Abstract
A novel gammaherpesvirus was identified in a large flying fox (Pteropus vampyrus) with conjunctivitis, blepharitis, and meibomianitis by nested polymerase chain reaction and sequencing. Polymerase chain reaction amplification and sequencing of 472 base pairs of the DNA-dependent DNA polymerase gene were used to identify a novel herpesvirus. Bayesian and maximum likelihood phylogenetic analyses indicated that the virus is a member of the genus Percavirus in the subfamily Gammaherpesvirinae. Additional research is needed regarding the association of this virus with conjunctivitis and other ocular pathology. This virus may be useful as a biomarker of stress and may be a useful model of virus recrudescence in Pteropus spp.
Bats are classified within the second largest mammalian order, Chiroptera, with approximately 1,100 species making up more than 20% of extant mammals. 26 As such a large portion of mammalian biodiversity, in addition to the gregarious social characteristics of many bat species, they also have diverse associated microbiomes. 17 The large flying fox (Pteropus vampyrus) ranges from southern Myanmar and Thailand eastward to the Philippines and southward to Sumatra, Java, Borneo, and Timor. 13 Pteropus vampyrus is a social species, and colonies of over 15,000 individuals have been reported. 13 The bats are listed under the Convention on International Trade in Endangered Species of Wild Fauna and Flora appendix II, and are under significant pressure throughout their range due to both habitat loss and intensive hunting for food and traditional medicine. 13 Populations appear to be declining rapidly, and current pressures on the population are unsustainable. 7
Pteropus vampyrus is also an important reservoir host for Nipah virus (NiV), a paramyxovirus in the genus Henipavirus. 24 NiV has been responsible for epidemics of fatal encephalitis in human and other mammals. 24 While the ecologic strategy of many paramyxoviruses typically involves rapid replication and spread to naïve hosts, followed by death or immunity of the host, 4 henipaviruses appear to commonly maintain persistent or latent infections in infected bat hosts. 24 Pteropus vampyrus infected with NiV have been found to have virus recrudescence 10–11 months after antibody titers had initially waned. 24
There has been significant investigation that has gone into viral discovery in bats, with over 100 viruses detected from bats. 2 Despite this discovery effort, as well as the choice of P.vampyrus for the National Institutes of Health Mammalian Genome Project (http://www.genome.gov/25521745), knowledge of the viral microbiome of P. vampyrus beyond NiV is very limited. An additional member of the genus Henipavirus has been partially characterized from a P. vampyrus. 22 There is a report of gammaretrovirus sequences in the genome that are likely endogenous. 5
Members of family Herpesviridae are large DNA viruses with significant host fidelity that often appear to have codiverged evolutionarily along with their hosts. 15 All herpesviruses investigated to date are able to remain latent in their natural host; the viruses also frequently undergo active shedding without associated clinical symptoms, or with only mild symptoms. 15 Fatal infections are usually rare in the natural, immunocompetent host. 15 Four alphaherpesviruses, 7 betaherpesviruses, and 10 gammaherpesviruses have been identified from bats.6,10,14,19,28-31,33 Only 1 of these is known to use a host in the genus Pteropus, an alphaherpesvirus found in P. lylei. 19
An 11-year-old, intact, male large flying fox presented for ocular discharge and periocular pruritus. This individual was housed in a captive breeding facility (Gainesville, FL) where he received annual clinical examinations and routine preventative health care. His medical history was unremarkable.
On initial examination, the patient was bright and alert. Conjunctival hyperemia, chemosis, and periocular mucoid discharge from the right eye were the only abnormalities noted. A swab of the ocular discharge was obtained for cytology and aerobic bacterial culture. Cytologic examination using Wright–Giemsa stain revealed marked neutrophilic conjunctivitis with frequent intracellular small bacilli and cocci. Treatment was initiated with neomycin/polymyxin B/bacitracin ophthalmic ointment a (3-mm strip in the right eye [OD] twice daily) pending culture results. Heavy growth of Staphylococcus aureus and a nonhemolytic Staphylococcus sp. were sensitive to neomycin and polymyxin B. After 2 weeks, the ocular discharge was improved, but conjunctivitis was still present and had progressed to moderate periocular swelling. A firm swelling of the proximal right tibia was noted on physical exam, which histology confirmed as a suppurative bacterial osteitis with aggressive bony proliferation and cortical lysis. Treatment for the osteitis included azithromycin b (10 mg/kg by mouth [PO] once daily for 4 months), meloxicam c (0.12 mg/kg PO once daily for 3.5 weeks), and tramadol d (2 mg/kg PO twice daily for 4.5 months).
Three weeks after the onset of conjunctivitis, ophthalmic examination revealed nuclear sclerosis, and a focal corneal epithelial bulla was noted at the lateral aspect of the cornea. Blepharitis and meibomianitis as well as an anterior cortical incipient cataract were noted in the right eye. Hypertonic saline ointment e (3-mm strip in the left eye [OS] twice daily) was added to the treatments to address the corneal edema. The following week, the conjunctivitis appeared improved so ophthalmic medications were discontinued while azithromycin, meloxicam, and tramadol were continued for treatment of the osteitis. The right eyelid continued to have some thickening, but did not appear to have active inflammation. Fibrosis due to the previous chronic inflammation was suspected. Two weeks after discontinuation of ophthalmic medications, the conjunctiva of the right nictitating membrane became hyperemic; neomycin/polymyxin B/bacitracin ophthalmic ointment (3-mm strip OD once daily for 4 weeks) was prescribed. No significant improvement was noted; the ointment was changed to neomycin/polymyxin B/dexamethasone ophthalmic ointment (3-mm strip OD twice daily for 2 weeks).
Over the next several months, the patient was treated with several medications including pentoxifylline f (25 mg/kg PO twice daily for 16 weeks), doxycycline g (2.5 mg/kg PO twice daily for 8 weeks), and continued intermittent use of neomycin/polymyxin B/bacitracin or neomycin/polymyxin B/dexamethasone ophthalmic ointment. There was minimal to no grossly visible response of the blepharitis and conjunctivitis.
Due to a lack of response to any medication, 1 year after initial presentation, the patient was anesthetized, and a 3-mm full-thickness wedge biopsy of the upper eyelid margin was performed. A portion of the eyelid biopsy was fixed in formalin. Histology revealed an interstitial to periadnexal to occasionally perivascular, predominantly lymphoplasmacytic, blepharitis, meibomian adenitis, and myositis with myocyte degeneration. Multifocal rupture of meibomian glands was associated with pyogranulomatous inflammation. The conjunctival mucosa was irregularly hyperplastic and spongiotic, with mild lymphocytic and neutrophilic infiltration. Gomori methenamine silver stain, Brown and Brenn, and acid-fast stains did not reveal a fungal or bacterial etiology. A portion of the biopsy was not fixed and was submitted for herpesvirus polymerase chain reaction (PCR).
DNA was extracted from the biopsy using a commercial kit h following the manufacturer’s instructions. Nested PCR amplification of a partial sequence of the herpesvirus DNA-dependent DNA polymerase gene was carried out using consensus primers from a previously described protocol. 27 Additional sequence was obtained using a different seminested protocol with specific reverse primers designed from the sequence obtained using a previously described protocol 27 (round 1: HerpPVA-Rev2 5’-AAGGTGCACGGTG TAATATTTTC-3’; round 2: HerpPVA-Rev1 5’-TGCACCT TCCTTGGTACGTT-3’) and consensus forward primer DFA. 27 Products were separated by agarose gel electrophoresis and sequenced directly using DNA sequencers. i
Initial nested PCR amplification resulted in a product of 166 bp after primers were edited out. The modified protocol using specific reverse primers produced a sequence length of 472 bp after primers were edited out. The sequence was submitted to GenBank under accession number KC510287.
Amino acid sequences homologous to the predicted 157 amino acid sequence of the DNA-dependent DNA polymerase sequence were aligned using MAFFT. 11 To choose an amino acid substitution model, ProtTest 2.4 was used. 1 Human herpesvirus 1 (GenBank accession no. X14112) was designated as the outgroup. Bayesian analyses of the amino acid alignment was performed using MrBayes 3.1.2 21 on the CIPRES server (Miller MA, Pfeiffer W, Schwartz T: 2010, Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop (GCE), November 14, New Orleans, LA, pp. 1–8). Maximum likelihood (ML) analysis of the alignment was performed using RAxML on the CIPRES server. 25 Bootstrap analysis was used to test the strength of the tree topology. 8 Numbers of bootstrap replicates were determined using the previously described stopping criteria.
The best model of protein evolution selected by ProtTest 2.4 was LG +G+I, which was implemented in RAxML. For the Bayesian analysis, because of the lack of the LG model in MrBayes 3.1.2, the second best model available (RtRev +G+I+F) was used. Bayesian posterior probabilities were calculated from 1,000,000 iterations. The Bayesian tree is shown in Figure 1. Stopping criteria for ML bootstrapping were reached after 500 subsets. Bootstrap values as percentages from ML analysis are shown on the Bayesian tree (Fig. 1). The virus was distinct from all other herpesviral sequences in GenBank, and is consistent with a novel species. The posterior probability for this novel flying fox herpesvirus clustering within the genus Percavirus in the subfamily Gammaherpesvirinae was 99% and the ML bootstrap value was 93% in the analyses.
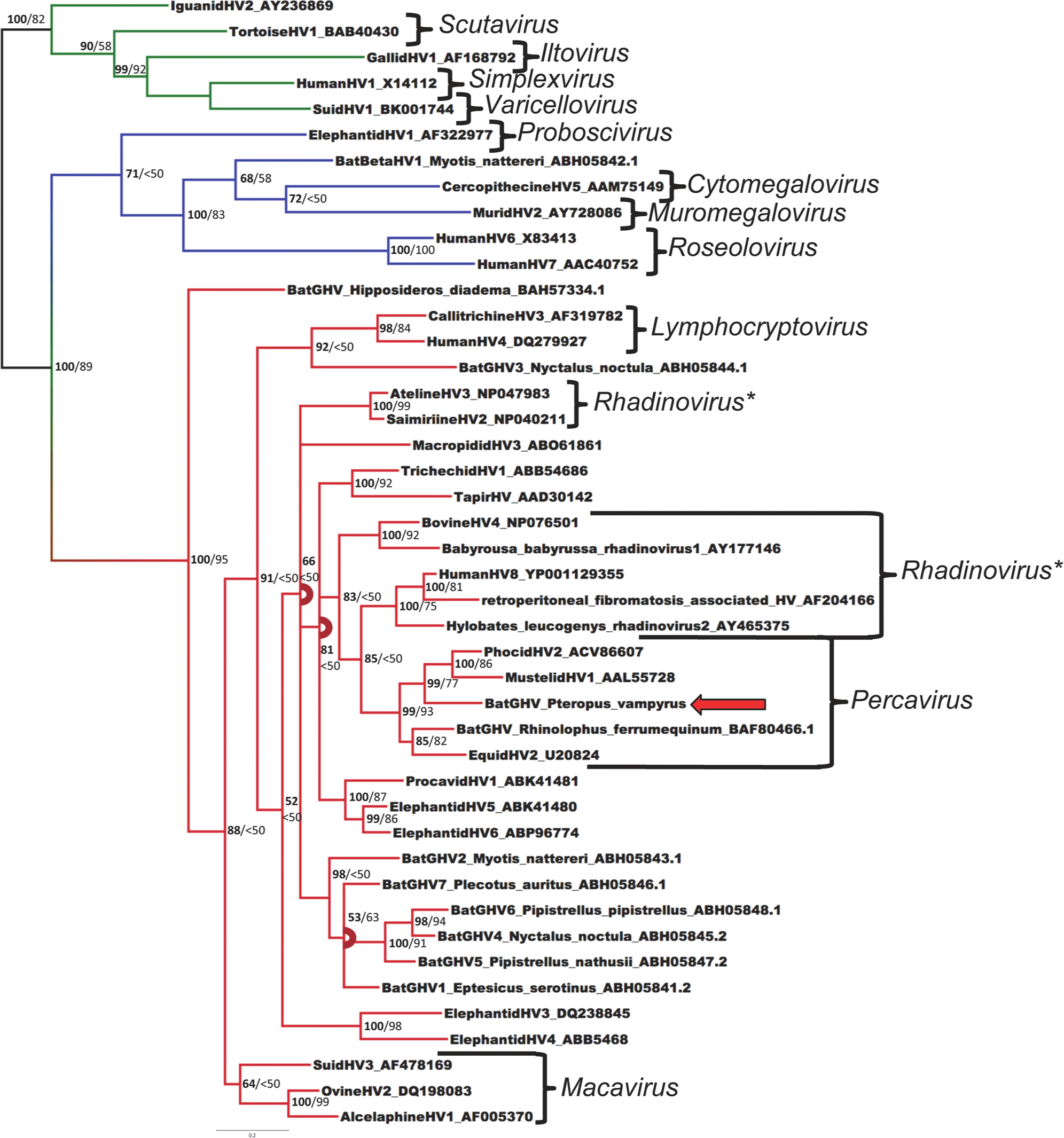
Bayesian phylogenetic tree of predicted amino acid sequences of herpesviral DNA-dependent DNA polymerase sequences based on MAFFT alignment. Multifurcations are marked with arcs. Bayesian posterior probabilities of clusters as percentages are in bold, and maximum likelihood bootstrap values for clusters based on 500 resamplings are given to the right. Human herpesvirus 1 was used as an outgroup. The Pteropus vampyrus gammaherpesvirus is indicated with an arrow. The subfamily Alphaherpesvirinae is marked in green, Betaherpesvirinae is in blue, and Gammaherpesvirinae is in red. Brackets demarcate genera. Accession numbers of sequences retrieved from GenBank are given after the name.
Cidofovir j (1 drop in both eyes [OU] twice daily for 7 weeks), a monophosphate nucleotide analogue that inhibits viral DNA synthesis, was prescribed as an antiviral; however, no significant improvement was noted. The cidofovir was discontinued, and prednisolone k (1 mg/kg PO once daily) was started to suppress inflammation. The prednisolone appeared to reduce the patient’s pruritus and conjunctivitis and was continued indefinitely to maintain the patient’s comfort. No further recurrences of the conjunctivitis have been noted although the eyelid continues to have mild swelling.
Although the novel gammaherpesvirus was identified in this flying fox with chronic conjunctivitis, it is not clear if the virus caused or contributed to the conjunctivitis or if it was an incidental finding. The patient did not have an apparent clinical response when treated with an antiviral. However, it is possible that topical application did not get good penetration, or that cidofovir does not have good activity against this virus. It is not known whether this herpesvirus is endemic in P. vampyrus. The bat had been housed in enclosures with other Pteropus species, including P. hypomelanus, P. giganteus, P. rodricensis, and P. poliocephalus. Pteropus hypomelanus is a sympatric species with P. vampyrus; the others are not.
Despite the discovery of a number of bat herpesviruses, there is relatively little information available on herpesviral disease in bats. Intraperitoneal inoculation of Suid herpesvirus 1, an alphaherpesvirus, into big brown bats (Eptesicus fuscus) resulted in clinical pseudorabies, although natural infections have not been reported. 20 An alphaherpesvirus isolated from 9 Malagasy straw-colored fruit bats (Eidolon dupraenum) is thought to be associated with keratitis: the virus was isolated when prevalence of keratitis in the bats was 10%, but could not be isolated the following year when keratitis was less prevalent. 19
A number of herpesviruses have been associated with conjunctivitis or keratitis in diverse species. An alphaherpesvirus isolated from the straw-colored fruit bat (Eidolon helvum) has been found to cause ocular disease in mice. 16 Meibomianitis has been attributed to Human herpesvirus 1. 23 In the genus Percavirus, Equid herpesvirus 2 has been associated with respiratory disease as well as keratoconjunctivitis in horses; however, it has also been isolated from clinically healthy horses. 12
It is also plausible that increased viral levels are resultant from, rather than causal to, concurrent disease. Stress is well documented as a cause of herpesvirus reactivation. 9 This factor may make the herpesvirus useful for studies as a biomarker; Panine herpesvirus 1 quantitative PCR has been used as a biomarker for social dominance–associated stress in chimpanzees. 32 Different stress factors have been shown to have different effects on herpesviral reactivation. 9 For some stress factors, herpesvirus recrudescence can be detected in experimental situations even when cortisol elevation cannot, 3 making it a potentially highly useful biomarker.
Understanding stress and virus recrudescence in Pteropus species is crucial. Anthropogenic stresses have the potential to cause NiV recrudescence in P. vampyrus. 18 This herpesvirus may represent a stress biomarker and a model for virus recrudescence in P. vampyrus that represents less risk to lab personnel than NiV.
In conclusion, a novel herpesvirus was identified from a large flying fox with blepharitis and meibomianitis. Further study is merited to examine the potential association of this virus with ocular clinical signs. The virus may also be useful for the study of stress and viral recrudescence in flying foxes.
Footnotes
Acknowledgements
The authors thank the Lubee Bat Conservancy for their support with this case. This project is published as Lubee Bat Conservancy Publication #163.
Notes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
