Abstract
Granulomatous colitis in dogs can be associated with infection of the colonic mucosa by invasive strains of Escherichia coli. To date, fluorescence in situ hybridization (FISH) is the gold-standard method to assess intramucosal and intracellular bacterial invasion. However, FISH requires expensive fluorescence microscopy equipment and is therefore not widely available. We investigated the use of immunohistochemistry (IHC) as an alternative method to detect invasive E. coli in dogs with granulomatous colitis. Archived paraffin-embedded blocks were selected from 26 dogs with colitis, in which FISH had been performed by an outside laboratory. Using a polyclonal antibody, IHC for E. coli was performed on sections cut from the same blocks, and the presence of invasive E. coli was recorded. All 11 specimens in which FISH had detected E. coli were also positive on IHC, with strong immunolabeling in the cytoplasm of macrophages and extracellularly in the lamina propria; all 15 specimens that were negative for invasive bacteria on FISH were also negative on IHC. We found that IHC is a sensitive technique for the detection of invasive E. coli in dogs with granulomatous colitis.
Escherichia coli–associated granulomatous colitis (GC), also known as histiocytic ulcerative colitis, was first described in Boxer dogs in 1965. 19 Since then, French Bulldogs have also been reported to develop the disease, and a few other single-case reports have described the condition in a Staffordshire Terrier, Doberman Pinscher, Rottweiler, Mastiff, Beagle, and Alaskan Malamute.2,11,17 The condition has also been reported, but is considered rare, in cats. 9
Clinically, dogs with GC have clinical signs consistent with large bowel diarrhea, such as increased frequency of defecation, mucoid stools, hematochezia, and tenesmus.11,16 Weight loss and hypoalbuminemia are often reported in dogs with severe forms of GC.4,16 Macroscopic findings include irregular thickening and ulceration of the colonic mucosa. 16 Microscopic lesions are characterized by infiltration of the lamina propria and submucosa by macrophages with abundant eosinophilic cytoplasm, which is positive on periodic acid–Schiff (PAS) stain, as well as by erosion or ulceration of the colonic epithelium.4,16
Adherent-invasive strains of E. coli (AIEC) were identified as the cause of GC in 2006. 16 Eradication of invasive E. coli with fluoroquinolone antimicrobials is correlated with clinical remission of GC.4,7,11,12 Resistance of E. coli to antimicrobials is significantly associated with a poor clinical outcome; antimicrobial susceptibility profiling guided treatment is associated with a positive long-term outcome of GC. 10
Fluorescence in situ hybridization (FISH) is a molecular approach used to detect and visualize a specific DNA or RNA sequence with fluorescence microscopy.8,13 FISH is considered the gold-standard method to assess colonic tissue samples for intramucosal and/or intracellular E. coli invasion in dogs. 16 However, FISH requires expensive fluorescence microscopy equipment and on-site interpretation by professionals with specific training, and therefore, it is not widely available as a diagnostic tool. 13 Immunohistochemistry (IHC) is an in situ technique that allows the detection of antigens in tissue sections through specific antibodies. 14 Whole-slide images can be shared digitally allowing remote interpretation. IHC has been used widely for the diagnosis of infectious diseases in fixed tissues, including a case of GC in a dog. 2
Our objective was to evaluate an immunoperoxidase IHC assay using a polyclonal anti–E. coli antibody as an alternative method to detect enteroinvasive E. coli in formalin-fixed, paraffin-embedded (FFPE) colonic samples from dogs with GC.
We used archived FFPE tissue blocks from endoscopic biopsy samples submitted to the histopathology service of the Gastrointestinal Laboratory at Texas A&M University (College Station, TX, USA) for diagnostic purposes. The records were reviewed, and canine cases with ≥5 colonic endoscopic biopsies in which FISH had been performed at an external laboratory (Simpson Laboratory, Cornell University, Ithaca, NY, USA) were selected and divided into a control and a GC group. At the external laboratory, samples were evaluated and interpreted by a veterinary professional with expertise in FISH. The control group consisted of dogs with colitis, negative PAS stain, and a negative FISH report for a universal eubacterial probe (EUB 338; ACTCCTACGGGAGCAGC). 16 The GC group consisted of dogs with granulomatous colitis with macrophages positive on PAS stain and positive FISH for both the EUB 338 and E. coli/Shigella probe (E. coli 16S rRNA, GCAAAGGTATTAACTTTACTCCC). 16
Stains (H&E, PAS) and IHC and FISH for E. coli were performed on colonic sections of the blocks that had been submitted for FISH and evaluated by a board-certified anatomic veterinary pathologist blinded to the FISH results. Slides stained with H&E were digitalized for evaluation and scoring of lesions according to the grading system (0 = normal, 1 = mild lesions, 2 = moderate lesions, 3 = severe lesions) developed by the World Small Animal Veterinary Association Gastrointestinal Standardization Group.1,5 Scores were assigned for each parameter (i.e., surface epithelial injury, crypt dilation, fibrosis, goblet cell count, intraepithelial lymphocytes, and lamina propria infiltrates of lymphocytes and/or plasma cells, eosinophils, neutrophils, and/or macrophages).1,5
For IHC, 4-µm thick sections were mounted on positively charged slides and dried overnight at 40°C. Before the assay, mounted sections were routinely deparaffinized and rehydrated. Heat-induced antigen retrieval was performed in a pressure cooker (Retriever for antigen unmasking; Electron Microscopy Sciences) by heating the slides to 121°C for 20 min in 1× citrate buffer, pH 4.5 (R-Buffer C 10×; Electron Microscopy Sciences) followed by endogenous peroxidase blocking with 3% hydrogen peroxide for 10 min and UV protein block buffer (UltraVision protein block; Richard-Allan Scientific) for 15 min. Tissue sections were then incubated for 1 h at room temperature with a polyclonal anti–E. coli antibody against O and K antigenic serotypes (Anti–E. coli, 1001; ViroStat) produced in rabbits and diluted at 1:200 with antibody diluent (Antibody diluent reagent solution; Life Technologies).
Intestinal samples from a piglet with colibacillosis confirmed by bacterial culture and histopathology were used as positive controls. For negative controls, the primary antibody was replaced with rabbit antiserum (Rabbit IgG isotype control; Invitrogen) diluted to the same concentration as the E. coli antibody in consecutive sections from each tissue. After incubation, slides were washed 3 times for 5 min each in 1× Tris-buffered saline with Tween 20 (TBST), and signal detection was performed using a horseradish peroxidase–linked polymer (UltraVision LP detection system; Epredia), followed by incubation for 2 min with a red peroxidase substrate chromogen (ImmPACT NovaRED; Vector). Slides were counterstained with Mayer hematoxylin, dehydrated, and mounted with a xylene-based non-aqueous mounting medium.
Slides were scanned at 40× with a slide scanner (NanoZoomer 2.0 HT; Hamamatsu Photonics) and viewed with NDP.view software (Hamamatsu Photonics) by a board-certified anatomic veterinary pathologist blinded to previous FISH results. Samples with clusters of immunolabeling in the cytoplasm of macrophages in the lamina propria were considered positive for enteroinvasive E. coli infection. Samples with immunolabeled bacteria on the mucosal surface, in the lumen of the crypts, or individualized extracellular bacteria in the lamina propria interpreted as sectioning carryover were considered negative. Areas with tissue folding or crushing artifacts were not considered for evaluation.
Cross-reactivity of the antibody was assessed for Helicobacter spp., a genus of commensal bacteria frequently found in the canine colonic crypts, 15 as well as the enteropathogenic bacteria Clostridium perfringens, Campylobacter sp., and Salmonella enterica. Samples included stomach from a dog with Helicobacter spp. infection confirmed by specific FISH, colon from a primate with campylobacteriosis, jejunum from a dog with clostridiosis, colon from a horse with salmonellosis, and colon and mesenteric lymph node from a pig with salmonellosis. The case of C. perfringens infection was confirmed by histopathology, positive culture, and detection of C. perfringens enterotoxin in intestinal contents. The diagnosis of campylobacteriosis and salmonellosis were based on histopathology findings and positive bacterial culture.
FISH for E. coli was repeated in-house following the same protocol used by the external laboratory, 16 utilizing appropriate negative and positive controls. Samples with clusters of fluorescent labeling in the lamina propria were considered positive; samples with labeling restricted to the mucosal surface or lumen of crypts were considered negative.
The control group was comprised of 15 dogs with colitis (4 intact males, 4 castrated males, 1 intact female, 6 spayed females; median age: 5 y, age range: 1–12 y). The most common breeds were Schnauzers (n = 2) and Boston Terriers (n = 2). The inflammatory infiltrate in the colon was neutrophilic (n = 2), lymphoplasmacytic (n = 2), or mixed (n = 11), which variably included macrophages, eosinophils, neutrophils, lymphocytes, and plasma cells. The median cumulative histologic score was 6 (range: 1–12).
The GC group included 11 dogs (4 intact males, 4 castrated males, 2 intact females, 1 spayed female; median age: 1 y, age range: 0.5–5 y). Breeds included French Bulldog (n = 6), Boxer (n = 4), and Rottweiler (n = 1). Histologic findings in all cases consisted of histiocytic inflammation in the lamina propria and/or submucosa (Fig. 1A) with a variable infiltrate of neutrophils, plasma cells, and lymphocytes, and erosion of the superficial epithelium. Infiltrating macrophages had abundant eosinophilic cytoplasm that was PAS positive in all cases (Fig. 1B). The median cumulative histologic score was 8 (range: 4–12).
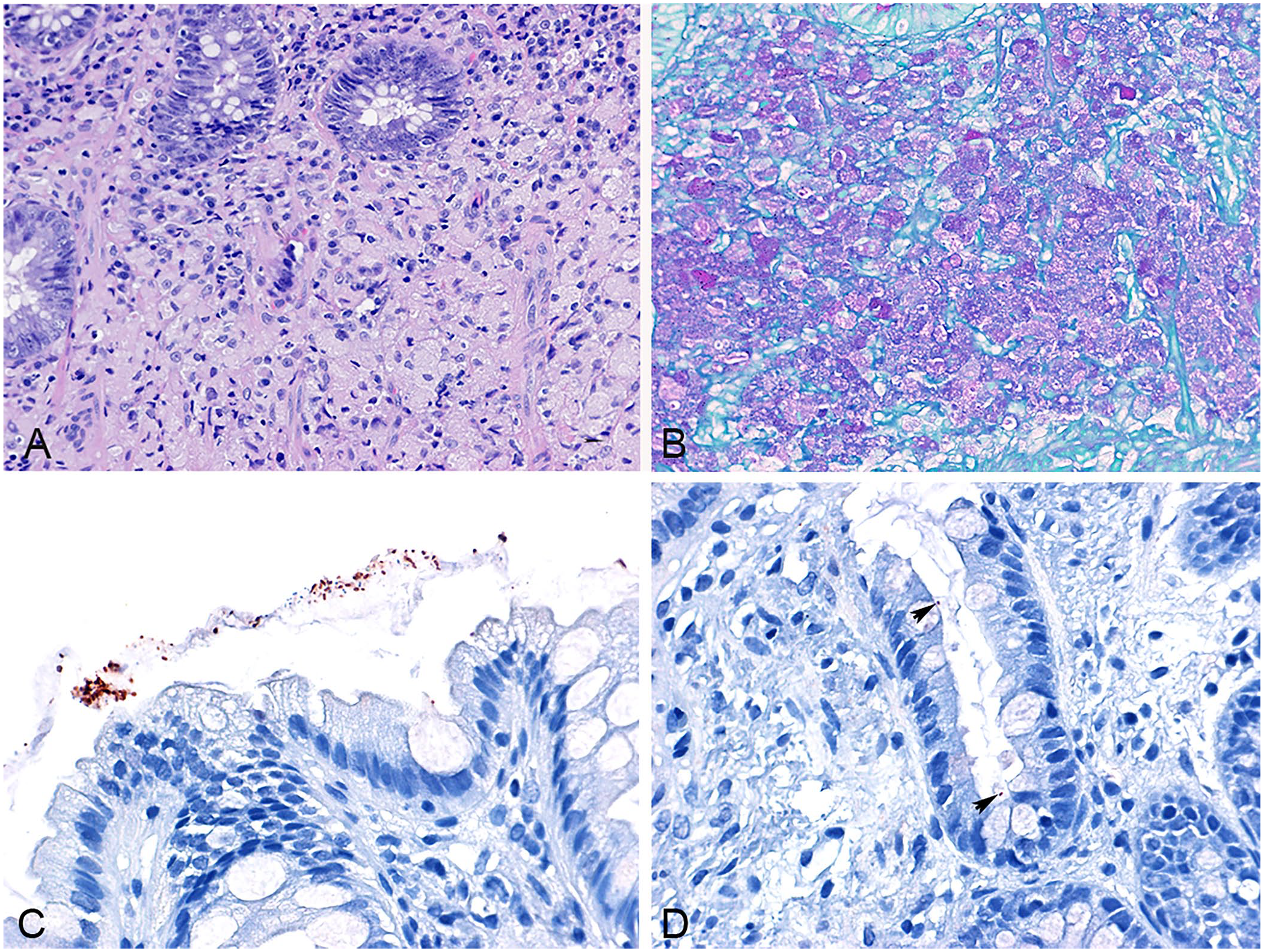
Canine Escherichia coli–associated granulomatous colitis (GC;
Immunolabeling for E. coli was not seen in the lamina propria or intracellularly in any of the 15 control cases, which were negative for invasive bacteria on FISH using the EUB probe at the external laboratory and in-house FISH for E. coli. Occasional bacteria immunolabeled as E. coli were seen on the mucosal surface with the intestinal contents (Fig. 1C) and in the lumen of the crypts (Fig. 1D), similar to the FISH technique.
All 11 GC cases in which FISH had detected invasive E. coli were positive on IHC for E. coli (Fig. 2A). Strong red clusters of immunolabeling were seen in the cytoplasm of macrophages (Fig. 2B) located in the lamina propria and/or submucosa along with extracellular immunolabeling in the lamina propria (Fig. 2C). As expected, the positive control for E. coli demonstrated strong immunolabeling. Our in-house FISH for E. coli detected invasive bacteria consistently in all 11 cases of GC (Fig. 2D).
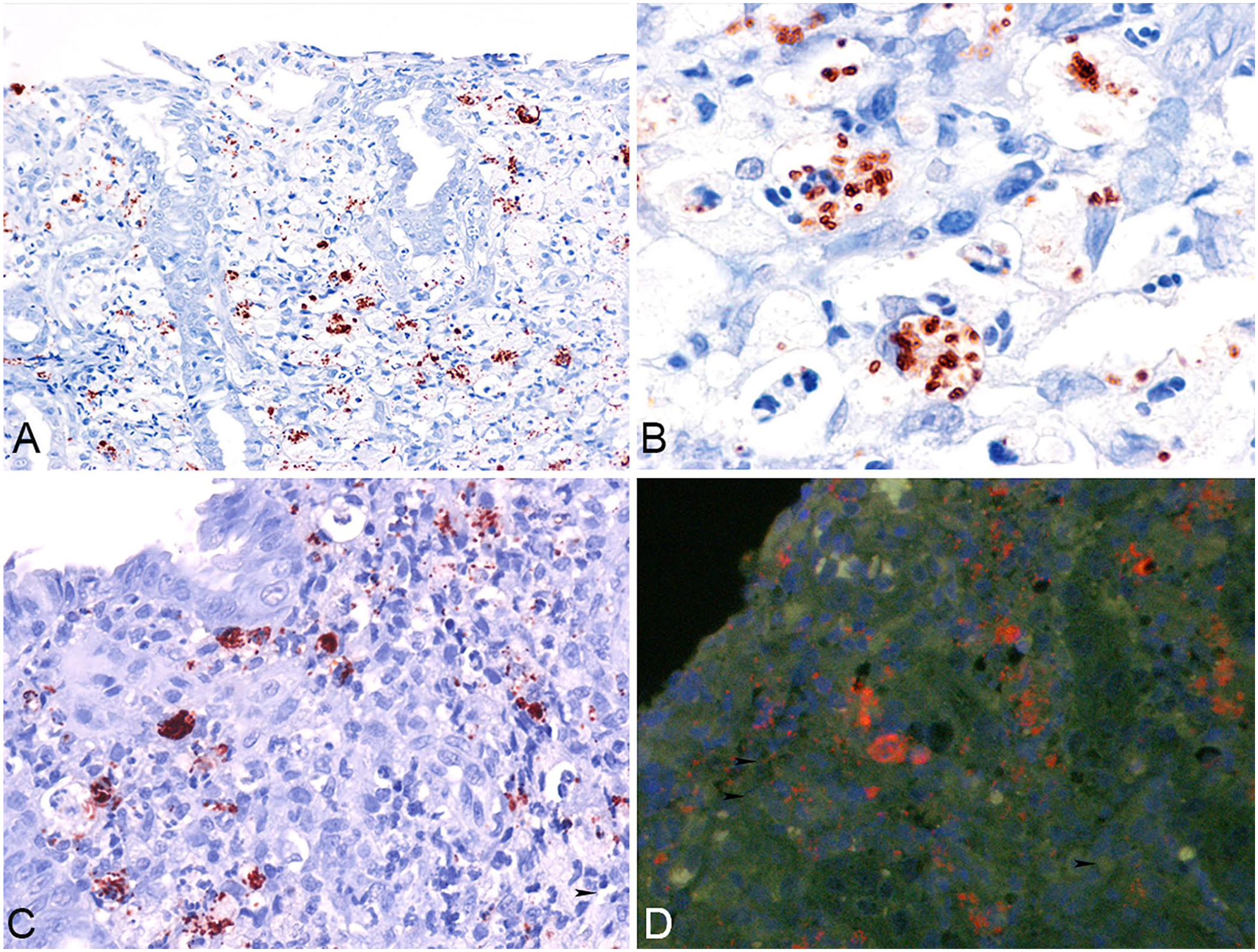
Canine Escherichia coli–associated granulomatous colitis.
The primary antibody for E. coli did not cross-react with Helicobacter spp. or C. perfringens. However, cross-reactivity occurred with Campylobacter sp. and S. enterica, with labeled bacteria located extracellularly in the lamina propria and on the mucosal surface of colonic lesions. In the case of mesenteric lymphadenitis in salmonellosis cases, labeled bacteria were seen extracellularly and intrahistiocytic within the sinuses.
Extraintestinal E. coli strains able to adhere to and invade colonic enterocytes, identified as AIEC, cause GC in predisposed dogs. 16 In Boxer dogs and French Bulldogs with GC, genetic susceptibility is linked to a region encoding the CD48/SLAM family of genes on chromosome 38, which has been associated with human inflammatory bowel disease. 6 The diagnosis of GC typically relies on characteristic histopathologic findings of PAS-positive macrophages in colonic biopsies, and FISH, to date, is considered the gold-standard method to demonstrate invasive E. coli within the mucosa. 16 We were able to detect E. coli antigen with IHC in all cases that were positive on FISH. In a reported case in which FISH was not available, the same polyclonal antibody was used to label invasive E. coli in a dog with GC caused by a strain within the EcoR group B2. 2
FISH is considered a specific and sensitive technique to detect invasive E. coli, and probes are known to cross-react with Shigella spp. 16 Disadvantages of FISH include the requirements of a costly fluorescence microscope and an on-site pathologist to read and interpret the slides immediately after the assay if fluorescence image slide scanners are not available. The fluorophore signals fade over time and usually exhibit low contrast for differentiation from tissue autofluorescence. An alternative to FISH is chromogenic in situ hybridization (CISH) in which the same probes used for FISH are applied, with the advantage of a stable signal that can be read with a regular light microscope. 20 However, sensitive CISH usually requires proprietary technologies with expensive reagents and time-consuming protocols.
Our IHC protocol allowed clear localization of strongly labeled bacterial antigen within the cytoplasm of macrophages in sections counterstained with hematoxylin. Other advantages of IHC include the ability to scan and store the slides for longer periods of time with preservation of the immunolabeling, reasonable reagent costs, assay automation, and accessible whole-slide scanning for remote interpretation by a pathologist and second-opinion consultation. Invasive bacteria were not identified in the control group of idiopathic colitis that had similar lesion severity assessed by the histologic scores.
A significant limitation of the polyclonal antibody that we used was cross-reaction with Campylobacter spp. and S. enterica. However, campylobacteriosis and salmonellosis are uncommon causes of enterocolitis in dogs and are not associated with histiocytic or granulomatous inflammation in the colon.3,18 Therefore, our IHC assay needs to be interpreted in the context of characteristic granulomatous inflammation with PAS-positive macrophages, and it is not recommended for other manifestations of enteric colibacillosis in dogs. It is important to note that most strains of E. coli are commensal bacteria that colonize the intestine of healthy humans and animals, 16 and mere isolation of E. coli from the feces or identification of the bacteria in the intestinal lumen are insufficient to make a diagnosis.
Footnotes
Acknowledgements
We thank Dr. Randi Gold for her technical assistance with FISH and Kelly Mallet for her support with histopathology. We also thank Dr. Carolyn Hodo for providing a sample from a case of campylobacteriosis.
Declaration of conflicting interests
Patricia Eri Ishii, Paula R. Giaretta, Jonathan A. Lidbury, Jan S. Suchodolski, and Joerg M. Steiner are employed by the Gastrointestinal Laboratory at Texas A&M University, which offers laboratory tests, including gastrointestinal histopathology services, on a fee-for-service basis.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
