Abstract
The objective of this study was to report a reliable real-time polymerase chain reaction assay compatible with the Roche LightCycler 2.0 capable of genotyping sheep for scrapie susceptibility at codon 171. The single nucleotide polymorphisms (SNPs) in the prion protein gene in sheep that may govern resistance to scrapie at codon 171 encode for lysine (K), histidine (H), glutamine (Q), and arginine (R). A modified proteinase K method for leukocytes or whole blood was used to isolate genomic DNA from sheep blood. Fluoresentric developed and optimized primers and probes for the codon 171 SNP assay. The assay was initially validated using 218 determinations from whole blood of known genotypes with 100% correct identity. The assay was further validated through a whole-blood check test provided annually by the National Veterinary Services Laboratory with a correct identification rate of 100%. From January 2005 to December 2006, 3,672 samples from blood were genotyped at codon 171. The genotypes were QR171 (n = 1,838, 50.05%). RR171 (n = 1,423, 38.75%), QQ171 (n = 407, 11.08%), HR171 (n = 2, 0.05%), and HQ171 (n = 2, 0.05%). The combination of this simple extraction method and the novel Fluoresentric assay is very accurate, is capable of identifying all 4 SNPs at codon 171 in one reaction, and has proven to be a useful tool for producers in their selective breeding programs.
Keywords
Scrapie is a terminal neurologic disease of sheep belonging to a family of prion diseases identified as transmissible spongiform encephalopathies. 1–7,16,17–19,21 The disease is a growing concern for sheep producers, who have incurred monetary loss estimated at $20–$25 million annually from scrapie (National Institute for Animal Agriculture: November 2001, What you as a producer need to know about the new scrapie eradication program. Accessed September 26, 2007. http://www.animalagriculture.org/scrapie/Pamphlets/WhatYouAsAProducer/NIAA%20Producer%20Kit.pdf). This prompted the National Institute for Animal Agriculture to promote a program designed to eradicate scrapie by 2010 (U.S. Department of Agriculture [USDA], Animal and Plant Health Inspection Service [APHIS]: December 2003, Distribution of genotypes at codon 171 in U.S. sheep. Accessed September 26, 2007. http://www.aphis.usda.gov/vs/ceah/ncahs/nahms/sheep/sheep01/Genotypes.pdf).
Genotyping sheep to determine susceptibility to scrapie is a technique used in selective breeding practices. Polymorphisms at codons 136, 154, and particularly 171 have been identified as having a significant effect on resistance to classic scrapie. 1,3–7,15,16,17–19 These sites code for the amino acids in the prion protein (PRNP) responsible for resistance or susceptibility to scrapie (USDA, APHIS, Veterinary Services [VS], National Animal Health Monitoring System: January 2004, Phase II: Scrapie: Ovine slaughter surveillance study 2002–2003. Accessed September 26, 2007. http://www.aphis.usda.gov/vs/ceah/ncahs/nahms/sheep/SOSSphase2.pdf). Genotypes are reported as the diploid amino acids of the codon tested (i.e., QR171). In the United States, genotyping focuses predominantly on codons 136 and 171. At codon 136, the amino acids alanine (A136) and valine (V136) affect susceptibility. At codon 171, the amino acids lysine (K171), histidine (H171), glutamine (Q171), and arginine (R171) affect susceptibility. 2,7,9–12,20 The genotype QQ171 is associated with natural scrapie infections in sheep of several breeds, notably Suffolk sheep in the United States and Japan. 1,4–6,16,17,21 The genotype RR171 appears to confer maximum resistance (USDA, APHIS, VS, CEAH. National Animal Health Monitoring System: n.d., The genetics of scrapie susceptibility. Accessed January 3, 2008. http://www.aphis.usda.gov/animal_health/animal_diseases/scrapie/downloads/scrapie_genetics.pdf) to naturally occurring scrapie infection. 1,4–6,15,16,17–19,21
Several PRNP genotyping assays are available. These include sequencing, restriction fragment analysis, mass spectrometry technology, and primer extension, all of which are accompanied by either multiple technical steps, staggering start-up costs, or both. The vast majority of genotyping assays center around polymerase chain reaction (PCR)-based technology. The Roche LightCycler 2.0 a uses fluorescence resonance energy transfer technology in a real-time PCR and offers a proven assay capable of directly testing extracted DNA (Roche Applied Science: June 2005, LightCycler 2.0 instrument operator's manual, software version 4.05. Chapter D: 92, 148–156). This allows the user to avoid unnecessary manipulation steps and keeps start-up costs significantly lower without sacrificing sensitivity/resolution. 13,19
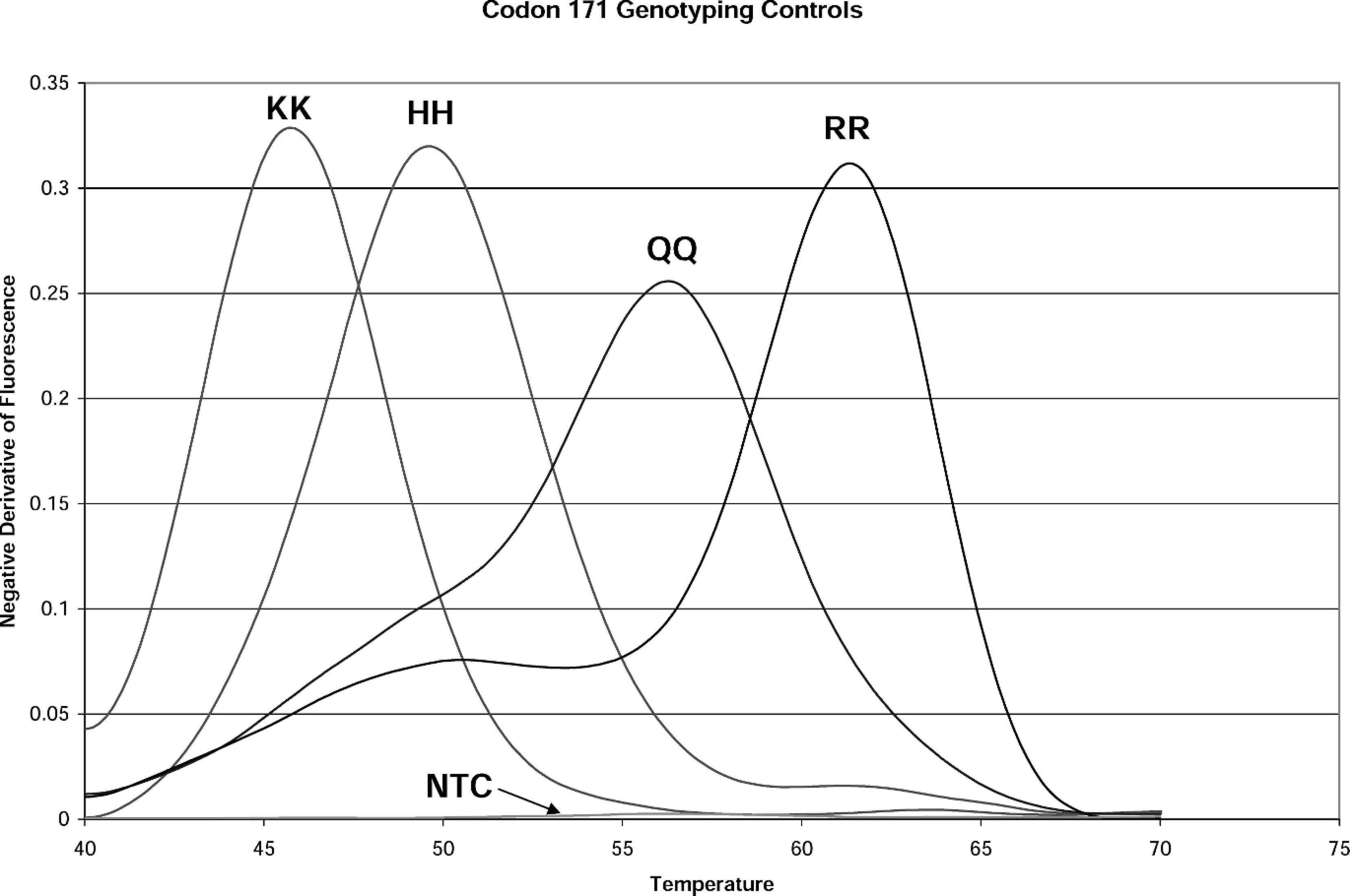
Melting peaks of known genotype controls from real-time polymerase chain reaction single nucleotide polymorphism assay at codon 171. KK = homozygous lysine peak; HH = homozygous histidine peak; QQ = homozygous glutamine peak; RR = homozygous arginine peak; NTC = no template control. An NTC control is run in all assays.
At the time of development, the Rocky Mountain Regional Animal Health Laboratory (RMRAHL) considered 2 platforms capable of performing the single nucleotide polymorphism (SNP) assay using real-time PCR technology: Applied Biosystems and Roche Applied Science. The Roche LightCycler 2.0 was chosen because of its speed 8 and capacity to identify all 4 alleles at codon 171 in one reaction. It uses a carousel and capillary system capable of running 32 reactions in 45 min for these reaction conditions. Genotyping occurs via melting temperature analysis, which generates melting peak profiles of samples and known genotype controls. LightCycler software displays each sample or control as a peak, allowing intuitive identification of the genotype. The LightCycler system, coupled with the LightCycler Multiplex DNA Master HybProbe a reagent, offers a fast, sensitive, and reliable platform. Although RMRAHL is capable of genotyping at both codon 136 and 171 with the LightCycler, this study focuses on codon 171 because of the unique aspects of this custom assay and the workload at the RMRAHL. The custom assay was developed with the help of Fluoresentric, b which optimized reaction conditions and maximized specificity and sensitivity of probes and primers at codon 171.
Blood samples were collected by venipuncture in tubes containing ethylenediamine tetra-acetic acid (EDTA). A process control was included with each group of samples to assess accurate isolation of DNA. The process control consisted of a 300-μl aliquot of whole blood (stored at −20°C until used) from a wether of genotype QQ171. The DNA isolation used a modified proteinase K extraction method. 14 In a 1.5-ml microtube, 300 μl of blood was washed twice with 1,000 μl of 1X TE (Tris-HCl pH 8.0, EDTA) buffer. Next, 10 μl of proteinase K c and 200 μl of lysis buffer K (pH 8.3; 10 mM Tris-HCl pH 8.0, 50 mM KCl, 2.5 mM MgCl, 1% sodium dodecyl sulfate [SDS]) were added to the pellet. Samples were then vortexed and incubated for 60 min in a 56°C water bath. After lysis. 90 μl of ammonium acetate d and 725 μl of 100% ethanol e was added to the sample. Samples were then vortexed and incubated at −20°C for 60 min, centrifuged, and washed twice with 200 μl of 70% ethanol. Samples were then incubated for 10 min at 100°C, rehydrated in 30 μl of 1X TE buffer, incubated an additional 5 min at 100°C, and immediately cooled in an ice-water bath.
The custom assay uses hybridization probes to exploit the FRET process. One probe is labeled with the fluorescent donor dye Fluorescein, 5′-CCCCAAC CAAGTGTACTAC-FITC-3′ (melting temperature [TM] 60.3°C); the other probe is labeled with an acceptor dye, 5′-705-ACCAGTGGATCGGTATAG-(P)-3′ (TM 58.3°C).
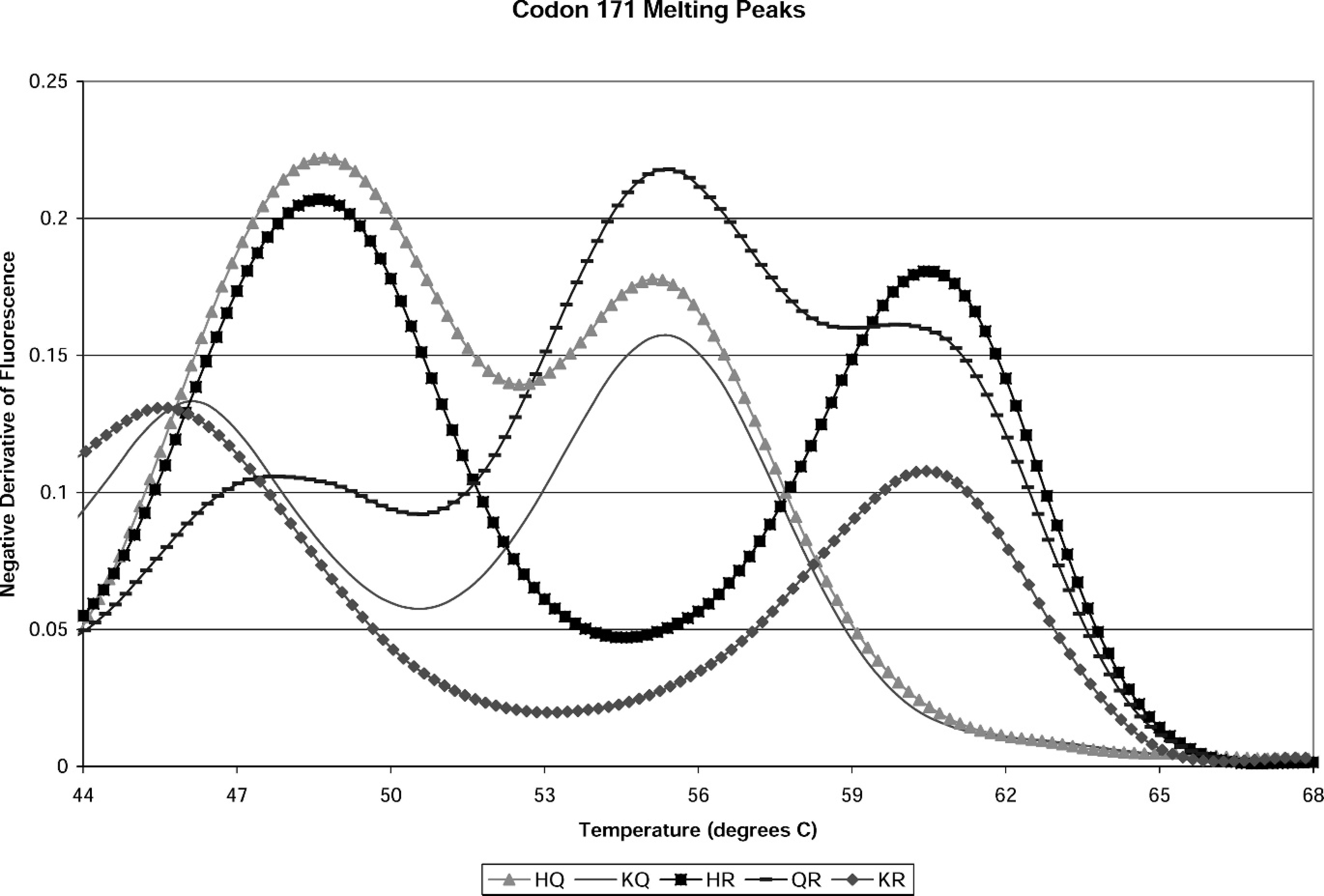
Melting peak pattern of proficiency panel genotypes from real-time polymerase chain reaction single nucleotide polymorphisms assay at codon 171.
The primers and probe were designed from GenBank accession number AY326330. It is important to note that the TMs of the 2 probes in this assay are lower than those of the primers. This arrangement keeps the probes from hybridizing with the template DNA until the melting curve analysis step. The hybridization of the probe pair in that step results in an energy transfer between the fluorescent dyes, allowing the donor dye to excite the acceptor dye. The fluorescence is read in this final step. The LightCycler takes continuous readings of the decrease in fluorescence as the probes are melted off according to the sequence of the target underlying the probes. Software analysis displays a peak or peaks corresponding to the genetic composition of that sample or control. 8
For this assay, each capillary contains 1 μl of the DNA sample or control and 9 μl of master mix containing LightCycler Multiplex DNA Master HybProbe, molecular grade H2O, MgCl2 at a reaction concentration of 2 mM and the optimized primer and probe mix. The sequence of the forward primer is 5′-GGCTACATGCTGGGAAG-3′ (Tm 64.2°C). The reverse primer is 5′-ATGTTGACA CAGTCAT(R)CAC-3′ (Tm 60.3°C). The arginine (R) is a mixture of purines adenine and guanine, presenting as a guanine in the sequence of the wild-type gene. After adding DNA template to the appropriate aliquot, the capillaries are positioned in a carousel and centrifuged for 15 sec in a carousel centrifuge. a A typical codon 171 assay consists of an initial step of 10 min at 95°C; a preamplification step of 10 sec at 95°C, 2 sec at 60°C, and 15 sec at 72°C; an amplification step of 45 cycles of 10 sec at 87°C, 2 sec at 60°C, and 15 sec at 72°C; a melting step of 30 sec at 95°C; cooling to 43°C for 1 min; and finally, the melting curve step, where the temperature rises to 85°C in 0.2°C/sec increments while in continuous acquisition mode (acquiring fluorescence data). At the end of the acquisition, the reaction is cooled to 40°C for 20 sec and melting peaks are analyzed.
The melting peak patterns of 4 homozygous controls used in the custom assay are shown in Figure 1. These are the standard to which unknown samples are evaluated in genotype determination. The custom assay is unique in that these controls do not come from sheep. The controls are synthetic template DNA produced by Fluoresentric, producing peaks at 47.5°C (KK), 49.9°C (HH), 55.8°C (QQ), and 60.4°C (RR; ± 1.5°C] for the 4 homozygous alleles possible at codon 171.
For the initial assay validation, 218 DNA isolates were obtained from blood samples provided in September 2004 by the National Veterinary Services Laboratory (NVSL; Ames, IA). The original genotypes were identified at NVSL via sequencing reactions. Concordant results for all 218 samples were obtained. Proficiency panels are obtained annually from the NVSL, and are blinded until after the test results are submitted and scored. The genotypes of past proficiency panels have included the following: HQ171. QR171, HR171, RR171, KQ171, KR171, and QQ171. These are shown in Figure 2. The assay has demonstrated a correct identification rate of 100% on 368 total proficiency panel SNP determinations as of October 2006.
Distribution of genotypes in sheep tested for single nucleotide polymorphisms at codon 171 of the prion gene
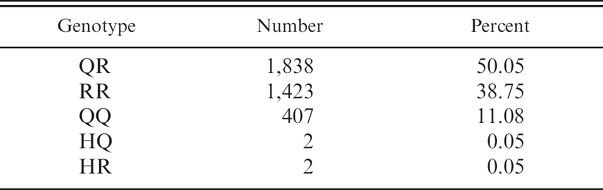
Using this method, 3,672 blood samples originating from various parts of the United States were genotyped at codon 171 from January 2005 to December 2006 (Table 1). Of the 3,672 sheep tested, 88.9% (n = 3,263) contained an R171 allele. The most common genotypes were QR171 (50.05%. n = 1,838) and RR171 (38.75%, n = 1,423). The least common genotypes were HR171 (0.05%, n = 2), HQ171 (0.05%, n = 2), and QQ171 (11.08%, n = 407). These numbers compare with the following values from the USDA APHIS fact sheet on genotype distribution at codon 171 in U.S. sheep: QR171 (44.1%), RR171 (16.0%), QQ171 (39.1%), and HH171/HQ171/HR171 (0.8%).
In summary, the method described is an accurate genotyping assay of sheep at codon 171 for susceptibility to scrapie. This assay fuses an inexpensive DNA extraction with SNP detection capable of precise identification of genotypes at codon 171. Eliminating a commercial DNA extraction kit reduces assay cost without compromising results.
Acknowledgements. The authors thank Ed Kline and Justin Niccoli for their diligent collection of a large portion of sheep blood samples and Kate McRoy and Stacy Romero for their effort on the figures and graphs.
Footnotes
a.
Roche Applied Science, Indianapolis, IN.
b.
Fluoresentric OaPRNP Codon 171 Genotyping Assay Kit. Fluoresentric, Park City, UT.
c.
Proteinase K Catalog # 3115828, Roche Applied Science. Indianapolis, IN.
d.
Ammonium acetate Catalog # A2706–100ML, Sigma-Aldrich, St. Louis, MO.
e.
Absolute ethanol Catalog # E7023-6X500ML, Sigma-Aldrich, St. Louis, MO.
