Abstract
Paragonimus heterotremus is a medically important lung fluke that causes human and animal paragonimiasis in Southeast Asia, including Thailand. In the current study, a real-time fluorescence resonance energy transfer polymerase chain reaction (real-time FRET PCR) with melting curve analysis was developed and evaluated to detect P. heterotremus eggs in the feces of experimentally infected cats. The detection limit of this method for the P. heterotremus DNA sequence was 3 × 102 copies of the positive control plasmid and 10−3 ng of P. heterotremus genomic DNA. The assay system could detect 10 eggs of P. heterotremus per gram of cat feces. No fluorescence signal was observed when DNA purified from 16 other organisms or genomic DNA from cats and human beings were tested. Real-time FRET PCR yielded positive results for all fecal samples from 17 P. heterotremus–infected cats and showed a negative relationship (r = −0.852, P < 0.001) between the number of parasite eggs in feces and the number of PCR cycles. The assay could detect genomic DNA from P. heterotremus, P. westermani, P. macrorchis, P. siamensis, P. harinasutai, and P. bangkokensis and can differentiate P. heterotremus from the other 5 species. The 6 Paragonimus species examined were divided into 4 groups by melting peak analysis. This assay can be useful for the detection of, and epidemiological studies on, P. heterotremus infection in endemic areas.
Keywords
Paragonimiasis is an important food-borne parasitic zoonosis caused by a lung fluke belonging to the genus Paragonimus.2,10 It is estimated that more than 20 million people are infected worldwide, 21 and it has been calculated that 293 million people are at risk. 9 Human beings and other mammals are infected by ingesting raw crustaceans containing metacercariae 22 and usually present with signs and symptoms in the lower respiratory tract (i.e., cough and hemoptysis). 2 Paragonimus westermani is the most widely distributed species in Asia and the most important human pathogen in China, Korea, and Japan.2,10 In Southeast Asia, however, P. heterotremus is the most important pathogen, and confirmed human cases have been found.2,10 To date, at least 7 lung fluke species have been documented in Thailand: Paragonimus heterotremus, P. siamensis, P. westermani, P. bangkokensis, P. macrorchis, P. harinasutai, and P. pseudoheterotremus.3,19,20 However, only P. heterotremus and P. pseudoheterotremus are proven human pathogens. 6
The standard diagnosis of paragonimiasis is based on the demonstration of the presence of the eggs of Paragonimus spp. in the sputum (by alkaline decontamination and centrifuge sedimentation technique) and/or feces (by formalin–ether concentration technique).1,22 However, species identification of parasite eggs in the sputum and/or feces by microscopic methods is tedious and requires well-trained personnel. Molecular methods based on the polymerase chain reaction (PCR) have been used for the detection and/or species discrimination of Paragonimus spp. (e.g., conventional PCR, 7 random amplified polymorphic DNA-PCR, 5 and multiplex PCR 17 ). As an effective molecular method, a real-time fluorescence resonance energy transfer PCR (real-time FRET PCR) has been developed as a diagnostic tool for the detection and species differentiation of various parasites (e.g., the detection of Opisthorchis viverrini, 16 Clonorchis sinensis, 16 and Schistosoma japonicum 18 ).
In the current study, a rapid and high throughput real-time FRET PCR assay combined with melting curve analysis was developed for the alternative detection of P. heterotremus eggs in the feces of experimentally infected cats. The applicability of the method for differentiation of P. heterotremus from 5 other Paragonimus spp. reported in Thailand was also tested. The detection limit and analytical specificity of the assay were evaluated.
Fecal samples were collected from 17 cats experimentally infected with 30 P. heterotremus metacercariae per cat and from 3 uninfected cats. The fecal samples and P. heterotremus eggs used in the present study were the samples remaining from a previous study 11 and were kept frozen at −80°C until use. All frozen fecal samples were thawed, and the condition of the preserved Paragonimus spp. eggs was reexamined using a simple wet-mount smear method. The number of eggs was counted using the modified formalin–ether sedimentation technique 1 for P. heterotremus eggs (range of eggs per gram of feces [EPG] = 50–2,950). For sensitivity testing, feces from uninfected cats were artificially mixed with various numbers of P. heterotremus eggs prior to DNA extraction. Frozen cat feces (100 mg each) were homogenized with disposable polypropylene pestles, a and genomic DNA (gDNA) was individually extracted using a commercial kit, b according to manufacturer’s protocol. Fecal DNA samples were eluted in 100 μl of distilled water, of which 5 μl was used for the real-time FRET PCR.
The metacercariae of P. heterotremus, P. westermani, P. macrorchis, P. siamensis, P. harinasutai, and P. bangkokensis were harvested from naturally infected fresh water crabs. The species of each Paragonimus metacercaria was determined based on microscopic morphology before DNA extraction. Metacercariae of each Paragonimus spp. were classified and identified by shape and size of cyst, cyst wall thickness, and larval body feature (i.e., shape, arrangement, color, and ventral sucker to oral sucker ratio).3,19,20 One metacercaria each of P. heterotremus, P. westermani, P. macrorchis, P. siamensis, P. harinasutai, and P. bangkokensis were separately homogenized with a disposable polypropylene pestle, and DNA was extracted using a commercial kit, c according to the manufacturer’s protocol. Genomic DNA from each Paragonimus spp. was eluted in 50 μl of distilled water, of which 5 μl was used for the real-time FRET PCR.
The PhITS2_F and PhITS2_R primers targeting the nuclear ribosomal second internal transcribed spacer (ITS2) region of Paragonimus spp. as well as the PhITS2_LC 640 and PhITS2_FL 530 probes were designed and selected based on partial consensus sequences of all 6 Paragonimus spp. using commercial software. d The genus-specific forward primer PhITS2_F (5’-TGGGGTGCCAGATCTATGG-3’) and the reverse primer PhITS2_R (5’-GGGTACTCACGTC TAATCCGAG-3’) e were expected to amplify a 231-bp fragment of the ITS2 sequence of P. heterotremus. A pair of adjacent oligoprobes, the first labeled at the 5’-end (PhITS2_LC 640; 5’ Red 640-TGGTCTGTGTCCGATGCTGACC TATATATGTGCC-Phosphate 3’) and the second at the 3’-end (PhITS2_FL; 5’-GTTCCGCTGTCCCGTCATCATC TATGGTTGAAGTTGCG-Fluo 530-3’) f were used to detect the Paragonimus-specific ITS2 product. For control plasmid preparations, the ITS2 PCR products of 6 Paragonimus spp. were obtained by conventional PCR using the PhITS2_F and PhITS2_R primers. This PCR was performed using 1 µl of template DNA, 0.2 µM of each primer, 2.5 µl of 10× PCR buffer (1.5 mM MgSO4 and 0.2 mM of each deoxyribonucleotide triphosphate), and 0.5 U Taq DNA polymerase. The amplification procedure was 95°C for 5 min; 35 cycles of 95°C for 30 sec, 58°C for 30 sec, and 72°C for 30 sec; and 72°C for 7 min. A 231-bp PCR product for P. heterotremus, a 228-bp PCR product for P. macrorchis, and 233-bp PCR products for P. westermani, P. siamensis, P. harinasutai, and P. bangkokensis were cloned into a commercial vector, g according to manufacturer’s instructions. The recombinant plasmids were transformed and propagated in Escherichia coli JM109, and these plasmids were used as the control plasmids. The nucleotide sequences of the inserted genes within plasmid were sequenced in both directions before using as the positive control of each species. The inserted sequences of P. heterotremus, P. macrorchis, P. westermani, P. siamensis, P. bangkokensis, and P. harinasutai control plasmids were found to be identical with those in GenBank, which had been used to design the primers in the current study (accession nos. AF159603, AF159608, AB354214, AB354222, AB248091, and AB354219, respectively).
The real-time PCR was performed using a commercial system. h Each glass capillary contained 1× commercial DNA mastermix, i 2 mM MgCl2, 0.3 μM PhITS2_F primer, 0.6 μM PhITS2_R primer, 0.2 µM each of the PhITS2_FL 530 and PhITS2_LC 640 probes, and each extracted DNA sample in a 20-μl total volume. A single initial denaturation step of 10 min at 95°C was followed by 45 cycles of repeated denaturation (10 sec at 95°C), annealing (30 sec at 50°C), and extension (15 sec at 72°C). The temperature transition rate was 20°C/sec. The amplification program was followed by a melting curve program of 95°C for 10 sec, 49°C for 20 sec, and then 49°C to 75°C at a rate of 0.2°C/sec with continuous monitoring of fluorescence. The color compensation for fluorescence signals was performed according to the manufacturer’s instructions. j Each sample was analyzed in duplicate. Sterile distilled water and a P. heterotremus positive control plasmid DNA (3 × 107 copies/reaction) were used as negative and positive controls, respectively, for the evaluation of the within-determination (intra-assay) and between-determination (interassay) variations in the melting temperature (Tm). The amplicons of the positive samples were sequenced with M13 universal primers in both directions by the Sanger method 15 and the sequence analyzed using commercial software. k
To verify the analytical sensitivity of the detection method, 5 μl of P. heterotremus control plasmid DNA samples containing between 3 × 107 and 3 copies, and samples of gDNA (1 μl) containing 10–10−5 ng of DNA extracted from a P. heterotremus adult were tested. To determine the sensitivity of the detection of eggs in feces, 5-μl aliquots of the DNA extracted from 100 mg of uninfected cat feces spiked with 1, 2, 4, 8, and 16 P. heterotremus eggs (equivalent to 10, 20, 40, 80, and 160 EPG, respectively) were also tested.
For the evaluation of the analytical specificity, gDNA (5 ng/μl) from various parasites and 1 bacterial species was analyzed. The panel of other organisms used for the real-time FRET PCR analysis included Schistosoma mekongi, S. japonicum, Haplorchis taichui, Fasciola gigantica, O. viverrini, Echinostoma malayanum, Clonorchis sinensis, Capillaria philippinensis, human hookworms, Strongyloides stercoralis, Taenia spp., Ascaris lumbricoides, Trichuris trichiura, lecithodendriid flukes, Giardia lamblia, and Mycobacterium tuberculosis. Cat and human gDNA as well as DNA from 3 samples of uninfected cat feces were also included.
For evaluation of the detection capacity of P. heterotremus eggs in feces, 5 μl of DNA extracted from the feces of 17 experimentally infected cats was tested. The correlation between the number of P. heterotremus eggs in 100 mg of cat feces and the threshold cycle (Ct) indicative of the presence of P. heterotremus DNA was analyzed using Spearman rank correlation test. l In addition, 5 μl of plasmid DNA (107 copies) and 5 μl (1 ng/μl) of gDNA from P. heterotremus, P. westermani, P. macrorchis, P. siamensis, P. harinasutai, and P. bangkokensis metacercariae were evaluated for species differentiation.
The detection limit of the ITS2 target DNA sequence was 3 × 102 copies of the positive control plasmid (Fig. 1A) and 10−3 ng of P. heterotremus gDNA (Fig. 1B), when using 35 cycles as the cutoff detection limit. The range, mean ± standard deviation (SD), and median of the Tm values for the P. heterotremus positive control plasmid and the gDNA were 68.28–68.51, 68.43 ± 0.08, 68.45 and 68.43–68.72, 68.54 ± 0.11, 68.52, respectively. A detection limit for P. heterotremus eggs in feces as low as 10 EPG of control uninfected cat feces was achieved (Fig. 1C). Moreover, no fluorescence signals (Ct values all >35) were observed for purified DNA from 15 parasites other than P. heterotremus, gDNA from M. tuberculosis, DNA from cats and human beings, and DNA extracted from the 3 samples of uninfected cat feces (Fig. 2A). Although the gDNA from several parasites (e.g., S. japonicum, C. philippinensis, S. stercoralis, and lecithodendriid flukes) were amplified as 231-bp bands (Supplementary figure), no specific fluorescence signal was detected by the melting peak analysis. The coefficients of the variation of this assay showed that all the tests were within the statistically acceptable range of day-to-day variation (data not shown).
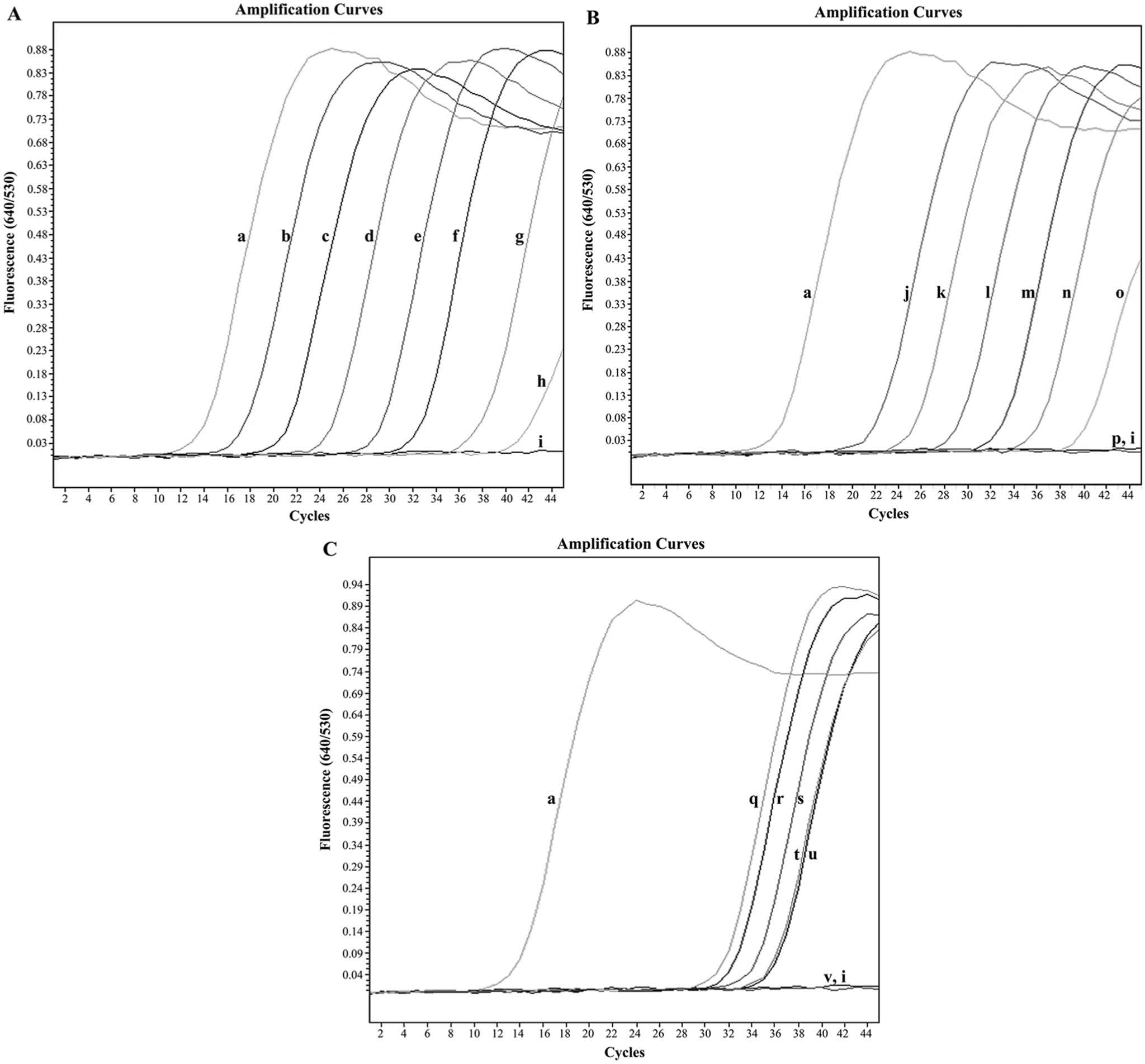
Amplification plots of fluorescence (y-axis) versus threshold cycle (Ct; x-axis) show the detection limit of the real-time fluorescence resonance energy transfer polymerase chain reaction assay.
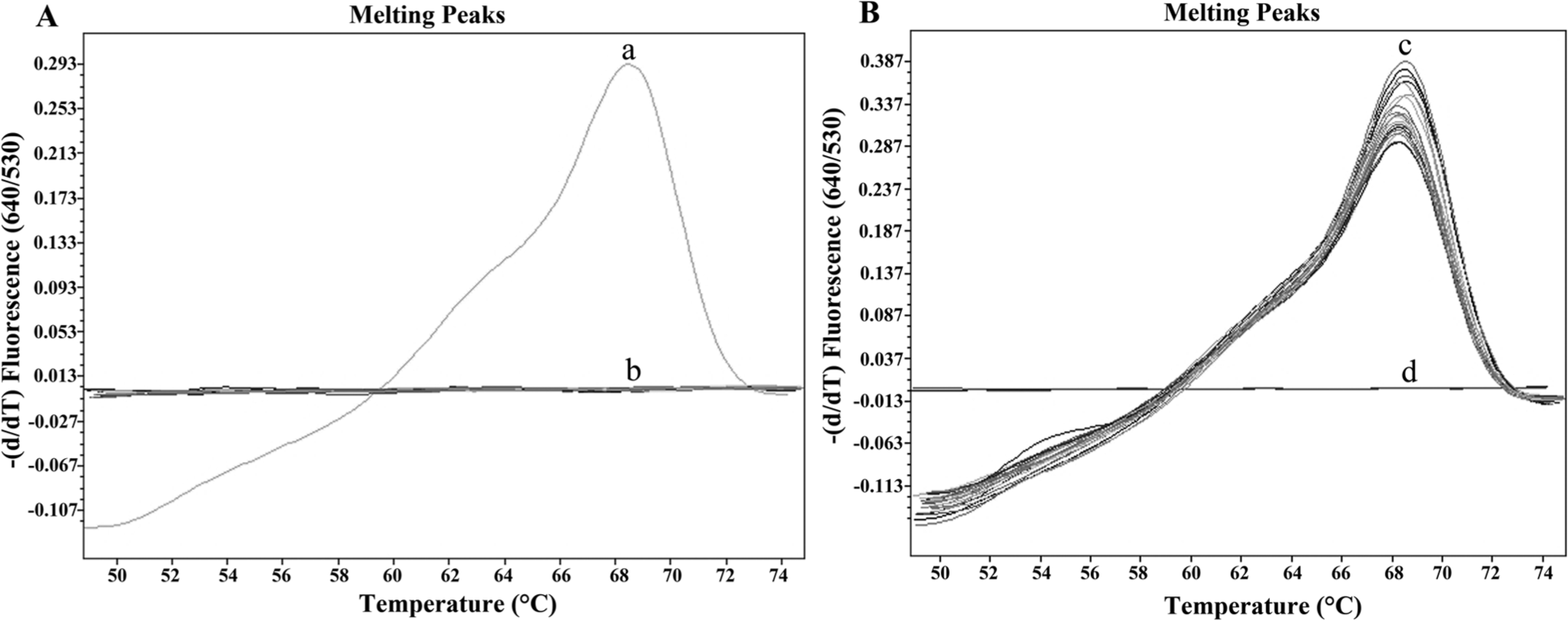
Specificity analysis of the real-time fluorescence resonance energy transfer polymerase chain reaction assay.
Real-time FRET PCR combined with melting curve analysis of the amplicon products was applied to detect DNA extracted from P. heterotremus eggs in infected cat feces. The assay yielded satisfactory results, with all 17 stool samples that were positive for P. heterotremus eggs by the simple smear method being positive by PCR. The fecal samples from 3 uninfected cats were negative. The results of the melting curve analyses for P. heterotremus DNA are shown in Figure 2B. The range, mean ± SD, and median of the Tm values for P. heterotremus DNA in the fecal samples were 68.23–68.46, 68.32 ± 0.08, and 68.30, respectively. Furthermore, a negative relationship (r = −0.852, P < 0.001) was observed between the number of P. heterotremus eggs in 100 mg of cat feces and the Ct indicative of the presence of P. heterotremus DNA. The 231-bp PCR amplicons observed for all infected fecal samples were sequenced in both directions, and the nucleotide sequences of all amplicons were identical to the reported P. heterotremus ITS2 sequence (AF159603).
When the plasmid and gDNA from P. heterotremus, P. westermani, P. macrorchis, P. siamensis, P. harinasutai, and P. bangkokensis were used for species differentiation using the real-time FRET PCR assay with the melting curve analysis, all 6 Paragonimus species could be detected, and were differentiated into 4 groups based on different Tm values (Fig. 3). Group 1 contained only P. heterotremus (Tm values of plasmid DNA = 68.40 and gDNA = 68.36). Group 2 contained P. macrorchis (Tm values of plasmid DNA = 62.04 and gDNA = 62.01). Group 3 consisted of P. siamensis (Tm values of plasmid DNA = 54.85 and gDNA = 54.82) and P. westermani (Tm values of plasmid DNA = 54.59 and gDNA = 54.66), which had almost equal Tm values. Group 4 consisted of P. harinasutai (Tm values of plasmid DNA = 53.59 and gDNA = 53.48) and P. bangkokensis (Tm values of plasmid DNA = 53.42 and gDNA = 53.39), which also had equal Tm values.
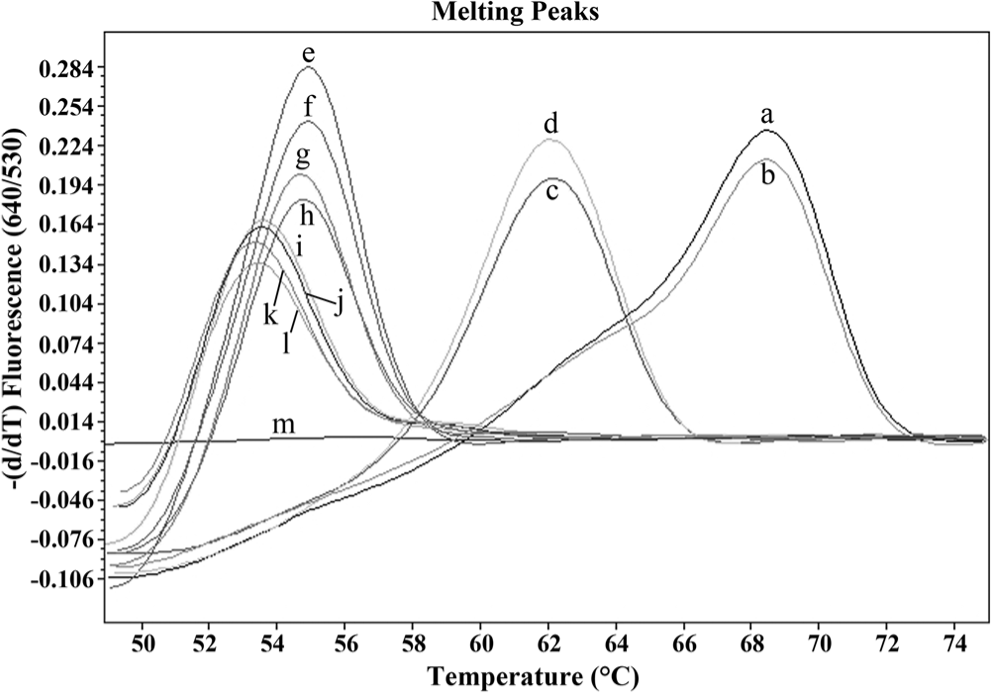
Representative melting curves constructed using 2 fluorophore-labeled probes hybridized to the amplification products of the second internal transcribed spacer from 107 copies of plasmid DNA (a, c, e, g, i, k) and 5 ng of genomic DNA (gDNA; b, d, f, h, j, l) from 6 species of Paragonimus. The melting curves for this experiment are shown as –(d/dT) fluorescence (640/530). Group 1: P. heterotremus plasmid DNA (a; melting temperature [Tm] = 68.40) and gDNA from P. heterotremus (b; Tm = 68.36); group 2: P. macrorchis plasmid DNA (c; Tm = 62.04) and gDNA from P. macrorchis (d; Tm = 62.01); group 3: P. siamensis plasmid DNA (e; Tm = 54.85), gDNA from P. siamensis (f; Tm = 54.82), P. westermani plasmid DNA (g; Tm = 54.59), and gDNA from P. westermani (h; Tm = 54.66); group 4: P. harinasutai plasmid DNA (i; Tm = 53.59), gDNA from P. harinasutai (j; Tm = 53.48), P. bangkokensis plasmid DNA (k; Tm = 53.42), and gDNA from P. bangkokensis (l; Tm = 53.39) were included. Distilled water (m) was used as a negative control in this study.
The current study was designed to develop a real-time FRET PCR assay combined with melting curve analysis to detect the ITS2 DNA sequence of P. heterotremus. The ribosomal DNA ITS2 sequences have been used for the detection and species identification of various Paragonimus species. 2 Using 35 cycles as the cutoff for positivity, the lowest detection limit for the P. heterotremus positive control plasmid was approximately 3 × 102 copies and 10−3 ng of P. heterotremus gDNA. The assay could also detect as few as 10 EPG of P. heterotremus eggs mixed in uninfected cat feces and also could detect P. heterotremus eggs in the feces of all experimentally infected cats. Although the detection ofP. heterotremus gDNA was 10 times less sensitive than the conventional PCR detection reported previously, 7 the detection limit of this method is quite similar to that for the detection of P. heterotremus eggs, for which the detection limit was at approximately 8–10 EPG. 7 Further improvement of the sensitivity of detection is needed.
In the current study, no fluorescence signal was observed for DNA samples from 15 parasites other than members of the genus Paragonimus, M. tuberculosis, cats, and human beings, showing the high specificity of this protocol. The metacercariae of different species of lung flukes can be found in the same freshwater crab species (e.g., P. bangkokensis and P. harinasutai metacercariae in Potamon smithianum4,13; P. heterotremus, P. bangkokensis, and P. harinasutai metacercariae in Potamon lipkei 14 ; and P. heterotremus, P. westermani, and P. harinasutai metacercariae in the freshwater crab Larnaudia larnaudii in Thailand 8 ). The species identification of the eggs and metacercariae of these lung flukes requires careful morphological examination by an expert. 12 The PhITS2_LC 640 and PhITS2_FL probes used in the current study were designed to detect the partial consensus sequences of the ITS2 of all 6 Paragonimus spp. occurring in Thailand. The partial ITS2 sequences of Paragonimus spp. were aligned using 2 probes that varied from the known sequence in the following manner: a 3-nucleotide difference for P. heterotremus (GenBank accession no. AF159603); a 10-nucleotide difference for P. macrorchis (AF159608), P. westermani (AB354214), and P. siamensis (AB354222); and a 12-nucleotide difference for P. bangkokensis (AB248091) and P. harinasutai (AB354219). The mismatches between the probes and the parasite DNA template resulted in different Tm values. The results of the melting point analysis show that the 6 Paragonimus spp. examined in the present study could be divided into 4 groups with different Tm values; P. heterotremus and P. macrorchis each form a distinct group, P. siamensis and P. westermani make up a group, and P. bangkokensis and P. harinasutai make up another group. This grouping is in agreement with the molecular phylogenetic tree analysis using ITS2 sequences performed in a previous study, with P. siamensis being grouped in the P. westermani complex 2 and P. bangkokensis and P. harinasutai being grouped in the same species complex. 4 The present results show that the Tm value can differentiate Paragonimus species at the species complex level. The method can be used not only for the detection of P. heterotremus eggs in cat feces but also for the differential detection of P. heterotremus and P. macrorchis among all 6 other Paragonimus spp.
In conclusion, a sensitive, specific, and reproducible real-time FRET PCR assay based on the ITS2 region was developed to detect P. heterotremus eggs in the feces of experimentally infected cats. This method can also differentiate P. heterotremus from 5 other Paragonimus spp.
Footnotes
Acknowledgements
Dr. Yukifumi Nawa provided valuable comments and suggestions on the manuscript.
a.
Polypropylene pestles, Bellco Glass Inc., Vineland, NJ.
b.
QIAamp DNA Stool Mini Kit, Qiagen GmBH, Hilden, Germany.
c.
Nucleospin Tissue kit, Macherey-Nagel GmbH & Co., Düren, Germany.
d.
The LC probe design software, Roche Applied Science, Mannheim, Germany.
e.
Sigma-Aldrich Pte. Ltd., Singapore.
f.
TIB MOLBIOL GmbH, Berlin, Germany.
g.
pGEM-T easy vector, Promega Corp., Madison, WI.
h.
Research products, Roche Applied Science, Mannheim, Germany.
i.
LightCycler FastStart DNA Master HybProbe, Roche Applied Science, Mannheim, Germany.
j.
Technical note no. LC19/2004, Roche Applied Science, Mannheim, Germany.
k.
MegaBACE 1000 DNA Analysis System, GE Healthcare, Piscataway, NJ.
l.
Spearman’s rank correlation, SigmaPlot version 9.01, Systat Software Inc., San Jose, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the National Science and Technology Development Agency (Discovery Based Development Grant), the Higher Education Research Promotion and National Research University Project of Thailand, the Office of the Higher Education Commission through the Health Cluster, Specific Health Problem in Greater Mekong Sub-region and Khon Kaen University, Thailand. Wanchai Maleewong and Pewpan M. Intapan are supported by Thailand Research Fund Senior Research Scholar Grant (Thailand Research Fund grant no. RTA5580004).
