Abstract
New tools to detect exposure of free-range Eurasian wild boar (Sus scrofa scrofa) to pathogenic mycobacteria would be valuable for improved disease surveillance and wildlife management. Two hundred sera from wild boar of known Mycobacterium bovis infection status were used to evaluate test suitability for the detection of antibodies against M. bovis and Mycobacterium avium subsp. paratuberculosis (or cross-reacting members of the M. avium complex). Two traditional enzyme-linked immunosorbent assays were evaluated using M. bovis purified protein derivative (bPPD) and paratuberculosis protoplasmatic antigen 3 (PPA3) as antigens, respectively, and a new point-of-care test format for bovine tuberculosis (bTB) that uses the innovative dual-path platform (DPP TB) test. The effect of individual factors (sex, age, lesions) on the diagnostic performance of the serologic tests was also determined. Although the DPP had a sensitivity of 89.6% and a specificity of 90.4%, for bPPD, the sensitivity was 79.2% and the specificity 100%. Both tests had a kappa agreement of 0.80. Sixty-five of 68 (95.6%) wild boar sera with antibodies against the PPA3 antigen corresponded to known M. bovis–infected wild boar. Significant differences were not observed in the bPPD and DPP readings among lesion categories or between age classes. A slight sex-related difference in sensitivity toward males in the DPP was found, but it was not detected in the bPPD enzyme-linked immunosorbent assay. The results support the use of antibody-based diagnostic tests for both large-scale and individual bTB testing of Eurasian wild boar and suggest that wild boar cannot be used as sentinels for infections caused by M. avium complex members.
Introduction
The Eurasian wild boar (Sus scrofa scrofa) is the most widespread free-ranging ungulate in large areas of Europe. Depending on habitat and management, wild boar numbers range from almost zero to up to 90 individuals per square km. 1 The continuing expansion of this species raises concerns regarding the control of diseases shared with livestock. 15 Due to their rooting habits, omnivorous diet, ability to cross barriers and contact with a broad range of wild and domestic animals, 7,16 and susceptibility to mycobacterial infections, 19 wild boar or their feral pig relatives have been used as disease sentinels. 22 In southwestern Europe, bovine tuberculosis (bTB), caused by Mycobacterium bovis, and paratuberculosis, caused by Mycobacterium avium subsp. paratuberculosis infection, are diseases of concern in wild and domestic ruminants. 24 Hence, setting up new tools to detect exposure of free-range wild boar to pathogenic mycobacteria would be a valuable tool for improved disease surveillance and wildlife management.
The mycobacterial species isolated from wild boar include M. bovis and other members of the Mycobacterium tuberculosis complex (MTBC); M. avium subsp. avium, M. avium subsp. hominissuis, and M. avium subsp. paratuberculosis of the M. avium complex (MAC); and other potentially pathogenic mycobacteria. 19 In southwestern Europe, M. bovis is the most frequently detected mycobacterial pathogen in wild boar, with local prevalence as high as 52% in certain areas. 13 The abundant populations living in Mediterranean habitats of southwestern Europe are considered true wildlife reservoirs for M. bovis infection. 21 In contrast, members of the MAC are only sporadically detected. 2,23 Moreover, although most M. bovis–infected wild boar develop characteristic gross lesions, 20 infections with MAC species often cause only minimal pathology, if any. 11
Bacterial culture is the gold standard technique for diagnosing mycobacterial infections. However, its sensitivity depends on the number and selection of tissues processed 10 and on sample quality. 9 Antemortem TB testing of wild boar has been attempted by means of serology, tuberculin skin test, and gamma interferon assay. The skin testing requires handling the dangerous wild boar twice and has a poor specificity, 17 whereas gamma interferon testing requires fresh blood samples, an equipped laboratory environment, and skilled personnel. 11 Detection of specific antibodies would be a useful alternative tool. Serum samples are easy to collect, transport, and store. Moreover, serology is faster, less expensive and complex, and more suitable for live animals than culture or other techniques. 26 Recently, a traditional enzyme-linked immunosorbent assay (ELISA), which uses M. bovis purified protein derivative (bPPD) as a coating antigen, has shown a fair sensitivity (73%) and good specificity (96%) in detecting infected wild boar. 3 Also, a lateral-flow rapid test a has demonstrated a sensitivity of 77% and specificity of 97% for detecting M. bovis infection in Eurasian wild boar. 18 More recently, a new point-of-care test format for bTB that uses the innovative dual-path platform (DPP) technology a has shown enhanced specificity in elephants and red deer. 6,14 These rapid tests have practical advantages over other diagnostic tools, because they are easy to perform, can be undertaken on site, and produce results typically within 20 min. 18
The present study evaluated the bPPD ELISA, the DPP TB test, and the paratuberculosis protoplasmatic antigen 3 (PPA3) ELISA for the detection of MAC using 200 sera collected from free-ranging wild boar and determined whether individual factors (sex, age, and the presence of bTB compatible lesions) potentially affect the diagnostic performance of the serologic tests.
Material and methods
Samples
The samples included 96 culture-confirmed, M. bovis–infected Eurasian wild boar from several sites in southern Spain and 104 culture-negative, lesion-negative wild boar from sites in northern Spain with no bTB history. Sex was known for 171 animals (83 females, 88 males). Based on tooth eruption patterns, 25 wild boar between 7 and 12 months of age were classified as yearlings (n = 29), those between 12 and 24 months as subadults (n = 43), and those older than 2 years as adults (n = 90). The presence or absence of bTB-compatible lesions was recorded in detail for all but 1 of the M. bovis–infected wild boar; 94 were also classified by the following bTB lesion scores 5,20 : zero for animals with no visible lesions (n = 6), 1 for animals with lesions smaller than 1 cm (n = 55), and 2 for animals with at least 1 lesion larger than 1 cm (n = 33). One animal with lesions could not be classified because data on lesion size were not available. Data on age and sex were available for all 96 infected wild boar. None of the wild boar was cultured for MAP. Additionally, 12 sera from experimentally M. avium subsp. avium–infected wild boar were tested by bPPD ELISA. 11
In-house ELISAs
Serum samples were tested for anti-PPD immunoglobulin antibodies by means of an ELISA using bovine tuberculin purified protein derivative (bPPD) b as antigen and protein G horseradish peroxidase c as a conjugate applying the previously described protocol 3 with the following modifications. Briefly, after coating the plates for 18 hr at room temperature, wells were washed with phosphate buffered saline (PBS) solution containing 0.05% Tween 20 (PBST) and blocked for 1 hr at 37°C with 200 μl of 5% skim milk in PBST. Sera were not adsorbed with Mycobacterium phlei saline suspension but added directly on plate (100 μl/well) at a dilution of 1:200 in PBS and incubated for 1 hr at 37°C. Samples, blanks, and positive and negative controls were tested in duplicate in each plate. Protein G was added (100 μl/well) at a dilution of 2.5 μg/ml in PBST and incubated at 37°C for 1 hr. After revealing, the reaction was stopped with 50 μl/well of sulfuric acid (H2SO4; 3N), and optical density (OD) was measured in a spectrophotometer at 450 nm. Pooled anti-PPD–positive serum was obtained from wild boar previously described as M. bovis culture positive and negative controls from TB-free wild boar previously described as M. bovis culture negative from bTB-free areas.
Sample results were expressed as an ELISA percentage (E%) that was calculated using the following formula: [sample E% = (mean sample OD/2 × mean negative control OD) × 100]. Cut-off values were defined as the ratio of the mean sample OD to the sum ODs of the negative controls. The cut-off with the best specificity was chosen. Serum samples with E% values greater than 100 were considered positive.
An ELISA using the PPA3 was performed to detect antibodies against MAP in the wild boar samples. The protocol was performed following the technique as previously described 24 but without the adsorption with M. phlei saline suspension. The PPA3 ELISA results were expressed as an ELISA percentage (E%) of the blanks that was calculated using the formula [sample E% = (mean sample OD/2 × mean blank ODs) × 100] because neither negative nor positive controls were available for wild boar.
Dual-path platform tuberculosis test
The DPP technology involves 2 nitrocellulose strips, which are connected in a “T” shape inside the cassette device, 14 which allows independent delivery of the test sample and the antibody-detecting reagent (protein A/G hybrid conjugated to colloidal gold particles). The DPP TB assay was performed using 5 μl of wild boar serum, 2 drops of buffer in the sample well, and 4 drops of buffer in the conjugate well, and it provided a result in 20 min. The presence and intensity of either of the 2 separate test lines (T1, MPB83 antigen; T2, CFP10/ESAT-6 fusion protein) were evaluated visually (presence or absence of a band of any intensity) and by a DPP optical reader, as described previously. 14 Reactivity of CFP10/ESAT-6 and/or MPB83 above the cut-off value of 5.0 relative light units was considered a positive result for the presence of antibody. Reactivity below 5.0 relative light units with the 2 test antigens was taken as an antibody-negative result.
Number of sera from known Mycobacterium bovis–free (culture-negative) and known M. bovis–infected (culture-positive) Eurasian wild boar testing positive to each of the serologic tests used. *
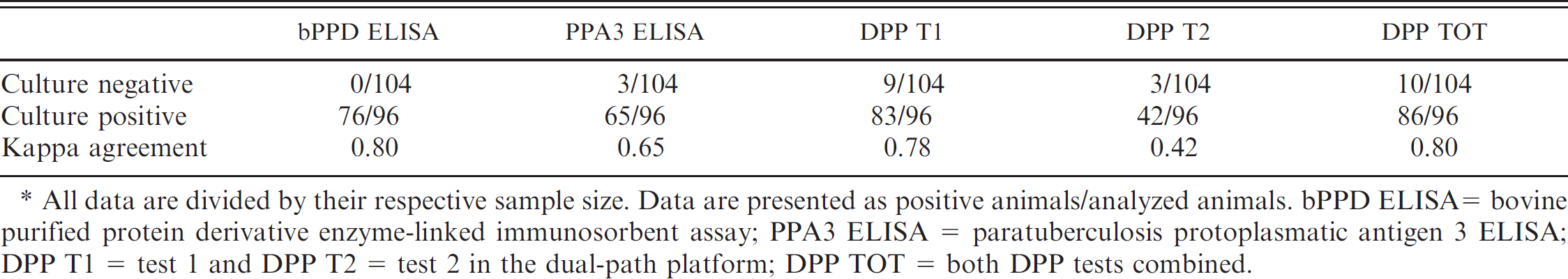
All data are divided by their respective sample size. Data are presented as positive animals/analyzed animals. bPPD ELISA = bovine purified protein derivative enzyme-linked immunosorbent assay; PPA3 ELISA = paratuberculosis protoplasmatic antigen 3 ELISA; DPP T1 = test 1 and DPP T2 = test 2 in the dual-path platform; DPP TOT = both DPP tests combined.
Statistical tests
A Kruskal–Wallis nonparametrical test was used to evaluate the effect of bTB lesion categories on the readings of the bPPD ELISA (E%) and the DPP (relative light units) for DPP T1 and T2. Chi-square tests were used for comparisons between age and sex classes. Expected frequencies were always greater than 5. The kappa agreement test was used to calculate the degree of agreement of each test with the gold standard (bacterial culture).
Results
Table 1 presents the positivity to each test of the known M. bovis–infected samples and of the culture- and lesion-negative wild boar. The DPP sensitivity (positives/known infected) was 86 of 96 (89.6%), and DPP specificity (negatives/known uninfected) was 94 of 104 (90.4%). The ELISA sensitivity was 76 of 96 (79.2%), and ELISA specificity was 104 of 104 (100%; Table 1). Kappa agreements are also shown in Table 1. Sixty-five of 68 (95.6%) wild boar sera with positive levels of antibodies against the PPA3 antigen corresponded to known M. bovis–infected wild boar. Figure 1 shows the serum antibody responses of 96 known M. bovis–infected wild boar to each of the studied antigens. None of the 12 sera from M. avium subsp. avium–infected wild boar were positive in the bPPD ELISA.
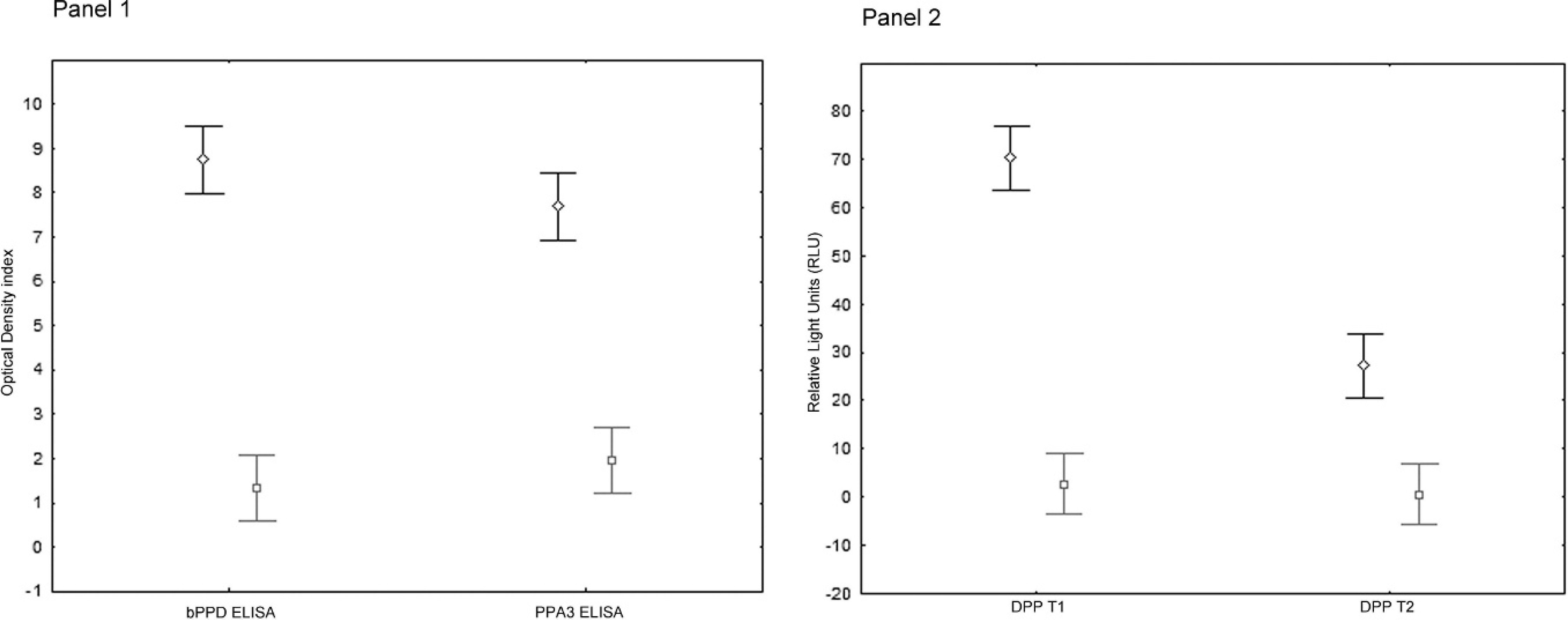
Mean Mycobacterium bovis purified protein derivative (bPPD) and paratuberculosis protoplasmatic antigen 3 (PPA3) optical density index (± standard deviation;
The 94 M. bovis–infected wild boar with known lesion status included 6 individuals with no visible lesions, 33 with lesions smaller than 1 cm, and 55 with larger lesions. Kruskal–Wallis tests did not reveal statistical differences in the readings of the bPPD ELISA (E%) and the DPP (relative light units) for DPP T1 and T2 by means according to bTB lesion categories (chi-square = 1.52, 2.14, 1.46, respectively; P > 0.05 in all cases; Fig. 2).
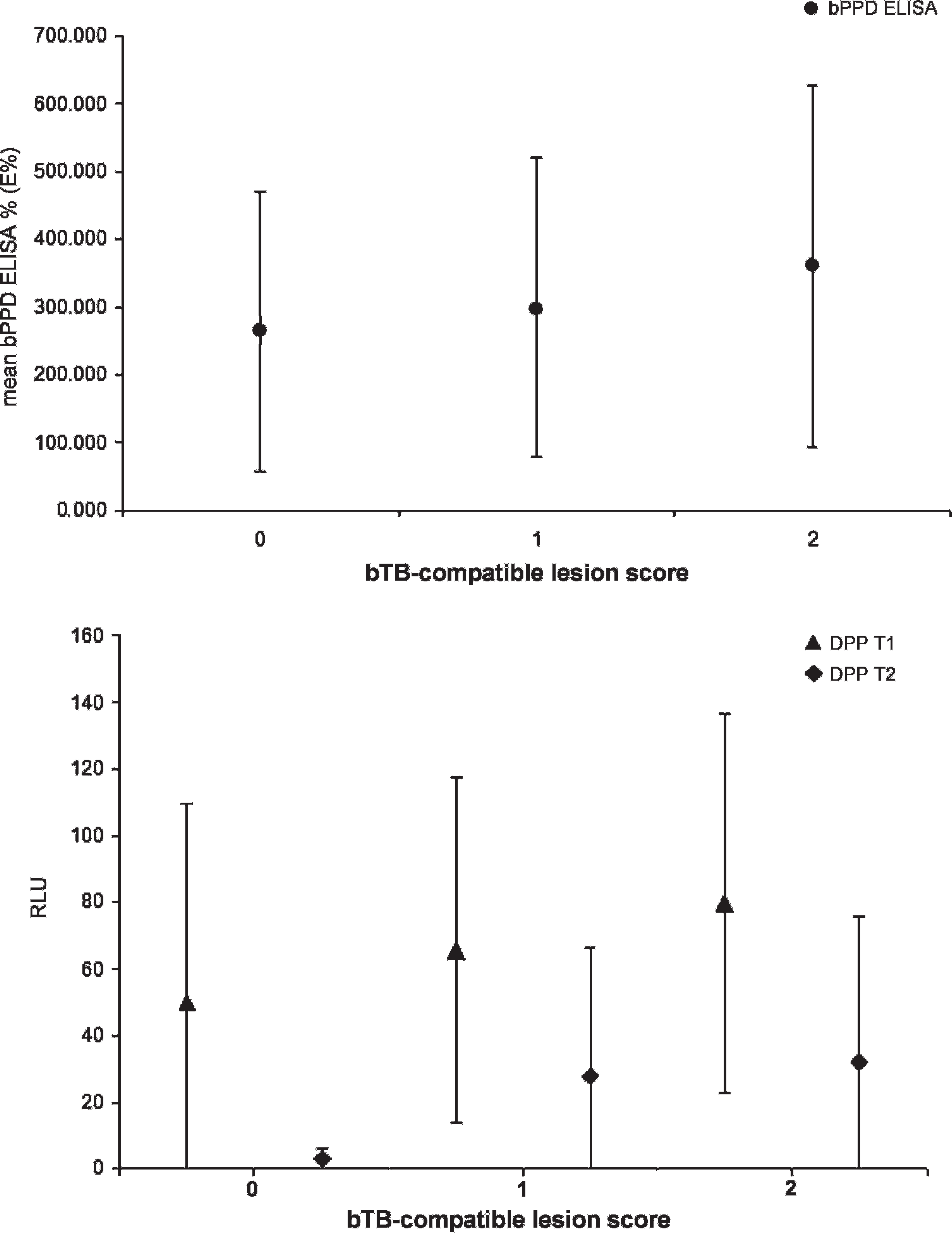
Mean Mycobacterium bovis purified protein derivative enzyme-linked immunosorbent assay (bPPD ELISA) percentage (
No age-related differences in sensitivity were observed for bPPD ELISA and DPP (chi-square = 1.36, 0.32, respectively; 2 degrees of freedom; P > 0.05 in both cases). No sex-related difference in sensitivity was recorded in the bPPD ELISA (chi-square = 1.975, P > 0.05). When the more sensitive DPP was used, sera from known infected male wild boar had a higher sensitivity than those from known infected female wild boar (chi-square = 4.2; 1 degree of freedom; P < 0.05; Fig. 3).
Discussion
The results confirm that in wild boar, in contrast to many other mammalian species, antibodies against M. bovis can be detected reliably with traditional and rapid serology tests. Both tests showed good agreement with bacterial culture. Although the bPPD ELISA was about 10% more specific than the DPP, the DDP test was about 10% more sensitive. As expected, sera from M. bovis–positive wild boar tended to react with the PPA3 antigen designed to detect MAC antibodies, and serum antibody positivity to PPA3 in M. bovis–negative wild boar was low The high specificity of the bPPD ELISA was further confirmed by the absence of positivity among 12 wild boar that had been infected with M. avium subsp avium.
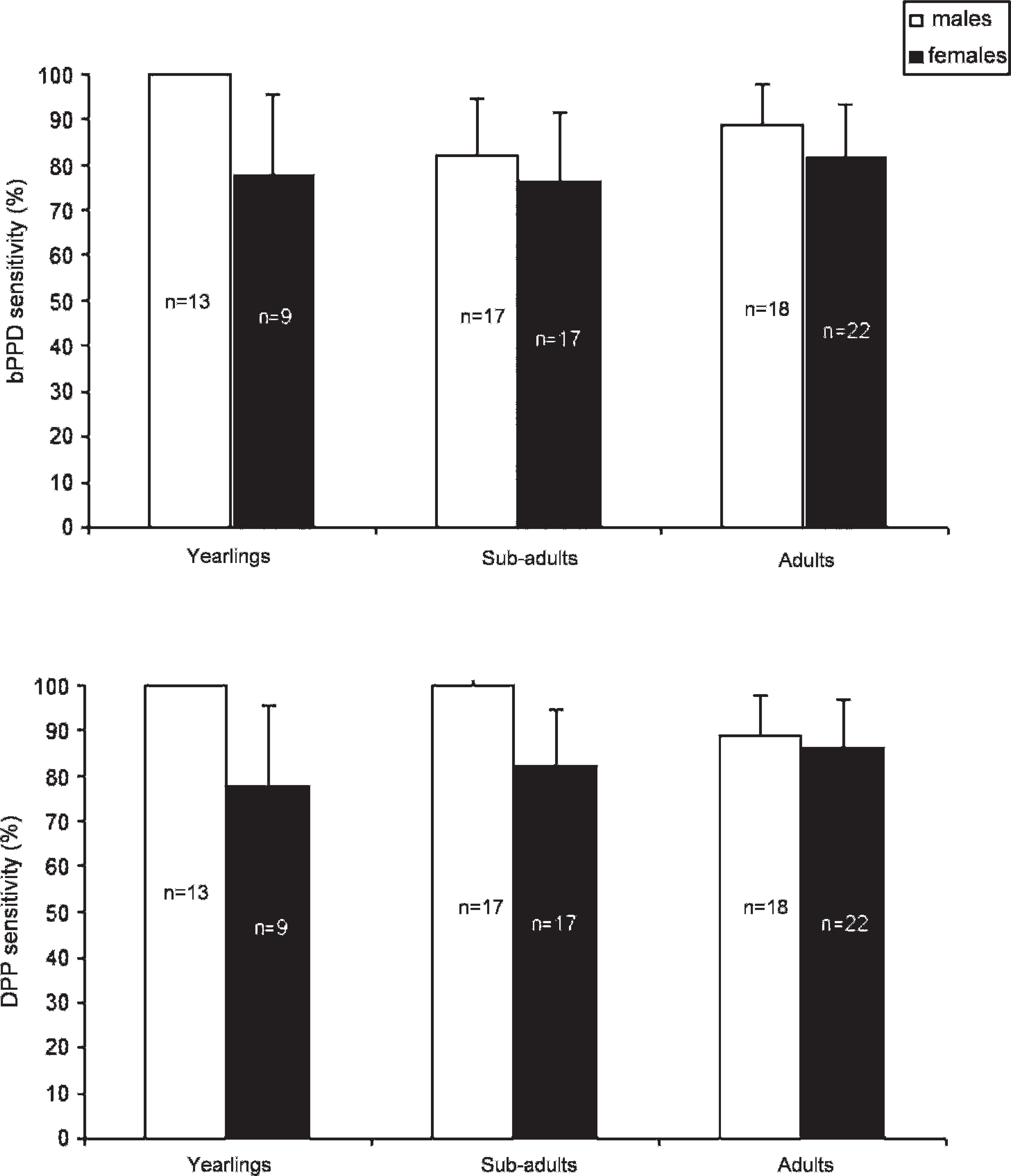
Mycobacterium bovis–purified protein derivative (bPPD) enzyme-linked immunosorbent assay percentage (
The ELISA is relatively inexpensive and can be easily automated to process large numbers of samples. In the current study, the decision was made to use the negative sample OD as reference for establishing the ELISA cut off,
24
rather than the positive control OD, because negative controls were expected to be more similar between laboratories than positive controls. Modifications in the cut-off value fairly reduced the specificity (11%, from 100 to 89), while a sensitivity increase was very low (5%, from 79 to 84). At the chosen cut off for bPPD ELISA, specificity was the maximum. Given its high specificity and fair sensitivity, this test is suitable for large-scale disease surveillance and disease monitoring in this host. It is also a potentially useful tool as a means to monitor the effects of eventual disease control strategies. Because mycobacterial diseases also infect domestic pigs,
In most species, the antibody response to M. bovis infection is limited, and, thus, most attempts to use antibody detection in bTB diagnosis have shown a rather limited sensitivity. 8 In swine, the existence of a dichotomy between the humoral and cellular immune response has been suggested. 27
The sera for PPA3 antibodies were tested to determine if wild boar could act as potential paratuberculosis sentinels, but the results obtained in the study could not support this possibility. First, the responsiveness of M. bovis–negative wild boar to this antigen was very low, suggesting no or little exposure to MAP (or other cross-reacting mycobacteria) in southwestern Europe. There was almost no indication of exposure to MAP in sera from wild boar collected in bTB-negative sites from northern Spain, although MAP presence in wild ruminants in these regions has been reported repeatedly. 4 Second, the vast majority of the PPA3 ELISA reactors (95%) were actually confirmed M. bovis–positive wild boar. Therefore, the study findings suggest that cross-reactions exist between mycobacteria rather than a genuine antibody response to MAP. Alternatively, the lack of responsiveness to PPA3 could also be a consequence of wild boar sera not reacting well with immunoglobulin G to MAP antigens or that the specific ELISA protocol used was not sensitive enough.
Regarding the individual factors affecting the response to the serologic tests, it was found that the differences encountered between the mean bPPD E% and the DPP readings among known infected wild boar with different lesion scores were not significant. In addition, no age-related differences in sensitivity for bPPD ELISA and DPP were observed. Finally, slight sex-related differences existed in sensitivity towards males in the DPP assay but not in the bPPD ELISA. The results of the current study support the suitability of both tests for any age or sex group and any lesion status. The finding of a slight trend towards a higher sensitivity in individuals with more severe lesions is in agreement with the stronger antibody response expected as disease progresses. 8 The finding of no age-related difference in sensitivity suggests that both tests have the same likeliness of detecting recently exposed juvenile wild boar as detecting exposed adults. The difference between males and females regarding the DPP sensitivity remains unexplained. In conclusion, the study results presented herein support the use of antibody-based diagnostic tests for both large-scale and individual bTB testing of the Eurasian wild boar and suggest that a role for wild boar as potential sentinels for infections caused by MAC members does not exist.
Acknowledgements
The current study is a contribution to MCINN Plan Nacional I+D+i research grant AGL2008-03875 and FEDER and to FP7 EU grant TB-STEP (212414). Studies on diseases shared between domestic animals and wildlife are also supported by Grupo Santander Fundación Marcelino Botín. Tania Carta acknowledges a grant from Regione Sardegna. Jose Luis Sáez (MARM) made valuable comments to the first draft.
Footnotes
a.
STAT-PAK™, DPP®, Chembio Diagnostic Systems Inc., Medford, NY.
b.
CZ Veterinaria SL, Porriño, Lugo, Spain.
c.
Sigma-Aldrich Química SA, Madrid, Spain.
