Abstract
A 75.9-kg, 3.5-year-old male Irish Wolfhound dog with a 2–3-week history of gagging and eating difficulties was referred to the University of Florida Veterinary Medical Hospital (Gainesville, Florida) for evaluation of a large cranial mediastinal mass suspected to be a thymoma or lymphosarcoma. The patient had 4 months of nearly 10 kg progressive weight loss with severe flank sensitivity and radiographically apparent lumbar vertebral changes interpreted as discospondylitis. Lab work revealed hyperglobulinemia, mild proteinuria, normal T4, negative Brucella canis titer, and negative blood and urine bacterial cultures. A thoracotomy revealed a nonresectable, destructive, space-occupying mediastinal mass resulting in euthanasia without surgical recovery. Biopsies from the mass were collected during surgery for histology. Microscopic examination revealed extensive granulomatous cellulitis and lymphadenitis characterized by central cavitated necrotic areas containing debris and degenerate neutrophils, intermediate zones of fibrovascular proliferation with marked mixed inflammation, peripheral fibrosis, frequent multinucleated macrophages, and scattered mineralization. The necrotic material contained dense mats of 2 µm wide by 8–15 µm long fungal hyphae with parallel walls, acute angle branching, frequent septae, and occasional bulb-like dilations. DNA sequencing and phylogenetic analysis of the internal transcribed spacer region confirmed the presence of a fungus in the Inonotus tropicalis group. Inonotus tropicalis is primarily a wood decay fungus that is found on dead wood from angiosperms in tropical and subtropical habitats. Isolates of the I. tropicalis group have been detected a few times from immunosuppressed human beings with X-linked granulomatous disease.
A 75.9-kg, 3.5-year-old male Irish Wolfhound dog was referred to the University of Florida Veterinary Medical Center (UF-VMC; Gainesville, Florida) for evaluation of a large cranial mediastinal mass observed on radiographs by the referring veterinarian 2 weeks prior and diagnosed as a probable thymoma or thymic lymphoma. The patient had been healthy prior to diagnosis of discospondylitis 4 months before referral for the mediastinal mass. The patient had presented to the referring veterinarian for lethargy, lumbar pain, hind limb weakness, and severe sharp pain in the abdomen or flank diagnosed as discospondylitis. The pain was progressive, associated with a total weight loss of 10 kg over 4 months, and unresponsive to antibiotic, nonsteroidal anti-inflammatory (carprofen and acetaminophen), and narcotic (fentanyl patches) therapies. Articular facet degenerative joint disease and spondylitis deformans were demonstrated on thoracic and lumbar radiographs twice during the 4-month period, supporting the diagnosis of discospondylitis. However, a thoracic mass was not detected. A complete blood cell count was unremarkable, and serum chemistry revealed increased total protein, elevated globulins, and a low normal protein albumin to globulin ratio. Blood and urine cultures were negative, the T4 level was normal, and the Brucella canis titer was negative.
The patient presented to the referring veterinarian with respiratory difficulty 2 weeks prior to referral to the UF-VMC. The owner reported an unusual choking or retching noise of approximately 2 weeks duration, and increased respiratory sounds were noted during the physical exam. A presumptive diagnosis of pneumonia was made at that time. On return 1 week later, however, the patient had lost an additional 1 kg, and the owner reported difficulty eating and drinking. Thoracic radiographs performed by the referring veterinarian demonstrated a large thoracic mass with caudal displacement of thoracic structures that was not present in radiographs from 3 months and 10 days earlier (Fig. 1). Complete blood cell counts were unremarkable but serum chemistry findings showed that serum globulins remained elevated. A urinalysis demonstrated a recent mild urine acidosis and mild proteinuria. The clients were referred to UF for an oncology consultation.
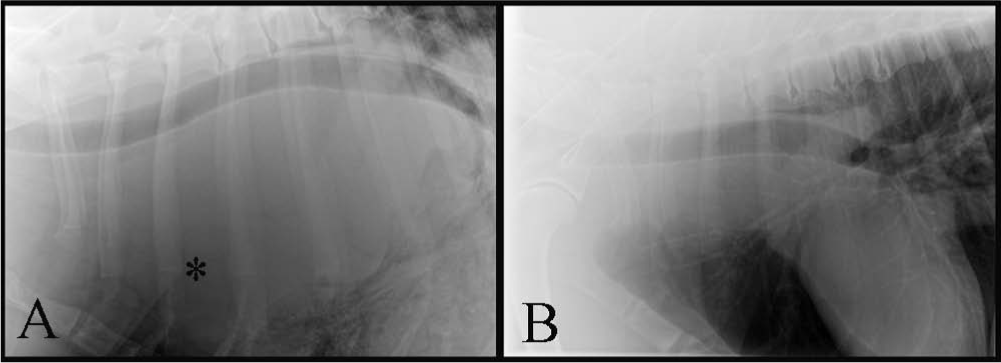
Lateral thoracic radiographs from a 3.5-year-old male Irish Wolfhound dog.
At UF, ultrasound-guided, fine-needle aspirates of the mass were attempted but the cytology was uninformative with a nonspecific mixture of blood and tissue elements. A core biopsy was not performed because of marked respiratory distress of the sedated patient. Complete blood cell count, serum chemistry, and urinalysis were largely unremarkable with no findings that suggested the origin of the mass. The globulins were moderately elevated with no corresponding elevation in total protein, and urine changes were the same as those documented by the referring veterinarian.
A thoracotomy was performed to attempt resection of the mass, and large areas of the mass were removed and submitted for histopathology to the UF-VMC Surgical Pathology Service. Euthanasia was performed during surgery because of poor prognosis based on the disruption of thoracic structures with incorporation of many large blood vessels, making it nonresectable, and the presence of megaesophagus. The owners declined a postmortem examination, thus it was not possible to conduct gross and microscopic evaluation of the entire mass, the vertebral column, and major organs. The clinicians did not submit surgical or postmortem samples for bacterial or fungal cultures because of the presumptive diagnosis of neoplasia and because there was no need for further therapeutic intervention.
Three tissue samples from the mass were received by the UF-VMC Surgical Pathology Service. The light tan to pink, soft, fluctuant multinodular samples measured 13.5 cm × 6 cm × 5.5 cm, 7.5 cm × 6.5 cm × 4.5 cm, and 11.5 cm × 10 cm × 5.7 cm. The tissue was fresh at the time of submission allowing for a gross description of the unfixed features. The samples contained many palpable fluid-filled cystic areas and were partially covered by a 2–4 µm firm, pink, fibrous, capsule-like coating. On cut surface, the specimens ranged from light tan to yellow, and were often soft or fluctuant; frequent poorly defined cystic areas were filled with thick cloudy fluid varying from yellow or tan to red-brown. Some areas contained abundant friable light tan to pink soft gelatinous material. There were solid areas that were light tan, soft, and homogenous, and others that were softer with a tan streaked pattern and yellow friable purulent material. The areas of fluid and purulent material ranged in size from pinpoint to 4 mm × 1 mm × 1 mm.
Representative sections from throughout the samples were placed in cassettes and allowed to completely fix in 10% neutral buffered formalin. The samples were routinely processed, paraffin embedded, and prepared as 4 µm, hematoxylin and eosin–stained sections on glass slides for examination by light microscopy. Histology of the sections revealed a varied pattern (Fig. 2) of inflammation, necrosis, cystic areas, and bands of fibrosis. Some areas retained the markedly disrupted and effaced remnants of lymph node architecture characterized by a partial capsule, lymphoid centers, and a poorly defined medulla. The areas of inflammation varied in character, but often included large numbers of macrophages, multinucleated giant cells admixed with lymphocytes and neutrophils forming solid sheets, and multifocal coalescing nodules. The extensive granulomatous cellulitis and lymphadenitis was often organized as poorly defined nodules with central cavitated necrotic areas containing debris and degenerate neutrophils. These were centers bordered by intermediate zones of fibrovascular proliferation with marked mixed inflammation, and bands of fibrosis with scattered mineralization (Figs. 3, 4). Multinucleated macrophages were abundant in many areas. The necrotic material often contained clear tracts or cleft-like spaces and dense mats of embedded 2 µm wide by 8–15 µm long fungal hyphae visible in hematoxylin and eosin–stained sections and strongly argyrophilic with Gomori methenamine silver stain (Fig. 4). The hyphae had occasional intercalary and terminal bulb-like expansions that expanded up to 8 µm in diameter. These hyphal bulbs are similar to those seen in cultures of Inonotus tropicalis that have been isolated from human tissues. 14 The hyphae had parallel to slightly bulging walls, frequent acute angle branching, and were often septate. There was no indication of an underlying neoplastic process. The final diagnosis was chronic, severe, granulomatous mediastinitis with abundant intralesional fungal hyphae.
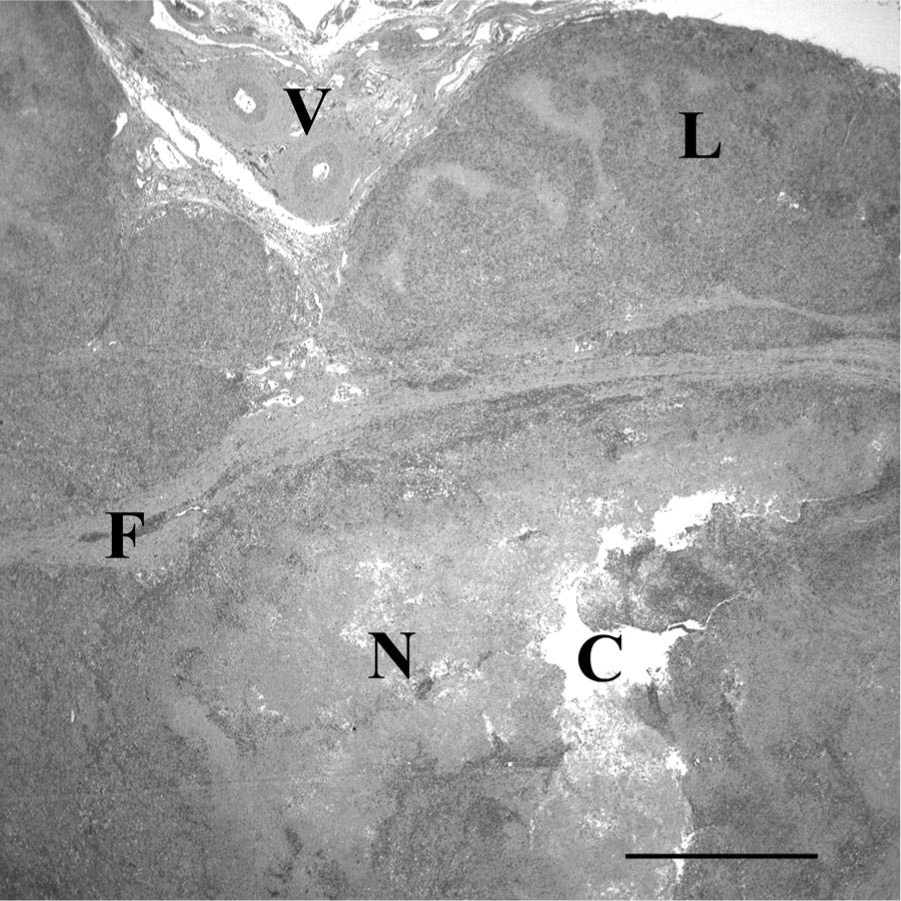
Low magnification histology of the mediastinal mass from a 3.5-year-old male Irish Wolfhound dog demonstrating large vessels (V), remnants of lymph node architecture (L), bands of fibrosis (F) and poorly defined nodules some of which contain abundant necrotic material (N) and a central clear tract or cavitation (C). Hematoxylin and eosin. Bar = 1 mm.
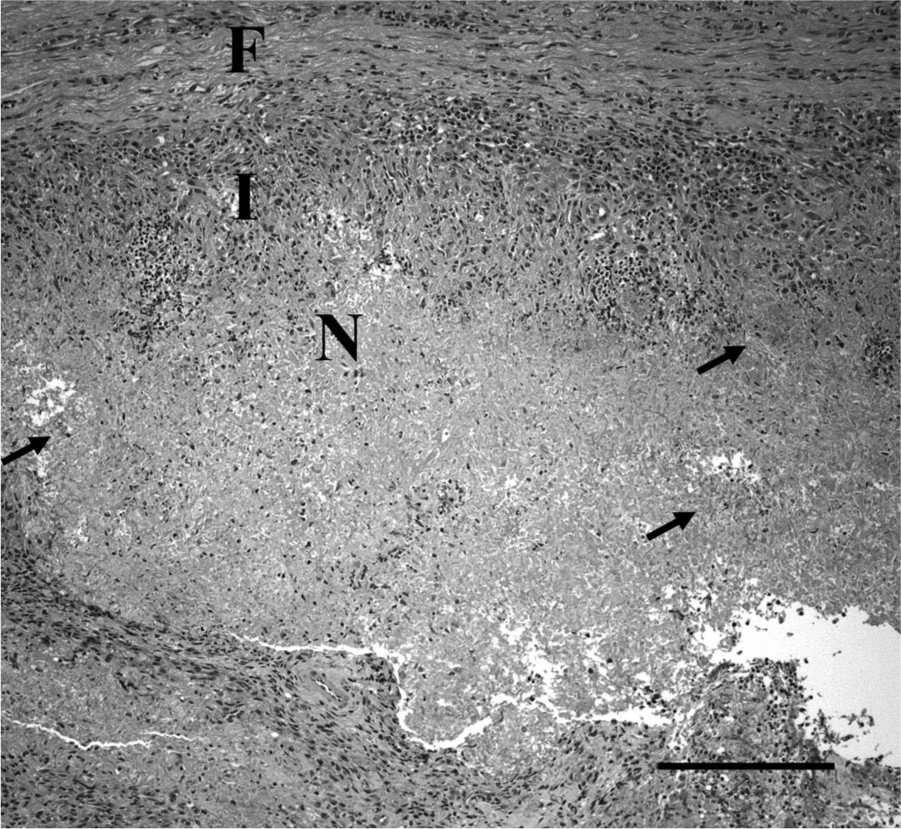
Histology of a nodule within the mediastinal mass from a 3.5-year-old male Irish Wolfhound dog having a central area of necrosis (N) bordered by a dense zone of inflammation (I) and fibrotic border (F). Large numbers of fungal hyphae (arrows) are within the necrotic material. Hematoxylin and eosin. Bar = 200 µm.
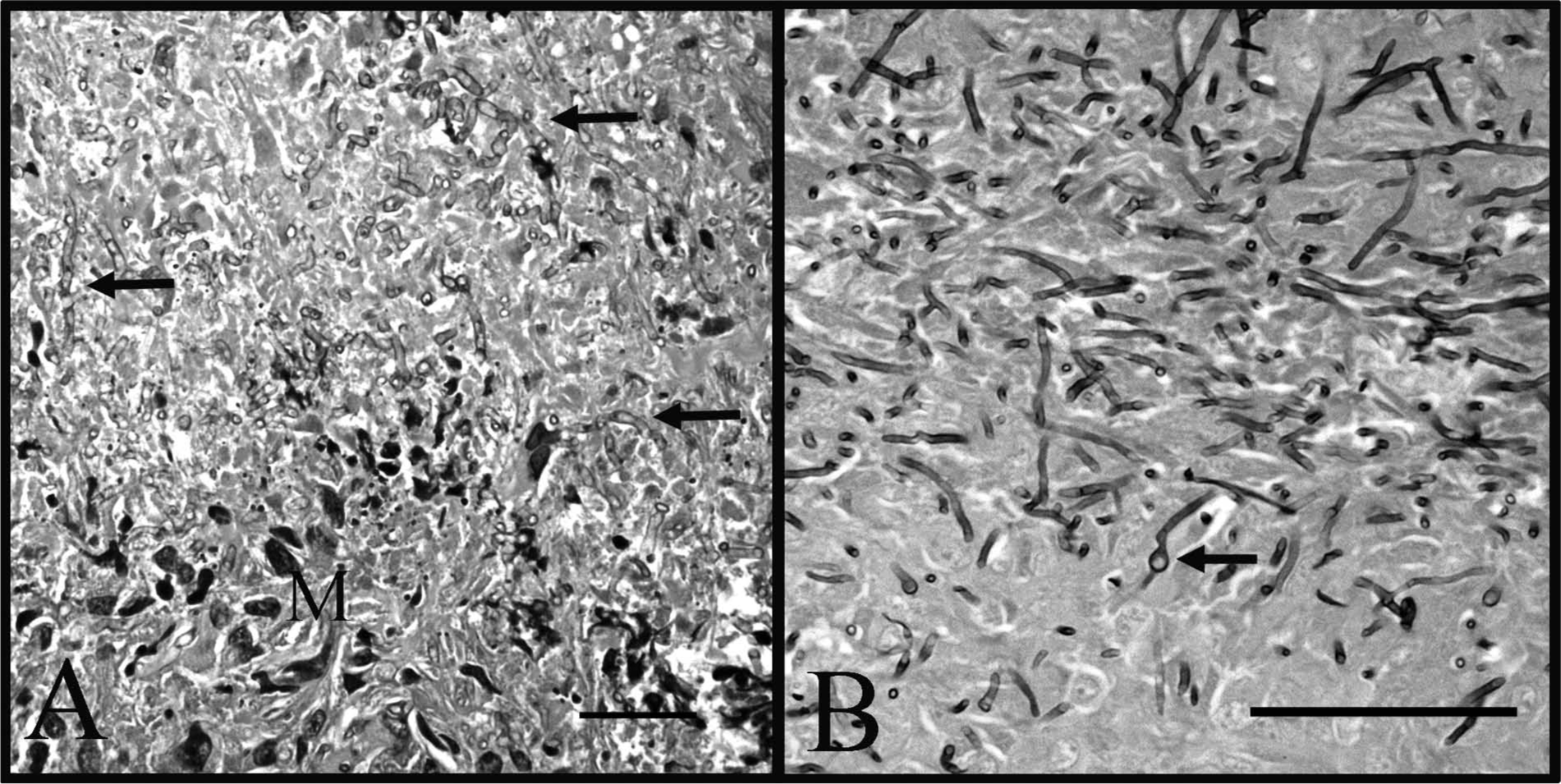
Fungal hyphae within the mediastinal mass from a 3.5-year-old male Irish Wolfhound dog as viewed with high magnification microscopy.
The identity of the unknown fungal pathogen was investigated by sequencing the internal transcribed spacer (ITS) ribosomal DNA (rDNA) region (ITS1-5.8s-ITS2) using fungal-specific primers. 6 This region has proved useful for molecular identification of fungi and was recently confirmed as an official DNA barcode marker for fungi. 12 The DNA was extracted from 15-µm scrolls cut from the formalin-fixed, paraffin-embedded specimens using a commercial kit a according to the manufacturer’s instructions. The ITS rDNA was amplified using the fungal-specific forward primer ITS1F 6 in combination with the universal eukaryotic reverse primer ITS4 18 using a previously published protocol from the Smith Mycology laboratory. 13 Amplicons were visualized on a 1.5% agarose gel stained with SYBR Green I. b The polymerase chain reaction products were prepared for sequencing using EXO and SAP enzymes. 7 Sequencing was performed at the UF Interdisciplinary Center for Biotechnology Research, and sequences were edited with commercial software. c To determine the closest relatives of the fungus, a nucleotide BLAST search against the National Center for Biotechnology Information GenBank database was conducted. All of the sequences with the highest homology in the BLAST comparison (i.e., sequences ≥80% similar across the ITS region) were fungi in the genera Phellinus, Inonotus, and closely allied groups. To determine the phylogenetic placement of the veterinary isolate, Mesquite 10 and MUSCLE 3 software packages to were used to generate an alignment that was 787 nucleotides in length and included 42 representative fungal taxa. A maximum parsimony (MP) analysis using PAUP* 15 and a maximum likelihood (ML) analysis using GARLI was then conducted (Zwickl D: 2006, Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maximum likelihood criterion. PhD dissertation, Department of Biology, University of Texas. Austin, Texas). Clade stability was estimated for both analyses using 1,000 bootstrap repetitions. The MP analysis generated 4 equally parsimonious trees of 393 steps; the ML analysis generated a single tree with a likelihood score of −ln 2797.59324 (Fig. 5). Both the ML tree (Fig. 5) and the MP consensus tree (data not shown) placed the isolate (Canine 1117 UF Veterinary Isolate) in a well-supported clade with I. tropicalis isolates from natural (AF534077) and clinical (AY641432, AY599487) settings. The DNA sequence from veterinary isolate 1117 was approximately 94–95% similar to the other 3 isolates of I. tropicalis, whereas the next most similar fungus, Phellinus repandus (AF534076), was only 85% similar. Inonotus tropicalis belongs to a taxonomically confusing group of fungi that includes species of Inonotus and Phellinus in the family Hymenochaetaceae.9,14,19 The taxonomy of wood decay fungi is based on molecular data and on fruiting bodies that do not typically form in pure culture. Thus, morphological studies of fungal cultures are not particularly useful for discriminating wood decay fungi at the genus or species level. 9
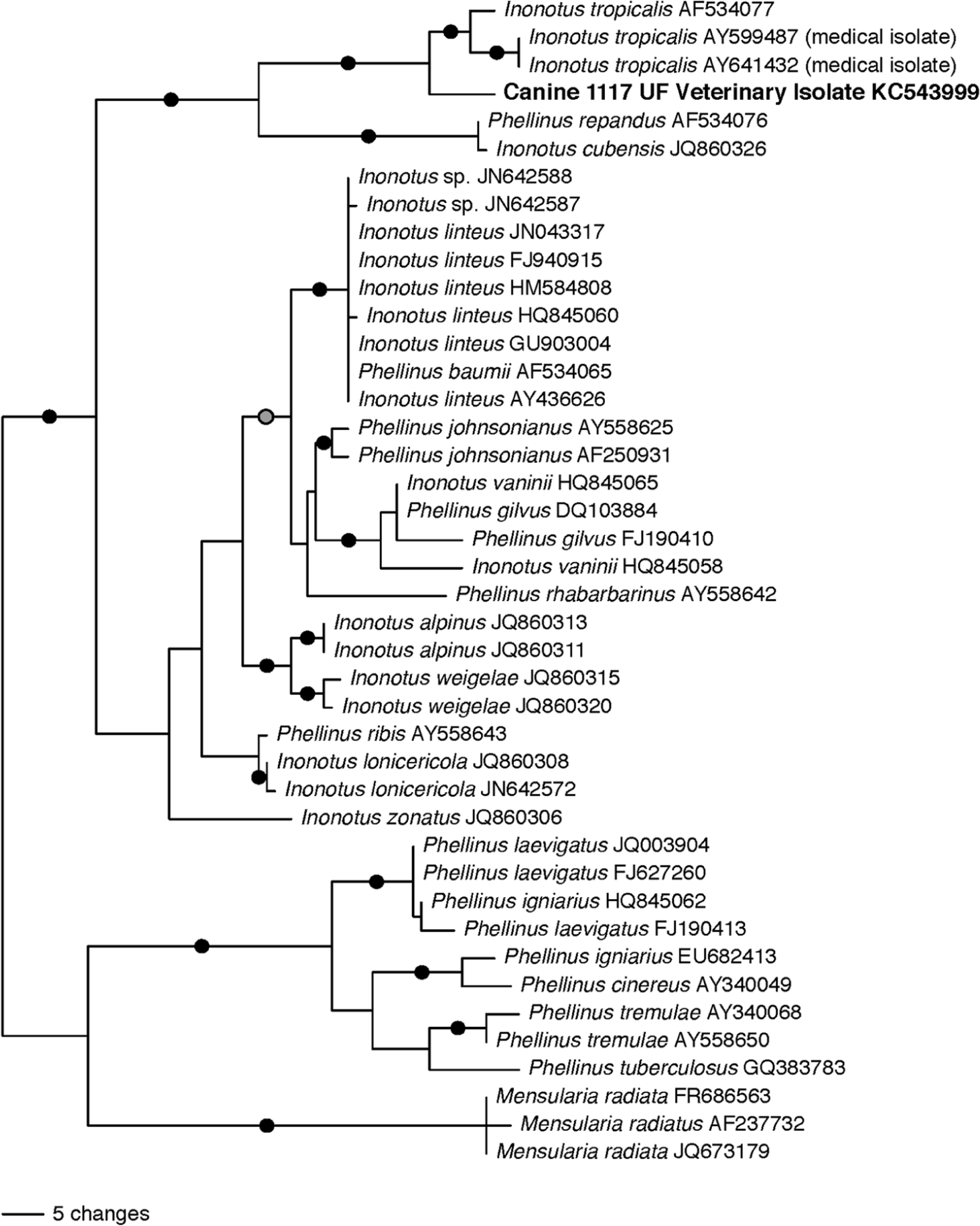
Midpoint rooted tree showing placement of veterinary isolate Canine 1117 UF Veterinary Isolate from the mediastinal mass of a 3.5-year-old male Irish Wolfhound dog based on maximum likelihood analysis of the internal transcribed spacer (ITS) ribosomal DNA. Black circles denote branches that received bootstrap support ≥75 in both maximum parsimony and maximum likelihood analyses. The gray circle denotes a branch that received support from maximum likelihood bootstrapping only.
The 4-month history of weight loss indicates the presence of a chronic condition resulting in the progressive loss of body condition. The most common causes for an intrathoracic mass within the cranial mediastinum in a middle-aged canine are trauma, neoplasia, or inflammation possibly associated with a foreign body or infectious agent.2,4 There was no known history of trauma in the Irish Wolfhound, and no clinical indication that fractures or additional soft tissue damage were present. The most common mediastinal tumors are thymoma and thymic lymphoma, although primary pulmonary carcinoma, branchial cyst, chemodectoma, neoplastic or ectopic thyroid tissue, and metastatic tumor are also possible. 20 Thymomas are rare in the canine and are composed of neoplastic epithelial tissue of thymic origin. 2 The tumors are often densely infiltrated by lymphocytes that are not neoplastic, but the presence of which can be misinterpreted as lymphoma. Aspirates of thymomas are diagnostically challenging because they often contain large numbers of lymphocytes and a paucity or complete lack of neoplastic epithelial cells. Thymomas are designated as benign or malignant predominantly based on their gross appearance and clinical behavior, indicating probable resectability rather than microscopic characteristics suggesting malignancy.2,20 Respiratory distress and paraneoplastic findings, including megaesophagus, have been associated with thymomas. Primary lung tumors tend to be solitary masses most often within the caudal lung lobes, although milliary and multifocal nodular patterns are also encountered. Most primary lung tumors are carcinomas, usually with adenocarcinoma differentiation, central cavitation due to necrosis, and pleural effusion. 21 In the current case, the identification of a solitary cranial mediastinal mass with a grossly cystic character in combination with megaesophagus was strongly suggestive for a thymoma. Definitive diagnosis of the tumor type requires biopsy of several areas due to the heterogeneous character of the mass as the oncology protocols differ significantly for carcinoma compared with thymic lymphoma. 21
Microscopic examination revealed an inflammatory pattern with no indication of neoplasia. Primary pulmonary granulomas or abscesses may occur with infections by genera Nocardia, Actinobacillus, Actinomyces, Mycobacterium, or fungi, and can be introduced by a foreign body entering the thoracic cavity by way of the digestive tract, trachea, or penetration of the body wall. A mass effect within the thorax may also be observed with lymphomatoid granulomatosis. 1 Blood and urine cultures were negative, diminishing the probability of bacterial intrathoracic lymphadenopathy or cranial lobar pneumonia. Pneumocystis carinii pneumonia in dogs diffusely affects the lung, and if this organism had been present in the current case, abundant 8 µm round to crescent, intracellular, and extracellular yeast forms would have been found. 2 Similarly, yeast forms associated with genera Blastomyces, Ajellomyces (Histoplasma), and Coccidioides were not present.
The finding of abundant fungal hyphae within a thoracic granulomatous mass is uncommon, but should be considered for a large mediastinal mass. Published agents for this type of lesion include Aspergillus spp. and Mucor spp., among others.2,4 In the present case, a fever was noted only on 1 occasion, and the only consistent blood work change was an elevation in serum globulins. It is unknown how long the mass had been present or its rate of growth, but it was not identified on earlier radiographs that identified the vertebral lesions.
It is unknown if the vertebral lesions and abdominal sensitivity were also due to fungus-associated inflammation and destruction in the vertebral column and abdominal organs because only the mediastinal mass was available for postmortem examination. A large intrathoracic mass, regardless of whether it is neoplastic or inflammatory, can be associated with bone lesions. However, the bone lesions of hypertrophic pulmonary osteopathy are characterized by organized periosteal new bone proliferation beginning in the distal extremities. 5 The clinical presentation of warm, swollen, painful metacarpal and metatarsal region bones and joints was not observed in the current case for which the clinically documented skeletal findings were limited to the vertebral column. Lesions of the cervical and lumbosacral spinal column in a large breed dog are more consistent with discospondylitis or osteomyelitis of the vertebrae due to hematogenous dissemination of a bacterium or fungus. 5 A variety of bacteria have been associated with vertebral osteomyelitis, including Staphylococcus aureus, Staphylococcus intermedius, Brucella canis, and Streptococcus spp. Similarly, a wide variety of fungi have been reported from vertebral osteomyelitis, including species of Aspergillus, Mucor, and Fusarium as well as Paecilomyces variotii. 5 A Brucella canis titer and bacterial cultures of the blood and urine were negative. Destructive inflammatory lesions affecting the vertebra can also be caused by reactions to migration of foreign bodies introduced through the body wall, respiratory, or digestive tracts. This scenario cannot be ruled out in the current case because it would explain the cranial mediastinal mass as a primary site of inoculation for the fungus from a foreign body. A mobile foreign body may have migrated to the cervical spine thereby initiating a secondary site of infection within the vertebral column. Migration of a foreign body and associated inflammation with infection through the paraspinal tissues, including entrapment of spinal nerve roots, is also consistent with the extreme pain response displayed by the patient when the flank was lightly touched. 8
In nature, I. tropicalis is a wood decay fungus that is normally detected as brown, resupinate (crust-like), fruiting bodies that emerge directly on the surfaces of hardwood logs. This fungus has been reported from fruiting body specimens collected in several different tropical countries (e.g., Costa Rica and Brazil) 9 and as clinical isolates from Texas. 14 Although this fungus is not frequently collected, it is likely that fungi in the I. tropicalis group are common in tropical to subtropical regions around the globe. Although there is no way to determine how I. tropicalis was able to enter the body of the patient, it is suspected that the fungus gained entry by a penetrating foreign body or through ingestion or inhalation of fungal spores. Reports of Inonotus species (and species from the related genus Phellinus) as pathogens of human beings are rare and thus far have involved immunosuppressed individuals.14,19
Identification of the wood decay fungus in a dog is evidence that the current lists of veterinary disease agents need to be updated. A report published in 2012 investigated the first documented case of infection by Oxyporus corticola from a dog with generalized lymphadenopathy. 11 Fungal hyphae exhibit very few morphological characters when they are viewed in tissue section, so diagnoses of fungal diseases in animals are often vague and potentially inaccurate. Work in the authors’ laboratory suggests that DNA sequencing of the ITS rDNA followed by direct comparison against GenBank can provide rapid, inexpensive, and accurate identification of fungi from diseased animal tissues.
Most fungi do not grow well in mammals because of a combination of factors, such as the difficulty of penetrating the skin, an elevated body temperature, and a successful immune response by the host. Thus, most of the serious fungal pathogens of mammals (e.g., Coccidioides immitis, Ajellomyces capsulata [syn. Histoplasma capsulatum], and Blastomyces dermatitidis) enter the body through the lungs, grow well at high temperatures, and have specialized mechanisms to avoid mammal immune systems.16,17 However, many fungal species are decomposers that can grow on a wide variety of carbon sources, including inside the nutrient-rich bodies of immunocompromised mammals.
Footnotes
Acknowledgements
The authors thank Mimi Barton for technical assistance.
a.
QIAamp DNA FFPE Tissue Kit, Qiagen GmbH, Hilden, Germany.
b.
SYBR Green I, Molecular Probes, Eugene, OR.
c.
Sequencher v.4.1 software, Gene Codes Inc., Ann Arbor, MI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
