Abstract
Yolk sac tumors (YSTs) are rare neoplasms of germ cell origin. In humans, the tumors primarily occur in the testes or ovaries, but occasionally develop at other sites. The neoplastic cells of YSTs form many histological patterns resembling embryonal structures, and the World Health Organization classification lists 11 such patterns: reticular, macrocystic, endodermal sinus, papillary, solid, glandular-alveolar, myxomatous, sarcomatoid, polyvesicular vitelline, hepatoid, and parietal. Among domestic animals, only 2 cases of YST, which were of testicular and abdominal cavity origin, have been reported in calves. In both cases, neoplastic cells had epithelial properties and disseminated metastases in the abdomen. In the present study, the enlarged testis of a newborn calf, which was subsequently diagnosed as YST and exhibited myxomatous, reticular, and polyvesicular vitelline histological patterns, is described. There was no metastasis in this case, and histological and immunohistochemical features varied from previous cases of YST.
Yolk sac tumors (YSTs) are primitive germ cell neoplasms derived from the embryonic yolk sac, allantois, or extraembryonic mesenchyme and usually develop in the testes or ovaries.7,14 Human YSTs show diverse histological features, and 11 such patterns are listed in the World Health Organization classification of tumors: reticular (microcystic, vacuolated, honeycomb), macrocystic, endodermal sinus (perivascular, festoon), papillary, solid, glandular-alveolar (including intestinal and endometrioid-like), myxomatous, sarcomatoid (spindle cell), polyvesicular vitelline, hepatoid, and parietal.7,14
Yolk sac tumors are extremely rare in domestic animals, and there have been only 2 reports of YSTs occurring in calves.4,8 One of these cases was a testicular YST, which was histologically determined to belong to the endodermal sinus type. 4 The other case involved a YST that developed in the abdominal cavity, and did not exhibit any testicular abnormalities. 8 The neoplastic cells in the latter case consisted of epithelioid cells and large atypical cells containing hyaline droplets and/or vacuoles. 8 In both cases, there were disseminated metastases in the abdominal cavity.4,8 In the present study, a case of testicular YST in a newborn calf, which exhibited a histological pattern completely different from the 2 cases previously diagnosed, is described. In addition, tissue samples from the present case and a previous case of testicular YST 4 in a calf were subjected to immunohistochemical staining in order to detect various proteins; differences were found between the protein expression profiles of each histological pattern.
A newborn Holstein calf presented with scrotal swelling, and a firm mass was found inside its scrotum on palpation. A fine-needle biopsy examination performed on the second day of life demonstrated the presence of myxoid tissue in the mass, but a definitive diagnosis could not be made. On the 8th day, the mass was surgically excised and confirmed to be an enlarged right testis. The resected testis was approximately 13 cm × 7 cm × 6 cm, but was structurally normal. It had a spermatic cord and mesorchium and was folded within the tunica vaginalis, which was partially anchored to the testis. The epididymis was located alongside the attachment to the tunica vaginalis and mesorchium. Cut sections revealed the presence of amber gelatinous tissue and blood-filled cystic cavities, which were separated by fibrous septae (Fig. 1). The entire right testis was submitted for histological examination. At the owner’s request, the calf was euthanized on the 61st day of life. The necropsy findings included suppurative bronchopneumonia in the cranial lobes and an abscess in the root of the tongue. The left testis was approximately 3 cm × 1.5 cm × 1.5 cm. There were no abnormal findings in the other genitourinary organs. The organs were fixed in 10% neutral buffered formalin, embedded in paraffin wax, and cut into 4-μm sections. The sections were stained with hematoxylin and eosin and Alcian blue at pH 2.5.
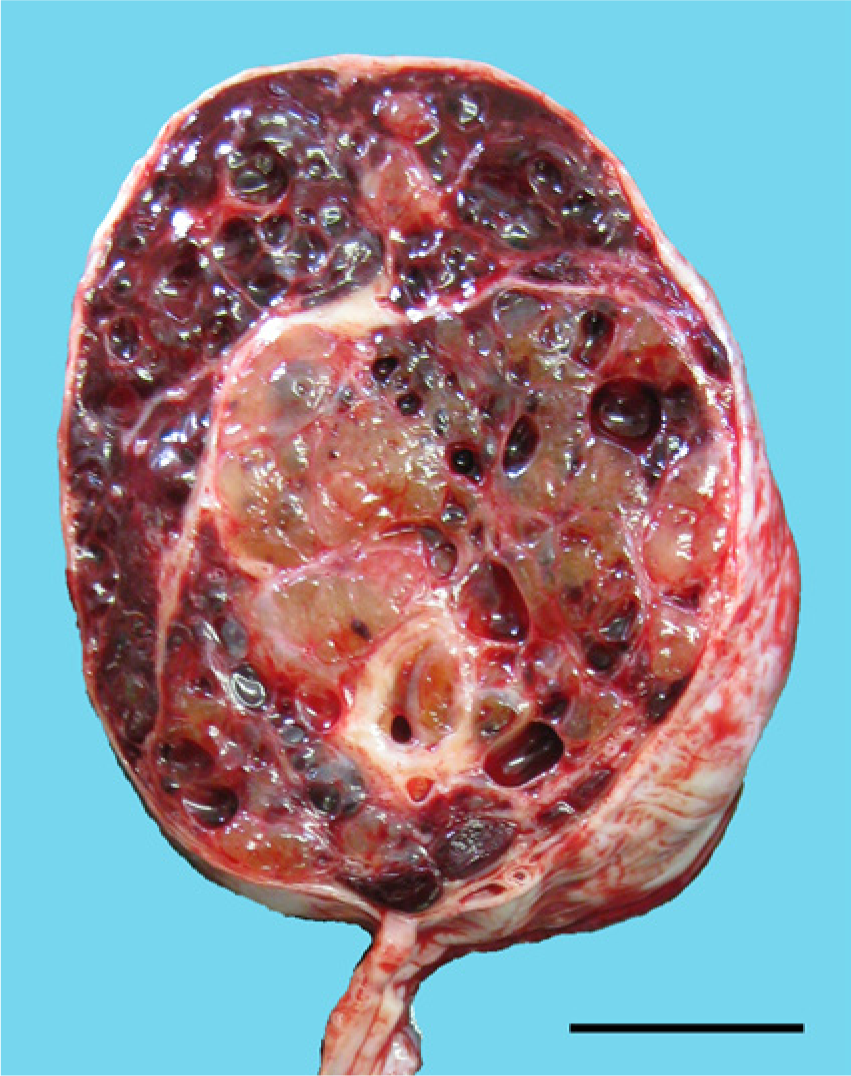
Cut surface of the testicular mass. Note the gelatinous tissue and multiple cystic cavities filled with blood. Bar = 2 cm.
Histologically, the gelatinous tissue in the excised right testis was composed of variably sized regions of myxoid tissue separated by fibrovascular stroma (Fig. 2). The number of cells in each myxoid compartment varied. In the compartments that displayed low cellularity, spindle-shaped or stellate cells were sparsely distributed (Fig. 3); whereas in the compartments that exhibited high cellularity, thin cords of neoplastic cells had anastomosed to form microcystic spaces or accumulated to form small cell clusters (Fig. 4). The cells had slightly eosinophilic cytoplasm with indistinct cell boundaries and a round to ovoid nucleus. Mitotic figures were infrequently observed. The myxoid substance was stained by Alcian blue stain. The grossly recognized cystic spaces filled with blood were lined by flattened cells and surrounded by fibrous stroma (Fig. 5). Occasionally, smaller cysts containing red blood cells were found within the myxoid compartments (Fig. 6).
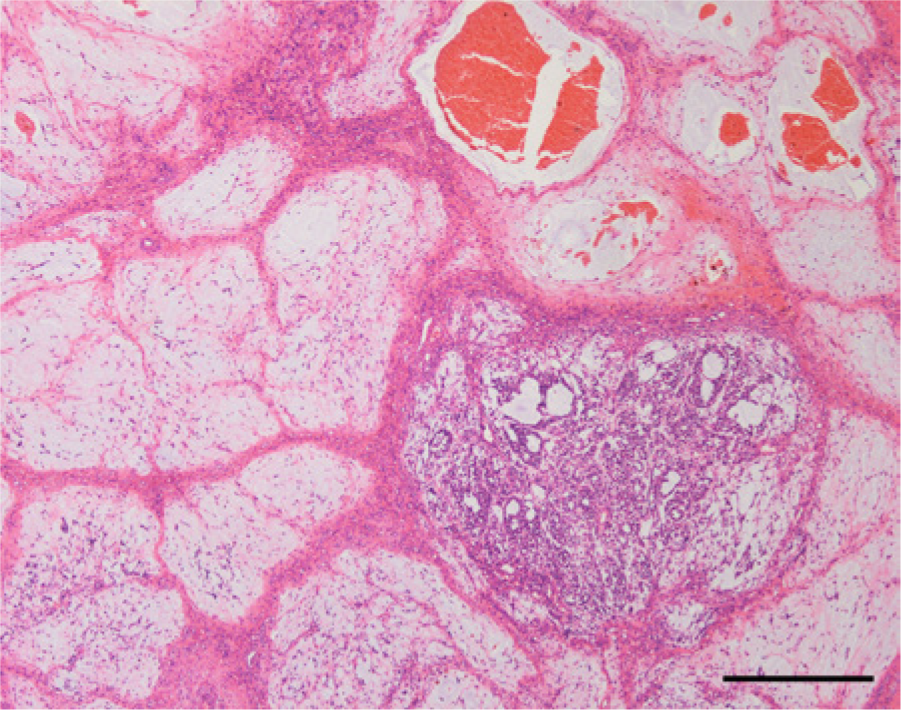
The gelatinous tissue was composed of myxoid tissue compartments with various cell densities separated by fibrovascular stroma. Hematoxylin and eosin. Bar = 500 μm.
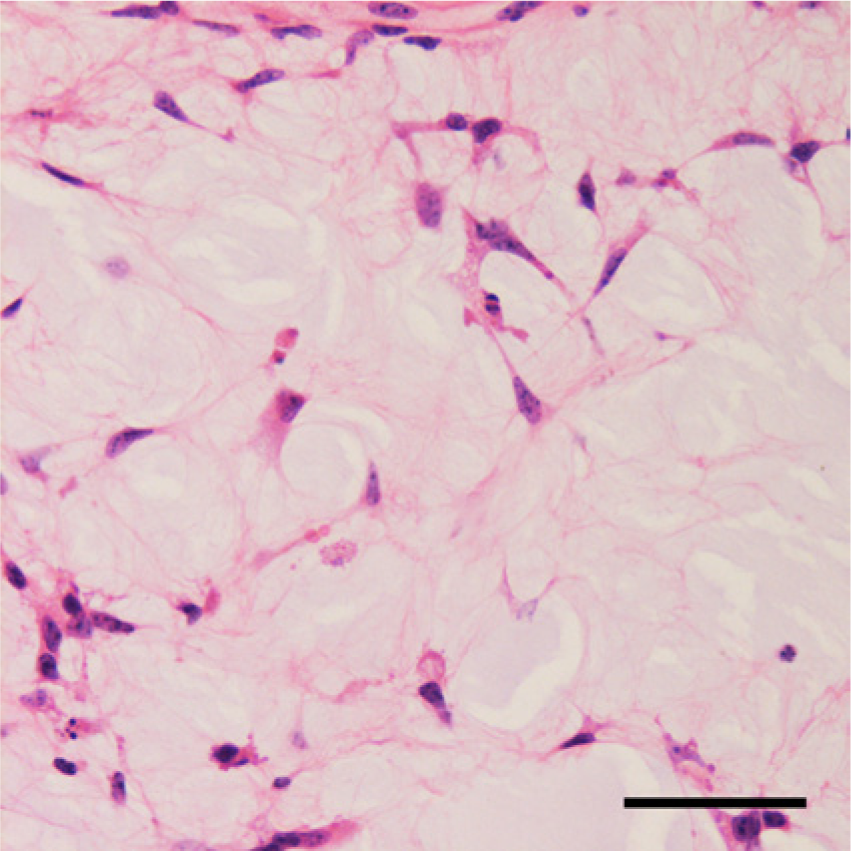
Higher magnification of a myxoid compartment with low cellularity. Spindle-shaped or stellate cells were sparsely distributed throughout the myxoid background. Hematoxylin and eosin. Bar = 50 μm.
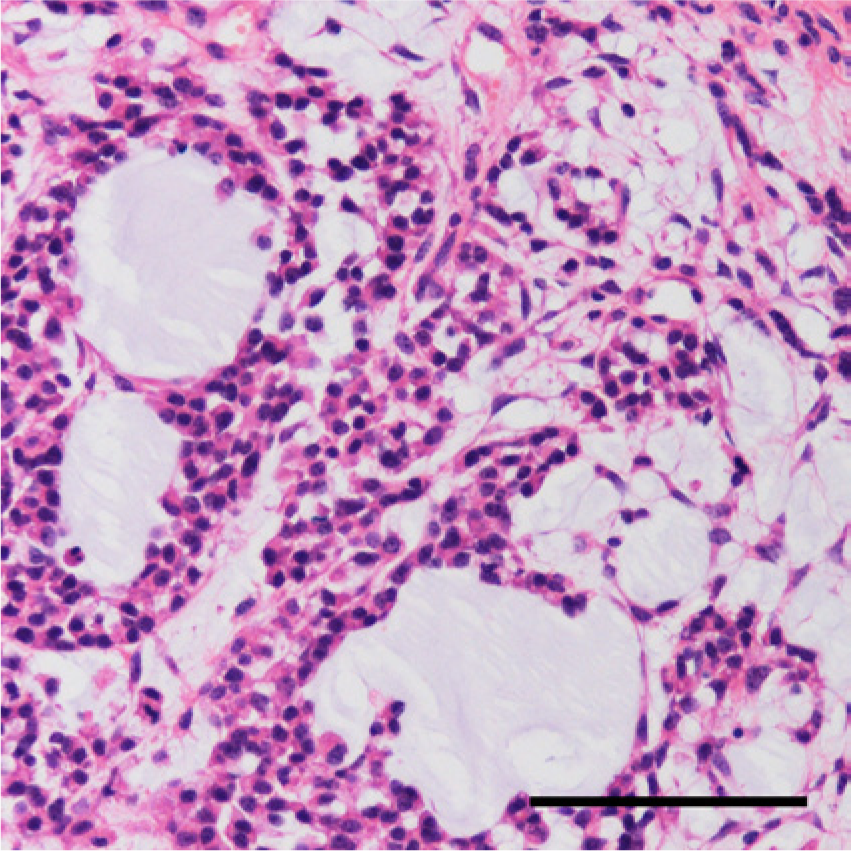
Higher magnification of a myxoid compartment with high cellularity. Neoplastic cells arranged to form microcystic spaces of varying size. Hematoxylin and eosin. Bar = 100 μm.
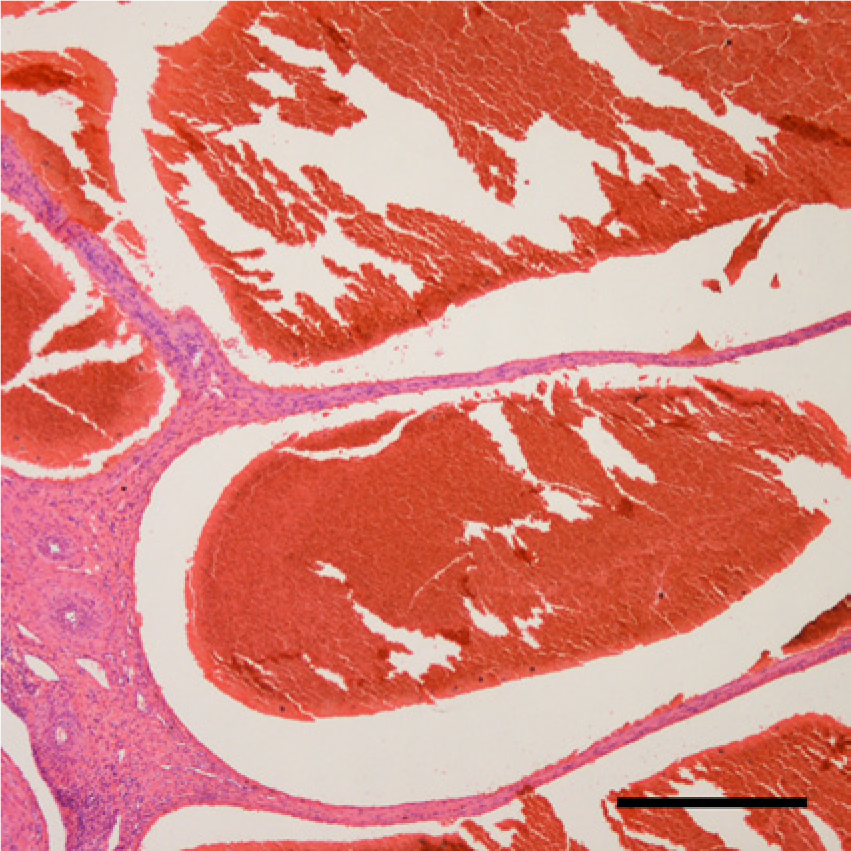
Large cystic cavities filled with red blood cells were surrounded by fibrous stroma. Hematoxylin and eosin. Bar = 500 μm.
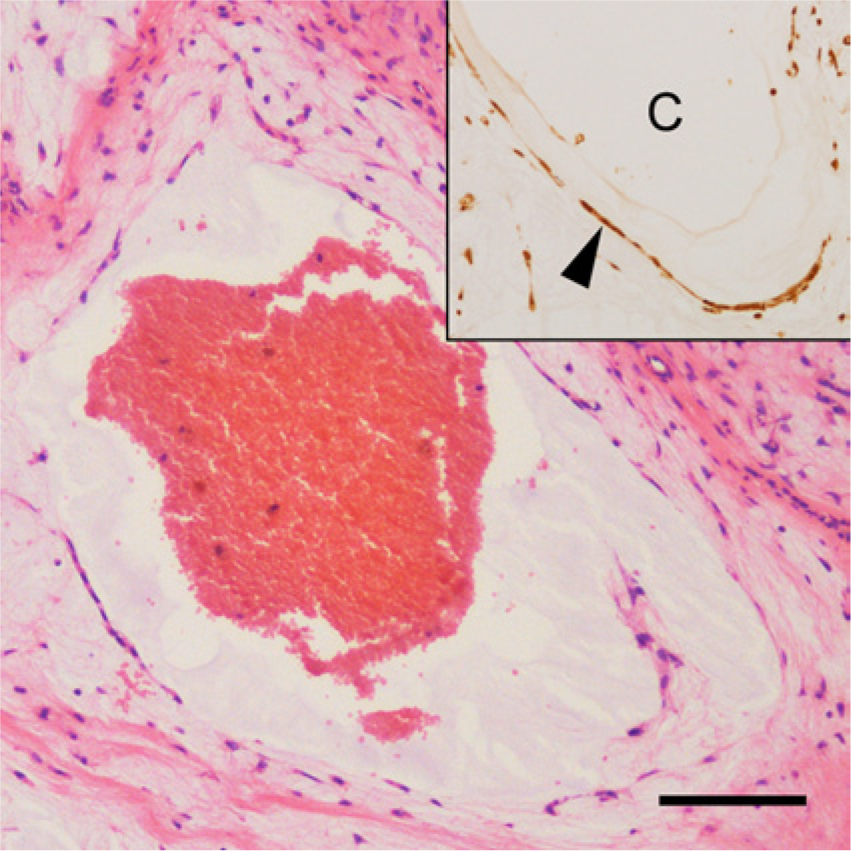
A small cyst containing red blood cells was lined by flattened cells and located within the myxoid compartment. Hematoxylin and eosin. Bar = 100 μm. Inset: cells lining the cavity (C), which were stained for α-fetoprotein (arrowhead). The immunohistochemical examination was performed with DAB (3,3′-diaminobenzidine) as a chromogen and hematoxylin as a counterstain.
In the testicular parenchyma adjacent to the epididymis, the rete testis and a small number of seminiferous tubules were observed. The efferent ductules and epididymal and seminal ducts were also confirmed to be present. The testicular tissue was surrounded by a collagen layer and lined by a continuous single layer of mesothelial cells. A histological examination did not detect any abnormal findings in the other genitourinary organs, including the left testis, spermatic ducts, prostate, vesicular glands, bulbourethral glands, bladder, ureter, and kidneys. Cortical to medullary sinuses were filled with a moderate number of histiocytes and neutrophils in the medial and lateral iliac lymph nodes.
Tissue specimens from the present case and the previous case of testicular YST in a calf 4 were subjected to immunohistochemical examinations using the avidin–biotin–peroxidase complex method. a The antibodies used included 2 types of rabbit antisera against α-fetoprotein (AFP)b,c; rabbit polyclonal antibodies against glial fibrillary acidic protein (GFAP), c c-kit, c S100, c and factor VIII–related antigen (factor VIII) b ; mouse monoclonal antibodies against vimentin, d desmin, e and inhibin α f ; and a rabbit monoclonal antibody against placental alkaline phosphatase (PLAP). g To detect cytokeratin, 2 antibody clonesh,i were used. DAB (3,3′-diaminobenzidine) was used as a chromogen. For negative controls, phosphate buffered saline was substituted for the primary antibodies. Positive reactions for AFP, inhibin α, and PLAP were detected in the control paraffin-embedded bovine tissues: fetal hepatocytes, granulosa cells in the ovary and chorionic epithelium, respectively. The 2 antibodies for AFP produced similar staining results. The antibody for c-kit was previously demonstrated to react with bovine tissue. 10 In the present case, the cells in the myxoid substance and those lining the blood-containing cavities exhibited positive staining for AFP (Figs. 6, 7) and vimentin (Fig. 8); positive reactions for cytokeratin and desmin were detected in a few populations of these cells. However, these cells were negative for GFAP, c-kit, S100, factor VIII, inhibin α, and PLAP. In the previous case of endodermal-type testicular YST, 4 the neoplastic cells were positively stained for AFP and cytokeratin, but negative for vimentin, desmin, GFAP, c-kit, S100, factor VIII, and inhibin α. In addition, focal positive staining for PLAP was detected in the cytomembranes of the neoplastic cells.
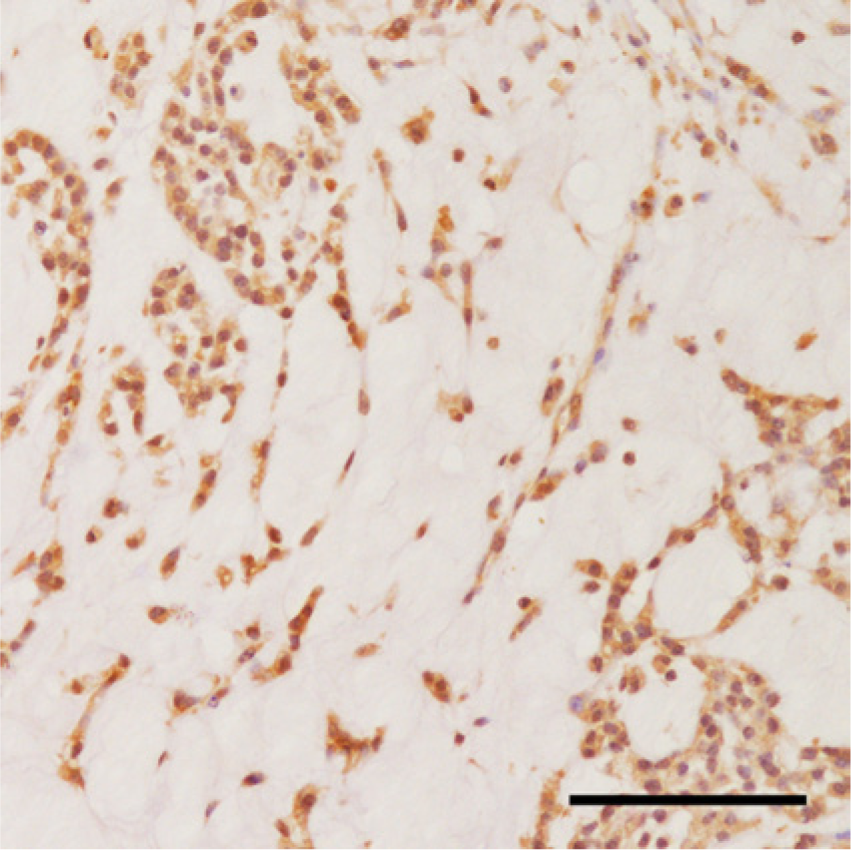
Positive reaction to α-fetoprotein was observed in the cells within the myxoid compartment. The immunohistochemical examination was performed with DAB (3,3′-diaminobenzidine) as a chromogen and hematoxylin as a counterstain. Bar = 100 μm.
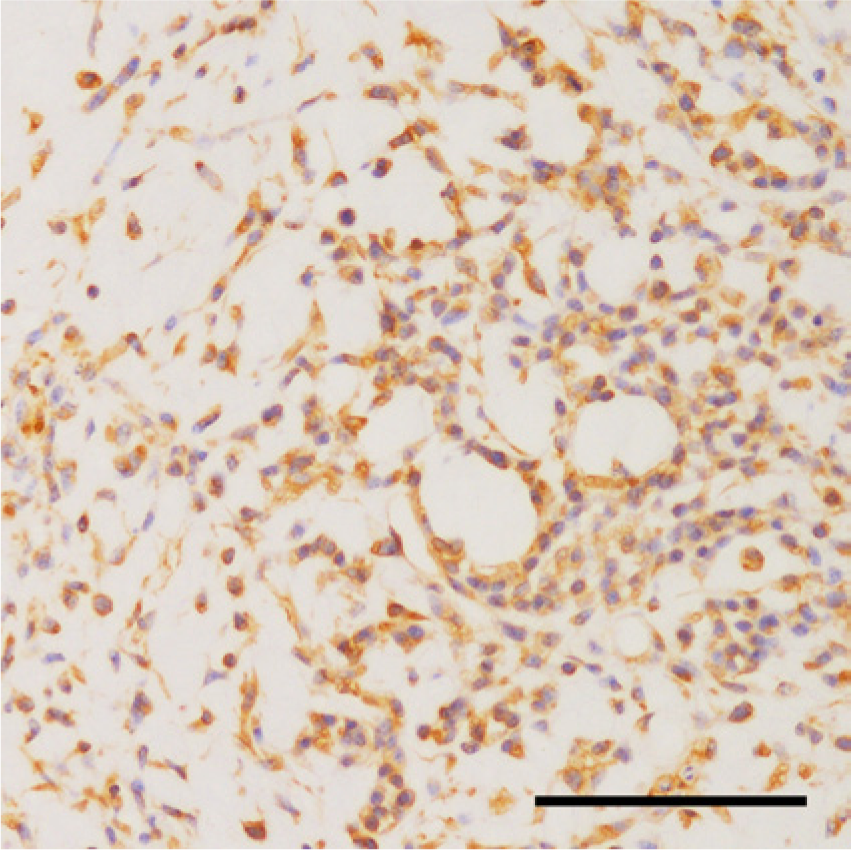
Positive reaction to vimentin was observed in the cells within the myxoid compartment. The immunohistochemical examination was performed with DAB (3,3′-diaminobenzidine) as a chromogen and hematoxylin as a counterstain. Bar = 100 μm.
Based on anatomical, histological, and immunohistochemical findings, the present case was diagnosed as YST that originated in the right testis. According to the histological classification of YST used in human medicine, sparsely distributed spindle-shaped or stellate cells, anastomosing cords or clusters of cells on a myxoid background, and cysts of various sizes correspond to the myxomatous, reticular, and polyvesicular vitelline patterns, respectively.7,14 Schiller–Duval bodies and hyaline globules, which are considered to be pathognomonic structures for human YST and were also present in the previous 2 calf cases of YST, were not found in the present case.3,4,7,8,14 In fact, the histological findings of the present case have not been reported in any domestic animals to the authors’ knowledge. A transition between the myxomatous and microcystic patterns was observed in the present case, as has been found in human YST. 12 The aggregation of microcysts is considered to result in a macrocystic or polyvesicular vitelline pattern in humans, 1 and small cysts lined by AFP-positive cells were located within the myxoid compartment in the present case. Therefore, although the histological components of the present case were unique, they could be considered to belong to a histological continuum.
In humans, immunohistochemical examinations facilitate the diagnosis of testicular tumors (e.g., YST is usually positive for AFP, whereas germinoma is usually negative for AFP, and many YSTs also express cytokeratin).1,6,9,13,15 PLAP and c-kit are detected in most cases of germinoma and some cases of YST, but are not found in Sertoli cell tumors. 2 Inhibin α is found in Sertoli cell tumors, but not in YST, germinoma, or embryonal carcinoma.13–15 In the current study, similar to human testicular YST, both cases of bovine testicular YST were positive for AFP and negative for c-kit and inhibin α. PLAP was expressed in the neoplastic cells of the previous endodermal-type testicular YST, but was not found in the present case. Furthermore, a diffuse positive reaction for cytokeratin was observed in the endodermal-type testicular YST, but a scattered distribution was detected in the present case; vimentin was detected in the current case, but not in the previous endodermal-type testicular YST. Vimentin expression is rare in human YST, but both cytokeratin and vimentin have been detected in the neoplastic cells in the mesenchyme-like component of the myxomatous type of YST.5,6 Positive staining for desmin was also detected in these cells, 5 as was found in the current case. From these observations, the present case appeared to be purely composed of mesenchyme-like components without the epithelial structures seen in the previous 2 cases of YST involving calves.
In human medicine, adult testicular YST usually presents as a component of a mixed nonseminomatous germ cell tumor, while pure YST is one of the most common childhood testicular neoplasms.3,14 Most cases in both groups present with testicular enlargement.3,14 In adults, the spread of the tumor and its prognosis depend on the other germ cell components of the tumor, and metastases usually first develop in the parailiac and paraaortic lymph nodes. Adult patients are suggested to have worse prognoses than children. 11 One possible reason for this is that metastasis is uncommon at presentation in children.1,14 The 2 previously reported cases of YST affecting calves, which originated in the testes and abdominal cavity, respectively, were biologically malignant and exhibited disseminated metastases.4,8 As with childhood testicular YST, the current case only presented with testicular enlargement without metastasis. In conclusion, the enlarged testis of a newborn calf was diagnosed as YST; however, the histological features of this case, including its immunohistochemical characteristics and biological behavior, were completely different from those of the 2 previously reported cases of YST involving calves.4,8
Footnotes
a.
Vectastain Elite ABC Kit, Vector Laboratories Inc., Burlingame, CA.
b.
Nichirei Corp., Tokyo, Japan.
c.
Dako Denmark A/S, Glostrup, Denmark.
d.
V9, Dako Denmark A/S, Glostrup, Denmark.
e.
D9, Progen Biotechnik GmbH, Heidelberg, Germany.
f.
R1, AbD Serotec, Oxford, UK.
g.
SP15, Thermo Fisher Scientific Inc., Waltham, MA.
h.
AE1/AE3, Nichirei Corp., Tokyo, Japan.
i.
CAM5.2, BD Biosciences, San Jose, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
