Abstract
Detection of antibodies against Bovine viral diarrhea virus (BVDV) in serum and milk by enzyme-linked immunosorbent assay (ELISA) is a crucial part of all ongoing national schemes to eradicate this important cattle pathogen. Serum and milk are regarded as equally suited for antibody measurement. However, when retesting a seropositive cow 1 day after calving, the serum was negative in 6 out of 9 different ELISAs. To further investigate this diagnostic gap around parturition, pre- and postcalving serum and milk samples of 5 cows were analyzed by BVDV antibody ELISA and serum neutralization test (SNT). By ELISA, 3 out of the 5 animals showed a diagnostic gap in the serum for up to 12 days around calving but all animals remained positive in SNT. In milk, the ELISA was strongly positive after birth but antibody levels decreased considerably within the next few days. Because of the immunoglobulin G (IgG)1-specific transport of serum antibodies into the mammary gland for colostrum production, the IgG subclass specificity of the total and the BVDV-specific antibodies were determined. Although all 5 animals showed a clear decrease in the total and BVDV-specific IgG1 antibody levels at parturition, the precalving IgG1-to-IgG2 ratios of the BVDV-specific antibodies were considerably lower in animals that showed the diagnostic gap. Results showed that BVDV seropositive cows may become “false” negative in several ELISAs in the periparturient period and suggest that the occurrence of this diagnostic gap is influenced by the BVDV-specific IgG subclass response of the individual animal.
Keywords
Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) is a major cattle pathogen and, because of its economic importance, eradication programs are ongoing in many European countries. 19 The key to successful eradication is the identification and removal of persistently infected (PI) animals. 14 A PI animal is the result of the transient infection of the dam between the second and fourth month of gestation. Persistently infected animals spread the virus throughout their lives and are the most important source of infection. 20 Upon contact with a PI animal, seronegative cattle may become transiently infected and show a wide range of usually mild clinical signs such as respiratory disease, diarrhea, and reduced reproductive rate. 2 Normally, the virus is cleared quickly, and a long-lasting humoral and cellular immune response prevents further infections. Therefore, a high proportion of antibody-positive animals in a herd is an indication of the possible presence of a PI animal. Hence, measuring antibodies against BVDV in milk or serum to detect either herds with PI animals or to verify the absence of the virus is a crucial part of every eradication program.15,19
To compare the suitability of serum and milk for the determination of BVDV antibody status, sera were sampled from the lactating animals of a dairy herd and screened for BVDV antibodies using a commercial antibody-capture enzyme-linked immunosorbent assay (ELISA). a Positive sera were further verified by serum neutralization test (SNT) as described previously. 1 Six weeks after the initial screening, serum and milk from all seropositive animals were sampled in order to compare the antibody levels in the 2 types of material with 9 different ELISAs (i.e., 8 commercially available kits and 1 “in-house” test developed at the authors’ institute). 6 Of the commercial tests, 3 were indirect ELISA kitsa–c and 5 had blocking formats.d–h The in-house test was an indirect ELISA. 6 Surprisingly, the serum of 1 animal that was classified as seropositive gave a negative result in 6 out of the 9 tests: in 4 out of 5 blocking ELISAsd,e,g,h and in 2 out of 4 indirect tests,a,c including the one used for the initial testing. However, the milk of the same animal was strongly positive in all of the ELISAs. Further investigations revealed that this cow had just given birth the day prior to sampling. Unfortunately, the animal was sold shortly after sampling and follow up of any further development of the antibody levels was impossible. All other animals were in “normal” lactation, and their sera proofed to be antibody positive in all of the 9 tests applied.
It is known that the total immunoglobulin (Ig) concentrations in serum are subject to variations during the periparturient period, mainly because of colostrum production.4,9 Furthermore, cows infected with Neospora caninum have been reported to turn seronegative in serum at parturition in an indirect fluorescent antibody test. 12 However, a drop in BVDV antibodies that leads to a diagnostic gap was unexpected as serum and milk have been regarded as reliable materials for antibody detection throughout the entire lactation. 18 To analyze whether this effect was transient and reproducible in other animals, serum and milk samples were repeatedly collected from 5 cows before and after parturition in the subsequent months. Sampling was performed weekly to biweekly and as soon as possible after the calving time point. All serum and milk samples were tested using a commercial BVDV antibody-capture ELISA, a which is used for routine diagnostics at the authors’ institute. In addition, 1 serum sample from before, close to, and after parturition was tested by SNT.
Antibody levels measured by ELISA became negative or indeterminate in sera of 3 out of the 5 animals around parturition (Fig. 1C–1E). Even though the titers were lower around parturition, all sera remained clearly positive when analyzed by SNT (Fig. 1). In the 2 animals that stayed ELISA positive, relative optical density (OD) levels dropped around parturition but not below the positive cutoff of the test (Fig. 1A, 1B). Because the sampling time points in correlation to the time of calving differed between the animals, direct comparison of the duration of the antibody decrease was difficult. However, the kinetics of the antibody loss seemed to vary substantially between animals. Animal 5 (Fig. 1E) was negative or indeterminate for at least 12 days (i.e., from 6 days antepartum [a.p.] to 5 days postpartum [p.p.]). This period was much shorter for the other 2 animals (3 and 4). In animal 5, the lowest antibody level was already reached at day 6 a.p., whereas animals 3 and 4 were still ELISA positive at days 7 and 9 a.p. In all cases, the antibody levels in milk were highest in the first sample postpartum and dropped very quickly thereafter (Fig. 1).
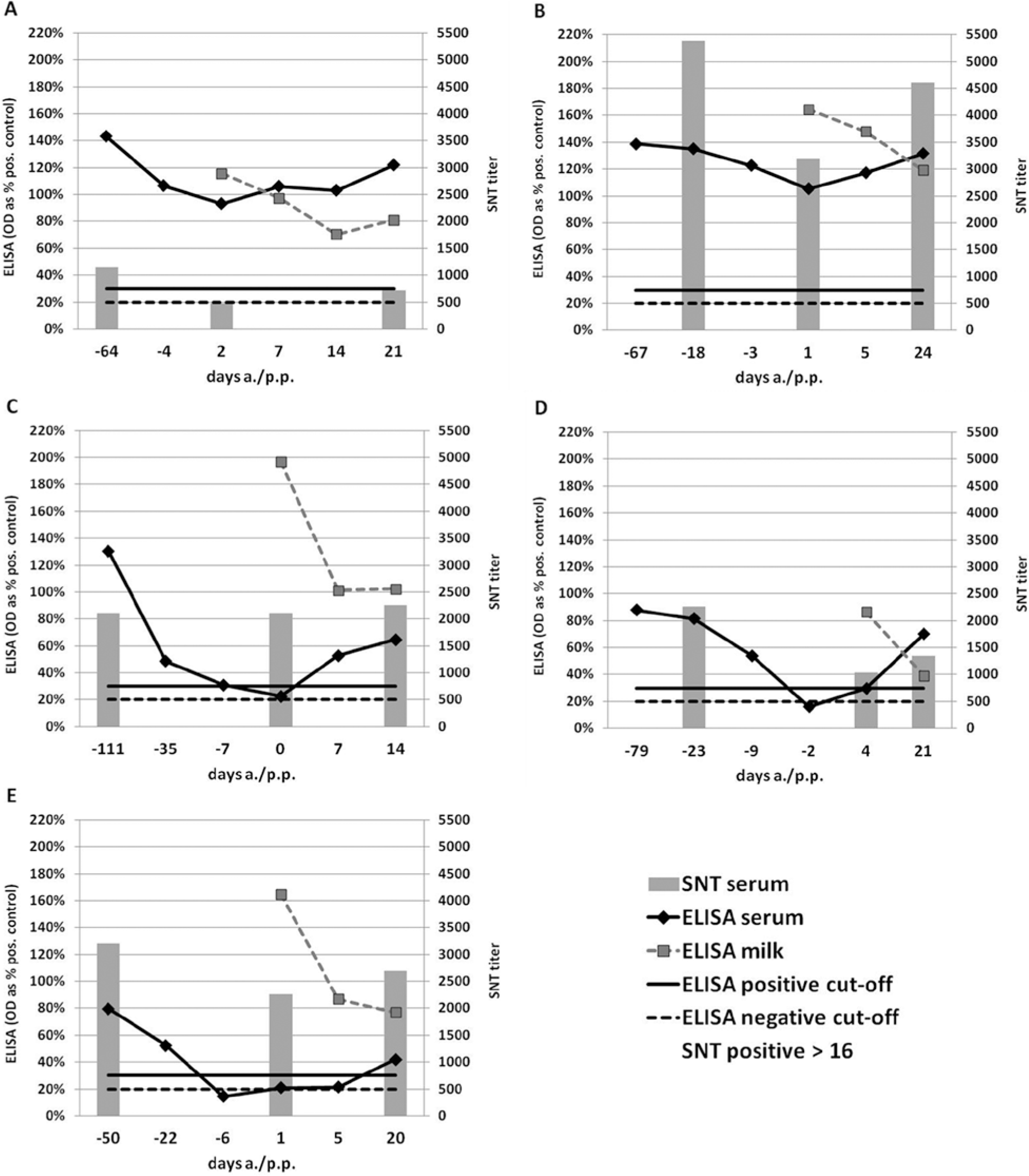
Bovine viral diarrhea virus (BVDV) antibody enzyme-linked immunosorbent assay (ELISA)
a
results (lines) and serum neutralization test (SNT) titers (bars, secondary vertical axis) of 5 cows around parturition. The positive cutoff for the SNT is 16. Animals 1 (
The generally lower relative OD values measured by ELISA at least 1 month prior to calving in 2 animals that turned antibody negative (animals 4 and 5) compared with the animals that stayed positive may explain the negative ELISA results around parturition. However, animal 3 had high relative OD values antepartum, similar to the animals that stayed positive. In addition, the SNT titers were not intrinsically lower in the animals that turned ELISA negative than in the animals that remained positive throughout.
Immunoglobulin G is the most abundant Ig in bovine serum (approximately 85–90%) and is divided into 2 subclasses with slightly more IgG1 than IgG2. 11 The majority of Ig in colostrum is blood derived. 4 Because of the subclass-specific nature of the receptor-mediated transport, IgG1 is the predominant Ig in colostrum. 5 As a result of hormonal changes, the transport is massively upregulated shortly before parturition, leading to an accumulation of IgG1 in colostrum and reduced concentrations of IgG1, but not IgG2, in the serum.4,7–9,13 Hence, it was hypothesized that individual differences in the IgG1 and IgG2 concentrations could be the reason for the different susceptibility of the cows to become negative in BVDV serum ELISA around parturition. Therefore, the total IgG, IgG1, and IgG2 concentrations of 1 sample before, shortly after, and 2–3 weeks after parturition (same time points as used for SNT) were quantified using a bovine IgG quantitation ELISA set i and subclass-specific detection antibodies. The time points were chosen based on serum samples that were available in sufficient volume to be used for all analyses. Briefly, 96-well flat-bottom microtiter plates were coated with the sheep anti-bovine IgG coating antibody for 1 hr, washed, and blocked overnight using 5% cold water fish gelatin. The serum samples were analyzed in duplicates, and a dilution series of the reference serum were incubated for 1 hr at room temperature. Purified bovine IgG1 l and IgG2 m were used to control the specificity of the test. After a wash step, the 1:4,000 diluted horseradish peroxidase–conjugated sheep anti-bovine IgG, i anti-bovine IgG1, j or anti-bovine IgG2 k detection antibodies were added. Tetramethylbenzidine (TMB) was used as substrate for the peroxidase, and the OD was measured at a wavelength of 450 nm. A standard curve was generated and the IgG concentrations of the samples calculated.
With the exception of animal 1, the IgG1 concentrations in serum were lower than the IgG2 concentrations (Fig. 2). According to the literature, the 2 subclasses should be present in approximately equal concentrations.3,8,21 No cross reactivity of the subclass-specific detection antibodies was detected. However, it cannot be excluded that they differ slightly in avidity. As expected, the total IgG and the IgG1 concentrations in the serum dropped at parturition in all cases (Fig. 2A, 2B), whereas IgG2 concentrations increased (Fig. 2C). There was a tendency for sera that showed the diagnostic gap to show higher IgG2 concentrations around parturition (Fig. 2C, animals 3–5). However, neither the total IgG or IgG1 concentrations nor the IgG1-to-IgG2 ratios showed any correlation to the susceptibility of the single animals to turn BVDV ELISA negative. Indeed, the IgG1-to-IgG2 ratio was similar in all animals except for animal 1 (Fig. 2D). However, because the IgG1-to-IgG2 ratio of BVDV antibodies may be different from the ratio of the total antibodies, the IgG specificity of the anti-BVDV antibodies using the in-house BVDV antibody ELISA together with the subclass-specific detection antibodies from the IgG quantitation ELISA kit was determined.
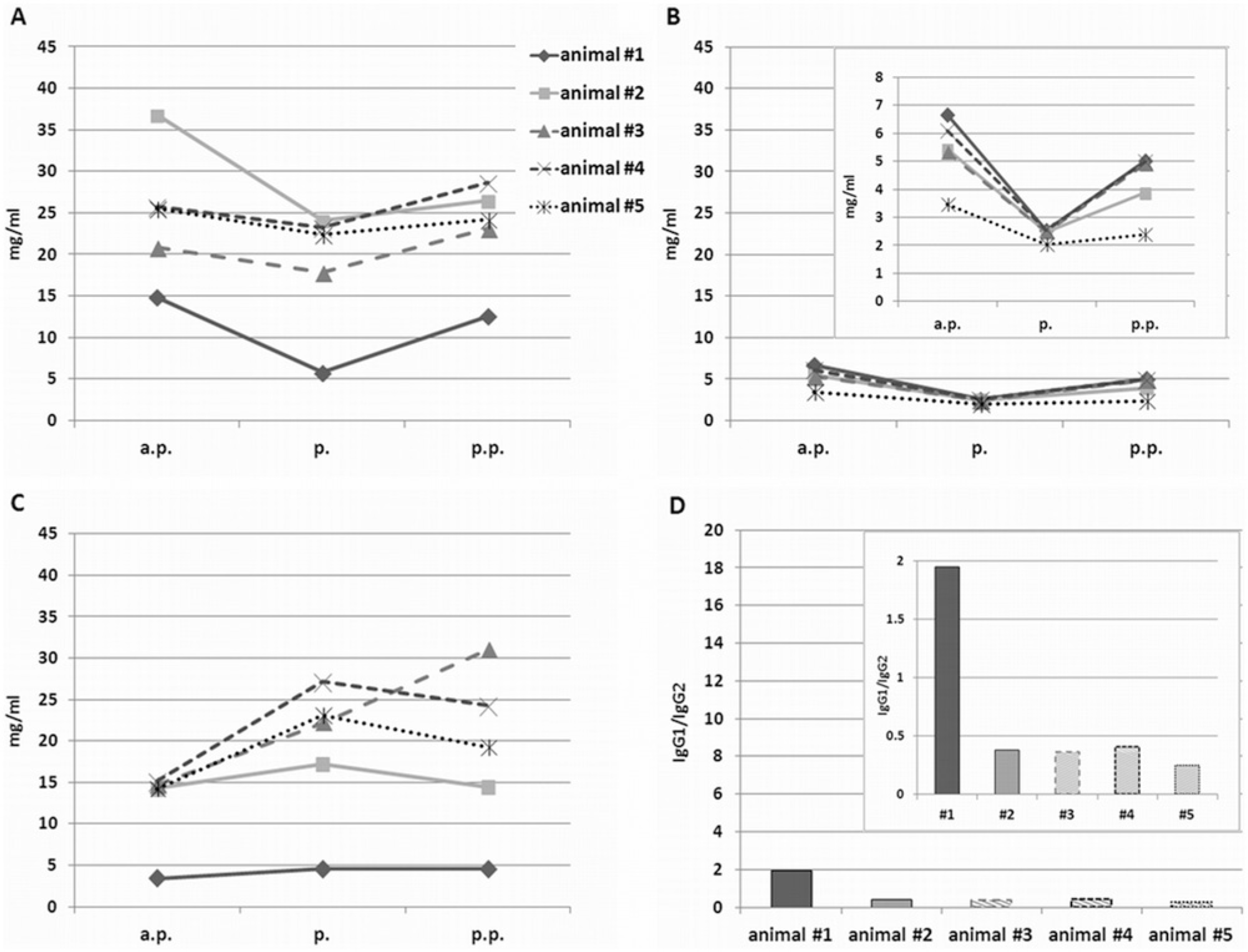
Bovine immunoglobulin G (IgG) concentrations in serum of 5 cows antepartum (a.p.), at birth (p.), and postpartum (p.p.). For the detailed sampling time points, see Figure 1 (serum neutralization test [SNT]). The total immunoglobulin (IgG) (
The kinetics of the BVDV-specific antibody levels showed the same IgG1 decrease and IgG2 increase around parturition as observed using the IgG quantitation ELISA. However, animals that turned BVDV ELISA negative at parturition tended to have lower BVDV-specific IgG1 but higher IgG2 concentrations than the animals that stayed positive (Fig. 3B, 3C). Even though the IgG1-specific OD values in the antepartum samples did not differ much between the animals, the IgG1-to-IgG2 ratio was considerably lower in animals that turned ELISA negative at parturition (Fig. 3D). In the 2 animals that remained positive, the ELISA OD values of the IgG1 BVDV antibodies were 8 and 18 times higher, respectively, than the IgG2 BVDV antibodies. In contrast, the IgG1 OD values of the 3 animals that turned negative or indeterminate in the BVDV ELISA at parturition were only 1.5 and 1.8 times higher or even lower than the IgG2-specific values.
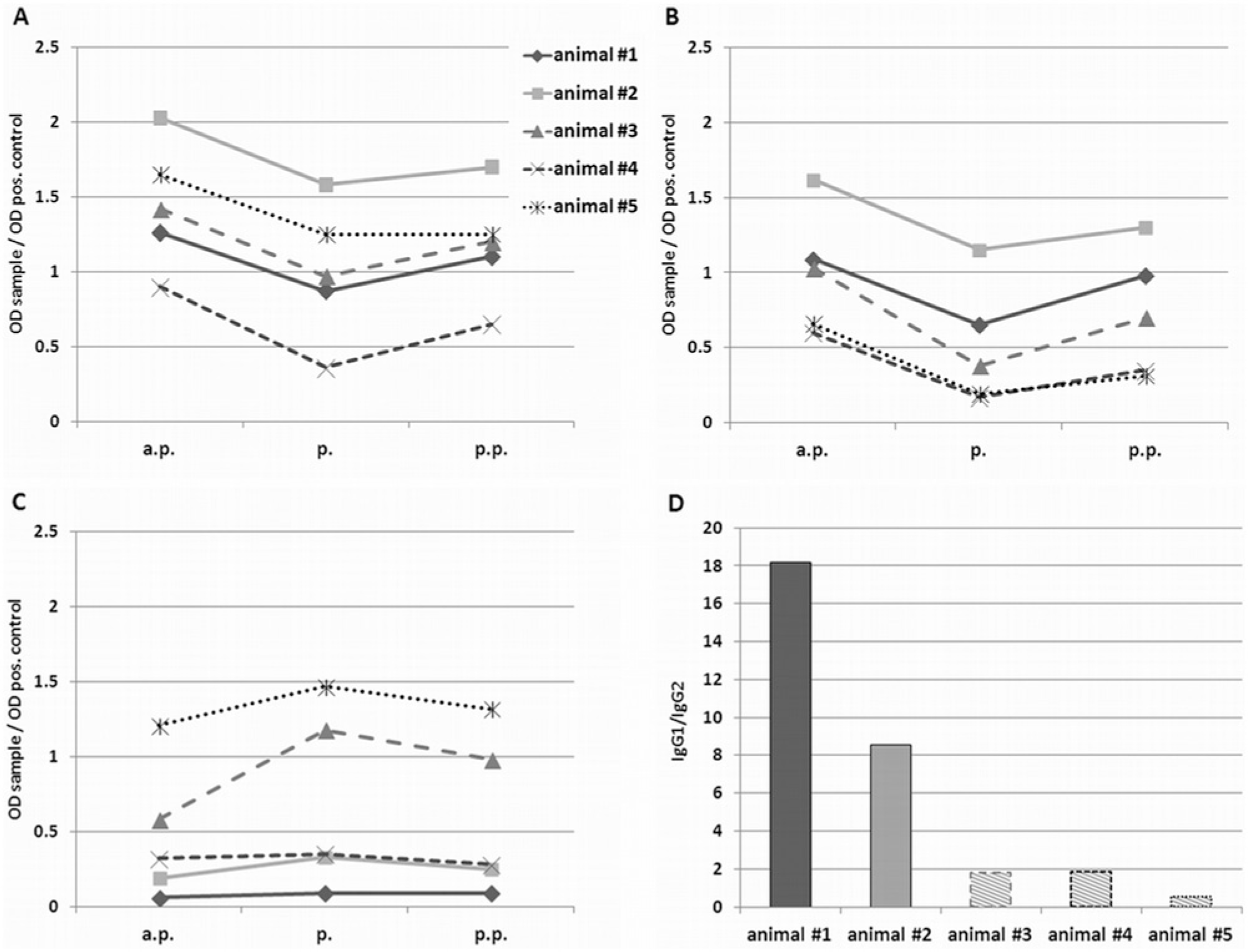
Immunoglobulin G (IgG) class-specific Bovine viral diarrhea virus (BVDV) antibody enzyme-linked immunosorbent assay (ELISA) results (relative optical density [OD] values) in serum of 5 cows antepartum (a.p.), at birth (p.), and postpartum (p.p.). For the detailed sampling time points, see Figure 1 (serum neutralization test [SNT]). The total anti-BVDV IgG (
Because of the small number of animals tested, statistical analysis of the results is not applicable. However, the results may provide an indication that the IgG specificity of the antibody response upon BVDV infection influences the risk of a particular animal to become “false” negative in BVDV ELISA around parturition. Regardless of the total concentration of antibodies, animals with a pronounced BVDV-specific IgG1 response appear less likely to “lose” the majority of the BVDV antibodies for colostrum production. As the extent of the IgG1 drop in serum around parturition and the periparturient changes in the immune response are generally reported to be influenced by the breed of the cows,9,16,17 a genetic predisposition for the diagnostic gap in BVDV serology around parturition is likely. In the current study, all animals, with and without diagnostic gap, were of the Swiss Fleckvieh (syn. Simmental × Holstein) breed, with the exception of animal 1, which was a Jersey cow. Further analyses will be necessary to investigate the occurrence of the diagnostic gap in other breeds. However, the occurrence of the diagnostic gap also depends on the diagnostic test used. The ELISA seems to be more susceptible than SNT. Whether other diagnostic tests used to detect BVDV antibodies, such as the agar gel immunodiffusion test, display this diagnostic gap, is not known. To the authors’ knowledge, all blocking BVDV antibody ELISAs and most indirect ELISAs detect antibodies that are directed against conserved antigens in the viral nonstructural protein NS3, albeit this could not be confirmed for 2a,b out of the 9 ELISAs used in the current study. In contrast, SNT measures neutralizing antibodies that are predominantly directed against the rather variable envelope glycoprotein E2. The IgG subclass proportions may be different between these 2 types of antibodies and hence the effect of colostrum-dependent loss may vary. In addition, the detection of antibodies by SNT is not IgG subclass–specific, as non-neutralized viruses—not the antibodies themselves—are measured. By contrast, in the case of indirect ELISAs, the detection antibody may preferably bind to bovine IgG1 or IgG2. Preliminary experiments exchanging detection antibodies between different indirect ELISAs support this hypothesis (data not shown). Furthermore, differences in the way the antigen is prepared may play a role (direct coating or by monoclonal antibodies).
It remains to be determined how frequently this diagnostic gap occurs. In the present study, 3 out of 5 cows became “false” negative; it may thus be a rather common phenomenon. The duration of the diagnostic gap requires further investigation, but results point to a gap of up to 2 weeks. It may be even longer in some cases, as the general immunological suppression in cows around parturition is reported to last from 2–3 weeks a.p. until 3 weeks p.p.7,10,16 It would also be interesting to analyze if antibody levels against other pathogens react similarly. The lack of evidence in the literature (except for N. caninum 12 ) might be explained by the transient nature of the phenomenon and the fact that emphasis of studies on the development of antibody levels is not typically set on cows around calving time. However, as the transport of IgG1 into the colostrum is not known to display any antigen specificity, a diagnostic gap might similarly occur for other antigens.
To determine the antibody status of cows shortly before calving, serum will be used for analysis. To miss a few seropositive animals may not be dramatic for the determination of a BVDV herd status. However, in case of a spot test or if determining the antibody status of a single animal, a false-negative status might be relevant as it masks a previous virus contact. In addition, if such an animal is retested several weeks after parturition, the change of the serological status from (false) negative to positive may indicate a very recent seroconversion, which might lead to costly herd investigations. Similarly, when investigating a possible infectious cause for a case of late abortion or stillbirth, the use of a paired serum sample with a false-negative result in the first sampling time point around parturition and a positive result a few weeks later might pretend a very recent, acute BVDV infection as the culprit of the reproductive loss. The use of serum to determine the BVDV antibody status by ELISA should therefore be avoided within at least 1 week before and after parturition.
Footnotes
Acknowledgements
The authors would like to thank the farmers and personnel of the agricultural college “Inforama Rütti” for their cooperation, and are indebted to Hans-Rudolf Vogt for help with sampling and Ruth Parham for linguistic improvement of the article.
a.
IDEXX HerdChek BVDV Ab (indirect) ELISA, IDEXX Switzerland AG, Liebefeld-Bern, Switzerland.
b.
Svanovir BVDV-Ab (indirect) ELISA, Svanova Biotech AB, Uppsala, Sweden.
c.
Bio-X BVDV BIO K004 (indirect) ELISA, Bio-X Diagnostics SPRL, Jemelle, Belgium.
d.
Svanovir BVDV p80-Ab (blocking) ELISA, Svanova Biotech AB, Uppsala, Sweden.
e.
Bio-X BVDV BIO K230 (blocking) ELISA, Bio-X Diagnostics SPRL, Jemelle, Belgium.
f.
PrioCHEK BVDV Ab (blocking) ELISA, Prionics AG, Schlieren-Zurich, Switzerland.
g.
SERELISA BVD p80 Ab Mono Blocking ELISA, Synbiotics SAS, Lyon, France.
h.
Pourquier ELISA BVD/MD/BD P80 Antibodies Screening, Institute Pourquier SAS, Montpellier, France.
i.
Bovine IgG Quantitation ELISA Set, Bethyl Laboratories Inc., Montgomery, TX.
j.
Sheep anti-bovine IgG1 antibody, HRP conjugated, Bethyl Laboratories Inc., Montgomery, TX.
k.
Sheep anti-bovine IgG2 antibody, HRP conjugated, Bethyl Laboratories Inc., Montgomery, TX.
l.
Purified bovine IgG1, Bethyl Laboratories Inc., Montgomery, TX.
m.
Purified bovine IgG2, Bethyl Laboratories Inc., Montgomery, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Canton of Bern and by the Swiss Veterinary Office (project no. 1.08.15).
