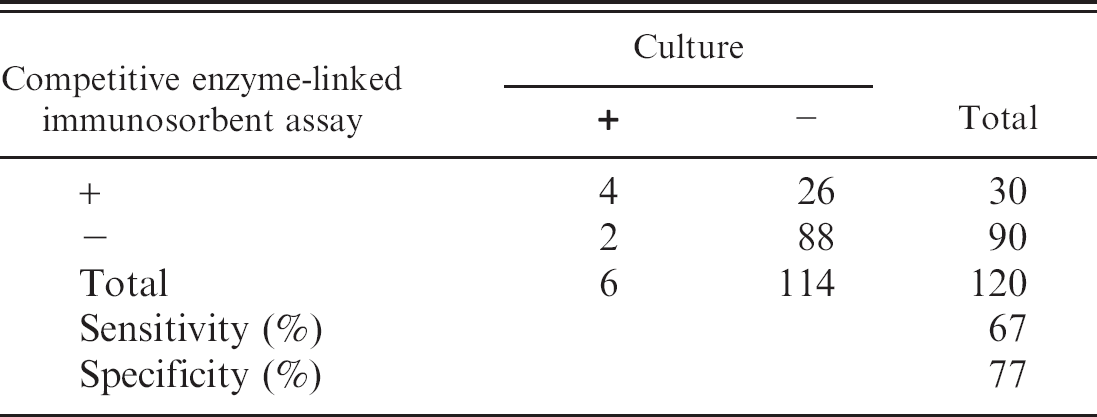
The serologic results for the culture-positive and culture-negative bottlenose dolphins are shown in Figure 1. Based on these results, the cutoff values for the cELISA were determined as positive ≥30.0%, suspect 25.0–29.9%, and negative ≤24.9%. In addition, by using the cutoff values determined from the cetacean samples, serologic results obtained from a small group of culture-positive pinnipeds are displayed (Fig. 2). Coefficient of variation (CV) for intra-assay variability was determined by repeated testing of 3 individual cetacean serum samples that represented 3 levels of antigenic reactivity (strong positive, positive, and negative), and the positive control goat anti-Brucella plasma (Table 1). The CV for interassay variability was determined by using the previously described positive control and negative control goat plasma (Table 2). Microbiologic culture for Brucella was used as the criterion standard to determine values of specificity and sensitivity for cELISA testing in cetacean (Table 3) and pinniped (Table 4) species, respectively. Brucella abortus—positive sera (n = 3) also tested positive on the marine cELISA, as did the B. melitensis—positive serum (n = 2). Both of the Y09—positive sera (n = 2) were negative, which suggests no cross-reaction of these antibodies with the harbor seal antigen.
Test comparison
Serologic comparison of the marine-origin cELISA results with B. abortus test methods for the wild bottlenose dolphin serum samples were performed. The Card, BAPA, and Rivanol tests yielded a total of 19, 18, and 12 seropositive samples of 48, respectively. A consensus method, previously used by others, 7 requires that a sample must be positive on all tests to be considered a true positive. When this method is applied, the number of (consensus) seropositive samples decreased to 10 of 48. The marine-origin cELISA detected 32 seropositive samples of 48 total samples.
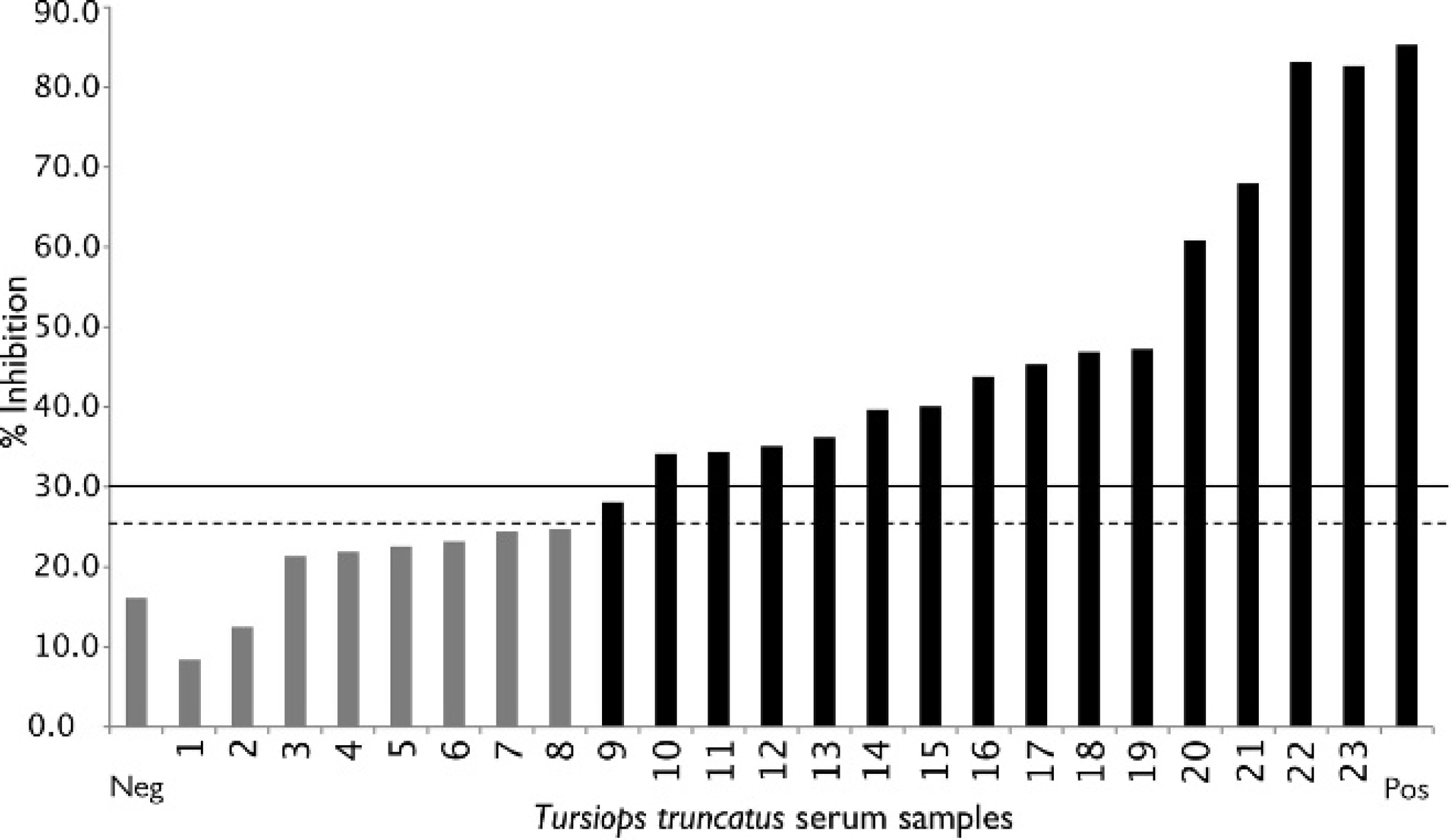
Percent inhibition results from 15 serum samples of culture-positive bottlenose dolphins (Tursiops truncatus; black bars), and 8 serum samples from culture-negative bottlenose dolphins (gray bars) used in developing cutoff values. The negative and positive control sera are also shown. The solid black line demarcates the positive cutoff value: a positive result lies above the line. The suspect range lies between the dashed and solid lines, and a negative result below the dashed line.
When Hawaiian monk seal serum samples were tested by the marine-origin cELISA, the results indicated that 16 of 28 samples were positive (Fig. 3). The previously reported results from this same set of samples revealed that 14 of 28 samples were seropositive when B. abortus test methods were applied. 26 All of the samples that tested positive on the B. abortus tests also tested positive with the marine-origin cELISA. One of the 2 serum samples that did not show agreement (Fig. 3; sample labeled “x”) had inconsistent results with the B. abortus tests, testing positive on all but one of the B. abortus test methods. Although it this serum sample (labeled “x”) tested positive on 3 of 4 B. abortus tests, the consensus method the authors applied required that this sample be reported as seronegative.
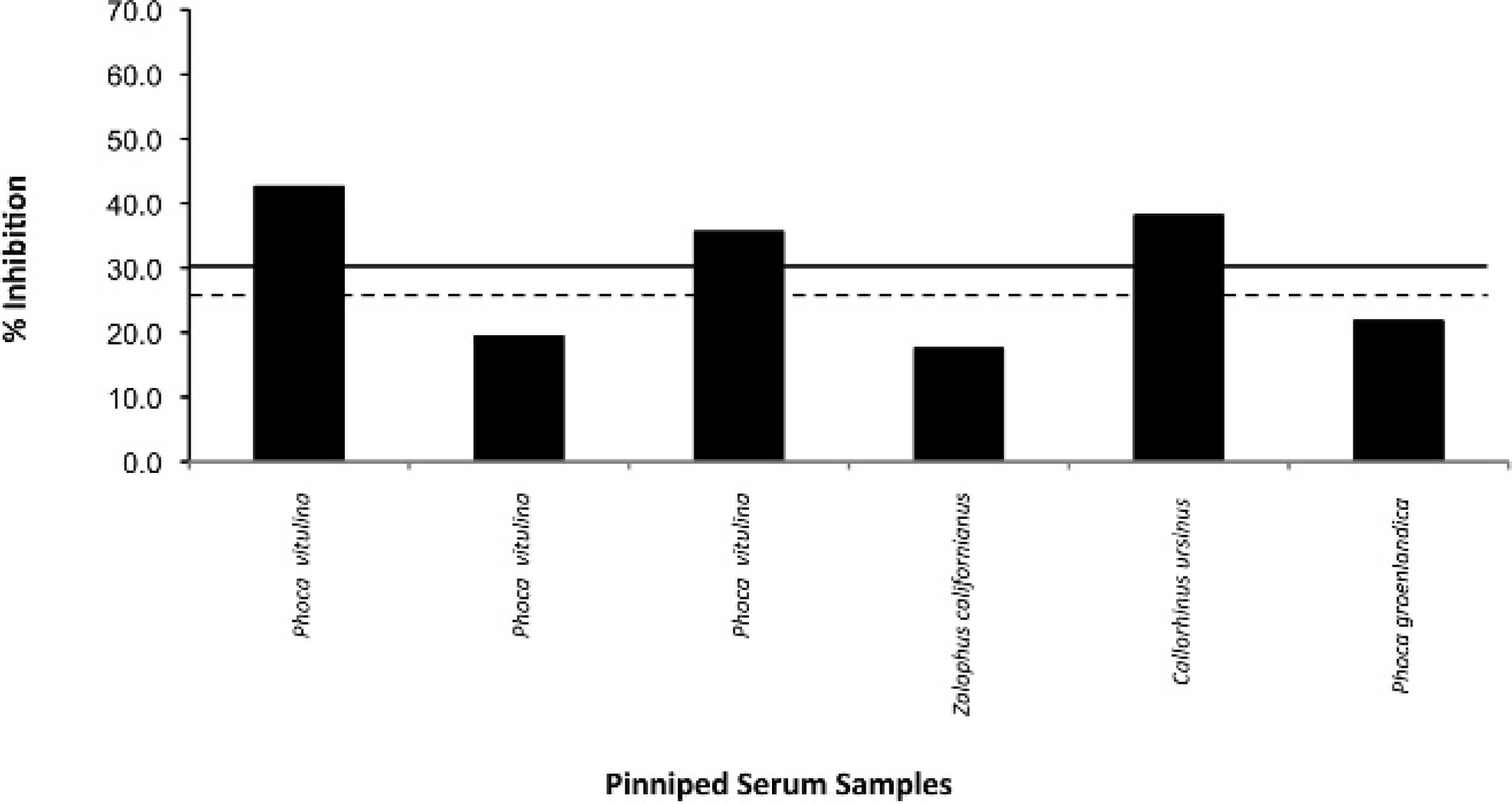
Serologic results from culture-positive pinnipeds. A positive test result lies above the solid line. The suspect range lies between the dashed and solid lines, and a negative result below the dashed line.
Intra-assay variability for the competitive enzyme-linked immunosorbent assay by using various test serum samples and positive and negative control sera.
Discussion
A cELISA that uses a whole-cell marine Brucella sp. isolated from a harbor seal and designed for screening marine mammal blood samples was successfully developed. The cELISA is effective and sensitive in detecting Brucella sp. antibodies for pinniped and cetacean species, and further testing is currently being performed to evaluate its utilization for additional marine mammal species.
The CVs for both intra- and interassay variability were less than 10% for positive samples. The CV for both intra- and interassay testing for the negative samples were relatively higher because of the lower overall inhibition levels, but absolute standard deviation for both tests were comparable with positive samples. Overall, the CV values indicate that the cELISA is both precise and consistent; the relative increase in variation in the lower end of the testing range emphasizes the importance of including a suspect category for animals perhaps in need of continued monitoring.
Specificity and sensitivity of the cELISA when applied to cetacean sera revealed high sensitivity but lower specificity. When used on pinniped sera, the specificity and sensitivity were both reduced. Previous reports have also demonstrated a lack of seroconversion in pinniped species with confirmed Brucella sp. infections. 21,26,34 The data in the present study support these previously reported findings, which may explain the lower sensitivity. In addition, it has been reported that Brucella sp. are often isolated from tissues of pinnipeds without corresponding significant associated gross or histopathologic lesions. 11
Interassay variability for the competitive enzyme-linked immunosorbent assay by using positive and negative control sera.
Specificity and sensitivity determined with cetacean serum samples.
Recently, it has been suggested that different marine Brucella isolates have varying degrees of pathogenicity based on their genetic structure (Maquart M, Zygmunt MS, Cloeckaert A: 2008, Marine mammal Brucella isolates with different genomic backgrounds display a differential response to human macrophage infection. Brucellosis International Research Conference, Egham, Surrey, England). Alternatively, each marine mammal species being surveyed may respond to the bacteria in different immunologic ways, depending on the location and severity of infection, pathogenesis, and individual immune responses.
Ideally, validation data and analysis should be performed on each marine mammal species that is evaluated. However, because of limited sample sizes of confirmed Brucella-positive control blood samples for each species examined, combined with legal, logistical, and ethical problems associated with producing laboratory populations of marine mammals infected with this zoonotic pathogen, it becomes extremely difficult to use these indices of validation with confidence. 26 It is important to note that, in the current study, the number of serum samples paired with successful microbiologic isolates was low and not ideal for determining statistical data. Therefore, until more information is obtained regarding species variations with infection and antibody production, sensitivity and specificity information is limited and difficult to interpret.
Specificity and sensitivity determined with pinniped serum samples.
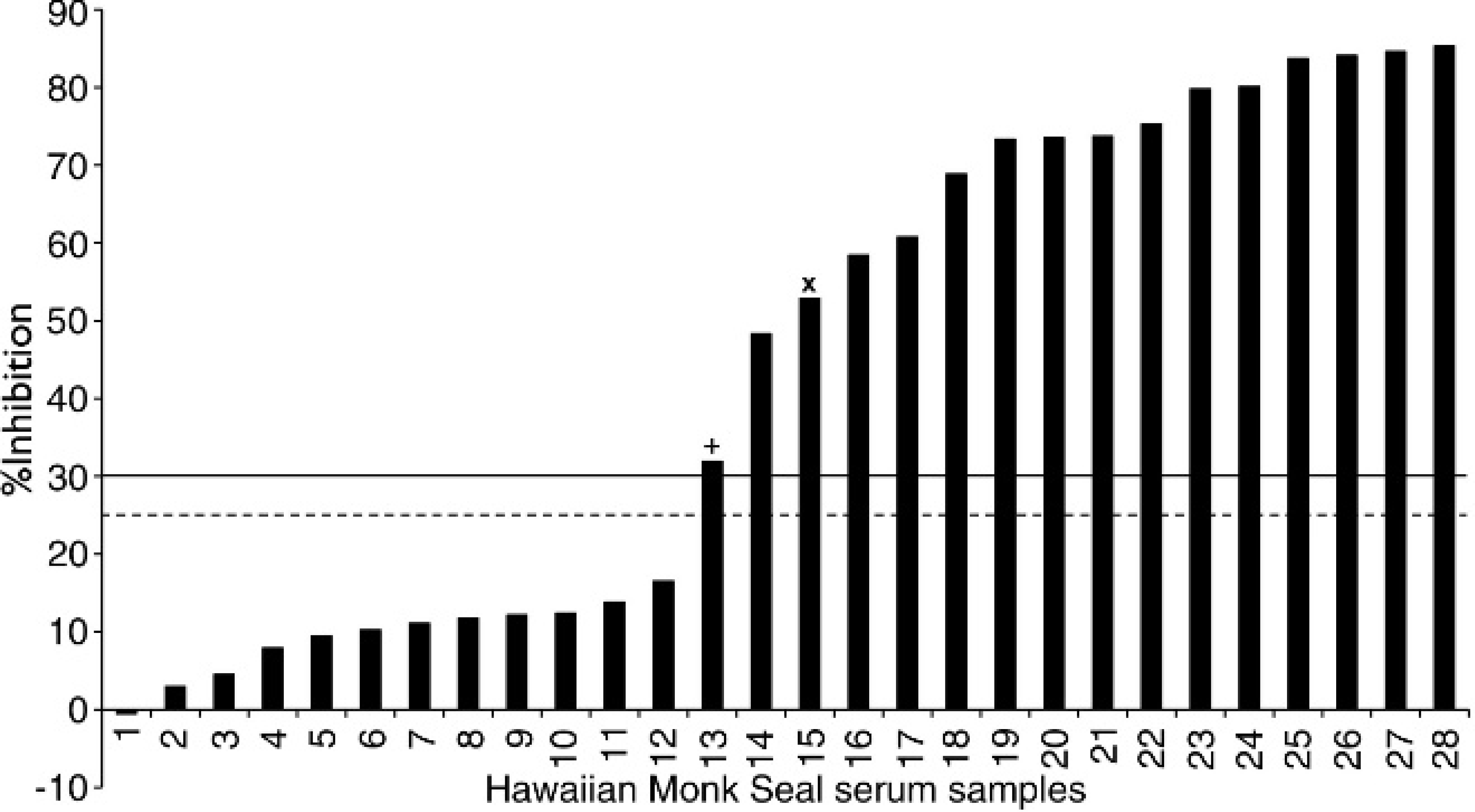
Serologic results from Hawaiian monk seal (Monachus schauinslandi) serum samples obtained from the marine-origin competitive enzyme-linked immunosorbent assay (cELISA). Positive values are indicated above the solid line. The suspect range lies between the dashed and solid lines, and a negative result lies below the dashed line. The samples labeled with a “+” sign and an “x” sign indicate serum samples that tested positive with the cELISA but negative with the terrestrial test methods.
The cELISA described in the present study uses a whole-cell Brucella antigen, which is in contrast to using isolated cell wall components, specifically the LPS portion often used for terrestrial Brucella serologic methods. 25 A recently developed indirect ELISA was designed 16 specifically for testing odontocete serum. This indirect ELISA uses LPS as the antigen; however, it uses terrestrial B. abortus and B. melitensis LPS rather than marine-origin LPS as the antigen. Further investigation is warranted to evaluate utilization of various cell wall components as antigens.
Utilization of a whole-cell antigen—based assay may be useful because certain outer membrane proteins specific for determining the antigenic nature of terrestrial Brucella to its host have also been identified in marine mammal strains. 4,5,35 The genes omp2a/b, omp25, omp28, and omp31 have been identified from various cetacean and pinniped isolates, and divergence in protein expression has been described within marine mammal strains when compared with terrestrial strains. 3–5,35 These antigenic outer membrane proteins may be unique to marine mammal Brucella biovars and useful for antibody detection assays.
The use of a polyclonal antibody combined with a whole-cell antigen may increase sensitivity at the cost of specificity, with a risk of an increase in the number of false-positive results occurring through nonspecific binding with antibodies present in the test sera against similar Gram-negative organisms. However, more cross-reactivity can occur when IgM is measured; therefore, the use of an IgG, as in this assay, is ideal. 25 Previous studies have noted cross-reactivity of Y09 with B. abortus serologic tests because of common antigenic epitopes of the smooth LPS O chain between these species. 13,19,24 Although marine mammal infection with Y09 has not yet been reported, and a previous study did not detect Y09 antibodies in serum from various marine mammals, 33 the potential for transmission theoretically exists. Knowledge of potential cross-reaction of the marine cELISA with Y09 antibodies will help differentiate between these pathogens. Although only two Y09-positive serum samples were available for testing, these samples tested negative on the marine cELISA, which suggests that cross-reaction is absent. In addition, B. abortus—and B. melitensis—positive serum tested positive on the marine cELISA, which confirmed cross-reaction of these Brucella spp. with the marine antigen.
Overall, the comparison testing performed in the current study reveals that the marine-origin cELISA is a more sensitive assay than the classic B. abortus methods for detecting anti-Brucella antibodies in both cetacean and pinniped species. Despite this higher sensitivity, the cutoff ranges for the newly developed cELISA were determined conservatively (Fig. 1). More importantly, the classic B. abortus tests failed to detect a seropositive result, but the marine-origin cELISA confirmed the presence of antibodies. 21,22 Results of testing showed that use of the current cELISA compared favorably with results obtained from multiple terrestrial Brucella sp. tests and indicates that this assay can be used successfully in lieu of multiple assay consensus testing in a variety of marine mammal species. The newly developed marine-origin cELISA is a reliable, rapid screening assay capable of evaluating multiple samples per plate and makes it useful for health screenings and as a diagnostic tool for clinical case situations.
Acknowledgements
The Brucella Project is funded by National Oceanic and Atmospheric Association (NOAA) Oceans and Human Health Initiative Grant NA04OAR4600209 and has received previous support from the NOAA Fisheries Prescott Grant Award NA03NMF4390408. The wild Hawaiian monk seal blood samples were collected under Scientific Research and Enhancement Permit to Take Marine Mammals 848-1335. The wild bottlenose dolphin blood samples were collected under the National Marine Fisheries Service Scientific Research permit 522-1569 and 522-1785 issued to Dr. Randall Wells of the Sarasota Dolphin Research Program (SDRP) in Sarasota Bay, Florida; and 998-1678 issued to Dr. Greg Bossart as part of the Bottlenose Dolphin Health and Risk Assessment Project conducted in the Indian River Lagoon, Florida; and the estuarine waters of Charleston, South Carolina. Funding was partially provided by the Harbor Branch Oceanographic Institution's Project Wild Dolphins license tag, NOAA, National Marine Fisheries Service, Dolphin Quest, and Disney's Animal Programs. Brian Balmer's expertise in preparing and processing samples for the SDRP is much appreciated. The authors also thank John McGiven of the Veterinary Laboratory Agency (Surrey, UK) for supplying the terrestrial Brucella and Yersinia serum samples. This work constitutes scientific contribution 181 from the Sea Research Foundation.
Footnotes
a.
10× PBS stock (pH 7.2, product 71003-073), Invitrogen Corp., Carlsbad, CA.
b.
DNeasy® Tissue Kit, Qiagen Inc., Valencia, CA.
c.
Antibody Production Project PAB-105-1413A, Rockland Immunochemicals Inc., Gilbertsville, PA.
d.
EZ-Link® Sulfo-NHS-Biotin kit (product 21335), Pierce Biotechnology Inc., Rockford, IL.
e.
Slide-A-Lyzer® Dialysis Cassettes (product 66807), Pierce Biotechnology Inc., Rockford, IL.
f.
Nunc-Immuno™ MaxiSorp 96-well plates (product 442404), Thermo Fisher Scientific Inc., Waltham, MA.
g.
ELx405™ Select, BioTek Instruments Inc., Winooski, VT.
h.
Bovine Serum Albumin (product A7030), Sigma-Aldrich, St. Louis, MO.
i.
ExtraAvidin®-labeled Alkaline Phosphatase (product E2636), Sigma-Aldrich, St. Louis, MO.
j.
Diethanolamine Substrate Buffer 5× (product 34064), Pierce Biotechnology Inc., Rockford, IL.
k.
ρ-nitrophenyl phosphate 5 mg tablets (product S0942), Sigma-Aldrich, St. Louis, MO.
l.
EL800™ Universal Microplate Reader, BioTek Instruments Inc., Winooski, VT.
m.
Kineticalc for Windows NT 5.0 version 2.6, BioTek Instruments Inc., Winooski, VT.
n.
Buffered acidified plate antigen test for detection of antibodies to Brucella abortus. SOP #: SEROPRO1024.02, effective date: March 1, 2004; Rivanol precipitation plate agglutination test for detection of antibodies to Brucella abortus. SOP: SEROPRO1025.03, effective date: April 29, 2004; Card test for detection of antibodies to Brucella abortus. SOP: SEROPRO1027.02, effective date: February 19, 2003.
