Abstract
In New Zealand, an arbovirus surveillance program has been operating for more than 20 years, which includes testing of cattle with the
Keywords
The New Zealand livestock population is free from
Laboratory diagnosis of Akabane disease is generally based on serology. The VNT is considered the reference method (gold standard) but is rather laborious and time consuming, requiring live virus and cell culture (Della-Porta AJ, White JR, Gard GP, Kirkland PD: 1993, Akabane disease—histopathology, virology and serology. Available at: http://www.scahls.org.au/__data/assets/pdf_file/0007/1280815/akabane_diseases.pdf). Commercial enzyme-linked immunosorbent assay (ELISA) kits are available, are easier to perform, and could be a good alternative to the VNT. The current study reports the comparative evaluation of 2 commercial ELISA kits on cattle serum samples from noninfected New Zealand herds, and infected Australian herds, with the aim of identifying an ELISA kit that would be best suited for AKAV surveillance on the New Zealand target population. 6
In the current study, the VNT was performed according to the Australian standard diagnostic procedure.
4
Samples with titers <1/4 were considered negative and titers ≥1/4 as positive. ELISA-1,
a
a commercial blocking ELISA with specificity for the viral glycoprotein Gc (formerly G1; Lunt RA, White JR, Newberry KM, et al.: 2007, Development of a commercial ELISA for the detection of serum antibody to Akabane virus.
The cattle numbers to be tested were based on previously published recommendations 6 and known diagnostic sensitivity (DSE) and diagnostic specificity (DSP) values, which were 94% and 98%, respectively, for ELISA-1 and for ELISA-2 (manufacturer’s information). Subsequently, a total of 334 serum samples from noninfected cattle from New Zealand were used as negative population from the sentinel cattle herds used for arbovirus surveillance. 10 The gold standard for absence of infection was the lack of detectable AKAV VNT antibody in New Zealand. A total of 548 cattle serum samples were collected from 3 herds in a region of Australia where AKAV is recognized to be endemic and were submitted to the Elizabeth Macarthur Agriculture Institute (Camden, New South Wales, Australia). Because Akabane disease is not present in New Zealand, and Australia is the most likely country from where the virus could spread, this population was considered the closest to the New Zealand target population. Of the 548 animals, a total of 378 had tested positive in the VNT at a cutoff of 1/4 and were considered the infected reference panel.
Frequency histograms for noninfected (panel 1) and infected (panel 2) cattle were generated for each ELISA to display the distribution of %I results in both classes of cattle with the aid of a spreadsheet program. Statistical analyses were undertaken using Epitools (Sergeant ESG: 2009, Epitools epidemiological calculators. AusVet Animal Health Services and Australian Biosecurity Cooperative Research Centre for Emerging Infectious Disease. Available at: http://epitools.ausvet.com.au). Diagnostic sensitivity and DSP for each test method were calculated by using the Epitool “test evaluation against a gold standard.” Receiver operating characteristic (ROC) analysis was performed over a range of possible cutoff points, based on ELISA %I values for the noninfected New Zealand cattle (panel 1) and the infected Australian cattle (panel 2) by using the Epitool “calculate test sensitivity and specificity and ROC curves” (Sergeant ESG: 2009).
The DSP was high for each of the 3 serological test methods evaluated. The DSP for the VNT performed at Investigation and Diagnostic Centre, Animal Health Laboratory, Upper Hutt, New Zealand, was 99.4%. Two samples had shown titers close to the cutoff of 1/4 (i.e., titers of 1/6 [intermediate titer of duplicates] and 1/4, respectively). These samples tested negative in both commercial ELISA kits and in a confirmatory VNT performed at the Australian Animal Health Laboratory (Geelong, Victoria, Australia). ELISA-1 possessed a DSP of 99.7%, as 1 sample tested with a suspect result (%I = 60.7). This sample was negative in the VNT and in ELISA-2. The DSP of ELISA-2 was 100%, with all 334 samples from the noninfected serum panel testing with negative results.
Test results for the VNT and the commercial ELISA kits for the infected herds’ serum panel were diverse. With the VNT as reference method (
In Figure 1, frequency histograms are shown for the 2 commercial ELISAs for infected (VNT-positive) and noninfected cattle. ELISA-1 presents a typical histogram for a well-performing test method where the infected and noninfected populations are fairly well separated. ELISA-2 shows a major peak in the positive %I range and, in addition, a shallow peak in the negative %I range. The noninfected population is clustered around low %I values. Again, these results could be interpreted with the presence of cattle with antibodies other than for AKAV, which are detected by ELISA-1 as positives, and which form the shallow peak in ELISA-2 in the negative range. It is known that even the VNT can suffer from cross-reactivity with other Simbu genogroup viruses because of the antigenic relatedness of these viruses 7 and the occurrence of reassorted Simbu genogroup viruses in nature. 12
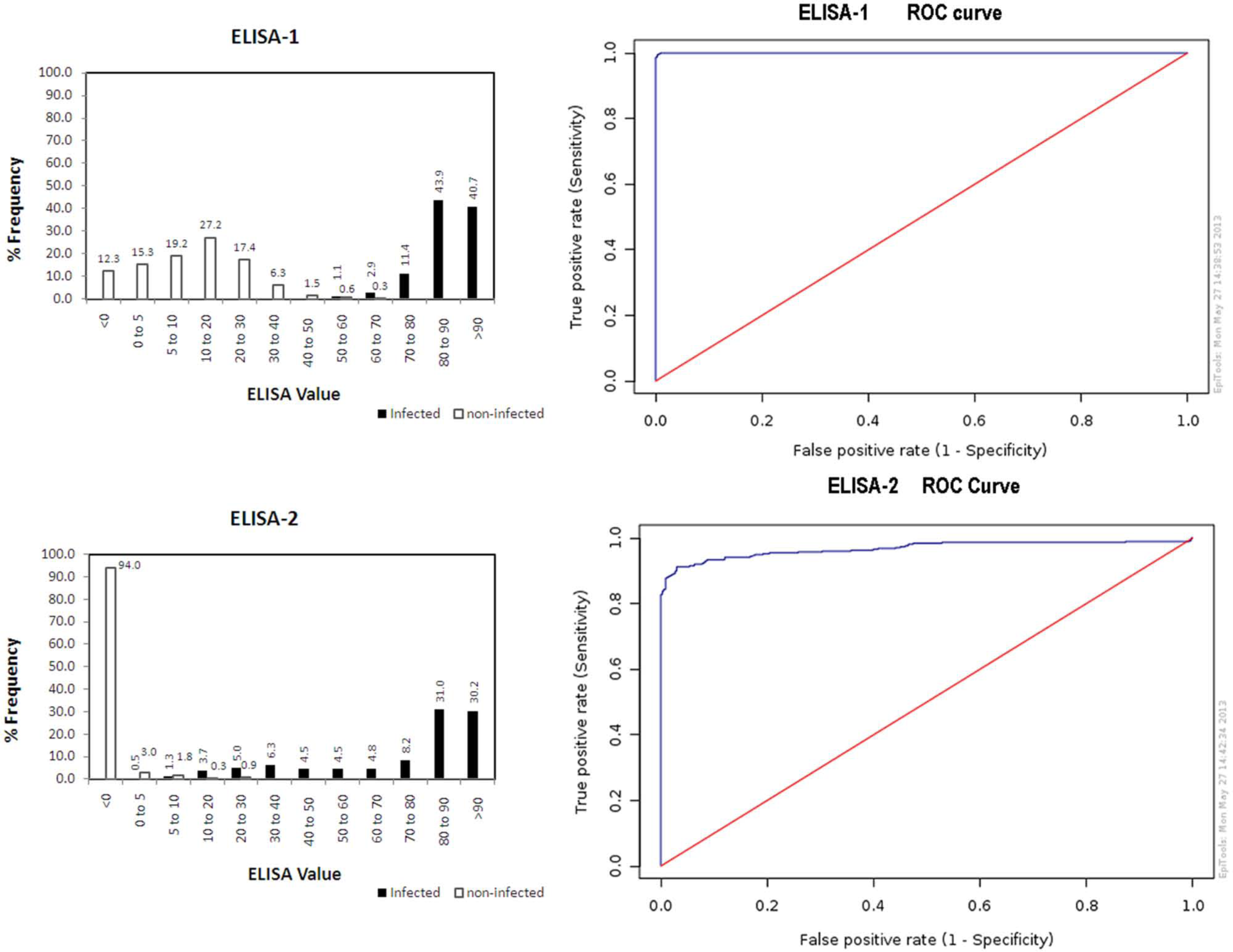
Frequency histograms and receiver operating characteristic (ROC) curves for 2 commercial blocking
The ROC analysis results for the commercial ELISAs are also shown in Figure 1. ELISA-1 presents a curve that is typical for a well-performing test method, where the curve climbs rapidly toward the upper left hand corner of the graph. This can be interpreted that the true positive rate is high and the false negative rate is low. ELISA-2 shows a less favorable curve. The slow rise indicates a problem with sensitivity on the population investigated.
Another possible explanation for the differences in the commercial ELISA results may lie in the different AKAV strains that were used for making the antigen for each ELISA. ELISA-1 uses antigen prepared from the Australian virus strain B8935; ELISA-2 uses antigen prepared from the Japanese virus strain OBE-1. A publication reporting on a dot immunobinding assay showed different patterns for the Australian virus strain B8935 (pattern 2) and the Japanese virus strain OBE-1 (pattern 1), 14 thus confirming antigenic strain differences. It has been shown by others that antigenic diversity exists among AKAV isolates.1-3 Furthermore, phylogenetically, both strains are quite distant, with B8935 belonging to genogroup III and OBE-1 to genogroup II. 9 Perhaps OBE-1 lacks epitopes that are present in AKAV strains circulating in Australia or the monoclonal antibody 19B-4 in ELISA-2 does not block serum antibodies against certain epitopes present on Australian virus strains. For both assumptions, animals with antibody responses to such epitopes would not be detected by ELISA-2. Further investigations would be necessary to answer these questions.
In conclusion, the current study has highlighted the importance of evaluating test methods for the target population for which they will be used. Even though evaluation data existed for other populations, and DSP values were comparable, DSE values for the various populations turned out to be quite different. In regard to fitness for purpose for the New Zealand arbovirus surveillance program, both commercial ELISA kits could be used. In view of the higher sensitivity of ELISA-1, this ELISA should be the preferred test method. As it is more likely that false-positive results will occur with ELISA-1, such results could be clarified by using the VNT as a confirmatory test method.
Footnotes
Acknowledgements
The authors would like to thank all staff from the laboratories involved in testing the sample panels, as well as the Laboratory Manager, Animal Health Laboratory, Upper Hutt, and other Ministry for Primary Industries, Surveillance Team, reviewers for valuable input and discussions. The authors are very grateful to AusVet Animal Health Services for making their evaluation software easily available. Furthermore, the authors would like to acknowledge the contribution made by IDvet Laboratories for making their ELISA kits available at reduced cost.
a.
ID Screen Akabane Competition ELISA kit, IDvet, Montpellier, France.
b.
Akabane ELISA kit, Chisso Corp., Yokohama, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this study was provided by the Ministry for Primary Industries New Zealand.
