Abstract
Influenza virus causes acute respiratory disease in pigs and is of concern for its potential public health significance. Many subtypes of influenza virus have been isolated from pigs, and the virus continues to evolve in swine populations. Current antibody assays have limited antigenic recognition, and accurate, broad-spectrum, high through-put screening tests are needed to detect infections in swine herds and to aid in the implementation of control measures. In the current study, a commercial blocking enzyme-linked immunosorbent assay (ELISA) developed for the detection of Influenza A virus nucleoprotein antibodies in avian species was evaluated for the detection of anti-influenza serum antibodies in swine. Serum samples used to evaluate the test were archived samples from influenza research conducted at the U.S. Department of Agriculture–Agricultural Research Service–National Animal Disease Center and included samples from influenza-inoculated pigs (H1N1, H1N2, H2N3, and H3N2), contact-infected pigs, vaccinated pigs, and negative controls. Based on samples of known status (n = 453), a receiver operating characteristic (ROC) curve analysis of the ELISA results estimated the optimized diagnostic sensitivity and specificity at 96.6% (95% confidence interval [CI]: 92.3, 98.9) and 99.3% (95% CI: 97.6, 99.9), respectively. By using the cutoff established in the ROC analysis, the assay was evaluated in pigs infected with 2 isolates of the 2009 pandemic H1N1 virus. Overall, the assay showed excellent diagnostic performance against the range of influenza subtypes investigated and could serve as a useful screening assay for swine.
Introduction
Influenza is a globally important pathogen of economic, veterinary, and public health concern. Swine influenza viruses (SIVs) of the H1N1 subtype have been continually circulating in pigs in the United States for at least 70 years. 12 In addition to the classic swine lineage H1N1, a triple reassortant H3N2 emerged in 1998 and spread rapidly in the swine population. 10 Since the introduction of the triple reassortant H3N2 in 1998, 3 predominant SIV subtypes have circulated in U.S. swine: H1N1, H1N2, and H3N2. The H1N1 viruses contain hemagglutinin (HA) and neuraminidase (NA) from the classic swine virus and the internal genes from the triple reassortant H3N2 viruses (rH1N1); the H1N2 viruses contain HA from the classic swine virus and contain NA and internal genes from the triple reassortant H3N2 viruses. 8,20
Since the introduction of the triple reassortant internal gene (TRIG) cassette, an increase in the rate of genetic change in North American swine influenza isolates appears to have occurred in H1 virus subtypes, and distinct genetic and antigenic clusters have evolved. 19 In Canada, in 2003 and 2004, H1N1 and H1N2 viruses with human-like HA and NA were isolated from pigs. 7 These viruses were wholly human or reassortants with internal genes of classic swine virus lineage. Since that time, reassortant viruses with human-like HA and NA, but with the TRIG cassette, were isolated from pigs across the United States. 18 These human-like H1 viruses have become endemic in the U.S. pig population and circulate concurrently with the contemporary SIV described above as a newly emerged genetic and antigenic cluster of H1 SIVs. 18
Serologic assays for SIV are crucial to the diagnosis of clinical disease, immunologic herd profiling for timing of vaccination, monitoring vaccine compliance, and epidemiologic studies based on seroprevalence. Current serum antibody assay formats include hemagglutination inhibition (HI), enzyme-linked immunosorbent assay (ELISA), and serum-virus neutralization (SVN). Although these assays show satisfactory diagnostic performance for the detection of antibodies homologous to antigens in the assay system, all demonstrate deficiencies related to detection of antibodies against heterologous influenza viruses. In addition, test performance of HI and SVN assays are affected by variation in nonstandardized biological reagents and the level of technical expertise.
The emergence of the human pandemic Influenza A virus 2009 H1N1 with genes of swine origin 4,6 further highlighted the need for improved, rapid diagnostic tests to investigate, surveil, and monitor the global swine population for zoonotic events. Fulfillment of this objective will require a serologic assay capable of detecting antibodies against a broad range of influenza subtypes, including the newly emerged SIV subtypes and reassortants in North America and Europe. 1
The commercial avian influenza multispecies ELISA a (AI-MS ELISA) was developed and validated for the detection of antibodies against influenza virus in avian species. The assay uses a monoclonal antibody generated against human Influenza A virus subtype H1N1 21 and recognizes a highly conserved epitope of influenza A nucleoprotein (NP). The objective of the current study was to evaluate the diagnostic performance of the AI-MS ELISA by using serum from pigs infected with influenza subtypes H1N1, H1N2, H2N3, and H3N2 under experimental conditions.
Materials and methods
Experimental design
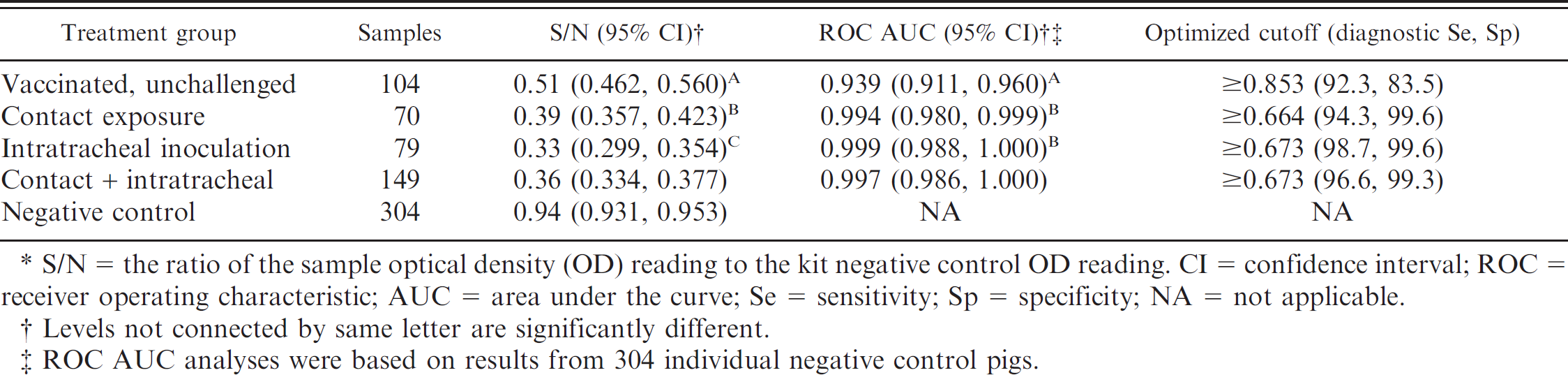
In study 1, the diagnostic performance of the AI-MS ELISA was evaluated by using 453 samples derived from previous influenza research conducted at the U.S. Department of Agriculture–Agricultural Research Service–National Animal Disease Center (USDA-ARS-NADC) 9,15–19 and additional unpublished experiments. The panel included 149 serum samples from animals exposed to Influenza A virus subtypes H1N1, H1N2, H3N2, or H2N3 in 6 separate experiments and 304 samples from negative control animals (Table 1). All samples were tested by using the AI-MS ELISA, and the results were evaluated by receiver operating characteristic (ROC) analysis. 23 In addition, 104 samples from vaccinated, unchallenged animals were assayed for purposes of comparison but were not included in the set of results used to evaluate assay performance.
In study 2, sera derived from pigs experimentally challenged with 2 different isolates of pandemic Influenza A virus 2009 H1N1 were evaluated by the AI-MS ELISA with positivity determined by using the cutoff value established in study 1. The panel included samples from 40 pigs collected at multiple time points pre- and post-exposure.
Viruses
Swine influenza viruses were isolated from outbreaks of respiratory disease in pigs and grown in Madin–Darby canine kidney cells or chicken embryos by using standard techniques to produce challenge or vaccine stocks, as previously described. 9,15–19 Viruses included in the live-exposure experiments were the following: A/SW/IA/15/1930 H1N1 (IA30), A/SW/MN/00194/2003 H1N2 (MN03), A/SW/IA/00239/2004 rH1N1 (IA04), A/SW/OH/511445/2007 H1N1 (OH07), A/SW/MN/07002083/07 huH1N1 (MN07), A/SW/MO/4296424/2006 H2N3 (MO06), A/SW/TX/4199-2/98 H3N2 (TX98), and the pandemic influenza viruses A/CA/04/2009 H1N1 (CA09) and A/Mexico/4108/09 H1N1 (MX09). Additional viruses included in the inactivated vaccine studies were the following: A/SW/MN/1192/2001 H1N2 (MN01), A/SW/NC36883/2002 rH1N1 (NC02), A/SW/KS/00246/2004 H1N2 (KS04), A/SW/IA/1945 H1N1 (IA45), A/SW/WI/1/1968 H1N1 (WI68), A/SW/IA/1973 H1N1 (IA73), A/SW/MN/37866/1999 H1N1 (MN99), A/SW/NC/02023/2008 H1N1 (NC08), A/SW/OH/02026/2008 H1N1 (OH08), A/SW/MO/02060/2008 H1N1 (MO08), A/SW/IA/02096/2008 H1N1 (IA08), A/SW/KY/02086/2008 H1N1 (KY08), A/SW/MN/02011/2008 H1N1 (MN08), A/SW/MN/02093/2008 H1N1 (MN08–2), A/SW/MN/02053/2008 huH1N1 (MN08–3), A/SW/NE/02013/2008 H1N1 (NE08), A/SW/NC/02084/2008 H1N1 (NC08), A/SW/TX/01976/2008 H1N2 (TX08), and A/SW/IA/02039/2008 huH1N1 (IA08-hu).
Animals, treatments, and serum samples
Four-week-old cross-bred pigs were obtained from a herd free of both SIV and Porcine reproductive and respiratory syndrome virus (PRRSV) infections. The animals were group-housed in individual isolation rooms and were cared for in compliance with the NADC Institutional Animal Care and Use Committee. All the pigs were administered ceftiofur crystalline-free acid b at the beginning of the acclimation period. Before inoculation or vaccination, SIV was not detected in nasal swab samples and anti-influenza antibodies were not detected by the serum HI assay. Thus, all animals were demonstrated to be free of SIV.
In study 1, a total of 453 serum samples were available for the evaluation of the AI-MS ELISA a (Tables 1, 2). The sample set was composed of 304 serum samples from 304 individual influenza-negative control animals and 149 serum samples from 84 influenza-infected animals. Experimentally infected animals (n = 44) received 2 ml of approximately 1 × 105 50% tissue culture infectious dose (TCID50) per milliliter of influenza virus intratracheally while under anesthesia, as previously described. 9,15–19 The remaining 40 influenza-infected pigs were exposed to SIV by contact with intratracheally inoculated animals beginning 2 or 3 days after inoculation. Influenza subtypes, isolates, exposure route (intratracheal or contact), number of pigs, and days of serum collection are listed in Table 1.
Blocking enzyme-linked immunosorbent assay a response by virus isolate, exposure, and day postinoculation (DPI).
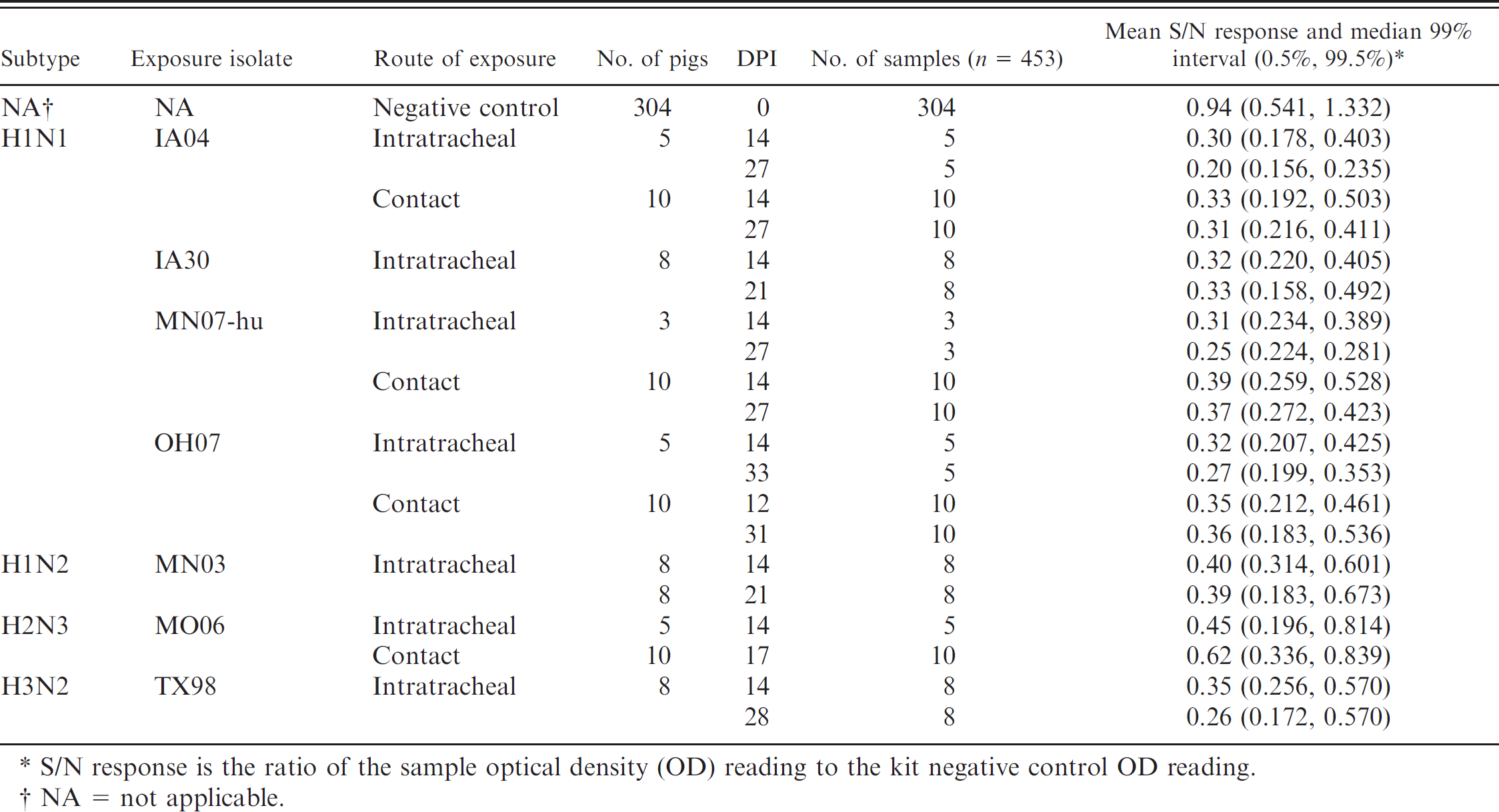
S/N response is the ratio of the sample optical density (OD) reading to the kit negative control OD reading.
NA = not applicable.
Serum samples (n = 104) from influenza-vaccinated and unchallenged animals were assayed but were not included in the cumulative ROC analysis, which established AI-MS ELISA a cutoffs and corresponding performance estimates. Vaccinated pigs received intramuscular injections of 1 × 106 TCID50 per milliliter or approximately 64–128 HA units of ultraviolet-inactivated influenza virus combined with a commercial adjuvant, c followed by a booster dose 2–3 weeks later. Serum was collected after the initial dose (approximately 14 days postvaccination) and again after the booster dose (approximately 28 days after initial vaccination). Vaccine isolate, the number of pigs, and the days of serum collection are listed in Table 2.
In study 2, to evaluate the performance of the AI-MS ELISA with sera collected from pigs infected with 2009 pandemic H1N1, assays were run by using the cutoff established by analysis of study 1 samples. Experimentally infected animals (n = 40) received 2 ml of approximately 1 × 105 TCID50 per milliliter of pandemic influenza virus intratracheally, as described above. Subsets of pigs were subsequently bled at days 0, 3, 5, 7, 14, and 21 postinfection.
Assays
Hemagglutination inhibition. Sera were heat inactivated at 56°C and treated to remove nonspecific agglutinators with a 20% suspension of kaolin, d followed by adsorption with 0.5% turkey red blood cells (RBC). The HI assays were performed with turkey RBCs by using homologous virus and anti-sera with standard techniques (WHO Manual on Animal Influenza Diagnosis and Surveillance, http://www.wpro.who.int/NR/rdonlyres/EFD2B9A7-2265-4AD0-BC98-97937B4FA83C/0/manualonanimalaidiagnosisandsurveillance.pdf).
AI-MS ELISA. The AI-MS ELISA a is licensed for the detection of antibody to avian influenza virus in chicken, turkey, duck, ostrich, and goose serum. Samples were assayed in duplicate according to the manufacturer's protocol for testing avian sera by using 15 μl of porcine sera diluted 10-fold in diluent provided by the manufacturer. The diluted serum was dispensed onto the NP-coated plate and allowed to bind to the antigen. An anti-NP antibody conjugate with horseradish peroxidase (HRP), followed by 3,3′,5,5′-tetramethyl benzidine (TMB) substrate, was then used as a color indicator. If present, antibody in the sample competitively bound to the epitope and blocked binding of the HRP conjugate. Color development, inversely proportional to the amount of anti-influenza virus antibodies in the test sample, was determined by using an ELISA plate reader c at a wavelength of 650 nm. Data management and calculations were performed by using software provided by the manufacturer. a Results were reported as the ratio of the sample optical density (OD) reading to the kit negative control OD reading (S/N).
Analysis
Descriptive statistics were generated by using the statistical software program JMP (version 7.0.2) f in study 1. The experimental design precluded a full factorial analysis by analysis of variance (ANOVA g ) at the level of isolate, subtype, treatment, or day post-inoculation (DPI), because the data were unbalanced across factors, and many combinations of factors contained few responses. Therefore, ANOVA g was conducted only at the level of treatment groups (intratracheal exposure, contact exposure, vaccinated, and unchallenged). Receiver operating characteristic analyses were conducted for each treatment group by using MedCalc version 10.4.0.0. h The influenza-negative data in each ROC analysis were composed of the S/N result from each of 304 individual negative control animals. In study 2, a ROC analysis was not conducted because of the small number of animals. Therefore, descriptive statistics with mean and median 99 percentile were generated, and positivity cutoff was based on ROC from study 1.
Blocking enzyme-linked immunosorbent assay a response in unexposed, vaccinated animals by day post-vaccination (DPV).
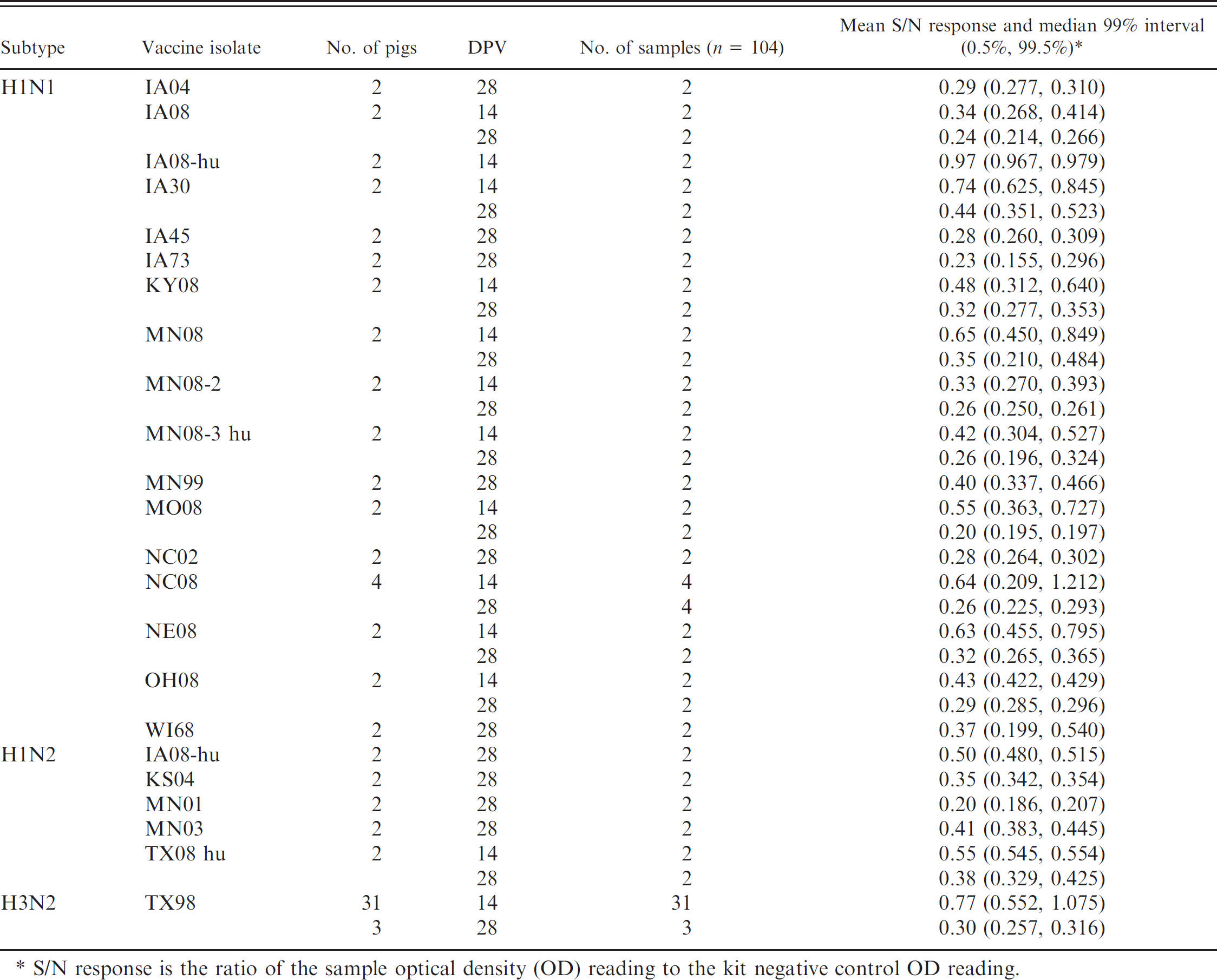
S/N response is the ratio of the sample optical density (OD) reading to the kit negative control OD reading.
Results
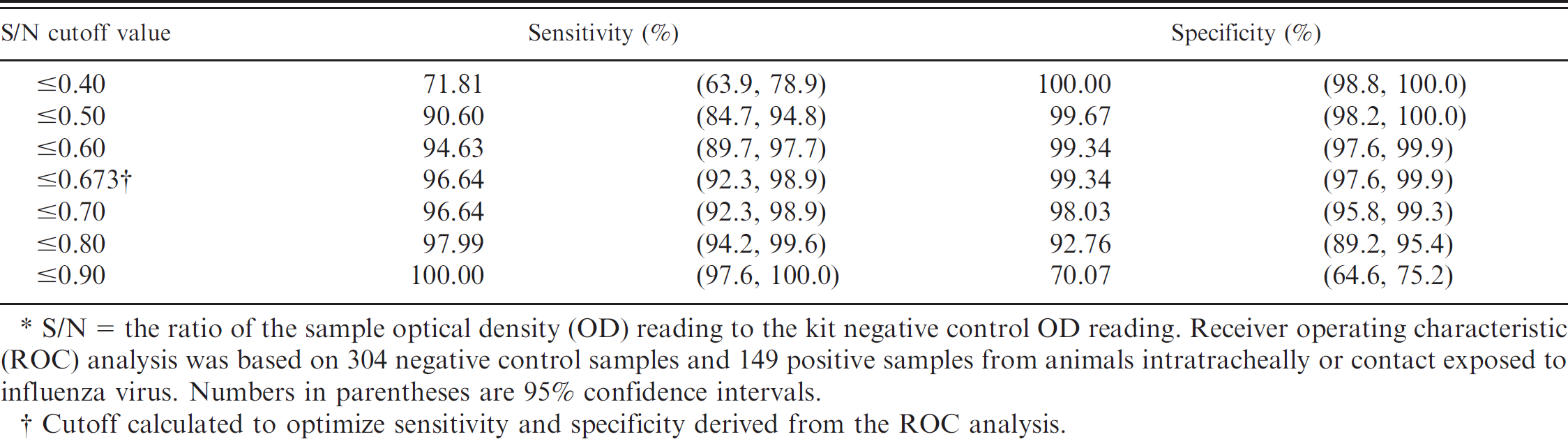
In study 1, sera from known positive animals as previously determined by HI were used in the ELISA. a Mean S/N responses and median 99% intervals (0.5%, 99.5%) for infected and vaccinated pigs are reported in Tables 1 and 2, respectively. To account for the sparse data points within some combinations of factors, the distribution of responses was summarized as the median 99% interval. The S/N means and ROC area under the curve estimates and the optimized S/N cutoffs with associated diagnostic sensitivity and/or specificity estimates are presented in Table 3. The ANOVA g analysis showed statistically significant differences in S/N response among intratracheally exposed, contact exposed, and vaccinated and unchallenged groups. However, the ROC analysis found no significant difference between the intratracheal and contact exposure groups. A ROC analysis based on 149 samples from influenza-infected pigs (intratracheal inoculated + contact groups) and 304 negative control pigs estimated the optimized AI-MS ELISA a cutoff as S/N ≥ 0.673, with corresponding diagnostic sensitivity and specificity estimated at 96.6% (95% confidence interval [CI]: 92.3, 98.9) and 99.3% (95% CI: 97.6, 99.9), respectively (Table 4, Fig. 1).
In study 2, by using the cutoff of 0.673 established above, seroconversion in 2009 pandemic H1N1-infected pigs was detected as early as 5 DPI in 1 pig and in 9 of 10 pigs on 7 DPI. By 14 DPI, all pigs experimentally challenged with CA09 or MX09 had S/N values below the positive cutoff. The mean S/N values for pigs infected with pandemic H1N1 are presented in Table 5.
S/N = the ratio of the sample optical density (OD) reading to the kit negative control OD reading. CI = confidence interval; ROC = receiver operating characteristic; AUC = area under the curve; Se = sensitivity; Sp = specificity; NA = not applicable.
Levels not connected by same letter are significantly different.
ROC AUC analyses were based on results from 304 individual negative control pigs.
Discussion
The AI-MS ELISA a used in the current study is an epitope-blocking assay in which a labeled monoclonal antibody directed against influenza A NP is blocked from binding to the avian influenza NP-coated microtiter plate in the presence of sample antibody to NP. The monoclonal antibody used in the test was generated by using human Influenza A virus subtype H1N1 21 and recognizes a highly conserved epitope of influenza A NP. Because the basis of the test is the ability of sample antibody to bind and block a conserved immunodominant epitope on the avian influenza NP-coated solid phase, the assay format does not require the use of species-specific reagents. This makes the assay broadly applicable for any species that generates an antibody response to the NP protein of Influenza A virus.
Epitope-blocking assays are a preferred diagnostic method when the pathogen has broad multispecies transmissibility and when a defined conserved immunodominant epitope and paired monoclonal are available. For example, multispecies blocking tests for antibody detection have been developed and evaluated for Foot-and-mouth disease virus, 3 West Nile virus, 2 Avian pneumovirus subtype C, 14 and Vesicular stomatitis virus. Most influenza A multispecies blocking tests reported in the literature use NP detection as the basis of the assay. 5,11,13,22 In a previous study, 5 a blocking ELISA format was successfully used to detect anti-NP antibodies in humans, ferrets, swine, horses, chickens, ducks, guinea pigs, mice, and seals.
To the authors' knowledge, the current study is the first report of an influenza ELISA developed and licensed for avian serum samples used to detect NP antibodies in serum from pigs infected or vaccinated with North American influenza viruses. The AI-MS ELISA a showed excellent performance and detected anti-influenza antibodies generated by the pig after exposure to all SIV isolates tested as early as 5 days postexposure (earliest sample collected). In contrast to the 0.500 S/N cutoff recommended for avian serum by the manufacturer, ROC analysis by using the available panel of serum samples determined the optimum cutoff for swine serum samples to be 0.673. Because the ELISA is a blocking format, samples ≤0.673 would be considered positive and >0.673 considered negative.
S/N = the ratio of the sample optical density (OD) reading to the kit negative control OD reading. Receiver operating characteristic (ROC) analysis was based on 304 negative control samples and 149 positive samples from animals intratracheally or contact exposed to influenza virus. Numbers in parentheses are 95% confidence intervals.
Cutoff calculated to optimize sensitivity and specificity derived from the ROC analysis.
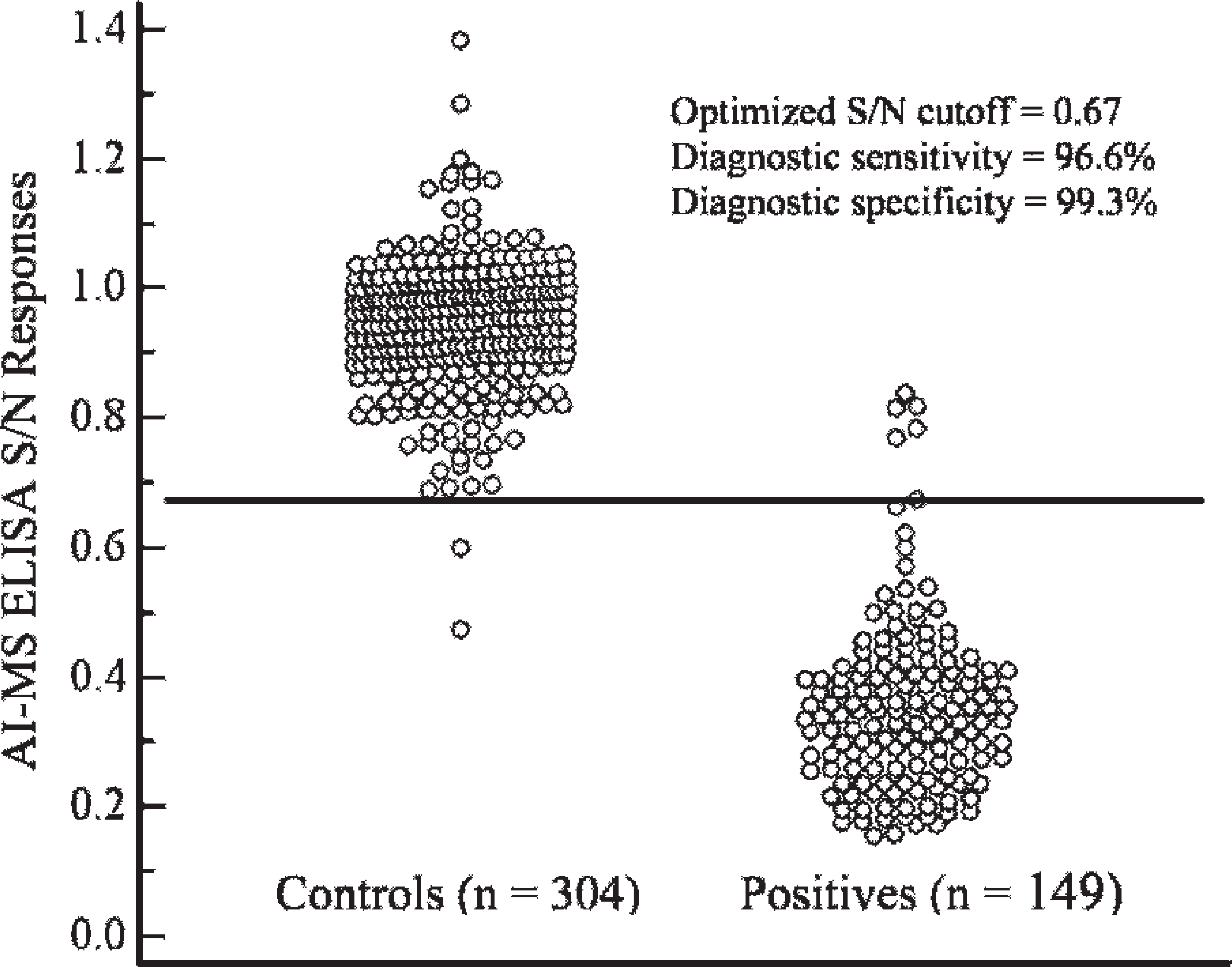
Results from the receiver operating characteristic analysis on samples from influenza-infected pigs (intratracheal inoculated + contact groups) and negative control pigs, which estimated the optimized commercial avian influenza multispecies enzyme-linked immunosorbent assay a (AI-MS ELISA) cutoff as S/N ≥ 0.673, with corresponding diagnostic sensitivity and specificity estimated at 96.6% and 99.3%, respectively.
Serum samples from pigs vaccinated with whole-virus vaccine demonstrated S/N responses in the positive range, often after the first dose (14 days post-vaccination [DPV]) and more reliably after the booster dose (28 DPV). The delayed response to NP in vaccinated animals is likely because of limited NP in the inactivated vaccine preparation compared with live challenge with virus replication in the host. Nonetheless, the data suggest that the assay could be useful for monitoring vaccine responses and on-farm compliance of vaccine administration as well. In addition, the assay has the potential to differentiate infected from vaccinated animals if split-virus or subunit vaccines that exclude NP are used. An area of future investigation of the applicability of this assay is for its potential to monitor maternal antibody decay in piglets for vaccination timing.
The recent emergence of the 2009 H1N1 human influenza pandemic virus 4,6 has led to global awareness of the need to increase surveillance in swine. The current study demonstrates that the AI-MS ELISA can rapidly detect antibody induced by infection with pandemic H1N1 in addition to endemic North American SIV. The assay can be incorporated as a tool for diagnosing and monitoring outbreaks with H1N1 or other influenza A viruses. The AI-MS ELISA a provides an assay with standardized reagents in a format compatible with the technology readily available in most veterinary diagnostic laboratories around the world. By extension, broad implementation of this assay could improve both the breadth and the uniformity of serologic data for influenza virus at a global level. Timely investigations should be undertaken to determine the detection limitations of the assay.
Acknowledgements
The authors thank Michelle Harland and Hillary Horst for technical assistance, Dr. Pablo Lopez from IDEXX Laboratories Inc. for comments and suggestions and for providing the ELISA kits; Dr. Marie Gramer (University of Minnesota), Dr. Alexander Klimov (CDC) for providing influenza A isolates; and Dr. Ray Waters (USDA-ARS-National Animal Disease Center) for critical review of the manuscript. The mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Blocking enzyme-linked immunosorbent assay a response by virus isolate and day postinoculation (DPI) in pandemic H1N1-infected pigs.
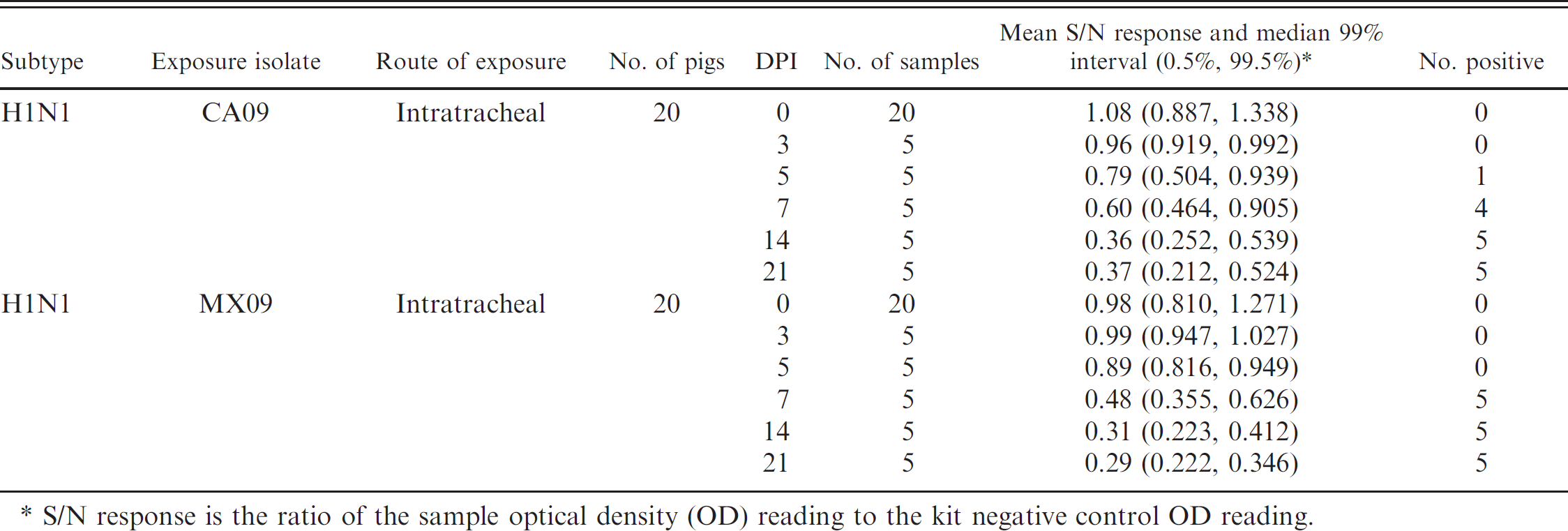
S/N response is the ratio of the sample optical density (OD) reading to the kit negative control OD reading.
Footnotes
a.
FlockChek® Avian Influenza MultiS-Screen Antibody Test Kit, IDEXX Laboratories Inc., Westbrook, ME.
b.
Pfizer Animal Health, New York, NY.
c.
Emulsigen, MVP Laboratories Inc., Ralston, NE.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Molecular Devices Corp., Sunnyvale, CA.
f.
SAS Institute Inc., Cary, NC.
g.
OriginLab, Northampton, MA.
h.
MedCalc Software, Mariakerke, Belgium.