Abstract
Two captive female Parma wallabies (Macropus parma) died after a history of flaccid paraplegia. On postmortem examination, granulomatous and suppurative osteomyelitis involving the left ischium and the lumbosacral region, with meningeal extension at the cauda equina, and caseonecrotic mastitis were the most significant changes. Multiple small nodules in the liver and spleen, and an enlargement of some lymph nodes with central caseous necrosis were also observed. Microscopically, a disseminated granulomatous inflammation with numerous multinucleate giant cells was seen. Numerous acid-fast bacilli were detected in macrophages, in multinucleated giant cells, and free in the central necrosis and suppurative exudate. After culture, polymerase chain reaction assays were carried out to detect the 65-kDa heat shock protein (Hsp65) and insertion sequences (IS)1245 and IS900. The causative agent was identified as Mycobacterium avium subsp. avium.
Mycobacterium avium is a major opportunistic pathogen of humans, especially in immunocompromised patients, 15 and of animals. This aerobic acid-fast bacillus is subdivided into 4 subspecies, namely M. avium subsp. hominissuis, M. avium subsp. silvaticum, M. avium subsp. paratuberculosis, and M. avium subsp. avium.23,37 The latter is considered a ubiquitous environmental saprophyte that generally does not spread from animal to animal. 2 While M. avium subsp. avium infection is the main cause of avian mycobacteriosis, it has been more sporadically described in farm,8,13 pet,9,25 exotic,7,35 or marine animals. 12 The current report documents 2 cases of M. avium infection in female Parma wallabies (Macropus parma), in which the lesions were mainly observed in the mammary gland and the vertebral bones. These animals were housed in the same enclosed parcel in a private domain. According to the owner, 1 or 2 wallabies, exclusively females, have died annually for several years. The infection seems to be enzootic. All the affected animals had signs of paraplegia prior to death.
Case 1 was a 3-year-old intact female Parma wallaby referred to the Pathology Diagnostic Laboratory of the Nantes-Atlantic College of Veterinary Medicine and Food Sciences (ONIRIS; LUNAM University, Site de la Chantrerie, France) for histological examination. The liver, spleen, kidney, lungs, and lymph nodes were collected and fixed in a 10% solution of phosphate buffered formalin, routinely processed, and embedded in paraffin wax. Sections (4 μm) were stained with hematoxylin and eosin, and selected sections were stained with Fite acid-fast stain for light microscopic examination. Microscopically, well-demarcated granulomas were present in the liver, lymph nodes, and spleen. The infiltrates were multifocal and marked for the first 2 tissues, focal and mild for the third one. Granulomas were composed of central necrosis with dystrophic calcification surrounded by epithelioid macrophages, Langhans-type multinucleated giant cells, lymphocytes, plasma cells, and fibroblasts. Fite acid-fast stains of the liver and the lymph node sections revealed myriads of acid-fast bacilli, mainly free in the central necrosis, as well as in the cytoplasm of the macrophages and multinucleated giant cells. In the liver, a marked bile duct hyperplasia was also observed. The lungs and the kidneys were unremarkable. Samples from the lymph nodes were sent to a commercial bacteriology laboratory. a After sequencing, a portion of the 16S ribosomal DNA, the resulting amplicon sequence (a 500–base pair fragment), was compared with sequences in a commercial database. b The obtained sequences were shared among M. avium subsp. avium and M. avium subsp. paratuberculosis. Given the lack of both clinical signs (e.g., chronic diarrhea) and microscopic intestinal lesions (e.g., diffuse granulomatous enteritis) related to Johne’s disease, 29 M. avium subsp. avium was considered as the most likely cause.
Case 2 was an adult intact female Parma wallaby with a 2-week history of hindlimb paralysis. Because of the poor prognosis, the wallaby was euthanized 3 weeks after the death of case 1. A complete autopsy was performed at the Department of Pathology of ONIRIS. Grossly, longitudinal sections of the axial skeleton showed a multifocal to coalescing chronic granulomatous and suppurative osteomyelitis, involving the lumbosacral region. A similar lesion was observed in the left ischium bone (Fig. 1). An abundant creamy yellow purulent material, lying against the dorsal aspect of the cauda equina, was observed. The liver and spleen had several white small nodular lesions, 1–4 mm in diameter, randomly distributed throughout the parenchyma. The tracheobronchic, pancreaticoduodenal, and retromammary lymph nodes were markedly enlarged with central caseous necrosis visible in the cut section. Severe diffuse caseonecrotic mastitis was present (Fig. 2). No significant lesion was found in the other organs.
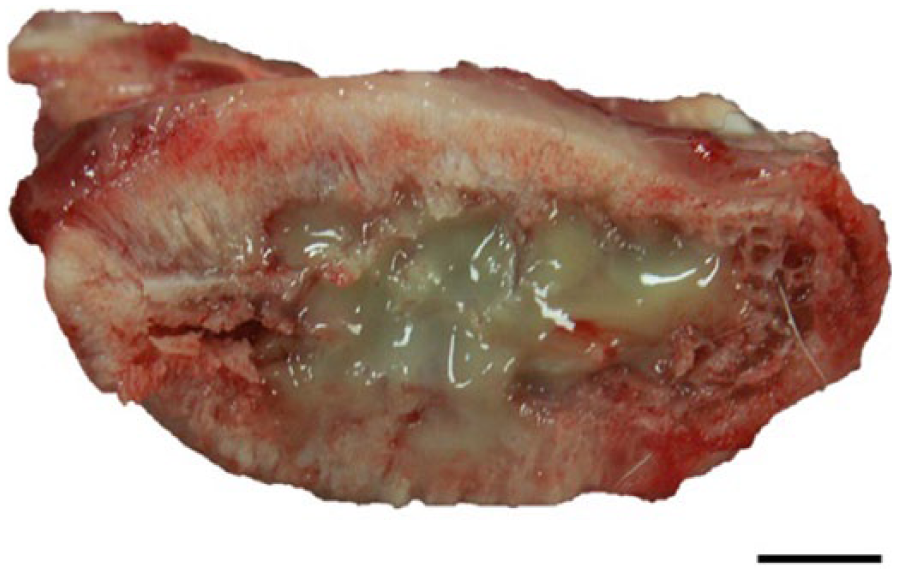
Left ischium; severe diffuse caseonecrotic and suppurative mycobacterial osteomyelitis. Bar = 5 mm.
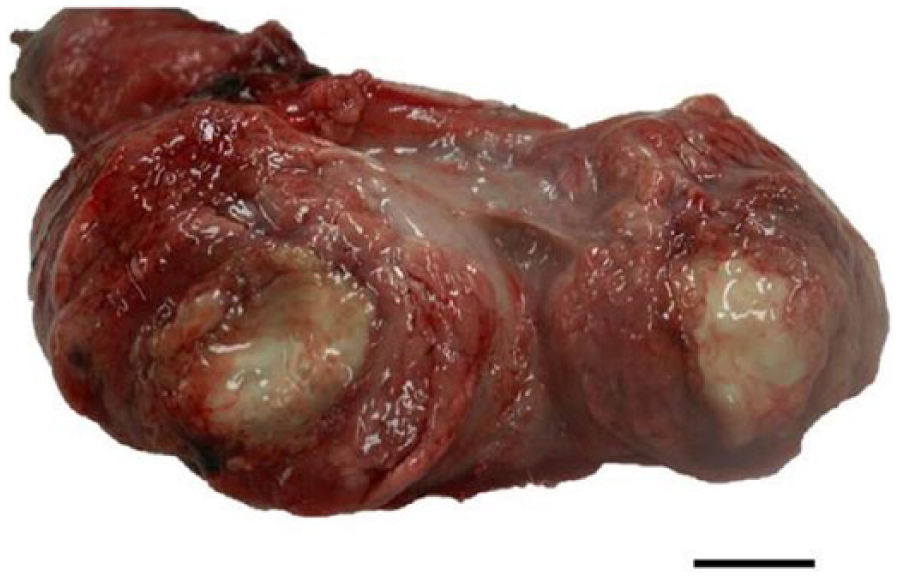
Mammary gland; severe diffuse caseonecrotic and suppurative mycobacterial mastitis. Bar = 10 mm.
Representative tissue sections from brain, spinal cord, lung, liver, spleen, kidney, adrenal glands, lymph nodes, stomach, intestine, pancreas, mammary gland, and bones were processed as previously mentioned. Samples of the most affected organs (i.e., liver, vertebrae, and mammary gland) were sent to a commercial bacteriology laboratory a for culture, using standard techniques. Blood agar and chocolate agar plates were examined after 24–48-hr incubation at 37°C. For assessment of Mycobacterium spp., the protocol NF U 47-104 was used. Briefly, ~2 g of pooled samples was crushed and decontaminated with 4% H2SO4 for 10 min. The suspension was inoculated on Löwenstein–Jensen and Coletsos slants at 37 ± 2°C. If no contamination was observed after 7 days, growth was monitored every 2 weeks for 3 months. The Mycobacterium isolates obtained by culture were evaluated by a polymerase chain reaction (PCR) assay for 65-kDa heat shock protein (Hsp65) and insertion sequences (IS)1245 and IS900. The tests were performed at the French Agency for Food, Environmental and Occupational Health & Safety (ANSES) Animal Health Laboratory (Maisons-Alfort, France).
Microscopic examination revealed multifocal to coalescing, variably encapsulated granulomas and diffuse neutrophil-rich exudation in between the granulomatous foci. The infiltrate was severe in the bones, mammary gland, and lymph nodes; moderate in the liver and meninges of the distal spinal cord; and minimal in the spleen (Fig. 3). In the cauda equina, rare lymphocytic and plasmacytic perivascular cuffing, 1–2 cell thick, at the Virchow–Robin spaces were observed, as well as some spheroids. A marked osteoclastic osteolysis was visible in the vertebrae and in the left ischium. Numerous acid-fast bacilli were present in the lesions (Fig. 4). Bacterial culture of the mammary gland yielded Citrobacter freundii and Aeromonas hydrophila, while the lumbosacral vertebrae did not yield any rapid-growing bacteria. PCR revealed the presence of Hsp65, IS1245, and IS900 in the pooled sample containing liver, mammary gland, and lumbosacral vertebrae, confirming the presence of M. avium subsp. avium.
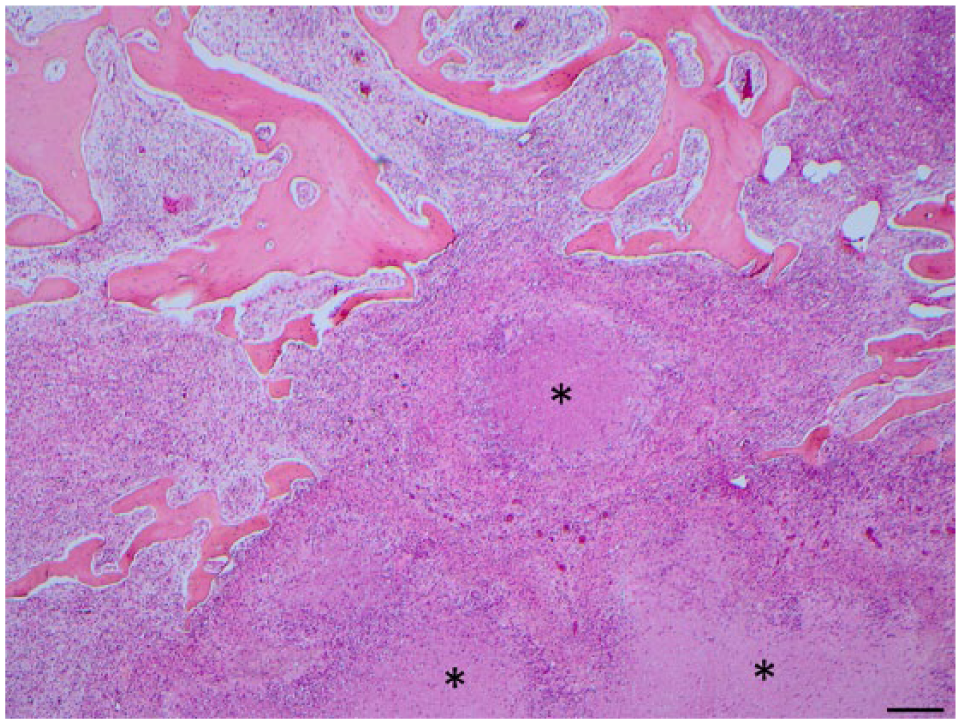
Section of lumbosacral vertebrae; severe multifocal to coalescing granulomatous osteomyelitis with multifocal central necrosis (*). Hematoxylin and eosin. Bar = 200 nm.
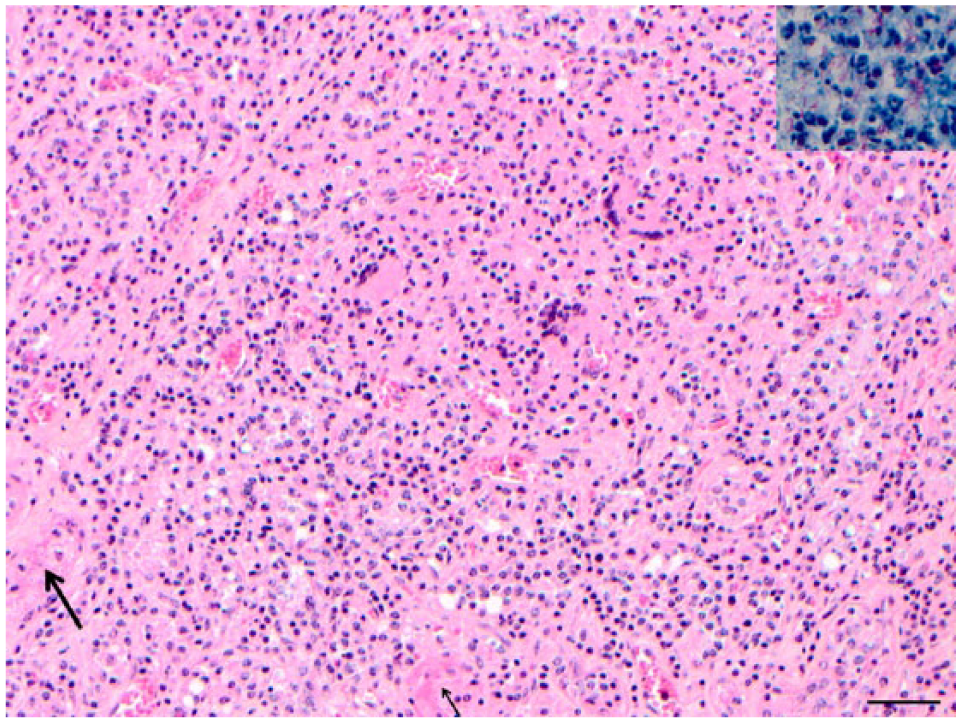
Section of the mammary gland; diffuse granulomatous mastitis with numerous macrophages, multinucleate giant cells, and some polynuclear leucocytes, leaving few remnants of identifiable glandular tissue (arrow). Hematoxylin and eosin. Bar = 200 nm. Inset: numerous acid-fast bacilli, free or in the cytoplasm of macrophages and giant cells. Fite stain.
Based on histological examination, Fite stain, culture prior to PCR, and 16S rDNA sequencing, both cases were diagnosed as disseminated mycobacteriosis caused by M. avium. Marsupials, especially from captive populations, are more susceptible to mycobacterial infection than eutherian mammals. 5 Moreover, a particular affinity of M. avium for the skeletal system has been recognized in marsupials. Mycobacterial osteomyelitis has been thus described in 4 Bennett’s wallabies (Protemnodon rufogrisea frutica), 14 1 tree kangaroo (Dendrolagus matschiei), 19 1 Parma wallaby (Macropus parma), and 1 long-nosed rat kangaroo (or long-nosed potoroo; Potorous tridactylus), 21 and 1 brush-tailed bettong (or woylie; Bettongia penicillata). 31 The clinical signs are variable, ranging from weakness and lethargy to paraplegia. In our report, the disorders of locomotion (i.e., posterior paresis), present in all affected animals, can be easily explained by both the severe granulomatous and suppurative vertebral osteomyelitis found at autopsy and the granulomatous meningitis at the cauda equina. Considering the localization of the bone lesions (i.e., the lumbosacral region and the left ischium), a direct extension of the infection in the medullary canal from the vertebral osteomyelitis is highly suspected. Spinal cord involvement by atypical mycobacteriosis is considered rare in domestic and wildlife animals.20,28 In marsupials, only 2 reports have been recorded.34,38 In both cases, no concomitant osteomyelitis was reported.
In humans, nontuberculous mycobacteria are an unusual cause of vertebral infection. 33 It is commonly related to trauma, 6 surgical treatment, or a compromised immune system (e.g., a low number of CD4+ T lymphocytes in AIDS patients or inherited gene defects related to the interleukin-12 interferon-γ tumor necrosis factor-α axis).4,10 However, multiple osteomyelitis in an immunocompetent individual caused by M. avium has been described. 27 Concomitant involvement of the central nervous system is also scarce. Interestingly, bacteria of M. avium complex are the most commonly isolated species in nontuberculous mycobacterial osteomyelitis 30 and meningitis 11 of human beings. Furthermore, the spine is the most common localization in adult hematogenous osteomyelitis, including those caused by M. avium complex. 17 The high frequency of vertebral osteomyelitis during bacteremia is consistent with our observation.
In the present case, an unexpected caseonecrotic mastitis associated with a retromammary granulomatous lymphadenitis was also observed in the wallaby in which mammary gland sampling was done. Taking into account the severity of this lesion and the gender predisposition (only females were reported to be affected in the flock), a mammary source was considered the likely route of infection and dissemination. To our knowledge, there are no reports in marsupials of the involvement of the mammary gland as a portal of entry for a disseminated mycobacteriosis caused by M. avium subsp. avium. This bacterium is known for widespread environmental distribution, the main sources being water and soil. 25 It might remain viable for several months. 16 No specific infectious source has been identified in the private domain where wallabies live. However, they share the same parcel with numerous wild captive birds. Some of these birds might be asymptomatic carriers, and fecal contamination of the soil or water should be considered.
Mycobacterial mastitis has been reported in a Chihuahua dog with concurrent cutaneous lymphoma 26 and in 1 alpaca affected by a disseminated Mycobacterium bovis infection. 32 Previous clinical mastitis was considered to be a predisposing factor in a dairy herd without systemic involvement. 36 Culture of the mammary gland in case 2 revealed C. freundii and A. hydrophila. Both are Gram-negative bacteria in the Enterobacteriaceae family and are usually considered opportunistic pathogens. In our case, a significant pathogenic role of these bacteria is very unlikely, as they have been rarely isolated from clinical bovine mastitis,1,18 and have never been reported in marsupial mastitis, to the authors’ knowledge. In human beings, the incidence of breast tuberculosis accounts for <0.1% of all breast lesions in Western countries, but it reaches 3–4% in tuberculosis-endemic countries. 22 Extension to the bone is rare but, in 2014, a sternum osteomyelitis secondary to tuberculous mastitis, via pectoral fascia invasion, has been described in a 40-year-old female. 3 Thus, the mammary gland should always be investigated as a potential primary source of infection in a case of disseminated mycobacteriosis.
Marsupials are known to be particularly susceptible to mycobacteriosis. A high prevalence of M. avium complex infection has been reported in a retrovirus-free breeding colony of tree kangaroos (Dendrolagus matschiei) as a result of a lowered cell-mediated immune reactivity. 24 A dysfunction of the T helper cell population may allow the expression of M. avium as primary rather than opportunistic pathogens in tree kangaroos. The T cell–mediated immunity in both wallabies in the current study was not explored. Neither underlying disease nor cutaneous traumatic wound was previously reported or found during autopsy and histological examination. The wallabies had never received any treatment. The main predisposing factor for M. avium infection in our study was thus considered to be the overpopulation of untreated captive birds in the domain, potentially associated with an environmental contamination.
In conclusion, the cases described in the present report are of special interest because of the suspected enzootic character of the M. avium infection, the clinical presentation manifested by paralysis with severe vertebral osteomyelitis and extension to the spinal cord, and the likely mammary source of infection. It is concluded that captive marsupials kept outdoors have a risk of M. avium infections, particularly in animals displaying paralysis as the main clinical manifestation. Taking into account that M. avium subsp. avium infection is the most common cause of disseminated bacterial infection among AIDS virus–infected individuals in developed countries, the results reinforce that care should be taken by owners and people manipulating wallabies.
Footnotes
Acknowledgements
We extend our sincere thanks to Mrs. F. Cappelier, to the histology laboratory staff at the Nantes-Atlantic College of Veterinary Medicine and Food Sciences (ONIRIS) for the histological processing of the samples (Mrs. C. Guereaud and Mr. B. Fernandez), to Drs. Cheval and Boschiroli for the microbiological data, and to Dr. De Bisschop for the clinical data.
Authors’ contributions
O Albaric contributed to conception of the study. C Robveille and J Abadie contributed to conception and design of the study. C Robveille drafted the manuscript. O Albaric, N Gaide, and J Abadie critically revised the manuscript. All authors contributed to acquisition, analysis, and interpretation of data; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Inovalys Nantes, Nantes, France.
b.
MicroSeq 500 16S rDNA Bacterial Libraries (Applied Biosystems, version 2013), Inovalys Nantes, Nantes, France.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
