Abstract
Bovine coronavirus (BCV; Betacoronavirus 1) and Bovine respiratory syncytial virus (BRSV) are significant causes of enteric and respiratory disease in beef and dairy cattle throughout the world. Indirect enzyme-linked immunosorbent assays are widely used to detect serum antibodies for herd monitoring and prevalence studies. In dairy herds, milk is more readily collected than serum. Hence, in order to investigate the test agreement between serum and milk, both serum and milk samples from 105 cows in 27 dairy herds were analyzed in parallel for presence of immunoglobulin G antibodies to BCV and BRSV. The Bland–Altman analyses of data demonstrated good agreement between serum and milk antibody titers for both viruses. The results indicate milk samples are sufficient for surveillance of antibodies to BCV and BRSV.
Keywords
Bovine coronavirus (BCV; Betacoronavirus 1, order Nidovirales, family Coronaviridae, subfamily Coronavirinae, genus Betacoronavirus) and Bovine respiratory syncytial virus (BRSV; order Mononegavirales, family Paramyxoviridae, subfamily Pneumovirinae, genus Pneumovirus) are significant causes of enteric and respiratory disease in beef and dairy cattle throughout the world.6,8,14,16 These viruses spread readily within herds and usually infect all naïve animals.4,11,16 Infections can be diagnosed either directly, by detection of virus or viral nucleic acid in fecal samples (BCV) or nasal swabs (BRSV), or indirectly, by antibody detection in milk or serum. Detection of antibodies by indirect enzyme-linked immunosorbent assays (iELISAs) are widely used for epidemiological research such as prevalence studies and herd monitoring.11,14,15 For example, pooled milk samples from primiparous cows have been successfully used in the Swedish eradication program for Bovine viral diarrhea virus (BVDV). 13 The relation between milk and serum antibodies for BRSV has previously been evaluated, 9 with the conclusion that there was a good agreement of the parallel analyses; a similar comparison of sample types has not been performed for BCV-specific antibodies. The aim of the current study was to evaluate the relationship between serum and milk antibodies for BCV and BRSV, using commercially available iELISAs. Antibody detection in milk would be a convenient way of monitoring herd infection status in, for example, future control programs for BRSV or BCV.
The blood and milk samples were obtained from 105 Swedish dairy cows. Sixty-three first-lactation cows from 26 dairy farms were sampled during the winter of 2009–2010, and 42 cows of various ages from a single dairy herd in 2011. All cows included were clinically healthy and were not vaccinated against BCV or BRSV. Milk was collected in test tubes containing the preservative agent bronopol (2-bromo-2-nitropropane-1.3-diol). a The blood samples were collected in 10-ml evacuated tubes without anticoagulant and allowed to clot, then centrifuged at 1,008 rcf for 10 min and the sera separated. All samples were stored at −20°C until tested. The samples were analyzed for presence of immunoglobulin G antibodies to BCV 1 and BRSV 9 using commercially available iELISAsb,c according to the manufacturers’ instructions. The optical density (OD) at 450 nm was corrected by subtraction of the negative control antigen OD of each well of the 96-well plate. To adjust between day-to-day variation, the percent positivity (PP) was calculated as: (corrected OD/positive control corrected OD) × 100. For both serum and milk samples, a PP value of <10 was considered negative; at that cutoff value, the sensitivity (Se) and specificity (Sp) for BCV was estimated to be 84.6% and 100%, respectively, and, for BRSV, 94.6% and 100%, respectively, according to the manufacturer for serum. Measurements of repeatability were computed using Bland–Altman plots. 5 Two alternative cutoffs were investigated for the milk samples where PP values of <5 and <15 were regarded as negative. The results from the serum analysis were considered the reference (i.e., if the serum was positive for BCV or BRSV antibodies, then the animal was considered positive). Concordance between serum and milk results was calculated as percent agreement and estimated by Cohen’s kappa value. The Se and Sp of the assays were calculated using the following formula: Se = (a/[a + c]) × 100 and Sp = (d/[b + d]) × 100, where a and d are the number of positive and negative results, respectively, by both sample types; b is the number of serum negative and milk positive results; and c is the number of serum positive and milk negative results. Statistical analyses were performed using commercial statistical software. d
A total of 105 serum and milk samples were analyzed in parallel for the presence of antibodies to BCV and BRSV. Seventy-two of the 77 cows with BCV antibodies in their sera were also positive for BCV antibodies in milk; the 5 cows with negative results for milk but positive for sera had PP values between 13 and 31 in sera. All cows that were antibody negative for BCV in sera were also antibody negative in milk. Eighty of the 82 cows with BRSV antibodies in their sera were also positive for BRSV antibodies in milk (Table 1); the 2 cows with negative results for milk but positive sera had PP values of 24 and 34 in sera, respectively. One cow was negative for BRSV antibodies in sera but was BRSV antibody positive in milk, with a PP value in milk of 61. The distributions of observed PP values are shown in Figure 1. The Bland–Altman analyses demonstrated good agreement between serum and milk antibody titers for both BCV and BRSV (Fig. 2). For an average PP value of 10 for BCV, the mean difference between serum and milk PP value was 5.5 with a 95% confidence interval of ±18. For an average PP value of 10 for BRSV, the mean difference between serum and milk PP value was 1.23 with a 95% confidence interval of ±26. The agreement between serum and milk PP value deteriorated with increasing average PP values of serum. The highest correctly classified number of results in milk compared to serum was accomplished with a PP <5 for both BCV (96.2%) and BRSV (99.1%) The corresponding kappa values (Table 2) were 0.90 for BCV and 0.97 for BRSV, indicating almost perfect agreement. 7
The results of 105 paired bovine serum and milk samples analyzed with a commercial indirect enzyme-linked immunosorbent assay for detection of antibodies to Bovine coronavirus (BCV) and Bovine respiratory syncytial virus (BRSV).*

A percent positivity (PP) of <10 was regarded as negative for serum samples, and cutoff at <5, <10, and <15 for milk samples.
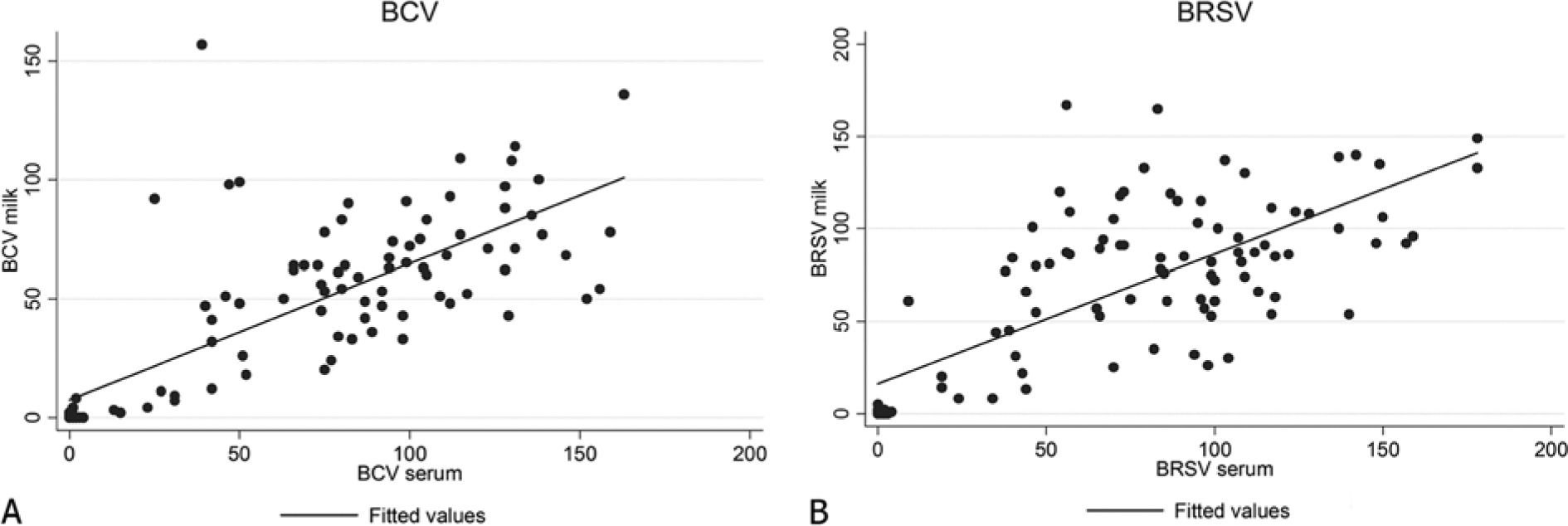
Antibody levels of 105 paired bovine serum and milk samples analyzed with commercial indirect enzyme-linked immunosorbent assays to (
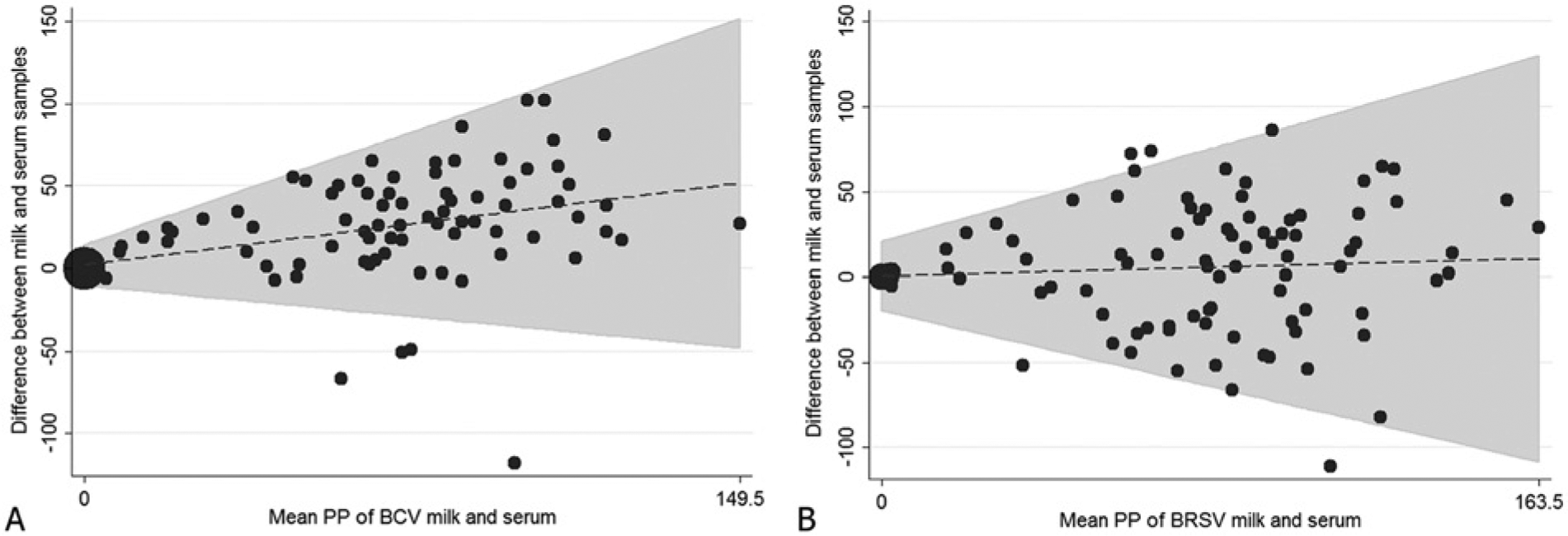
Bland–Altman, 95% limits of agreement, and mean difference of observations from 105 paired bovine serum and milk samples analyzed with commercial indirect enzyme-linked immunosorbent assays to Bovine coronavirus (BCV) and Bovine respiratory syncytial virus (BRSV) expressed as percent positivity.
Evaluation of test characteristics for 2 commercially available indirect enzyme-linked immunosorbent assays for detection of antibodies to Bovine coronavirus (BCV) and Bovine respiratory syncytial virus (BRSV), including 105 paired bovine serum and milk samples.*
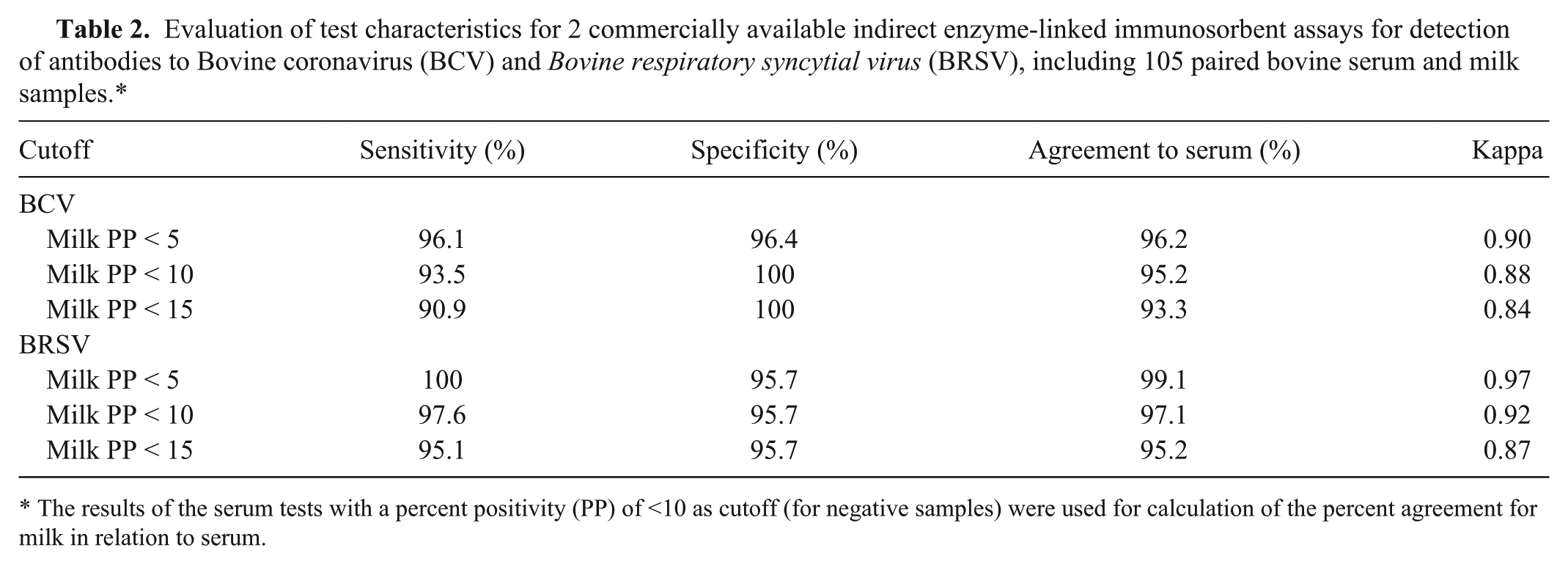
The results of the serum tests with a percent positivity (PP) of <10 as cutoff (for negative samples) were used for calculation of the percent agreement for milk in relation to serum.
Reliable, fast, and cost-effective diagnostic tools are required for the control of contagious viral infections such as BCV and BRSV in dairy herds. Indirect enzyme-linked immunosorbent assays are advantageous in these aspects, particularly in dairy herds if the tests can be applied to milk samples. The present study evaluates the relationship between individual serum and milk antibodies for BCV and confirms the relationship between individual serum and milk antibodies for BRSV established by others. 9 Bland–Altman plots were used to evaluate the variability between serum and milk antibody titers, providing a more useful way of evaluating repeatability than comparing measures with a paired t-test, as Bland–Altman plots quantify the agreement over the full range of results rather than just comparing the means.
There was close agreement between matched serum and milk samples for both BCV and BRSV. For BCV, the antibody level was generally lower in milk compared to serum, a pattern seen in previous studies on evaluating BRSV, 9 Bovine leukosis virus, 10 and BVDV antibodies. 12 In the previous studies, the authors have explained the discrepancy by lower immunoglobulin levels in milk compared to serum. However, in the current study, the levels of BRSV antibodies in milk and serum were similar and consistent with another study that showed similar levels of antibodies to BVDV in milk and serum samples. 2 One cow had a divergent result for BSRV (i.e., negative in serum but with a high titer in milk). This result is hard to explain biologically and could be due to a misidentification of the sample when it was taken or in the laboratory.
The use of milk samples for the detection of antibodies to BCV and BRSV is a more convenient sampling strategy compared to serum samples as a means of screening dairy herds as a part of potential control programs. From a practical view, the most important aspect of using milk samples instead of serum is that the agreement is good in the interval around the cutoff value. The current study demonstrates good agreement in the important lower interval, and the increasing divergence in the higher interval is of less practical importance. Depending on which test characteristics are desired, Se and Sp can be altered by changing the cutoff. In the current study, the test characteristics indicate that a cutoff at PP < 5 is optimal for milk samples. In conclusion, milk samples are suitable for the surveillance of antibodies to both BCV and BRSV.
Footnotes
Acknowledgements
The authors thank Prof. Stefan Alenius for fruitful discussions, Prof. Ulf Emanuelson and Dr. Tove Fall for epidemiological advice, Maj Hjort for skillful laboratory work, and the farmers for their collaboration.
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Swedish Research Council Formas (grant 22.0/2004-0421) and a fellowship grant for Isabel Blanco-Penedo from the Spanish Foundation of Pedro Barrié de la Maza.
a.
Bronopol (2-bromo-2-nitropropane-1.3-diol), National Veterinary Institute, Uppsala, Sweden.
b.
SVANOVIR BCV-Ab, Boehringer Ingelheim Svanova, Uppsala, Sweden.
c.
SVANOVIR BRSV-Ab, Boehringer Ingelheim Svanova, Uppsala, Sweden.
d.
Stata statistical software, Release11.0, StataCorp LP, College Station, TX.
