Abstract
The IDEXX Swine influenza virus H1N1 and H3N2 enzyme-linked immunosorbent assays (ELISAs) are used worldwide, but their capacity to detect antibodies to European Swine influenza viruses (SIVs) has not been documented. A total of 313 well-defined sera from SIV seronegative pigs and pigs experimentally infected with European SIVs were used to compare the performance of both ELISAs and the hemagglutination inhibition (HI) test. The ELISAs largely failed to detect pigs that had been infected with H1N1 (0/42 positive in H1N1 ELISA) or H3N2 only (9/18 positive in H3N2 ELISA; group 1). Higher ELISA detection rates were found after consecutive infection of pigs with either H1N1 or H3N2 and 1 other subtype (7/40 and 11/22 positive in H1N1 and H3N2 ELISA, respectively; group 2). Of 39 pigs that had been vaccinated twice with 1 of 4 commercial SIV vaccines (group 3), 25 tested positive in the H1N1 and 4 in the H3N2 ELISA. Pigs that had received a single vaccination after a prior infection with H1N1 and/or H3N2 (group 4) were more frequently positive than group 1 or 3 pigs (23/24 and 15/24 positive in H1N1 and H3N2 ELISA, respectively). Both the H1N1 and H3N2 ELISA showed a low sensitivity (39% and 35%, respectively) relative to the HI test. Because pigs in the field are frequently infected and/or vaccinated with multiple SIV subtypes and variants, they are more likely to test positive in the ELISAs. However, the interpretation of ELISA results will be difficult, and HI remains the method of choice.
Introduction
“Avian-like” H1N1 and “human-like” H3N2 Swine influenza viruses (SIVs) have been enzootic in the European swine population since the 1980s, and an H1N2 reassortant became widespread in the late 1990s. The hemagglutinin (HA) of most H1N2 viruses shows little antigenic and genetic relationship with that of the swine H1N1 viruses, and it has likely been derived from human H1N1 viruses circulating in the 1980s. Still, novel H1N2 reassortants with an avianlike HA have also been reported. 4,6 Genetic analyses have further shown that all 3 SIV subtypes share similar “avian-like” internal genes. All 3 SIV subtypes are widespread in swine-dense regions of Europe, and pigs frequently experience consecutive infections with 2 or even 3 different SIV subtypes. 11 To further complicate matters, part of the swine population is vaccinated against SIV. The SIV vaccines currently commercialized in Europe are inactivated, bivalent vaccines. These vaccines contain various H1N1 and H3N2 influenza strains, some of which are antigenically rather different from the current SIV field strains. 13 A trivalent vaccine containing H1N1, H3N2, and H1N2 strains has recently been developed but is not yet registered. The vaccines are used mainly in gilts and sows, which may have been infected with 1 or more SIV subtypes before the first vaccination or at later time points.
Serologic testing is used for the diagnosis of SIV, to monitor a herd's immune status, to optimize vaccination schedules, and for seroprevalence studies. The hemagglutination inhibition (HI) test is most frequently used for SIV serology and remains the “gold standard.” The test is highly specific, and antibody titers correlate with protection. However, it also yields variable results dependent on the test virus strain and is very labor intensive. Commercial enzyme-linked immunosorbent assays (ELISAs) for the detection of SIV antibodies have recently become available and they can test 46 samples per 96-well microtiter plate in just a few hours. All necessary reagents are provided with the kit and part of the test can be automated. The IDEXX Swine Influenza Virus H1N1 a and H3N2 b ELISAs were developed in North America and appear to be suitable for detection of antibodies in pigs exposed to North American H1N1 and H3N2 strains. The H1N1 ELISA has shown an excellent specificity and sensitivity in studies with sera from pigs vaccinated with SIV vaccines used in the United States (Carman S, McEwen B, Josephson G: 2001. Validation of IDEXX H1N1 swine influenza virus ELISA. AHL Newsletter 5(December):56. Available at: http://www.labservices.uoguelph.ca/units/ahl/news_archive.cfm; Erickson G, Rapp-Gabrielson V, Jackson T, et al.: 2002, Duration of HI and ELISA antibodies following vaccination against SIV. Proc 17th International Pig Veterinary Society Congress, p. 180). On the other hand, the ELISA was less sensitive than the HI test in experimental infection studies with U.S. H1N1 viruses. 10,18 The H3N2 ELISA was capable of detecting antibodies in pigs vaccinated with an experimental U.S. H3N2 SIV-derived modified live vaccine, but there are little data on its sensitivity and specificity. 9 Both tests are also used in veterinary diagnostic laboratories in Europe and for seroprevalence studies worldwide (Bottcher J, Gangl A, Alex M, et al.: 2007, Endemic circulation of swine influenza virus (SIV) in the absence of disease. Proc 5th International Symposium on Emerging and Re-emerging Pig Diseases, p. 255; Yoon JS, Han JH, Jeong HK, et al.: 2006, Sero-prevalence of swine influenza virus (H3N2) in Korea according to age of pigs and season. Proc 19th International Pig Veterinary Society Congress, p. 133). 1,7,16 The H1N1 and H3N2 SIV field and vaccine strains in Europe, however, are antigenically and genetically very different from their counterparts in the United States, 5 and there are concerns about the sensitivity of the ELISAs for the detection of antibodies to European SIVs. 3 Still, there are no extensive validation studies with sera to European SIVs.
Detection of H1N1 antibodies by commercial H1N1 Swine influenza virus (SIV) enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) test after experimental infection of pigs with 1 (group 1) or 2 (group 2) different SIV subtypes.
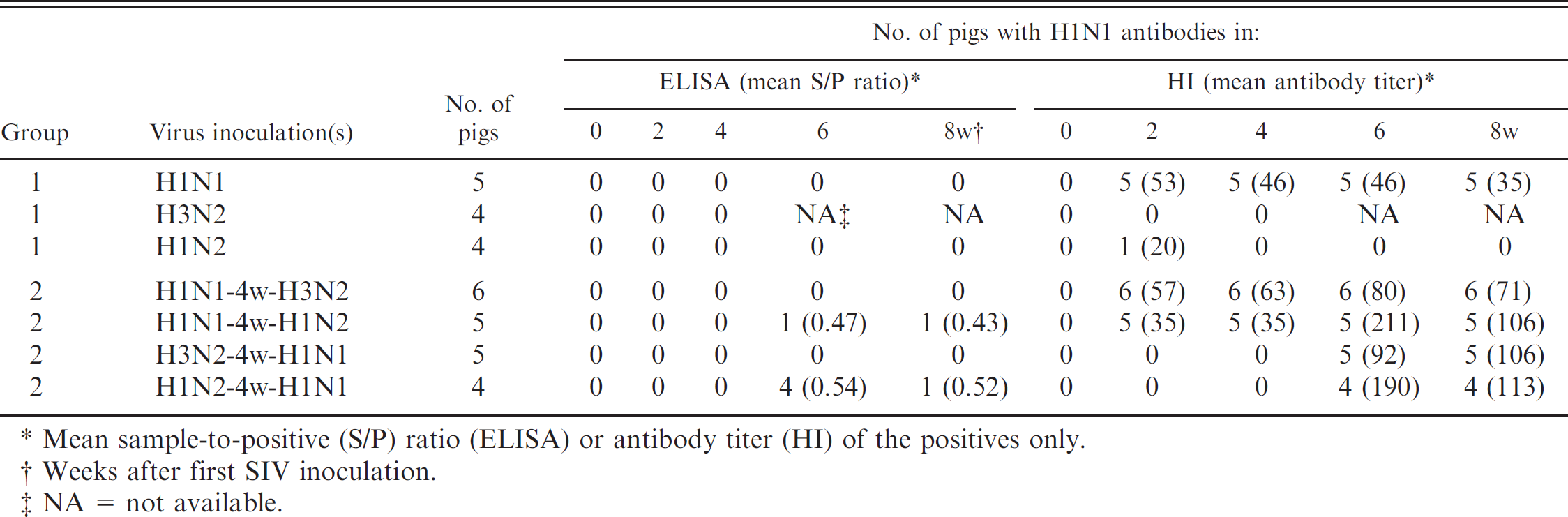
Mean sample-to-positive (S/P) ratio (ELISA) or antibody titer (HI) of the positives only.
Weeks after first SIV inoculation.
NA = not available.
The serum samples used in this study originated from other published and unpublished studies. 13,15 A total of 313 sera from 106 pigs were examined. The pigs belonged to 1 of 4 experimental groups that are described below. Tables 1 and 4, 2 and 5, and 3 and 6 show the different subgroups, the number of pigs in each subgroup, and the times of blood collection for groups 1 and 2, and 3 and 4, respectively.
Detection of H1N1 antibodies by commercial H1N1 Swine influenza virus (SIV) enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) test after a double vaccination of pigs with commercial SIV vaccines (group 3).
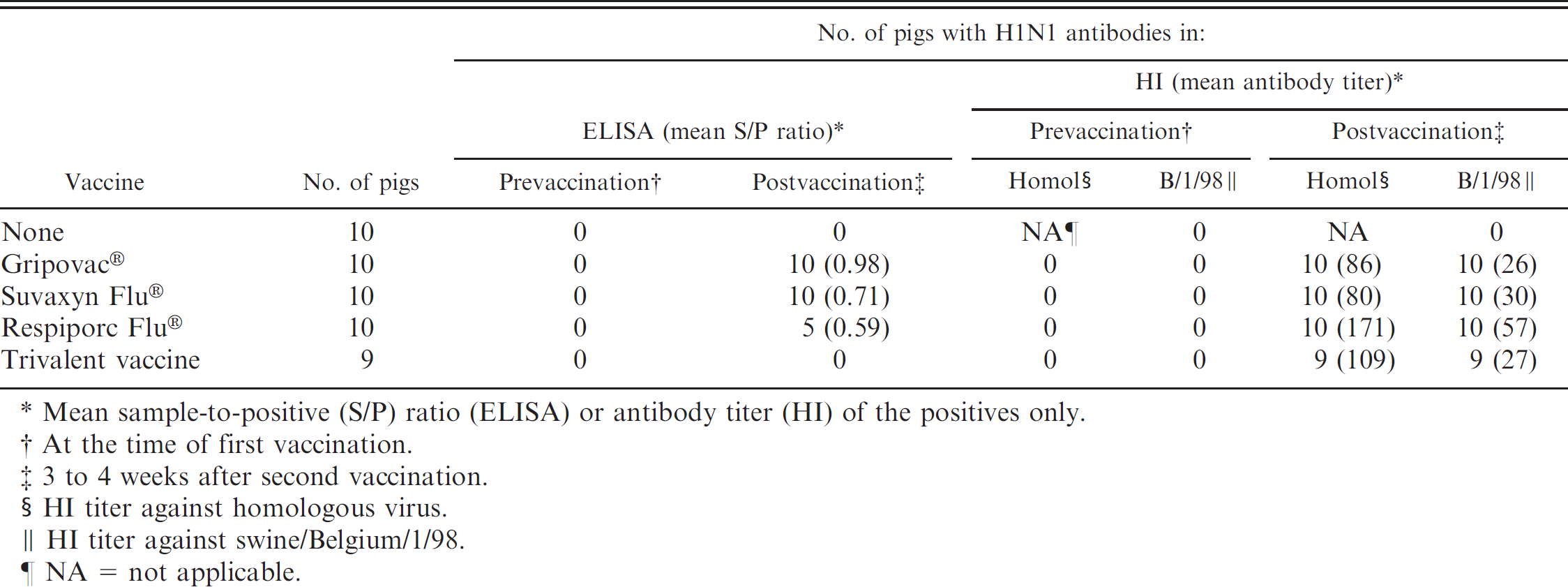
Mean sample-to-positive (S/P) ratio (ELISA) or antibody titer (HI) of the positives only.
At the time of first vaccination.
3 to 4 weeks after second vaccination.
HI titer against homologous virus.
HI titer against swine/Belgium/1/98.
NA = not applicable.
Detection of H1N1 antibodies by commercial H1N1 Swine influenza virus enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) test after a single vaccination* of pigs with infection immunity to H1N1, H3N2, or both subtypes (group 4).
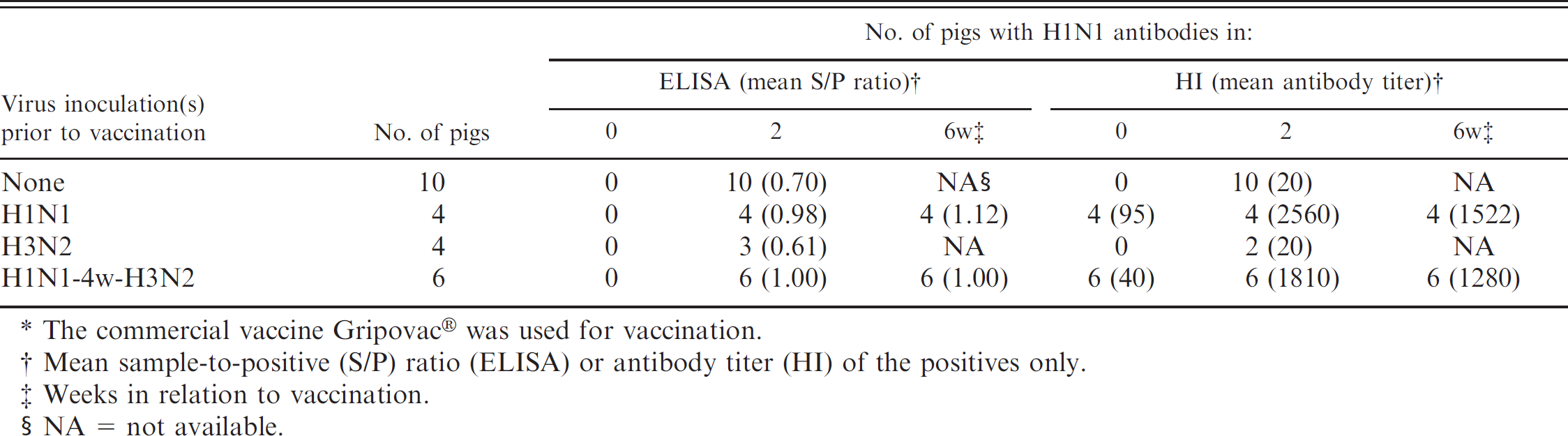
The commercial vaccine Gripovac® was used for vaccination.
Mean sample-to-positive (S/P) ratio (ELISA) or antibody titer (HI) of the positives only.
Weeks in relation to vaccination.
NA = not available.
Detection of H3N2 antibodies by commercial H3N2 Swine influenza virus enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) test after experimental infection of pigs with 1 (group 1) or 2 (group 2) different SIV subtypes.
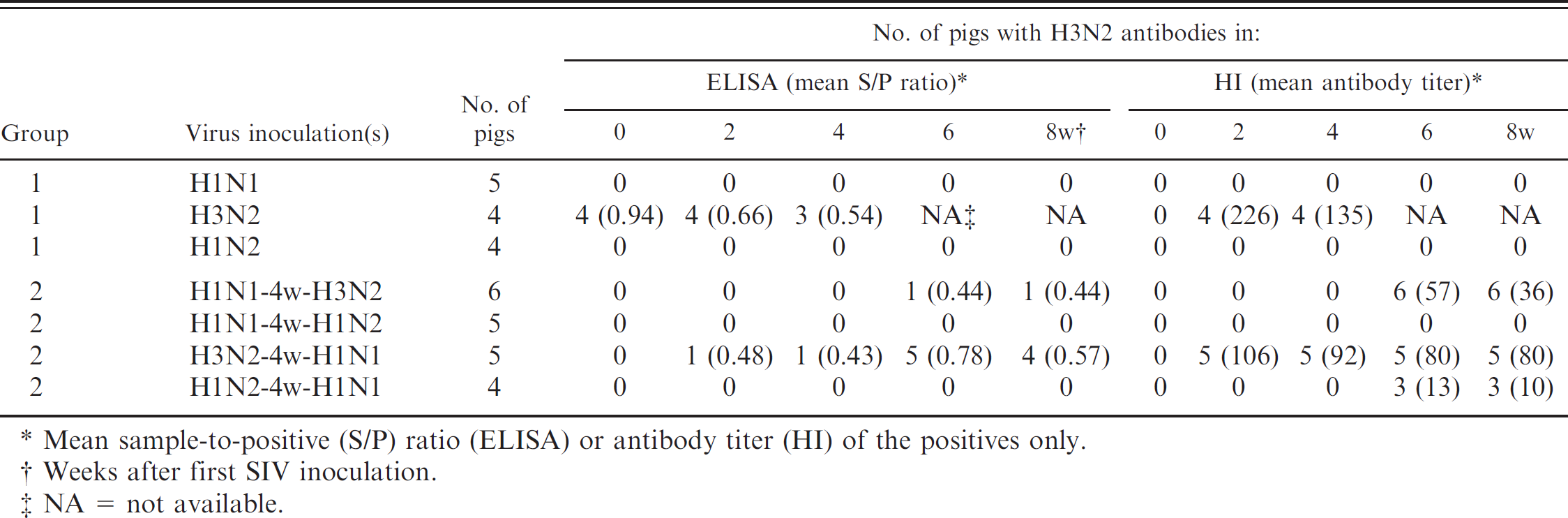
Mean sample-to-positive (S/P) ratio (ELISA) or antibody titer (HI) of the positives only.
Weeks after first SIV inoculation.
NA = not available.
Detection of H3N2 antibodies by commercial H3N2 Swine influenza virus (SIV) enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) test after a double vaccination of pigs with commercial SIV vaccines (group 3).
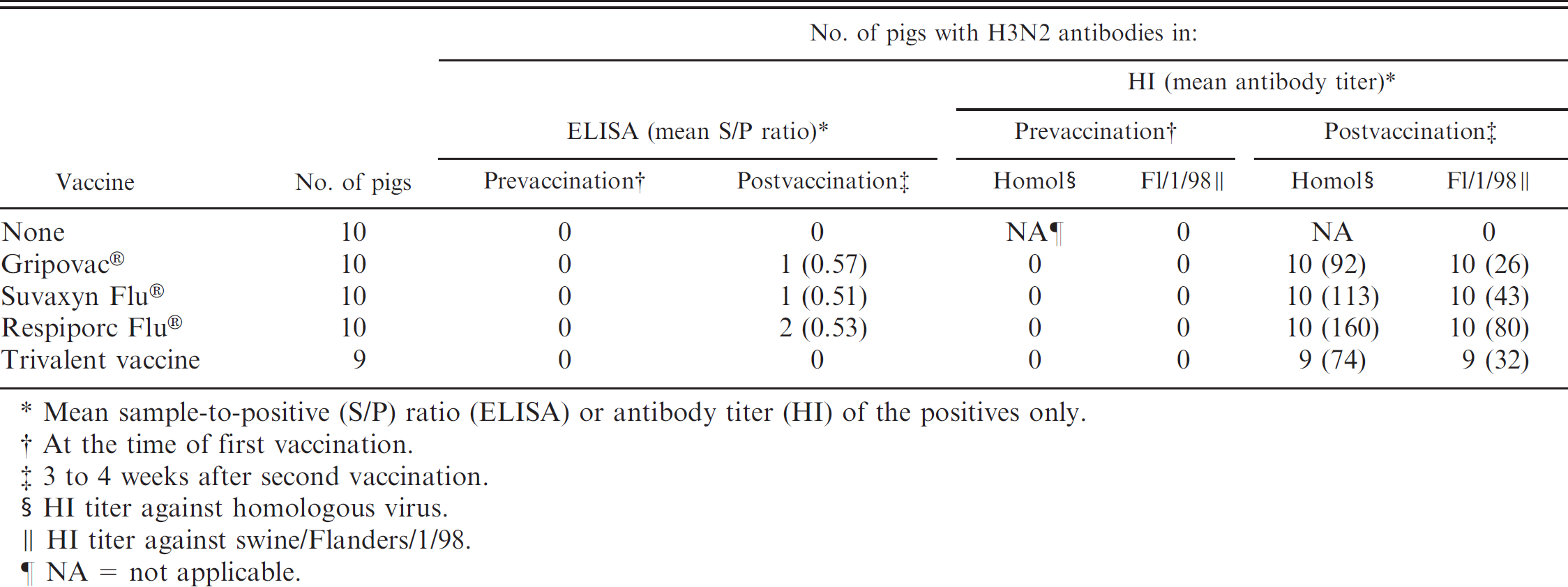
Mean sample-to-positive (S/P) ratio (ELISA) or antibody titer (HI) of the positives only.
At the time of first vaccination.
3 to 4 weeks after second vaccination.
HI titer against homologous virus.
HI titer against swine/Flanders/1/98.
NA = not applicable.
Detection of H3N2 antibodies by commercial H3N2 Swine influenza virus enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) test after a single vaccination* of pigs with infection immunity to H1N1, H3N2, or both subtypes (group 4).
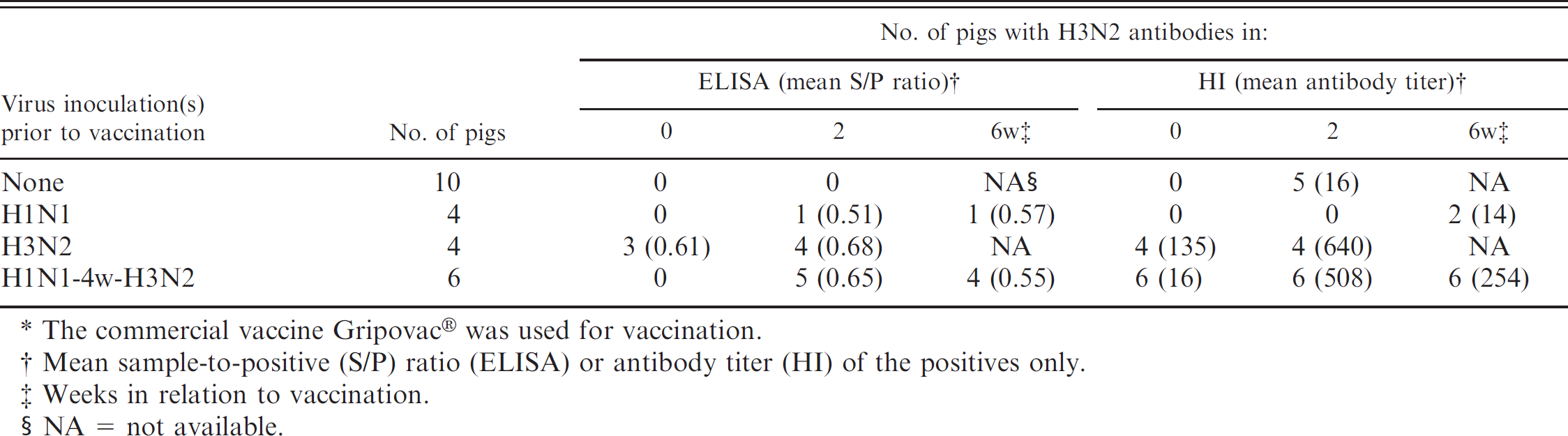
The commercial vaccine Gripovac® was used for vaccination.
Mean sample-to-positive (S/P) ratio (ELISA) or antibody titer (HI) of the positives only.
Weeks in relation to vaccination.
NA = not available.
Materials and methods
Serum samples
This study aimed to assess the capacity of the IDEXX H1N1 and H3N2 ELISAs to identify pigs infected with
European H1N1 or H3N2 SIV or pigs vaccinated with European SIV vaccines. Well-defined sera from previous experimental studies were tested in the ELISAs, as well as in HI tests to European H1N1 and H3N2 SIVs. Because various and complex situations may occur in the field, the study included sera from pigs that had been exposed to 1) a single SIV infection, 2) consecutive infections with 2 SIV subtypes, 3) a double vaccination with commercial SIV vaccines, and 4) infection followed by a single vaccination.
Group 1: single inoculation with H1N1, H3N2, or H1N2 SIV. Three groups of pigs had been inoculated intranasally with 107.0 EID50 of sw/Belgium/1/98 (H1N1), sw/Flanders/1/98 (H3N2), or sw/Gent/7625/99 (H1N2). The virus isolates are representative of H1N1, H3N2, and H1N2 viruses currently circulating in the European swine population, and they are described in detail elsewhere. 12 After inoculation, infection was confirmed by virus isolation from nasal swabs. Blood was collected 0, 2, 4, 6, and 8 weeks after virus inoculation.
Group 2: dual, consecutive inoculations with 2 SIV subtypes. Four groups of pigs had been inoculated consecutively with 2 SIV subtypes at a 4-week interval. The subtype combinations included H1N1 followed by H3N2 and vice versa, and H1N1 followed by H1N2 and vice versa. The mode and dose of virus inoculation and the virus isolates were identical to those in group 1 pigs. Infection with both subtypes was confirmed by virus isolation from nasal swabs. Blood was collected 0, 2, 4, 6, and 8 weeks after the first SIV inoculation.
Group 3: vaccination with commercial SIV vaccines. Three groups of 10 pigs each had been vaccinated twice intramuscularly with an interval of 3 weeks with 1 of 3 commercial SIV vaccines. The following commercial vaccines were used, the influenza virus strains in each vaccine are given between brackets: Gripovac® c (A/New Jersey/8/76 [H1N1] and A/Port Chalmers/1/73 [H3N2]), Suvaxyn Flu® d (sw/Netherlands/25/80 [H1N1] and A/Port Chalmers/1/73 [H3N2]), and Respiporc Flu® e (sw/Belgium/230/92 [H1N1] and sw/Belgium/220/92 [H3N2]). Another group of 9 pigs had been vaccinated with a trivalent SIV vaccine that is not yet registered. This vaccine contains sw/Haselunne/2617/03 (H1N1), sw/Bakum/1769/03 (H3N2), and sw/Bakum/1832/00 (H1N2) and is referred to as “trivalent vaccine.” f One group of 10 pigs was left unvaccinated. Blood was collected at the time of the first vaccination, and 3 to 4 weeks after the second vaccination.
Group 4: inoculation with H1N1 and/or H3N2 SIV followed by a single vaccination with commercial SIV vaccine. Three groups of pigs had been inoculated intranasally with H1N1 SIV (sw/Belgium/1/98), with H3N2 SIV (sw/Flanders/1/98) or with H1N1 followed 4 weeks later by H3N2. Four to 5 weeks after the last inoculation, they were given a single intramuscular vaccination with Gripovac®. One control group was left uninoculated and received a single administration of Gripovac® only.
Enzyme-linked immunosorbent assay
The IDEXX Swine Influenza Virus H1N1 and H3N2 ELISAs were used according to the manufacturer's instructions. a,b The serum samples were diluted 1/40 in sample diluent, and 100 μl of positive and negative controls as well as diluted samples were added in duplicate to the wells. Thereafter, plates were incubated for 30 min. After removing the samples, plates were washed thoroughly, and 100 μl of an anti-porcine horseradish-labeled conjugate was added to all wells. Plates were again incubated for 30 min. Next, substrate (tetramethylbenzidine) was added after a washing step, and 15 min later the reaction was stopped with a sodium dodecyl sulfate solution. Next, optical densities (OD) were read at 650 nm in an ELISA plate reader. Results were expressed as a sample to positive (S/P) ratio by using the following formula: (mean sample OD – mean negative control OD)/(mean positive control OD – mean negative control OD). Samples with S/P ratios ≥0.4 were considered to be positive for SIV antibodies. For the H3N2 ELISA, S/P ratios <0.4 but ≥0.3 were classified as suspect. All other S/P ratios were classified as negative. All samples and controls were tested in duplicate, and the test was performed at room temperature. Arithmetic mean S/P ratios of positive samples were calculated.
Hemagglutination inhibition test
All serum samples were also tested in the HI test using the same H1N1 and H3N2 strains that were used to inoculate the pigs, namely sw/Belgium/1/98 (H1N1) and sw/Flanders/1/98 (H3N2). Serum samples from group 3 animals were also tested for HI antibodies against H1N1 and H3N2 vaccine strains. The HI test was performed according to standard procedures. Briefly, sera were pretreated with receptor destroying enzyme from Vibrio cholerae g to remove nonspecific inhibitors of the agglutination and with a 50% chicken red blood cell (RBC) suspension to remove nonspecific agglutinins. Serial 2-fold dilutions (starting at 1:10 dilution) of the pretreated sera were made in phosphate buffered saline. After addition of 4 hemagglutinating units of the respective viruses and 1 hr of incubation, a 0.5% chicken RBC suspension was added. One hour later, HI titers were recorded as the reciprocal of the last sample dilution that completely inhibited hemagglutination. Geometric mean titers of positive samples were calculated.
Statistical analysis
Standard 2-sample Mann–Whitney U-tests were used to compare ELISA S/P ratios between groups. P < 0.05 was taken as the level of significance. Cohen's kappa coefficient was used to calculate agreement between HI test and ELISA.
Results
Performance of the H1N1 ELISA
The results of ELISA and HI tests for H1N1 SIV with sera collected after infection, vaccination, or combinations of both are shown in Tables 1, 2, and 3, respectively.
Groups 1 and 2: inoculation with 1 or 2 SIV subtypes (Table 1). All sera collected before SIV inoculation and all but 1 serum collected after inoculation with subtypes other than H1N1 were negative for H1N1 antibodies in the HI test, whereas all sera collected after H1N1 inoculation were positive. Individual HI antibody titers were between 20 and 160 in the pigs inoculated with H1N1 only or with H1N1 followed by H3N2. Higher antibody titers (80–320) were seen after H1N1 inoculation of previously H1N2 inoculated pigs or vice versa.
All sera collected before inoculation or after inoculation with H3N2 or H1N2 were negative in the ELISA. The sera collected after inoculation with H1N1 only (20 sera in group 1 + 22 sera in group 2) were also negative. With one exception, all H1N1-immune pigs remained negative after subsequent inoculation with H1N2 or H3N2. The pigs inoculated with H3N2 followed by H1N1 also tested negative. In the group inoculated consecutively with H1N2 and H1N1, all 4 pigs were positive at 2 weeks postinoculation with H1N1 and 1 pig was still positive at 4 weeks. Individual S/P ratios of positive pigs were between 0.43 and 0.65. All sera that were positive in the ELISA had HI antibody titers ≥160, although 13 of the total 20 sera with HI titers ≥160 were still negative in the ELISA.
Group 3: vaccination with commercial SIV vaccines (Table 2). The HI test was negative for all sera collected before vaccination or from unvaccinated pigs. All sera collected 3 to 4 weeks after vaccination were positive in the HI test against the homologous H1N1 virus and against sw/Belgium/1/98 (Table 2). HI antibody titers to the homologous H1N1 vaccine strains were higher than those to sw/Belgium/1/98.
The ELISA was negative for all prevaccination sera and for sera from unvaccinated controls. Postvaccination sera tested positive for all pigs vaccinated with Gripovac® or Suvaxyn Flu®, for 5 out of 10 pigs vaccinated with Respiporc Flu®, and for none of the pigs vaccinated with the trivalent vaccine. The S/P ratios ranged between 0.49 and 1.18 and were highest in the pigs vaccinated with Gripovac® (P < 0.05).
Group 4: inoculation with H1N1 and/or H3N2 SIV followed by a single vaccination with commercial SIV vaccine (Table 3). In the HI test, influenza naive control pigs and pigs previously infected with H3N2 tested negative for H1N1 antibodies before vaccination. A single vaccination resulted in an antibody response in all control pigs and 2 of the 4 H3N2-immune pigs, but antibody titers to sw/Belgium/1/98 remained very low (10–20). All pigs that had been previously infected with H1N1 or with H1N1 followed by H3N2 were HI antibody positive before vaccination and showed a dramatic booster of H1N1 HI antibodies after a single vaccination (320–5,120).
Prevaccination sera of the influenza naive control pigs and of the pigs previously infected with H3N2 were also negative by ELISA. A single administration of the commercial vaccine resulted in an ELISA antibody response in all control pigs and in 3 out of 4 H3N2-immune pigs. The pigs that had been previously infected with H1N1 or with H1N1 followed by H3N2 were ELISA antibody negative before vaccination, but all of them became positive after a single vaccination. Individual S/P ratios in the latter groups were between 0.84 and 1.44 and between 0.69 and 1.31, respectively, and they were higher than those in the control (0.49–0.86) and H3N2-immune groups (0.39–0.67; P < 0.05). The positive ELISA S/P ratios after vaccination of the H1N1 immune pigs corresponded to high HI antibody titers (320–5,120). However, pigs in the control and H3N2-immune groups generally had HI antibody titers ≤20 after the vaccination, and they were also ELISA positive.
Comparison between hemagglutination inhibition (HI) test and enzyme-linked immunosorbent assay (ELISA).*
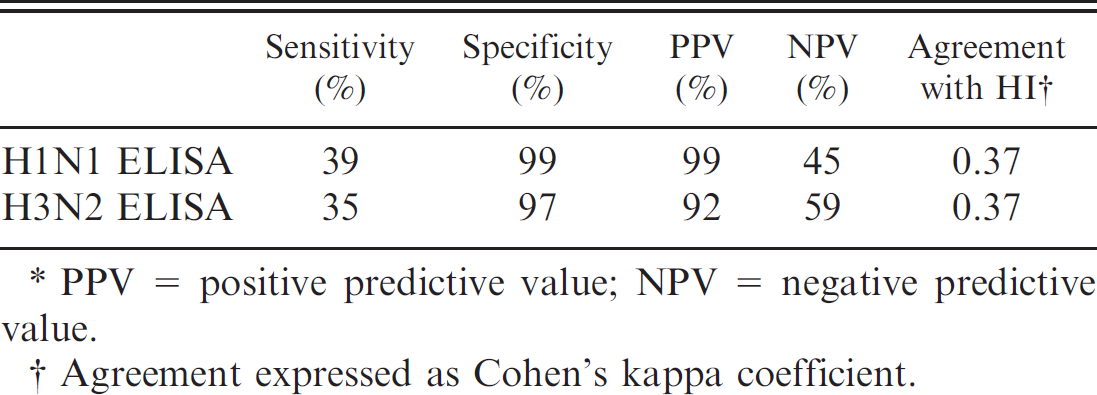
PPV = positive predictive value; NPV = negative predictive value.
Agreement expressed as Cohen's kappa coefficient.
Comparison between HI test and ELISA (Table 7). The overall relative sensitivity and specificity of the H1N1 ELISA was 39% and 99%, respectively. Agreement between ELISA and HI was low as illustrated by the Cohen's kappa coefficient of 0.37. In Belgium, the prevalence of SIV H1N1 antibodies in fattening pigs is about 66.7% (Labarque G, Vyt P, Van Reeth K, et al.: Seroprevalence ofdifferent swine influenza virus subtypes in swine in Belgium in 2001–2003. Proc 18th International Pig Veterinary Society Congress, p. 84). This gives a positive predictive value (PPV) of 99% and a negative predictive value (NPV) of 45%.
Performance of the H3N2 ELISA
The results of ELISA and HI tests for H3N2 SIV with sera collected after infection, vaccination, or combinations of both are shown in Tables 4, 5, and 6, respectively.
Groups 1 and 2: inoculation with 1 or 2 SIV subtypes (Table 4). All sera collected before inoculation and sera collected after inoculation with subtypes other than H3N2 were negative for H3N2 antibodies in the HI test, except for 3 out of 4 animals inoculated with H1N2 followed by H1N1, which had very low HI antibody titers (10–20). All sera collected after H3N2 inoculation were positive. Individual HI antibody titers were between 80 and 320 in the pigs inoculated with H3N2 only and between 20 and 160 in pigs inoculated with H3N2 in combination with H1N1.
Most preinoculation sera and all sera collected after inoculation with H1N1 or H1N2 were negative in the ELISA. Four of the total 33 preinoculation sera, however, were clearly positive in the ELISA (range: 0.83–1.13). All 4 sera were from the singly H3N2-inoculated subgroup in group 1, and S/P ratios decreased after the H3N2 inoculation. Only 1 of the 5 singly H3N2-inoculated pigs in group 2 was positive in the ELISA, whereas 5 out of 5 and 4 out of 5 pigs became positive at, respectively, 2 and 4 weeks after the subsequent inoculation with H1N1. Only 1 of the 6 H1N1-immune pigs became positive after inoculation with H3N2. Individual S/P ratios of the positive pigs in group 2 ranged between 0.43 and 1.02. All these sera had an HI titer ≥40.
Group 3: vaccination with commercial SIV vaccines (Table 5). The HI test was negative for all sera collected before vaccination or from unvaccinated controls. All sera collected 3–4 weeks after vaccination were positive in the HI test against the homologous H3N2 virus and against sw/Flanders/1/98 (Table 5). All vaccinated pigs had higher HI antibody titers to the homologous H3N2 vaccine strains than to sw/Flanders/1/98. The ELISA was negative on the prevaccination sera and on sera from unvaccinated controls. Only 1 or 2 out of 10 pigs vaccinated with Gripovac®, Suvaxyn Flu®, or Respiporc Flu®, and none of the pigs vaccinated with the trivalent vaccine, tested positive. Individual S/P ratios were ≤0.60.
Group 4: inoculation with H1N1 and/or H3N2 SIV followed by a single vaccination with commercial SIV vaccine (Table 6). In the HI test, influenza naive control pigs and pigs previously infected with H1N1 only tested negative for H3N2 antibodies before vaccination. Half of these pigs became positive after a single vaccination, but antibody titers to sw/Flanders/1/98 remained low (10–40). All pigs that were previously infected with H3N2 or with H1N1 followed by H3N2 were HI antibody positive before vaccination and showed a dramatic booster of H3N2 HI antibodies after a single vaccination (160–2,560).
Prevaccination sera of the influenza naive control pigs and of the pigs previously infected with H1N1 or with H1N1 followed by H3N2 were negative by ELISA. A single administration of the commercial vaccine resulted in an ELISA antibody response in none of the control pigs, only 1 of the H1N1-immune pigs, and 5 out of 6 (H1N1 + H3N2)-immune pigs. Three of the 4 pigs that had been previously infected with H3N2 were ELISA positive before the vaccination, and all 4 were positive thereafter. All 3 infection-immune groups had similar ELISA S/P ratios after vaccination, and individual values ranged from 0.40 to 0.91. Except for the serum of the H1N1-infection immune pig, all positive sera had HI antibody titers ranging between 80 and 2,560.
Comparison between HItest and ELISA (Table 7). The overall relative sensitivity and specificity of the H3N2 ELISA was 35% and 97%, respectively. Agreement between the 2 tests was low and similar to that of the H1N1 ELISA (Cohen's kappa coefficient of 0.37). In Belgium, the prevalence of SIV H3N2 antibodies in fattening pigs is about 51.2% (Labarque G, et al.: Seroprevalence of different swine influenza virus subtypes in swine in Belgium). This gives a PPV of 92% and a NPV of 59%.
Discussion
This is the first extensive validation study of the IDEXX H1N1 and H3N2 SIV antibody ELISA with well-defined sera from pigs that had been infected with European SIVs or vaccinated with European vaccines. These ELISAs are based on North American H1N1 and H3N2 SIVs that are antigenically different from their European counterparts. This raises the question as to whether the ELISAs are appropriate to detect antibodies against European SIVs, and a lack of sensitivity had already been reported in a limited European study. 3 The present data show that the ELISAs frequently fail to identify pigs infected with contemporary European SIVs or pigs vaccinated with commercial European vaccines, especially if they were previously SIV naive. An unexpected specificity problem was also encountered with the H3N2 ELISA, in which 4 out of 103 preinfection/prevaccination sera tested positive. These sera were shown to be negative in 3 other serological assays for SIV, including HI and virus neutralization tests and a subtype-unrestricted immunoperoxidase monolayer assay, and the nonspecific reactions cannot be explained. In contrast with the ELISAs, the HI test showed an excellent sensitivity and specificity in the present study. This is in part related to the use of identical H1N1 (sw/Belgium/1/98) and H3N2 (sw/Flanders/1/98) strains in the test as those used for experimental infection of the pigs. However, these strains are still appropriate for the serological diagnosis of the currently circulating SIVs and they resemble H1N1 and H3N2 SIVs in other European countries. In addition, all pigs vaccinated with commercial SIV vaccines showed HI antibodies to both the homologous vaccine viruses and the heterologous strains used in the test.
Pigs that had been consecutively infected with 2 of the 3 European SIV subtypes were more frequently positive in both ELISAs than singly H1N1- or H3N2-infected pigs. Similarly, most H1N1 and/or H3N2 infection–immune pigs tested positive in the ELISAs after a single administration of commercial SIV vaccine. The higher ELISA detection rates are likely due to increased antibody levels to the viral HA, as demonstrated by the high HI antibody titers in most pigs, or to other viral proteins. These observations agree with a study in which only hyperimmune sera, with HI titers ≥1,280 to the homologous virus, were invariably positive in the ELISAs (Schmoll F, Lang C, Dürrwald R, et al.: 2006, Restriction of commercial swine influenza ELISA kits for subtype-specific differentiation of swine influenza-A viruses. Proc 19th International Pig Veterinary Society Congress, p. 135). However, the IDEXX H1N1 and H3N2 ELISAs could not discriminate between hyperimmune sera to European H1N1, H3N2, or H1N2 strains in that study. Also, a second commercial ELISA kit showed a similar low specificity and sensitivity as the IDEXX ELISAs. It is noteworthy that in the field, pigs are frequently infected and/or vaccinated with multiple influenza virus subtypes and variants. This may explain why several field studies report relatively high seropositivity rates in the IDEXX ELISAs and, in some cases, a reasonable level of agreement between ELISA and HI test results. In one study in the United States, for example, there was an overall 85.5% agreement between the IDEXX H1N1 ELISA and the HI test. 10 In a field study in Austria, SIV seroprevalence rates for H1N1 and H3N2 did not differ significantly in the IDEXX ELISAs (38% and 15%, respectively) and in the HI test (42% and 19%, respectively). 3 The same investigators, however, had previously described low sensitivity and specificity of the ELISAs based on results with well-defined sera from experimentally infected animals. In addition, more than 1 out of 3 of the field sera yielded discrepant results in the IDEXX ELISA and the second commercial ELISA kit tested.
The poor sensitivity of the ELISAs may be due to antigenic differences between the strains in the test kits and the European H1N1 and H3N2 SIVs, to a low sensitivity of the tests as such, or to a combination of both. Some of the present results suggest that antigenic differences may play a role. Only 1 of the 4 SIV vaccines used (Gripovac®) contains a classical H1N1 virus like the IDEXX H1N1 ELISA, and pigs vaccinated with this vaccine had the highest ELISA positivity rates and S/P ratios. On the other hand, detailed information is lacking about the strains used in the ELISAs and their antigenic relatedness to the SIVs used in the present study, as well as data about the protein specificity of the ELISAs. Unlike the HI test, which exclusively detects antibodies against the distal, most variable part of the viral HA, 8 indirect SIV antibody ELISAs may also detect antibodies against conserved viral proteins like matrix proteins or the stem of the HA. More technical details and a different experimental setup are thus needed to determine the reason(s) for the low specificity. Interestingly, a low sensitivity of the IDEXX H1N1 ELISA has also been reported in 2 independent experimental infection studies with North American, classical H1N1 SIVs from 1992. 10,18 In these studies the ELISA detected antibodies in only 6 of 72 and 35 of 60 postinfection sera examined, whereas all sera were positive in HI tests. The ELISA clearly failed to identify H1N1-seropositive animals as effectively as the HI test, especially at the early stage of infection. Other investigators have tested pig hyperimmune sera against 10 antigenically different U.S. H1 SIVs isolated between 1930 and 2004. 17 These sera were obtained by a double vaccination of pigs with inactivated virus followed by a live virus challenge. All sera showed high HI antibody titers (≥640) to the homologous virus, but the ELISA was only marginally above the S/P positive cut-off ratio of 0.4 for some modern H1 strains. The authors therefore concluded that the HI assay with a panel of H1 and H3 viruses is a better diagnostic test for swine influenza than the ELISA.
Swine influenza virus serology is more challenging now than in the past. During the last 10 years novel SIV subtypes and reassortants have emerged in both Europe and North America. Besides, the H1N1, H3N2, and H1N2 SIVs on both continents have a different genetic constellation and antigenically distinct hemagglutinin proteins. 5 The value of antibody ELISAs based on whole SIVs is diminished in this complex epidemiological situation. In fact, there are currently only 2 options for the serological diagnosis of SIV. A first option is to perform HI tests using strains that are antigenically similar to the predominant SIVs in a given region. In most regions of Western Europe, at least 3 separate HI tests including SIVs with an avian-like H1, a human-like H1, and an H3 are required. Although serological cross-reactions cannot be completely excluded, the HI test can largely discriminate between antibodies to these 3 HAs in unvaccinated pigs. 15 A second option is to use one subtype-unrestricted universal antibody ELISA based on conserved influenza virus proteins. 2 The nucleoprotein appears to be the best candidate for this purpose, 2 and several NP-based, usually multispecies ELISA kits are on the market. This second approach is less complicated than the HI test, but it does not provide information about the subtype or antigenic characteristics of the infecting viruses. Caution must be exerted, however, because there are no published data about the performance of such commercial influenza antibody ELISAs with sera from infected or vaccinated pigs. In preliminary investigations with one of these kits and sera from the present study, pigs infected with 1 or with 2 different SIV subtypes (groups 1 and 2) were usually positive. Part of the vaccinated pigs (group 3), in contrast, tested negative, and further validation studies are certainly needed. Finally, ELISA tests based on the influenza virus nonstructural NS1 protein, which is expressed only during viral replication, appear to be suitable to differentiate pigs vaccinated with current killed vaccines from infected animals. 2 Such tests are not yet commercially available, and they could considerably improve the serological diagnosis of SIV.
In conclusion, the HI test remains the method of choice for the serological diagnosis of SIV, to monitor a herd's immune status, or to determine the prevalence of specific SIV subtypes in the European swine population. The IDEXX SIV ELISAs will likely detect part of the SIV-infected and/or -vaccinated pigs under field conditions, but the interpretation of positive ELISA test results will be difficult.
Acknowledgements
The authors thank Lieve Sys, Melanie Bauwens, and Nele Dennequin for technical assistance and Fernand De Backer for help with animal experiments. The authors would also like to thank IDEXX Laboratories Inc. for providing the ELISA kits. This study was in part supported by the “European Surveillance Network for Influenza in Pigs 2 (ESNIP2)” (SSPE-CT-2005–022749) and “Lab-on-site” (SSPE-CT-2004–513645), a coordination action and specific targeted research project respectively in the 6th framework program of the European Commission.
Footnotes
a.
IDEXX Swine Influenza Virus H1N1 ELISA, IDEXX Laboratories Inc., Westbrook, ME.
b.
IDEXX Swine Influenza Virus H3N2 ELISA, IDEXX Laboratories Inc., Westbrook, ME.
c.
Gripovac®, Merial, Lyon, France.
d.
Suvaxyn Flu®, Fort Dodge Animal Health, Madison, NJ.
e.
Respiporc Flu®, Impfstoffwerk Dessau-Tornau Biologika GmbH, Dessau-Roßlau, Germany.
f.
Trivalent vaccine, Impfstoffwerk Dessau-Tornau Biologika GmbH, Dessau-Roßlau, Germany.
g.
Receptor destroying enzyme from Vibrio Cholerae, Sigma-Aldrich, St. Louis, MO.
