Abstract
Aberrant migration of Parelaphostrongylus tenuis in camelids results in neurologic deficits, recumbency, and sometimes death. An antemortem diagnosis of P. tenuis in camelids is typically based upon the presence of characteristic asymmetric neurologic deficits, known exposure to white-tailed deer, cerebrospinal fluid (CSF) eosinophilia, and response to treatment. The diagnostic accuracy of CSF eosinophil percentage for the diagnosis of P. tenuis in camelids has not been critically examined. The objective of the current study was to determine the sensitivity (Se) and specificity (Sp) of CSF eosinophil percentage, CSF eosinophil concentration, total nucleated cell concentration, and protein concentration for the antemortem diagnosis of P. tenuis. Medical records of camelids admitted to Cornell University with clinical signs of neurologic disease, CSF analysis, and necropsy were examined from January 2000 through December 2009. Se and Sp were determined by receiver operating characteristic curves in camelids diagnosed with P. tenuis (n = 13) or other conditions (n = 24) based on postmortem examination. More than 17% of eosinophils in CSF had a Se of 85% and Sp of 92% for P. tenuis diagnosis (area under the curve [AUC]: 0.87; SE AUC: 0.07; P < 0.0001; 95% confidence interval [CI] AUC: 0.72–0.96), and >1.4 eosinophils/µl of CSF had a Se of 85% and Sp of 96% (AUC: 0.9; SE AUC: 0.06; P < 0.0001; 95% CI AUC: 0.76–0.97). Cerebrospinal fluid eosinophil percentage and concentration are sensitive and specific methods for diagnosing P. tenuis antemortem in camelids residing in regions endemic to white-tailed deer.
Introduction
Cerebrospinal nematodiasis of alpacas (Vicugna pacos) and llamas (Lama glama) in the northeastern United States occurs due to aberrant migration of the parasite Parelaphostrongylus tenuis, also known as the “meningeal worm.”4,5,7,24,29 Parelaphostrongylus tenuis is endemic to areas of the United States where there are large populations of its definitive host, the white-tailed deer (Odocoileus virginianus).10,25,44 Clinical signs of infection most commonly consist of asymmetric pelvic limb neurologic deficits that progress cranially. Scoliosis and cranial nerve deficits have also been reported.20,47 However, the disease often goes undetected until the camelid becomes recumbent. Experimental and natural infection with P. tenuis also occurs in sheep,1,21,35 goats,16,23,27 cattle,11,28 reindeer, 30 caribou, 2 antelope, 31 fallow deer,22,36 sika deer, 14 moose, 15 bison, 45 wapiti, 32 and elk.6,8 Aberrant migration of P. tenuis in horses causes verminous encephalitis, scoliosis, and intraocular infection.37,40,41,43
The lifecycle of P. tenuis within definitive and aberrant hosts has been described.4,27,29,34 In the camelid, ingestion of an intermediate host gastropod containing third-stage P. tenuis larvae is followed by larval migration from the gastrointestinal tract to the spinal cord over a minimum of 49 days.12,38 Once in the spinal cord, the larvae mature and migrate cranially, creating destructive tracts in the process. Histologically, these migration tracts correlate with random, asymmetric foci of rarefaction and neuroparenchymal loss with variably eosinophilic, granulomatous, or lymphoplasmacytic inflammation. Migration tracts are more common in the white matter of the spinal cord; however, lesions frequently occur in the spinal cord gray matter and within the cerebrum and cerebellum.20,28,41
A definitive diagnosis of P. tenuis is based on postmortem findings, specifically the presence of metastrongylid nematodes in tissue sections 41 accompanied by molecular diagnostic confirmation.28,41 However, such molecular techniques are not widely available, and nematodes are frequently absent on tissue sectioning. In these cases, the postmortem diagnosis is based on a high clinical index of suspicion and corroborative histologic findings of larval migration tracts.20,21,38 Antemortem diagnosis of P. tenuis in camelids is not definitive, but is typically based on the combination of classic neurologic examination findings, exposure to white-tailed deer, the presence of eosinophils in the cerebrospinal fluid (CSF), and response to treatment with anthelmintics (ivermectin and fenbendazole), anti-inflammatories (flunixin meglumine and corticosteroids), and other supportive neuroprotectants (vitamin E, thiamine, etc.).20,24,26,29,47 At present there are no commercially available antibody or DNA testing kits for the antemortem diagnosis of P. tenuis, and as patent infection does not develop in camelids, fecal evaluation for first-stage larvae is not useful in the diagnosis of P. tenuis in camelids or other aberrant hosts. 38
Increased CSF eosinophil percentage is a particularly consistent finding upon cytologic evaluation of CSF in camelids infected with P. tenuis and is often accompanied by an increased protein and nucleated cell concentration. Increased CSF eosinophil percentage strongly supports a presumptive clinical diagnosis of P. tenuis in camelids,5,12,20,26,38 but despite its common use in clinical practice as a means of tentatively diagnosing P. tenuis, the diagnostic accuracy of CSF eosinophilia has not been critically examined. The purpose of the current study is to determine the sensitivity (Se) and specificity (Sp) of CSF eosinophil percentage, eosinophil concentration, nucleated cell concentration, and protein concentration for the antemortem diagnosis of P. tenuis in camelids.
Materials and methods
A retrospective case study using a keyword-based search of the medical records information system database of Cornell University Equine and Farm Animal Hospital (CU-EFAH; Ithaca, New York) from between January 2000 and December 2009 was performed. Keyword search terms included the follow: South American camelid, alpaca, llama, CSF cytology, and necropsy. Camelids were included if neurologic symptoms were present and if both CSF cytology and postmortem examination with spinal cord and brain histopathology had been performed. Pathologists (HS Bender, DH Schlafer) retrospectively evaluated the postmortem results and were not blinded. Where the original diagnosis was in question, histopathology sections were reexamined to confirm a diagnosis of P. tenuis. Inclusion in the “P. tenuis” group required a postmortem diagnosis of P. tenuis based on gross and histologic findings. Gross necropsy findings were typically unremarkable, with no parasites identified on examination of the brain and spinal cord in any of the cases in this study. A histologic diagnosis was made if metastrongylid nematodes were identified in formalin-fixed sections of spinal cord or brain. Parelaphostrongylus tenuis were identified by their size (150–300 µm in diameter in cross-section); smooth, thin cuticle; accessory hypodermal cords; polymyarian–coelomyarian musculature; genital tract containing either sperm or eggs; and prominent intestinal tract lined by few multinucleate cells.13,41 In cases where organisms were not observed, criteria for histologic diagnosis of P. tenuis infection included multifocal, randomly distributed areas of rarefaction and gliosis, consistent with parasitic migration, accompanied by eosinophilic or lymphoplasmacytic encephalomyelitis, axonal swelling, and Wallerian degeneration. Parelaphostrongylus tenuis infection was excluded in the remaining camelids, which were placed in the “other diagnosis” group.
The following data were obtained from medical records: signalment, neurological examination findings, CSF cytological findings (total protein, total nucleated cell concentration, red blood cell concentration, eosinophil percentage, and eosinophil concentration), duration of clinical signs prior to CU-EFAH admission, anthelmintic administration before and during hospitalization, and necropsy and histologic findings.
Descriptive data and preliminary tests were generated using Statistix 9. a The Shapiro–Wilk normality test was used to determine that the data were non–Gaussian, requiring nonparametric methods. The Wilcoxon rank-sum test was used to compare median CSF eosinophil percentage, eosinophil concentration, total nucleated cell concentration, and protein concentration between camelids with and without a postmortem diagnosis of P. tenuis infection. The Se and Sp of these 4 CSF test results for the diagnosis of P. tenuis in camelids were evaluated. Receiver operating characteristic (ROC) curves and Se and Sp at different cut-off concentrations, with 95% confidence intervals (CI), were generated using MedCalc 11.2.0.0. b Differences were considered statistically significant at an overall P-value of 0.05; a Bonferroni correction was applied for multiple comparisons (0.05/4), reducing the P-value cut-off to 0.013.
Results
A total of 37 neurologic camelid cases met the inclusion criteria, consisting of 9 llamas and 28 alpacas. Two additional potential cases were excluded due to severe blood contamination in the CSF, complicating cytologic interpretation. Diagnosis of P. tenuis was made in 35% (13/37) of the cases, based on characteristic histologic findings of migratory tracts in 100% (13/13) and observation of a metastrongylid nematode within the spinal cord segments in 31% (4/13) of cases (Fig. 1A, 1B). Two of the camelids with larvae seen on cross-section of the spinal cord had no known anthelmintic administration prior to or during hospitalization. The camelids in the P. tenuis group had a median age of 4 years (range: 4 months to 10 years) and 92% female distribution (12 females, 1 male). Presenting neurological deficits in the P. tenuis camelids included recumbency in 77% (10/13) of cases, hind limb ataxia only in 15% (2/13) of cases, and ataxia of all limbs in 8% (1/13) of cases. A single camelid (8%, 1/13) in this group presented with cranial nerve deficits, and scoliosis was observed in another (8%, 1/13). One case included in the P. tenuis group was euthanized due to dilated cardiomyopathy approximately 1 year after P. tenuis diagnosis, at which time histological evidence of P. tenuis infection was still present in the spinal cord.
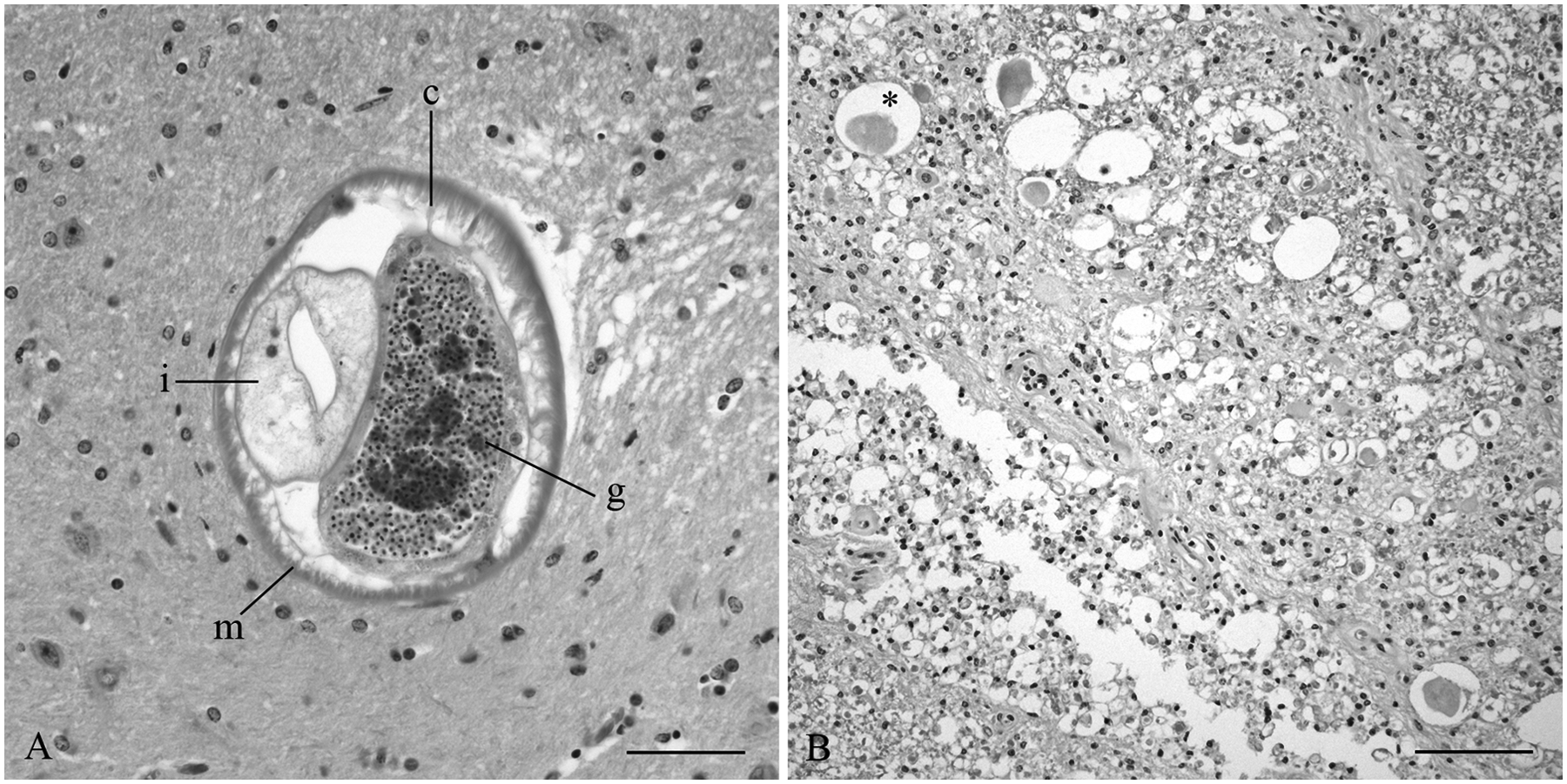
Llama (Lama glama).
Nineteen of the remaining 24 cases were given a postmortem diagnosis other than P. tenuis. These other diagnoses included diseases affecting the spinal cord (vertebral malformation, sarcocystosis, infarction, hematomyelia, trauma, myelomalacia, and degenerative myelopathy), diseases affecting the brain (lateral ventricular dilation, pituitary adenoma, hepatic encephalopathy, poliomalacia, fungal encephalitis, lymphocytic perivascular encephalitis, and toxoplasmosis), and non-neurologic diseases (septicemia, otitis media, and tetralogy of Fallot). A single case of each of these disorders was diagnosed, except for 2 cases of degenerative myelopathy and septicemia. A diagnosis was not obtained in 5 camelids. The median age in the other diagnosis group was 3.7 years (range: 1 month to 18 years), with a 75% female distribution (18 female, 6 male). Most of these cases (89%) were given anthelmintics within 1 week of postmortem examination. Presenting neurological deficits in the other diagnosis group included recumbency in 58% (14/24), hind-limb ataxia only in 4% (1/24), and ataxia of all 4 limbs in 8% (2/24). Cranial nerve deficits were seen in 33% (8/24) of camelids, and a single alpaca exhibited seizure activity.
The median eosinophil percentage, eosinophil concentration, and total nucleated cell concentration in the CSF of camelids diagnosed with P. tenuis on postmortem examination were significantly higher than camelids with a different diagnosis (Table 1). The ROC curve determined that a CSF eosinophil percentage >17% (the algorithm’s suggestion of optimal cut-off) yielded a Se of 85% (95% CI: 55–98) and Sp at 92% (95% CI: 73–99) for the diagnosis of P. tenuis in camelids in this population (Fig. 2A; Tables 2, 3). While this test retains its Se of 85% at eosinophil percentage cut-offs down to >2%, the Sp decreases to 88% (95% CI: 68–97; Fig. 2A; Tables 2, 3). Comparatively, the ROC curve for CSF eosinophil concentration showed that an absolute eosinophil concentration of >1.4 eosinophils/µl (the algorithm’s suggestion of optimal cut-off) yielded a Se of 85% (95% CI: 55–98) and Sp at 96% (95% CI: 79–100) for the diagnosis of P. tenuis in camelids in this population. This test retains its Se of 85% at cut-offs down to >0 eosinophils/µl; however, the Sp decreases to 83% (95% CI: 63–95; Fig. 2B; Tables 2, 4). The ROC curves (explicitly, the lower point estimates of the area under the curves [AUC]) for CSF total nucleated cell concentration and protein concentration show that these are less sensitive and less specific tests than the eosinophil percentage and concentration (Table 2). For example, a CSF total nucleated cell concentration >24 cells/µl (program-suggested cut-off) is 77% sensitive (95% CI: 46–95) and 88% specific (95% CI: 68–97) for the diagnosis of P. tenuis. Protein concentration in CSF was poorly diagnostic for P. tenuis infection, with a Se of 77% (95% CI: 46–95) and Sp of 62% (95% CI: 41–81) at the program-suggested cut-off of protein concentration >0.5 g/l. At the Bonferroni-adjusted P-value for significance (P < 0.013), neither the rank-sum test (Table 1, P = 0.048) nor the AUC (Table 2, P = 0.029) for protein indicated that increased CSF protein concentrations could differentiate P. tenuis camelids from those with other conditions.
Selected cerebrospinal fluid cytology results for camelids with Parelaphostrongylus tenuis infection and those with other diagnoses.
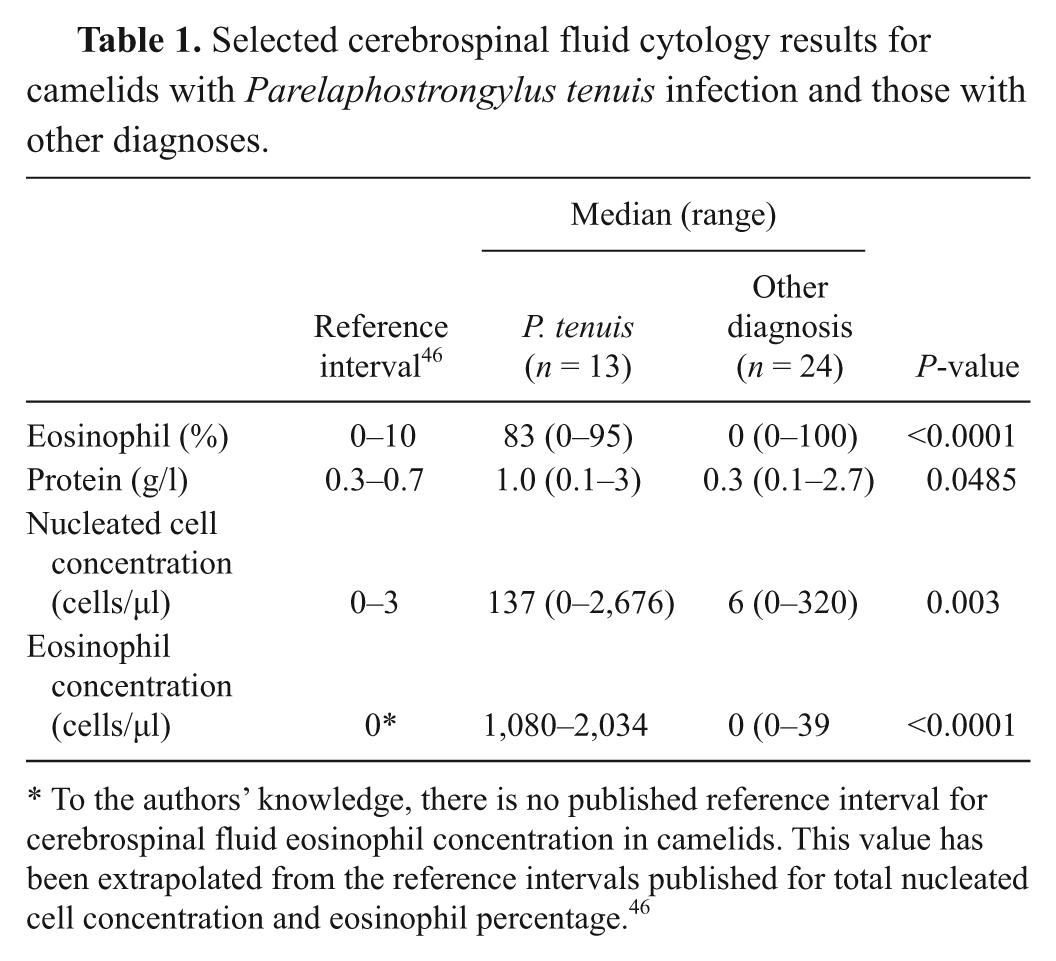
To the authors’ knowledge, there is no published reference interval for cerebrospinal fluid eosinophil concentration in camelids. This value has been extrapolated from the reference intervals published for total nucleated cell concentration and eosinophil percentage. 46
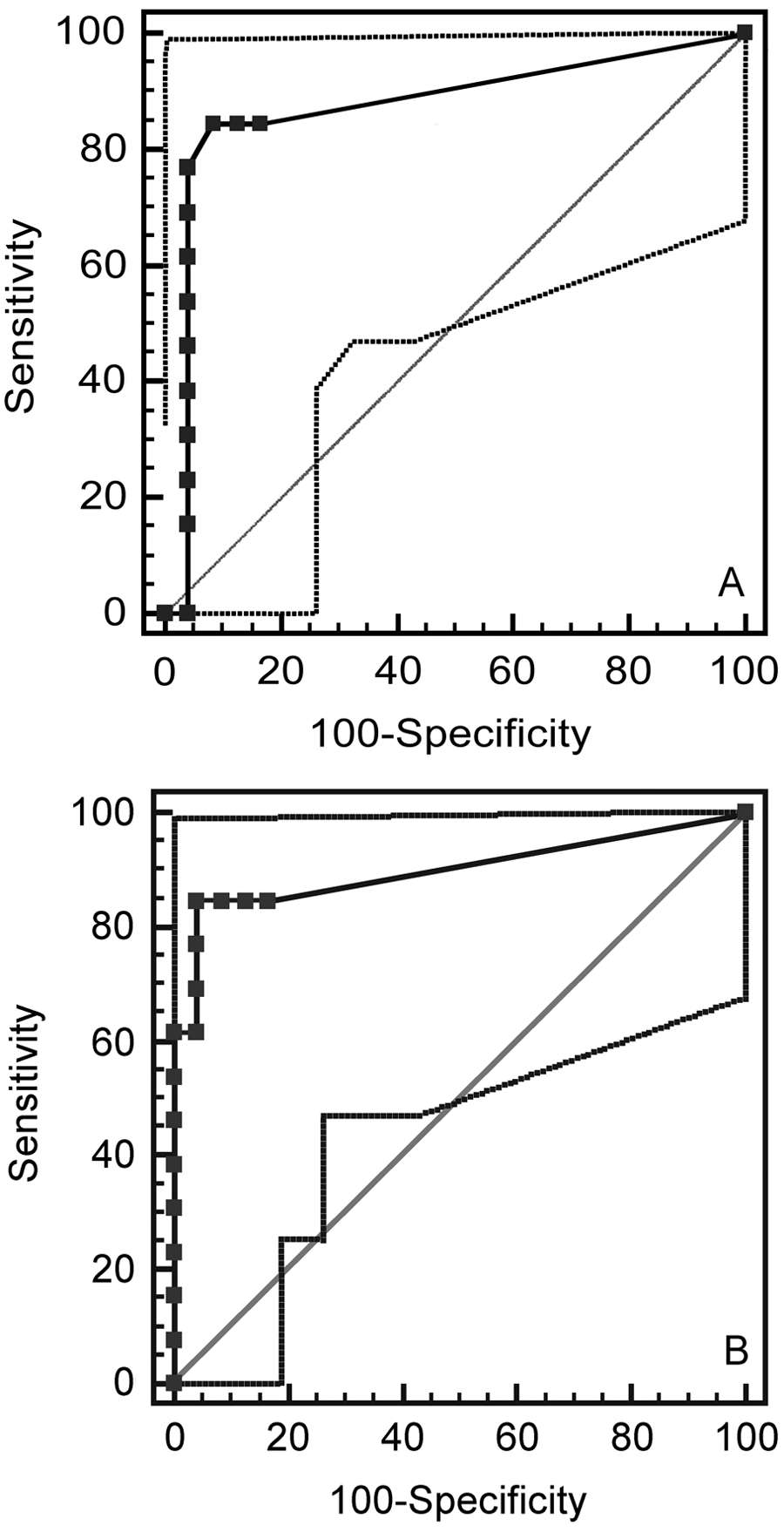
Receiver operating characteristic curve for cerebrospinal fluid: (
Area under the curve (AUC) as measured by receiver operating characteristic curves for selected cerebrospinal fluid (CSF) cytologic results in camelids.*
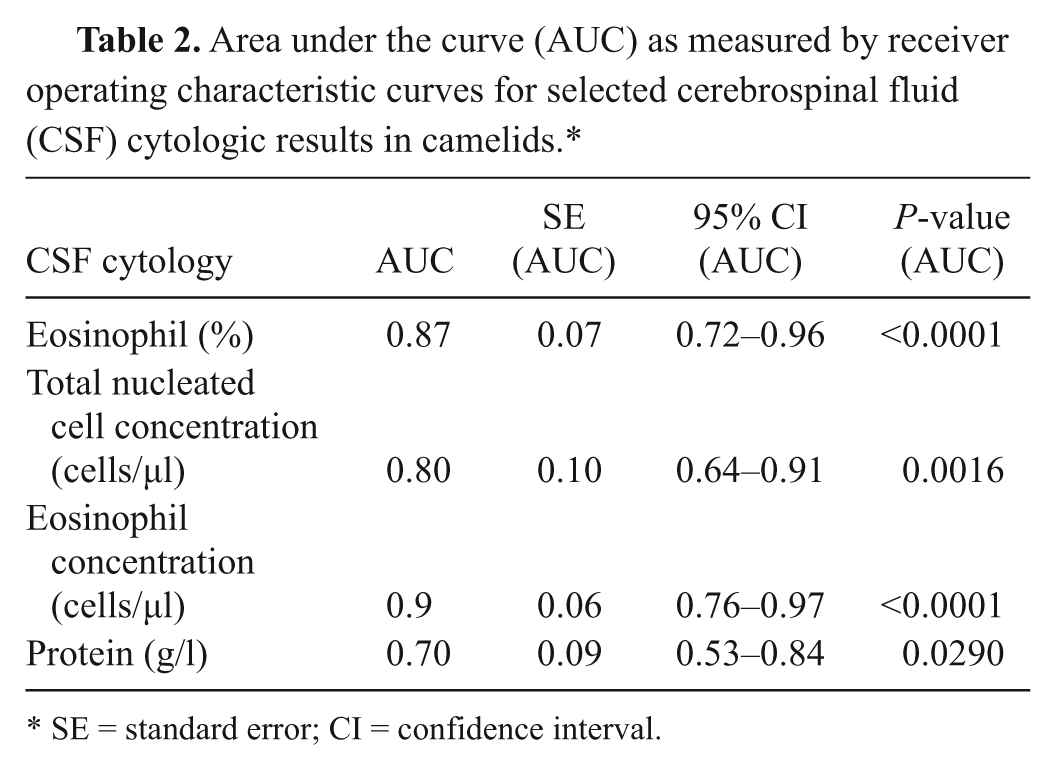
SE = standard error; CI = confidence interval.
Sensitivity and specificity (determined by receiver operating characteristic curves) of cerebrospinal fluid (CSF) eosinophil percentage for the diagnosis of Parelaphostrongylus tenuis in camelids.
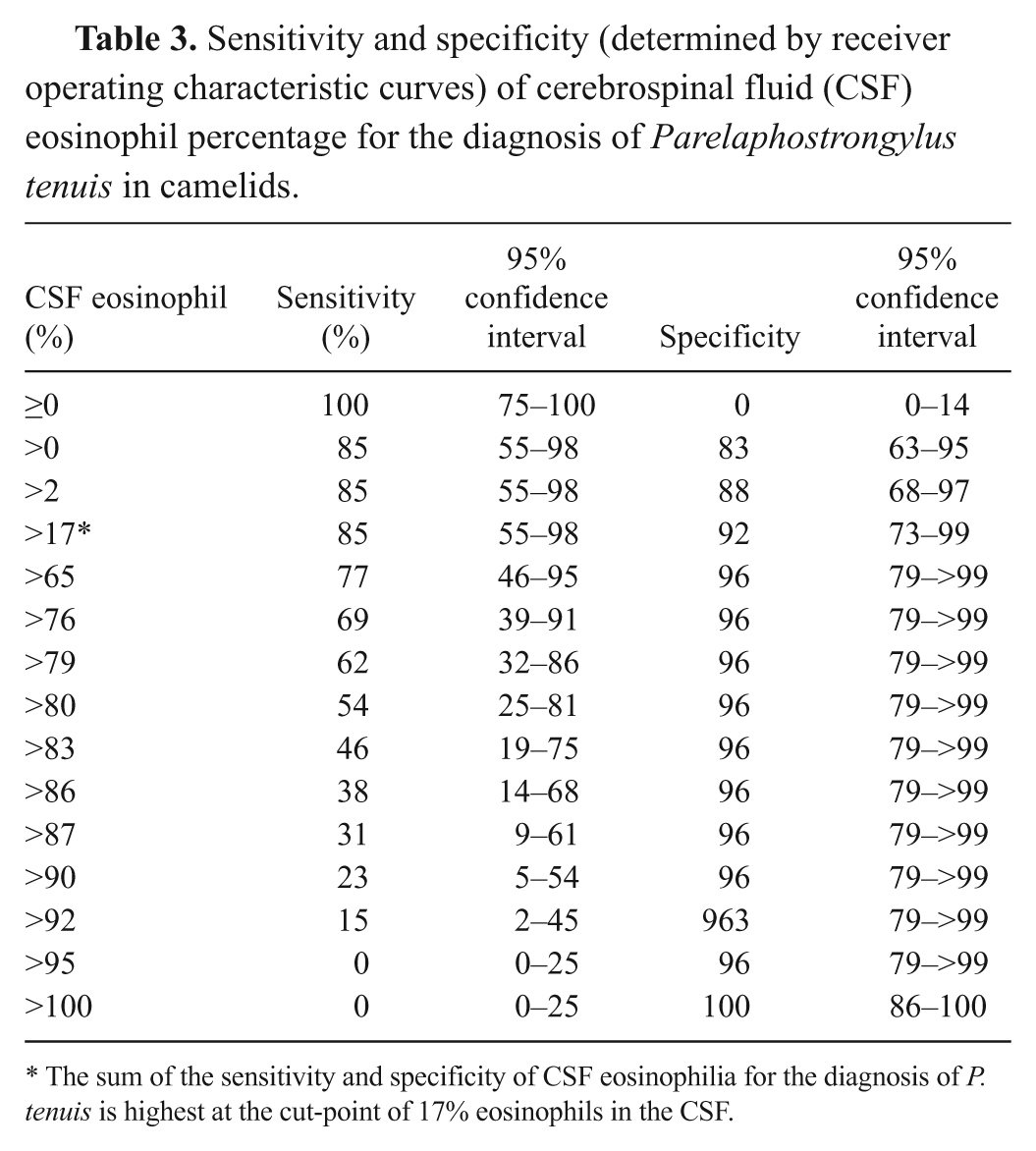
The sum of the sensitivity and specificity of CSF eosinophilia for the diagnosis of P. tenuis is highest at the cut-point of 17% eosinophils in the CSF.
Sensitivity and specificity (determined by receiver operating characteristic curves) of cerebrospinal fluid (CSF) eosinophil concentration for the diagnosis of Parelaphostrongylus tenuis in camelids.
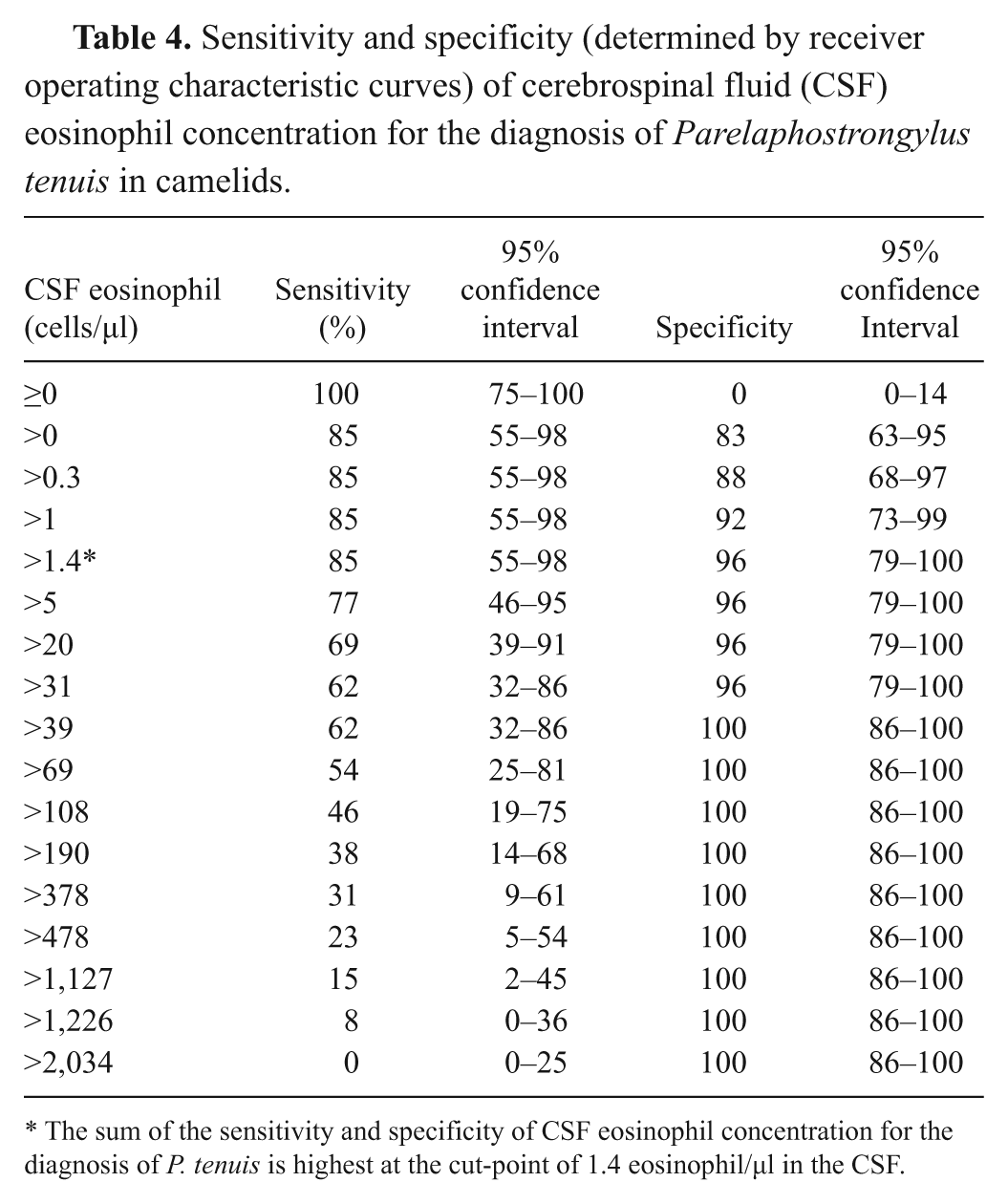
The sum of the sensitivity and specificity of CSF eosinophil concentration for the diagnosis of P. tenuis is highest at the cut-point of 1.4 eosinophil/µl in the CSF.
Two camelids in the “other diagnosis” group (2/24; 8%) had an increased percentage of CSF eosinophils (published reference interval: 0–10% 46 ) but were not diagnosed with P. tenuis at postmortem examination. The first case was an adult alpaca with a total nucleated cell concentration of 1 cell/µl in the CSF, with all cells being identified as eosinophils (100%). This alpaca presented with signs of marked depression, decreased pupillary light response, and seizure activity. Histopathology revealed small numbers of perivascular lymphocytes, neutrophils, and macrophages within the cerebrocortical white and gray matter. A conclusive diagnosis was not made in this alpaca. The second case was a 4-week-old alpaca cria diagnosed with Toxoplasma gondii infection at postmortem examination, with an increased total nucleated cell concentration of 60 nucleated cells/µl (published reference interval: 0–3 cells/μl 46 ), consisting of 65% eosinophils. In contrast to these 2 cases, 85% (11/13) of camelids with a postmortem diagnosis of P. tenuis had increased CSF total nucleated cell concentrations, absolute eosinophil concentrations, and eosinophil percentages, and 78% (10/13) also had increased CSF protein concentrations (published reference interval: 0.3–0.7 g/l 46 ).
Discussion
In the absence of commercially available molecular-based techniques for the diagnosis of P. tenuis in camelids, antemortem diagnosis can only be made presumptively, and histopathology is needed to provide a tentative diagnosis. The current study demonstrates that the presence of eosinophils in CSF, both the eosinophil percentage and concentration, are sensitive and specific means of reaching an antemortem diagnosis of P. tenuis in camelids in areas where white-tailed deer are endemic. Increased CSF protein and total nucleated cell concentration were not diagnostic due to their lower Se and Sp.
Eosinophilic inflammation within the CSF is a characteristic feature of cerebrospinal parasitism.5,20,29 Therefore, it is not surprising that the eosinophil percentage and concentration were highly sensitive for the diagnosis of P. tenuis. Despite the various durations of clinical disease (median: 3 days, range: 1–15 days) and prior treatment in the P. tenuis camelids described herein, CSF eosinophil percentage above the reported reference interval (0–10% 46 ) was a common finding. However, 2 camelids lacked CSF eosinophilia on cytology but were diagnosed with P. tenuis based upon postmortem histopathologic findings. Some reports have suggested that CSF cytology varies with the duration of P. tenuis infection, and some cases may not have high percentages of CSF eosinophils, resulting in a false-negative diagnosis. Cerebrospinal fluid mononuclear pleocytosis has been observed prior to definitive postmortem diagnosis of P. tenuis in llamas and goats27,38 and preceded eosinophilic pleocytosis in 2 out of 6 experimentally infected llamas. 38 Lymphocytic pleocytosis, without eosinophils, was also observed in chronic cases of P. tenuis in Limousin cattle, 28 suggesting that both acute and chronic infections with P. tenuis may not result in high percentages of CSF eosinophils. In addition, the effect of anthelmintic treatment on CSF eosinophil percentage in camelids with P. tenuis has not been thoroughly evaluated to the authors’ knowledge and could potentially reduce the proportion of eosinophils in the CSF. A differential diagnosis of P. tenuis should therefore not be excluded in camelids with characteristic clinical signs of infection, but that lack high percentages of eosinophils in CSF. In such cases, absolute eosinophil concentration in CSF may aid in reaching a diagnosis of P. tenuis. In cases with no increase in eosinophil percentages or concentrations, it would be worthwhile still treating for P. tenuis where there is known exposure to white-tailed deer and an alternative cause for the clinical signs cannot be found. It is important to note that the Se estimates in the current study may have been skewed by the strict requirement for a necropsy diagnosis of P. tenuis, which may bias included cases toward more severely affected animals. However, it is not known if CSF eosinophil percentage or concentration correlates to the severity of clinical signs in camelids with P. tenuis infection.
It is important to interpret the eosinophil percentage in light of the total nucleated cell concentration, a point highlighted by 1 case that was given an open diagnosis at postmortem, in which CSF was characterized by a normal nucleated cell concentration that consisted only of eosinophils. Because the total nucleated cell concentration was not increased in this camelid, the dominance of eosinophils was not interpreted as an abnormal finding. In addition, increased eosinophil percentage and concentration are unlikely to be specific for P. tenuis in camelids outside areas endemic to white-tailed deer. Aberrant migration of a variety of parasites within the central nervous system of other species can result in increased CSF eosinophil percentage and concentration, as seen in the case of toxoplasmosis in the present study. Other examples include Setaria spp. in cattle, Taenia multiceps in sheep, Halicephalobus gingivalis in horses (formerly Micronema deletrix), and Cryptococcus neoformans, Neospora caninum, and Baylisascaris procyonis in dogs.9,33,39,42,48 In Europe, camelids, sheep, and goats are susceptible to cerebrospinal nematodiasis and CSF eosinophilia due to Elaphostrongylus cervi and Elaphostrongylus rangiferi, which are closely related to P. tenuis in the Elaphostrongylinae subfamily.3,17–19 In addition, nonparasitic causes of CSF eosinophilia must be considered and have been reported in several noncamelid species (e.g., idiopathic eosinophilic meningoencephalomyelitis and acute intervertebral disc herniation in dogs 48 and neonatal meningitis and central nervous system abscesses in cattle). 39
Lumbosacral collection of CSF from camelids is somewhat invasive, but it is not difficult to perform. However, it appears to be infrequently performed by field practitioners, likely due to lack of experience with the procedure and sample handling and costs. Furthermore, most camelids with characteristic clinical signs of P. tenuis infection are frequently treated, with response to treatment becoming the means of antemortem diagnosis. However, because several camelids in the “other diagnosis” category presented with similar clinical signs to camelids with P. tenuis, collection of CSF would be useful to help exclude this diagnosis, particularly if there is known exposure to white-tailed deer. Furthermore, a CSF tap would be recommended if a camelid does not respond to conventional P. tenuis therapy.
The current study documents that most camelids diagnosed with P. tenuis (85%) have increased CSF eosinophil percentage, eosinophil concentration, nucleated cell concentration, and protein concentration. The present study also indicates that cut-offs of >17% eosinophils or >1.4 eosinophils/µl in CSF are sensitive and specific tests for the antemortem diagnosis of P. tenuis in camelids residing in regions populated by white-tailed deer.
Footnotes
a.
Analytical Software, Tallahassee, FL.
b.
MedCalc for Windows (version 11.2.0.0), MedCalc Software, Mariakerke, Belgium.
Declaration of conflicting interests
The author(s) declare no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
