Abstract
Rhodococcus equi infection in horses is common and is characterized by pyogranulomatous pneumonia and ulcerative enterocolitis. R. equi clinical disease in cattle, however, is rare and typically manifests as granulomatous lymphadenitis discovered in the abattoir. A 19-mo-old female Santa Gertrudis had a history of intermittent inappetence and weight loss for a 3-mo period before euthanasia. Gross and histologic examination revealed severe, chronic, ulcerative, and granulomatous inflammation in the tongue, pharynx, and small intestine. Also, the heifer had severe, granulomatous pharyngeal and mesenteric lymphadenitis. Bacterial cultures from the ileum, tongue, and liver yielded numerous-to-moderate numbers of R. equi. PCR analysis of the isolate detected the linear virulence plasmid vapN, which is often identified in bovine isolates (traA- and vapN-positive). The bacteria also lack the circular plasmids vapA and vapB that are associated with virulence in horses and swine, respectively. We report herein an atypical and unusual clinical presentation of R. equi infection in cattle, which has zoonotic potential.
Keywords
Rhodococcus equi is a ubiquitous, gram-positive, facultative intracellular nocardioform coccobacillus that has been associated with disease in a wide range of hosts.5,8,10,18 The pathogenicity of R. equi is attributed to a wide range of virulence factors that include cholesterol oxidase enzyme, cell wall mycolic acids, capsular polysaccharide, and virulence-associated proteins (vap).3,5,7 To date, 3 R. equi vap plasmids have been identified: the circular vapA and vapB plasmids, and the linear vapN plasmid.5,16,17,19 The vap gene products facilitate R. equi survival and replication within host macrophages.5,7,15,16 The circular plasmids vapA and vapB are associated with virulence in horses and swine, respectively. VapN is an inverton-like, linear plasmid that has been associated with cattle5,16 and goats. 12
In foals 1–5 mo old, R. equi causes severe pyogranulomatous pneumonia, and ulcerative and granulomatous enterocolitis. 18 The disease accounts for considerable economic losses in the equine industry worldwide. 19 R. equi infection in humans, mainly in acquired immunodeficiency syndrome patients, causes cavitating pulmonary abscesses, bacteremia, and lymphadenitis.8,9,13,17
Clinical disease associated with R. equi in cattle is rare and often manifests as mesenteric lymphadenitis discovered in the abattoir.2,20 R. equi has been cultured from rare cases of bronchopneumonia, mastitis, metritis, ulcerative lymphangitis, and septic arthritis in cattle, but has not been characterized by molecular analysis for virulence plasmids. 8 A case of disseminated R. equi infection with no involvement of the intestinal tract in a Japanese heifer has been reported. 6 We describe herein the gross and histologic findings of an unusual presentation of granulomatous and ulcerative enteritis caused by R. equi infection in a heifer, and provide detailed molecular analysis of the isolated R. equi.
A 19-mo-old Santa Gertrudis heifer was presented to a veterinary clinic initially because of intermittent inappetence and weight loss for a 3-mo period. Clinical examination detected ulcers on the tongue, for which she was treated with antibiotics and anti-inflammatory drugs for several days. The treatment regimen included oxytetracycline, tildipirosin, penicillin G, and steroidal and nonsteroidal anti-inflammatory and analgesic drugs. Periodic improvement and reduction in tongue swelling was observed initially. Two weeks later, however, the animal became completely inappetent for 10 consecutive days. Eventually, the heifer became weak and recumbent, and was euthanized.
On autopsy, the heifer was minimally autolyzed and weighed 235 kg. The heifer had pale mucous membranes, and was markedly emaciated with generalized muscle atrophy and increased prominence of bony processes. Two symmetrical, oval ulcers were located on the dorsolateral surfaces of the torus linguae of the tongue. The ulcers were well demarcated with yellow granular surfaces and measured 4 × 4.5 cm (left) and 4.5 × 5 cm (right; Fig. 1A). Ulcers of similar appearance were located in the nasopharynx and right lateral oropharynx. The pharyngeal lymph nodes were enlarged and contained pasty tan material. The distal ileum was markedly thickened and firm; the mucosal surface had variably sized, coalescing crateriform ulcers. The ulcers were more severe and numerous in the ileum but they were also distributed throughout the jejunum (Fig. 1B). Intestinal contents were bloody. Mesenteric lymph nodes were enlarged and contained pasty tan material.
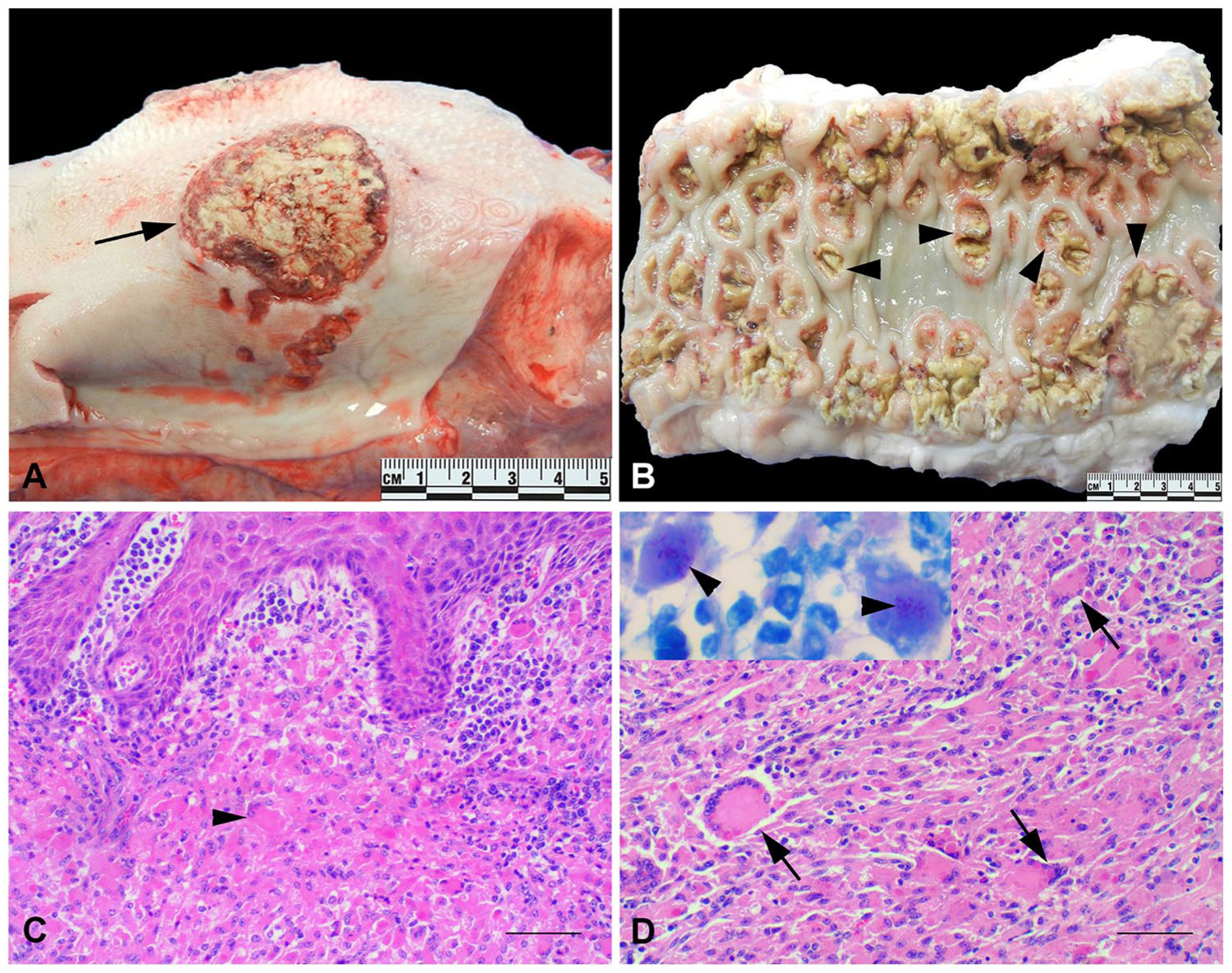
Ulcerative and granulomatous glossitis and enteritis caused by Rhodococcus equi in a heifer.
Formalin-fixed, paraffin-embedded sections were stained with hematoxylin and eosin (H&E) stain. The mucosa of the tongue was multifocally ulcerated, and the submucosa was infiltrated by large numbers of epithelioid macrophages and multinucleate giant cells (Fig. 1C). The mucosa of the ileum was multifocally ulcerated, and the underlying submucosa was markedly expanded by a dense population of epithelioid macrophages and large numbers of multinucleate giant cells (Fig. 1D). Many macrophages and giant cells contained phagocytosed bacteria. Small numbers of lymphocytes were distributed randomly throughout and formed small nests and aggregates. The centers of some of the larger inflammatory infiltrates were necrotic and contained mineralized foci. The architecture of the lymph nodes was disrupted by sheets of epithelioid macrophages with central necrosis and mineralized foci. Additional changes included marked reactive lymphoid hyperplasia and marked medullary histiocytic infiltration. Sections from the ileum and tongue were stained with Gram stain and Fite acid-fast stain; the bacteria seen on H&E within cytoplasmic vacuoles of macrophages were gram-positive and were partially acid fast (Fig. 1D, insert).
Tissue samples from the tongue, ileum, and lymph nodes were cultured aerobically using blood agar, and were incubated at 37°C. After 48 h, smooth, glistening, 2–4 mm diameter, non-hemolytic, mucoid colonies developed, which became red-orange to salmon-pink after 5–6 d of incubation.
A matrix-assisted laser desorption/ionization time-of-flight mass spectrometer (Microflex LT MALDI-TOF MS; Bruker Daltonics, Billerica, MA) was used to achieve genus- and species-level identification on the bacterial isolate. The MS was calibrated prior to sample testing using the bacterial test standard (Bruker Daltonics), as per the manufacturer’s recommendations. The colony morphologic features and MALDI-TOF MS analysis identified the isolates from the tongue, ileum, and lymph nodes as R. equi.
The isolated R. equi was submitted for molecular analysis to further characterize the virulence plasmids. The real-time PCR (rtPCR) assay targeted choE, a chromosomal gene that is highly conserved in R. equi, 3 and revealed that the isolated R. equi was choE+. Also, Mycobacterium avium subspecies paratuberculosis was not detected by rtPCR.
Detailed conventional PCR analysis for virulence plasmids showed that the bacteria were traA+/vapAB–/vapN+ (Fig. 2). Briefly, the isolated R. equi was cultured on trypticase soy agar slants and was incubated at 37°C with 10% CO2 for 48 h until the primary streak was visible. A 1-μL calibrated disposable inoculating loop (220215; Becton, Dickinson, Franklin Lakes, NJ) was used to inoculate 3 mL of brain–heart infusion broth with the sample. The culture was grown at 37°C with shaking (200 rpm) on a 2D orbital shaker for 12 h. The bacteria were pelleted, and genomic DNA was extracted (DNeasy blood & tissue kit; Qiagen, Hilden, Germany) with the protocol for lysis of gram-positive organisms. The isolate and the following controls (2 μL of template) were used in conventional PCRs: ATCC 33701 (vapA), clinical porcine isolate EIDL 99–213 (vapB), ATCC 6939 (avirulent), and TAMU 49-33 (vapN). PCR for the traA PCR was performed as described previously, 7 and PCR for the vapA, vapB, and vapN (425-bp product) was conducted as described previously.1,4,12,16 PCR products were electrophoresed in 1% agarose gel stained with GelRed (Biotium, Fremont, CA) at 100 V for 35 min. The isolated bacterium was traA- and vapN-positive (traA+/vapAB–/vapN+) and did not amplify vapA or vapB (Fig. 2). For vapN sequencing, an additional PCR was performed using previous methods1,12 to yield a longer, 625-bp segment that was purified (NucleoSpin gel and PCR clean-up kit; Macherey-Nagel, Düren, Germany) and sent for Sanger sequencing (Eton Biosciences, San Diego, CA). The consensus sequence was queried against submissions in GenBank using the Basic Local Alignment Search Tool-Nucleotide (BLASTn). The isolated R. equi PCR product had 100% identity with R. equi strain PAM2012 plasmid pVAPN2012 (KP851975.1), 100% identity with R. equi strain TAMU 49-33 vapN gene (KT211244.1), and 99% identity with R. equi strain PAM1571 plasmid pVAPN1571 (KF439868.1).
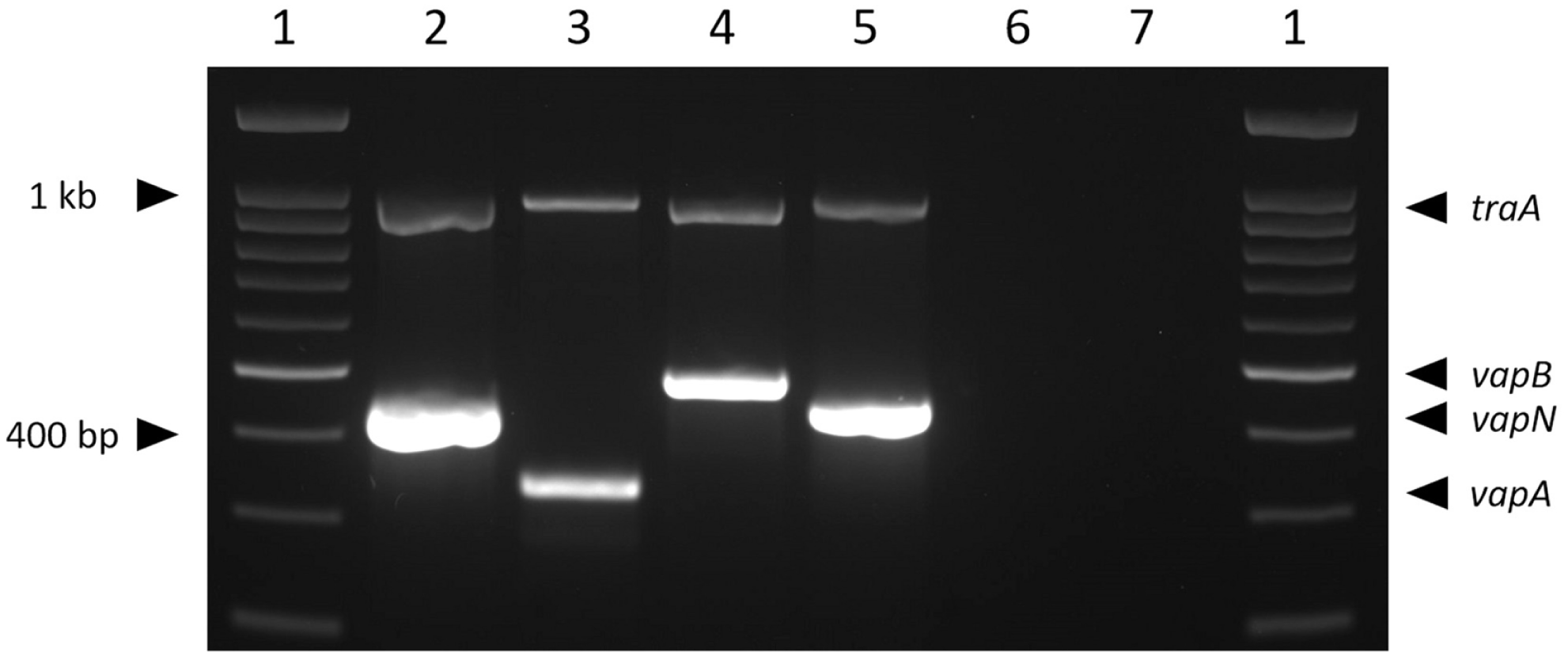
Agarose gel electrophoresis composite of conventional PCRs.1,5,12 Reactions were performed individually and then combined for the composite image. 1 = Biotium 100-bp DNA ladder (31040); 2 = bovine clinical isolate 1; 3 = ATCC 33701 (vapA-positive); 4 = clinical porcine isolate EIDL 99–213 (vapB-positive); 5 = clinical canine isolate TAMU 49–33 (vapN-positive); 6 = ATCC 6939 (avirulent, traA-negative); 7 = PCR reaction blank.
R. equi is an opportunistic, intracellular pathogen found in soil and as part of the normal intestinal flora of horses and other animals. This microorganism is a major pathogen of horses, typically causing suppurative bronchopneumonia in foals. Gastrointestinal lesions are identified in ~50% of foals with R. equi pneumonia, and intestinal involvement includes pyogranulomatous and ulcerative enterotyphylocolitis, pyogranulomatous mesenteric lymphadenitis, and large intra-abdominal abscesses.15,18
The gross and histologic findings in our heifer are reminiscent of R. equi–associated ulcerative enterocolitis in horses. Grossly, these ulcers often develop over Peyer patches in the ileum and have a raised periphery with a depressed ulcerated center (“volcano ulcers”). Similarly, the ulcers in the ileum in this bovid developed over Peyer patches and had a raised periphery caused by massive infiltration of the mucosa and submucosa by macrophages and multinucleate giant cells, many of which contained intracytoplasmic bacteria. Infection of the tongue may have followed teeth trauma to the tongue, which allowed this opportunistic microorganism to cause ulcerative and granulomatous glossitis.
In the intestine, the bacteria most likely infected the subepithelial dome via microfold cells (M cells) overlying submucosal lymphoid tissue and Peyer patches. Within macrophages, vapN inhibits phagosome–lysosome fusion, which enhances R. equi survival and replication within the host. Failure to clear the infection resulted in a robust, but ineffective, immune response and fulminant granulomatous inflammation in the ileum.
The gross changes in the tongue of our heifer resembled lingual actinobacillosis (“wooden tongue”), which is caused by the gram-negative Actinobacillus lignieresii. 15 The common histologic changes in lingual actinobacillosis are pyogranulomas often centered on club colonies of A. lignieresii. 15 In our case, the histiocytes and multinucleate giant cells in the submucosa contained gram-positive, partially acid-fast intracytoplasmic bacteria. Also, R. equi was cultured from the tongue.
Paratuberculosis (Johne’s disease) was a differential diagnosis in our case, given the marked thickening of the small intestine. A scraping from the ileum was negative by rtPCR for M. avium ssp. paratuberculosis.
Why our heifer developed this presentation is still unclear; immunosuppression as a result of stress or concurrent subclinical viral infection was suspected. The heifer was negative for bovine viral diarrhea virus by a fluorescent antibody test, however.
Molecular epidemiology and phylogenetic analysis of virulence plasmid markers supports studies that R. equi infection in humans is zoonotic in origin.7,11,14,18 VapN-positive R. equi has been isolated from ~43% of R. equi–infected human patients, 1 which raises public health concerns regarding consumption of undercooked meat.
Footnotes
Acknowledgements
We thank Mr. Stephan Locke from the bacteriology laboratory, and Dr. Erdal Erol, Dr. Li Zeng, and Jocelynn Morgan from the molecular diagnostic laboratory, at the UKVDL for their help in this case. We also thank Dr. Craig Carter, the director of the UKVDL, for his support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Support for the publication of this article was provided by the University of Kentucky Veterinary Diagnostic Laboratory, University of Kentucky, Lexington, KY.
